Abstract
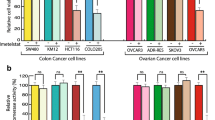
Telomerase activation is a key step in the development of human cancers. Interferon-β (IFN-β) signaling induces growth arrest in many tumors but the anticancer mechanism of IFN-β is poorly understood. In the present study, we show that IFN-β signaling represses telomerase activity and human telomerase reverse transcriptase (hTERT) transcription in ovarian cancer and suggest that this signaling is mediated by p21WAF1. IFN-β triggered down-regulation of telomerase activity and hTERT mRNA expression and also induced p21 expression, independently of p53 induction. Ectopic expression of p21 attenuated hTERT promoter activity. Murine embryonic fibroblasts (MEFs) genetically deficient in p21 (p21-/-) showed elevated (> 15 times) hTERT promoter activity compared to wild-type MEFs. Overexpression of p21 reduced the hTERT promoter activity of p21-/- MEFs and hTERT mRNA expression in HCT119 p21WAF1 null cell. These findings provide evidence that p21 is a potential mediator of IFN-β-induced attenuation of telomerase activity and tumor suppression.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Akiyama, M., Iwase, S., Horiguchi-Yamada, J., Saito, S., Furukawa, Y., Yamada, O., Mizoguchi, H., Ohno, T., and Yamada, H. (1999). Interferon-alpha repressed telomerase along with G1-accumulation of Daudi cells. Cancer Lett. 142, 23–30.
Anttila, M.A., Kosma, V.M., Hongxiu, J., Puolakka, J., Juhola, M., Saarikoski, S., and Syrjänen, K. (1999). p21/WAF1 expression as related to p53, cell proliferation and prognosis in epithelial ovarian cancer. Br. J. Cancer 79, 1870–1878.
Bae-Jump, V.L., Zhou, C., Gehrig, P.A., Whang, Y.E., and Boggess, J.F. (2006). Rapamycin inhibits hTERT telomerase mRNA expression, independent of cell cycle arrest. Gynecol. Oncol. 100, 487–494.
Bali, A., O’Brien, P.M., Edwards, L.S., Sutherland, R.L., Hacker, N.F., and Henshall, S.M. (2004). Cyclin D1, p53, and p21Waf1/ Cip1 expression is predictive of poor clinical outcome in serous epithelial ovarian cancer. Clin. Cancer Res. 10, 5167–5177.
Buchkovich, K.J., and Greider, C.W. (1996). Telomerase regulation during entry into the cell cycle in normal human T cells. Mol. Cell. Biol. 16, 1443–1454.
Buchynska, L.G., Nesina, I.P., Yurchenko, N.P., Bilyk, O.O., Grinkevych, V.N., and Svintitsky, V.S. (2007). Expression of p53, p21WAF1/CIP1, p16INK4A and Ki-67 proteins in serous ovarian tumors. Exp. Oncol. 29, 49–53.
Chawla-Sarkar, M., Leaman, D.W., and Borden, E.C. (2001). Preferential induction of apoptosis by interferon (IFN)-beta compared with IFN-alpha2: correlation with TRAIL/Apo2L induction in melanoma cell lines. Clin. Cancer Res. 7, 1821–1831.
Counter, C.M., Hirte, H.W., Bacchetti, S., and Harley, C.B. (1994). Telomerase activity in human ovarian carcinoma. Proc. Natl. Acad. Sci. USA 91, 2900–2904.
Crowe, D.L., and Nguyen, D.C. (2001). Rb and E2F-1 regulate telomerase activity in human cancer cells. Biochim. Biophys. Acta 1518, 1–6.
Damdinsuren, B., Nagano, H., Sakon, M., Kondo, M., Yamamoto, T., Umeshita, K., Dono, K., Nakamori, S., and Monden, M. (2003). Interferon-beta is more potent than interferon-alpha in inhibition of human hepatocellular carcinoma cell growth when used alone and in combination with anticancer drugs. Ann. Surg. Oncol. 10, 1184–1190.
De Maeyer, E., and De Maeyer-Guignard, J. (1998). Type I interferons. Int. Rev. Immunol. 17, 53–73.
