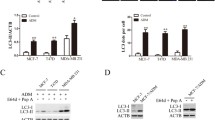
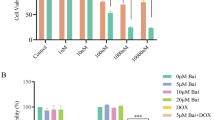
Abstract
The effectiveness of an apoptosis-targeting therapy may be limited in tumor cells with defects in apoptosis. Recently, considerable attention in the field of cancer therapy has been focused on the mammalian rapamycin target (mTOR), inhibition of which results in autophagic cell death. In our study using multidrug-resistant v-Ha-rastransformed NIH3T3 (Ras-NIH 3T3/Mdr) cells, we demonstrated that rapamycin-induced cell death may result from 2 different mechanisms. At high rapamycin concentrations (≥ 100 nM), cell death may occur via an autophagy-dependent pathway, whereas at lower concentrations (≤ 10 nM), cell death may occur after G1-phase cell cycle arrest. This effect was accompanied by upregulation of p21Cip1 and p27Kip1 expression via an autophagy-independent pathway. We also tested whether inhibition of mTOR with low concentrations of rapamycin and ectopic Beclin-1 expression would further sensitize multidrug resistance (MDR)-positive cancer cells by upregulating autophagy. Rapamycin at low concentrations might be insufficient to initiate autophagosome formation in autophagy but Beclin-1 overexpression triggered additional processes downstream of mTOR during G1 cell cycle arrest by rapamycin. Our findings suggest that these combination strategies targeting autophagic cell death may yield significant benefits for cancer patients, because lowering rapamycin concentration for cancer treatment minimizes its side effects in patients undergoing chemotherapy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arceci, R.J., Stieglitz, K., and Bierer, B.E. (1992). Immunosuppressants FK506 and rapamycin function as reversal agents of the multidrug resistance phenotype. Blood 80, 1528–1536.
Cao, Y., and Klionsky, D.J. (2007). Physiological functions of Atg6/Beclin 1: a unique autophagy-related protein. Cell Res. 17, 839–849
Dancey, J.E. (2005). Inhibitors of the mammalian target of rapamycin. Expert Opin. Investig. Drugs 14, 313–328.
Daniel, F., Legrand, A., and Pessayre, D. (2007). Beclin1 mRNA strongly correlates with Bcl-XL mRNA expression in human hepatocellular carcinoma. Cancer Invest. 25, 226–231.
Faivre, S., Kroemer, G., and Raymond, E. (2006). Current development of mTOR inhibitors as anticancer agents. Nat. Rev. Drug Discov. 5, 671–688.
Franken, N.A., Rodermond, H.M., Stap, J., Haveman, J., and van Bree, C. (2006). Clongenic assay of cells in vitro. Nat. Protoc. 1, 2315–2319.
Gottesman, M.M. (2002). Mechanisms of cancer drug resistanace. Annu. Rev. Med. 53, 615–627.
Guertin, D.A., and Sabatini, D.M. (2005). An expanding role for mTOR in cancer. Trends Mol. Med. 11, 353–361.
Hamacher-Brady, A., Brady, N.R., and Gottlieb, R.A. (2006). Enhancing macroautophagy protects against ischemia/reperfusion injury in cardiac myocytes. J. Biol. Chem. 281, 29776–29787.
Hartford, C.M., and Ratain, M.J. (2007). Rapamycin: something old, something new, sometimes borrowed and now renewed. Clin. Pharmacol. Ther. 82, 381–388.
Kawamata, S., Sakaida, H., Hori, T., Maeda, M., and Uchiyama, T. (1998). The upregulation of p27Kip1 by rapamycin results in G1 arrest in exponentially growing T-cell lines. Blood 91, 561–569.
Kim, K.W., Hwang, M., Moretti, L., Jaboin, J.J., Cha, Y.I., and Lu, B. (2008). Autophagy upregulation by inhibitors of caspase-3 and mTOR enhances radiotherapy in a mouse model of lung cancer. Autophagy 4, 659–668.
King, M.A., Hands, S., Hafiz, F., Mizushima, N., Tolsovsky, A.M., and Wyttenbach, A. (2008). Rapamycin inhibits polyglutamine aggregation independently of autophagy by reducing protein synthesis. Mol. Pharmacol. 73, 1052–1063.
Komata, T., Kanzawa, T., Takeuchi, H., Germano, I.M., Schreiber, M., Kondo, Y., and Kondo, S. (2003). Antitumour effect of cyclindependent kinase inhibitors (p16(INK4A), p18(INK4C), p19(INK4D), p21(WAF1/CIP1) and p27(KIP1)) on malignant glioma cells. Br. J. Cancer 88, 1277–1280.
Koneri, K., Goi, T., and Hirono, Y. (2007). Beclin1 gene inhibits tumor growth in colon cancer cell lines. Anticancer Res. 27, 1453–1457.
