Abstract
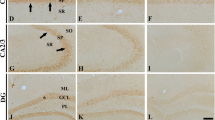
Phosphorylated extracellular signal-regulated kinase (pERK) mediates neuronal synaptic plasticity, long-term potentiation, and learning and memory in the hippocampus. In this study, we examined pERK1/2 immunoreactivity and its protein level in the gerbil hippocampus at various ages. In the postnatal month 1 (PM 1) group, very weak pERK1/2 immunoreactivity was detected in the hippocampus. In the CA1 region, pERK1/2 immunoreactivity was considerably increased in the stratum pyramidale in the PM 6 group. Thereafter, pERK1/2 immunoreactivity was decreased. In the CA2/3 region, pERK1/2 immunoreactivity increased in an age-dependent manner until PM 12. Thereafter, numbers of pERK1/2-immunoreactive neurons were decreased. However, in the mossy fiber zone, pERK1/2 immunostaining became stronger with age. In the dentate gyrus, a few pERK1/2-immunoreactive cells were observed until PM 12. In the PM 18 and 24 groups, numbers of pERK1/2-immunoreactive cells were increased, especially in the polymorphic layer. In Western blot analysis, pERK1/2 level in the gerbil hippocampus was increased with age. These results indicate that total pERK1/2 levels are increased in the hippocampus with age. However pERK1/2 immunoreactivity in subregions of the gerbil hippocampus was changed with different pattern during normal aging.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aikawa, R., Komuro, I., Yamazaki, T., Zou, Y., Kudoh, S., Tanaka, M., Shiojima, I., Hiroi, Y., and Yazaki, Y. (1997). Oxidative stress activates extracellular signal-regulated kinases through Src and Ras in cultured cardiac myocytes of neonatal rats. J. Clin. Invest. 100, 1813–1821.
Boulton, T.G., Nye, S.H., Robbins, D.J., Ip, N.Y., Radziejewska, E., Morgenbesser, S.D., DePinho, R.A., Panayotatos, N., Cobb, M.H., and Yancopoulos, G.D. (1991). ERKs: a family of proteinserine/threonin kinases that are activated and tyrosine phosphorylated in response to insulin and NGF. Cell 65, 663–675.
Duchemin, A.M., Ren, Q., Mo, L., Neff, N.H., and Hadjiconstantinou, M. (2002). GM1 ganglioside induces phosphorylation and acti vation of Trk and Erk in brain. J. Neurochem. 81, 696–707.
English, J.D., and Sweatt, J.D. (1997). A requirement for the mitogenactivated protein kinase cascade in hippocampal long term potentiation. J. Biol. Chem. 272, 19103–19106.
Frye, C.A., and Walf, A.A. (2008). Progesterone enhances performance of aged mice in cortical or hippocampal tasks. Neurosci. Lett. 437, 116–120.
Gooney, M., Messaoudi, E., Maher, F.O., Bramham, C.R., and Lynch, M.A. (2004). BDNF-induced LTP in dentate gyrus is impaired with age: analysis of changes in cell signaling events. Neurobiol. Aging 25, 1323–1331.
Grewal, S.S., York, R.D., and Stork, P.J. (1999). Extracellularsignal-regulated kinase signalling in neurons. Curr. Opin. Neurobiol. 9, 544–553.
Hara, H., Onodera, H., Kato, H., and Kogure, K. (1992). Effects of aging on signal transmission and transduction systems in the gerbil brain: morphological and autoradiographic study. Neuroscience 46, 475–488.
Harris, F.M., Brecht, W.J., Xu, Q., Mahley, R.W., and Huang, Y. (2004). Increased tau phosphorylation in apolipoprotein E4 transgenic mice is associated with activation of extracellular signal-regulated kinase: modulation by zinc. J. Biol. Chem. 279, 44795–44801.
He, W.B., Zhang, J.L., Hu, J.F., Zhang, Y., Machida, T., and Chen, N.H. (2008). Effects of glucocorticoids on age-related impairments of hippocampal structure and function in mice. Cell. Mol. Neurobiol. 28, 277–291.