Deiss, L.P., Feinstein, E., Berissi, H., Cohen, O., and Kimchi, A. (1995). Identification of a novel serine/threonine kinase and a novel 15-kD protein as potential mediators of the gamma interferon- induced cell death. Genes Dev. 9, 15–30.
el-Deiry, W.S., Tokino, T., Velculescu, V.E., Levy, D.B., Parsons, R., Trent, J.M., Lin, D., Mercer, W.E., Kinzler, K.W., and Vogelstein, B. (1993). WAF1, a potential mediator of p53 tumor suppression. Cell 75, 817–825.
Giandomenico, V., Vaccari, G., Fiorucci, G., Percario, Z., Vannuchi, S., Matarrese, P., Malorni, W., Romeo, G., and Affabris, G.R. (1998). Apoptosis and growth inhibition of squamous carcinoma cells treated with interferon-alpha, IFN-beta and retinoic acid are associated with induction of the cyclin-dependent kinase inhibitor p21. Eur. Cytokine Netw. 9, 619–631.
Hahn, W.C., Stewart, S.A., Brooks, M.W., York, S.G., Eaton, E., Kurachi, A., Beijersbergen, R.L., Knoll, J.H., Meyerson, M., and Weinberg, R.A. (1999). Inhibition of telomerase limits the growth of human cancer cells. Nat. Med. 5, 1164–1170.
Hendren, S.K., Prabakaran, I., Buerk, D.G., Karakousis, G., Feldman, M., Spitz, F., Menon, C., and Fraker, D.L. (2004). Interferon- beta gene therapy improves survival in an immunocompetent mouse model of carcinomatosis. Surgery 135, 427–436.
Henriksen, R., Strang, P., Wilander, E., Bäckström, T., Tribukait, B., and Oberg, K. (1994). p53 expression in epithelial ovarian neoplasms: relationship to clinical and pathological parameters, Ki-67 expression and flow cytometry. Gynecol. Oncol. 53, 301–306.
Hu, X., Bies, J., and Wolff, L. (2005). Interferon beta increases c- Myc proteolysis in mouse monocyte/macrophage leukemia cells. Leuk. Res. 29, 1307–1314.
Ichikawa, Y., Nishida, M., Suzuki, H., Yoshida, S., Tsunoda, H., Kubo, T., Uchida, K., and Miwa, M. (1994). Mutation of K-ras protooncogene is associated with histological subtypes in human mucinous ovarian tumors. Cancer Res. 54, 33–35.
Jackson, R.J., Engelman, R.W., Coppola, D., Cantor, A.B., Wharton, W., and Pledger, W.J. (2003). P21Cip1 Nullizygosity increases tumor metastasis in Irradiated mice. Cancer Res. 63, 3021–3025.
Jemal, A., Siegel, R., Ward, E., Murray, T., Xu, J., Smigal, C., and Thun, M.J. (2006). Cancer statistics. CA Cancer J. Clin. 56, 106–130.
Kar, R., Sen, S., Singh, A., Sharma, H., Kumar, S., Gupta, S.D., and Singh, N. (2007). Role of apoptotic regulators in human epithelial ovarian cancer. Cancer Biol. Ther. 6, 1101–1105.
Katayama, T., Saitoh, Y., Nemoto, C., Hirama, T., Isosu, T., and Murakawa, M. (2007). Effects of olprinone on neuromuscular blockade caused by vecuronium. Fukushima J. Med. Sci. 53, 61–69.
Kondo, S., Tanaka, Y., Kondo, Y., Hitomi, M., Barnett, G.H., Ishizaka, Y., Liu, J., Haqqi, T., Nishiyama, A., Villeponteau, B., et al. (1998). Antisense telomerase treatment: induction of two distinct pathways, apoptosis and differentiation. FASEB J. 12, 801–811.
Kyo, S., Kanaya, T., Takakura, M., Tanaka, M., Yamashita, A., Inoue, H., and Inoue, M. (1999). Expression of human telomerase subunits in ovarian malignant, borderline and benign tumors. Int. J. Cancer 80, 804–809.