Kuma, A., Hatano, M., Matsui, M., Yamamoto, A., Nakaya, H., Yoshimori, T., Ohsumi, Y., Tokuhisa, T., and Mizushima, N. (2004). The role of autophagy during the early neonatal starvation period. Nature 432, 1032–1036.
Lee, M., Ahn, J.-H., and Eum, K.-H. (2009). The differences in biological properties between parental and v-Ha-ras transformed NIH3T3 cells. Cancer Res. Treat. 41, 93–99.
Marinov, M., Fischer, B., and Arcaro, A. (2007). Targeting mTOR signaling in lung cancer. Crit. Rev. Oncol. Hematol. 63, 172–182.
Mashima, T., and Tsuruo, T. (2005). Defects of the apoptotic pathway as therapeutic target against cancer. Drug Resist. Updat. 8, 339–343.
Mazzanti, R., Platini, F., Bottini, C., Fantappie, O., Solazzo, M., and Tessitore, L. (2009). Down-regulation of the HGF/MET autocrine loop induced by celecoxib and mediated by P-gp in MDRpositive human hepatocellular carcinoma cell line. Biochem. Pharmacol. 78, 21–32.
Meric-Bernstam, F., and Gonzalez-Angulo, A.M. (2009). Targeting the mTOR signaling network for cancer therapy. J. Clin. Oncol. 27, 2278–2287.
Miracco, C., and Cosci, E. (2007). Protein and mRNA expression of autophagy gene Beclin1 in human brain tumours. Int. J. Oncol. 30, 429–436.
Moretti, L., Yang, E.S., Kim, K.W., and Lu, B. (2007). Autophagy signaling in cancer and its potential as novel target to improve anticancer therapy. Drug Resist. Updat. 10, 135–143.
Panwalkar, A., Verstovsek, S., and Giles, F.J. (2004). Mammalian target of rapamycin inhibition as therapy for hematologic malignancies. Cancer 100, 657–666.
Pawarode, A., Shukla, S., Minderman, H., Fricke, S.M., Pinder, E.M., O’Loughlin, K.L., Ambudkar, S.V., and Baer, M.R. (2007). Differential effects of the immunosuppressive agents cyclosporin A, tacrolimus and sirolimus on drug transport by multidrug resistance proteins. Cancer Chemother. Pharmacol. 60, 179–188.
Pop, I.V., Pop, L.M., Ghetie, M.A., and Vitetta, E.S. (2009). Targeting mammalian target of rapamycin to both downregulate and disable the P-glycoprotein pump in multidrug-resistant B-cell lymphoma cell lines. Leuk. Lymphoma 50, 1155–1162.
Sir, D., and Ou, J.H. (2010). Autophagy in viral replication and pathogenesis. Mol. Cells 29, 1–7.
Sun, Y., Liu, J.H., Jin, L., Lin, S.M., Yang, Y., Sui, Y.X., and Shi, H. (2010). Over-expression of the Beclin1 gene upregulates chemosensitivity to anti-cancer drugs by enhancing therapy-induced apoptosis in cervix squamous carcinoma CaSki cells. Cancer Lett. 294, 204–210.
Ullman, E., Fan, Y., Stawowczyk, M., Chen, H.M., Yue, Z., and Zong, W.X. (2008). Autophagy promotes necrosis in apoptosis-deficient cells in response to ER stress. Cell Death Differ. 15, 422–425.
Weppler, S.A., Krause, M., Zyromska, A., Lambin, P., Baumann, M., and Wouters, B.G. (2007). Response of U87 glioma xenografts treated with concurrent rapamycin and fractionated radiotherapy: possible role for thrombosis. Radiother. Oncol. 82, 96–104.
Wu, C.P., Calcagno, A.M., and Ambudkar, S.V. (2008). Reversal of ABC drug transporter-mediated multidrug resistance in cancer cells: evaluation of current strategies. Curr. Mol. Pharmacol. 1, 93–105.
Yousefi, S., Perozzo, R., Schmid, I., Ziemiecki, A., Schaffner, T., Scapozza, L., Brunner, T., and Simon, H.U. (2006). Calpainmediated cleavage of Arg5 switches autophagy to apoptosis. Nat. Cell Biol. 8, 1124–1132.
Yue, Z., Jin, S., Yang, C., Levine, A.J., and Heintz, N. (2003). Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc. Natl. Acad. Sci. USA 100, 15077–15082.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Eum, KH., Lee, M. Targeting the autophagy pathway using ectopic expression of Beclin 1 in combination with rapamycin in drug-resistant v-Ha-ras-transformed NIH 3T3 cells. Mol Cells 31, 231–238 (2011). https://doi.org/10.1007/s10059-011-0034-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-011-0034-6