Kyo, S., Takakura, M., Taira, T., Kanaya, T., Itoh, H., Yutsudo, M., Ariga, H., and Inoue, M. (2000). Sp1 cooperates with c-Myc to activate transcription of the human telomerase reverse transcriptase gene (hTERT). Nucleic Acids Res. 28, 669–677.
Lee, S.H., Kim, J.W., Lee, H.W., Cho, Y.S., Oh, S.H., Kim, Y.J., Jung, C.H., Zhang, W., and Lee, J.H. (2003). Interferon regulatory factor-1 (IRF-1) is a mediator for interferon-gamma induced attenuation of telomerase activity and human telomerase reverse transcriptase (hTERT) expression. Oncogene 22, 381–391.
Lee, S.H., Kim, J.W., Oh, S.H., Kim, Y.J., Rho, S.B., Park, K., Park, K.L., and Lee, J.H. (2005). IFN-gamma/IRF-1-induced p27kip1 down-regulates telomerase activity and human telomerase reverse transcriptase expression in human cervical cancer. FEBS Lett. 579, 1027–1033.
Lindner, D.J., and Borden, E.C. (1997). Effects of tamoxifen and interferon-beta or the combination on tumor-induced angiogenesis. Int. J. Cancer. 71, 456–461.
Maciejewski, J.P., Selleri, C., Sato, T., Cho, H.J., Keefee, L.K., Nathan, C.F., and Young, N.S. (1995). Nitric oxide suppression of human hematopoiesis in vitro. Contribution to inhibitory action of interferon-gamma and tumor necrosis factor-alpha. J. Clin. Invest. 96, 1085–1092.
Meden, H., Marx, D., and Rath, W. (1994). Elevated serum levels of a c-erbB-2 oncogene product in ovarian cancer patients and in pregnancy. Int. J. Gynecol. Patho. 13, 45–53.
Meyerson, M., Counter, C.M., Eaton, E.N., Ellisen, L.W., Steiner, P., Caddle, S.D., Ziaugra, L., Beijersbergen, R.L., Davidoff, M.J., Liu, Q., et al. (1997). hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell 90, 785–795.
Morrison, B.H., Bauer, J.A., Kalvakolanu, D.V., and Lindner, D.J. (2001). Inositol hexakisphosphate kinase 2 mediates growth suppressive and apoptotic effects of interferon-beta in ovarian carcinoma cells. J. Biol. Chem. 276, 24965–24970.
Morrison, B.H., Tang, Z., Jacobs, B.S., Bauer, J.A., and Lindner, D.J. (2005). Apo2L/TRAIL induction and nuclear translocation of inositol hexakisphosphate kinase 2 during IFN-beta-induced apoptosis in ovarian carcinoma. Biochem. J. 385, 595–603.
Nakamura, T.M., Morin, G.B., Chapman, K.B., Weinrich, S.L., Andrews, W.H., Lingner, J., Harley, C.B., and Cech, T.R. (1997). Telomerase catalytic subunit homologs from fission yeast and human. Science 277, 955–959.
Odaka, M., Sterman, D.H., Wiewrodt, R., Zhang, Y., Kiefer, M., Amin, K.M., Gao, G., Wilson, J.M., Barsoum, J., Kaiser, L.R., et al. (2001). Eradication of intraperitoneal and distant tumor by adenovirus-mediated interferon-beta gene therapy is attributable to induction of systemic immunity. Cancer Res. 61, 6201–6212.
Odaka, M., Wiewrodt, R., DeLong, P., Tanaka, T., Zhang, Y., Kaiser, L., and Albelda, S. (2002). Analysis of the immunologic response generated by Ad.IFN-beta during successful intraperitoneal tumor gene therapy. Mol. Ther. 6, 210–218.
Oh, S.T., Kyo, S., and Laimins, L.A. (2001). Telomerase activation by human papillimavirus type 16E6 protein: induction of human telomerase reverse transcriptase expression through Myc and GC-rich Sp1 binding sites. J. Virol. 75, 5559–5566.
Sanceau, J., Hiscott, J., Delattre, O., and Wietzerbin, J. (2000). IFN-beta induces serine phosphorylation of Stat-1 in Ewing’s sarcoma cells and mediates apoptosis via induction of IRF-1 and activation of caspase-7. Oncogene 19, 3372–3383.
Shats, I., Milyavsky, M., Tang, X., Stambolsky, P., Erez, N., Brosh, R., Kogan, I., Braunstein, I., Tzukerman, M., Ginsberg, D., et al. (2004). p53-dependent down-regulation of telomerase is mediated by p21waf1. J. Biol. Chem. 279, 50976–50985.
Shay, J.W., and Bacchetti, S. (1997). A survey of telomerase activity in human cancer. Eur. J. Cancer 33, 787–791.
Shiratsuchi, M., Muta, K., Umemura, T., Nishimura, J., Nawata, H., and Kozuru, M. (1999). Telomerase activity in myeloma cells is closely related to cell cycle status, but not to apoptotic signals induced by interferon-alpha. Lymphoma 34, 349–359.
Takakura, M., Kyo, S., Kanaya, T., Tanaka, M., and Inoue, M. (1998). Expression of human telomerase subunits and correlation with telomerase activity in cervical cancer. Cancer Res. 58, 1558–1561.
Varras, M.N., Sourvinos, G., Diakomanolis, E., Koumantakis, E., Flouris, G.A., Lekka-Katsouli, J., Michalas, S., and Spandidos, D.A. (1999). Detection and clinical correlations of ras gene mutations in human ovarian tumors. Oncology 56, 89–96.
Wan, M., Li, W.Z., Duggan, B.D., Felix, J.C., Zhao, Y., and Dubeau, L. (1997). Telomerase activity in benign and malignant epithelial ovarian tumors. J. Natl. Cancer Inst. 89, 437–441.
Wang, J., Xie, L.Y., Allan, S., Beach, D., and Hannon, G.J. (1998). Myc activates telomerase. Genes Dev. 12, 1769–1774.
Won, J., Yim, J., and Kim, T.K. (2002). Opposing regulatory roles of E2F in human telomerase reverse transcriptase (hTERT) gene expression in human tumor and normal somatic cells. FASEB. J. 16, 1943–1945.
Wu, K.J., Brandori, C., Amacker, M., Simon-Vermont, N., Polack, A., Lingner, J., and Dalla-Favera, R. (1999). Direct activation of TERT transcription by c-MYC. Nat. Genet. 21, 220–224.
Xu, D., Erickson, S., Szeps, M., Gruber, A., Sangfelt, O., Einhorn, S., Pisa, P., and Grander, D. (2000a). Interferon alpha down-regulates telomerase reverse transcriptase and telomerase activity in human malignant and nonmalignant hematopoietic cells. Blood 96, 4313–4318.
Xu, D., Wang, Q., Gruber, A., Chen, Z., Zaid, A., Selivanova, G., Peterson, C., Wiman, K.G., and Pisa, P. (2000b). Down regulation of telomerase reverse transcriptase mRNA expression by wild type p53 in human tumor cells. Oncogene 29, 5123–5133.
Zhang, X., Mar, V., Zhou, W., Harrington, L., and Robinson, M.O. (1999). Telomere shortening and apoptosis in telomerase-inhibited human tumor cells. Genes Dev. 13, 2388–2399.
Zhu, X., Kumar, R., Mandal, M., Sharma, N., Sharma, H.W., Dhingra, U., Sokoloski, J.A., Hsiao, R., and Narayanan R. (1996). Cell cycle dependent modulation of telomerase activity in tumor cells. Proc. Natl. Acad. Sci. USA 93, 6091–6095.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work
About this article
Cite this article
Lee, JH., Lee, SY., Lee, JH. et al. p21WAF1 is involved in interferon-β- induced attenuation of telomerase activity and human telomerase reverse transcriptase (hTERT) expression in ovarian cancer. Mol Cells 30, 327–333 (2010). https://doi.org/10.1007/s10059-010-0131-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-010-0131-y