Hu, Y., Schett, G., Zou, Y., Dietrich, H., and Xu, Q. (1998). Abundance of platelet-derived growth factors (PDGFs), PDGF receptors and activation of mitogen-activated protein kinases in brain decline with age. Mol. Brain Res. 53, 521–528.
Kelleher, III. R.J., Govindarajan, A., and Tonegawa, S. (2004). Translational regulatory mechanisms in persistent forms of synaptic plasticity. Neuron 44, 59–73.
Kim, H.G., Hong, S.M., Kim, S.J., Park, H.J., Jung, H.I., Lee, Y.Y., Moon, J.S., Lim, H.W., Park, E.H., and Lim, C.J. (2003). Agerelated changes in the activity of antioxidant and redox enzymes in rats. Mol. Cells 16, 278–284.
Klesse, L.J., and Parada, L.F. (1999). Trks: signal transduction and intracellular pathway. Micr. Res. Tech. 45, 210–216.
Kurino, M., Fukunaga, K., Ushio, Y., and Miyamoto, E. (1995). Activation of mitogen-activated kinase in cultured rat hippocampal neurons by stimulation of glutamate receptors. J. Neurochem. 65, 1282–1289.
Kuroki, Y., Fukushima, K., Kanda, Y., Mizuno, K., and Watanabe, Y. (2001). Neuroprotection by estrogen via extracellular signalregulated kinase against quinolinic acid-induced cell death in the rat hippocampus. Eur. J. Neurosci. 13, 472–476.
Kyriakis, J.M., and Avruch, J. (1996). Protein kinase cascades activated by stress and inflammatory cytokines. Bioessays 18, 567–577.
Loskota, W.A., Lomax, P., and Verity, M.A. (1974). A stereotaxic atlas of the Mongolian Gerbil Brain (Meriones unguiculatus) (Michigan, Ann Arbor Science).
Lukiw, W.J., Pappolla, M., Pelaez, R.P., and Bazan, N.G. (2005). Alzheimer’s disease—a dysfunction in cholesterol and lipid metabolism. Cell. Mol. Neurobiol. 25, 475–483.
Lynch, A.M., and Lynch, M.A. (2001). The age-related increase in IL-1 type I receptor in rat hippocampus is coupled with an increase in caspase-3 activation. Eur. J. Neurosci. 15, 1779–1788.
Maher, F.O., Nolan, Y., and Lynch, M.A. (2005). Downregulation of IL-4-induced signalling in hippocampus contributes to deficits in LTP in the aged rat. Neurobiol. Aging 26, 717–728.
Mo, L., Ren, Q., Duchemin, A.M., Neff, N.H., and Hadjiconstantinou, M. (2005). GM1 and ERK signaling in the aged brain. Brain Res. 1054, 125–134.
Namura, S., Iihara, K., Takami, S., Nagata, I., Kikuchi, H., Matsushita, K., Moslowitz, M.A., Bonventre, J.V., and Alessandrini, A. (2002). Intravenous administration of the MEK inhibitor UO126 affords brain protection against forebrain ischemia and focal cerebral ischemia. Proc. Natl. Acad. Sci. USA 98, 11569–11574.
Ortiz, J., Harris, H.W., Guitart, X., Terwilliger, R.Z., Haycock, J.W., and Nestler, E.J. (1995). Extracellular signal-regulated protein kinases (ERKs) and ERK kinase (MEK) in brain: regional distribution and regulation by chronic morphine. J. Neurosci. 15, 1285–1297.
Park, E.M., Joh, T.J., Volpe, B.T., Chu, C.K., Song, G., and Cho, S. (2004). A neuroprotective role for extracellular signal-regulated kinase in N-acetyl-O-methyldopamine-treated hippocampal neurons after exposure to in vitro and in vivo ischemia. Neuroscience 123, 147–154.
Rosen, L.B., Ginty, D.D., Weber, M.J., and Greenberg, M.E. (1994). Membrane depolarization and calcium influx stimulate MEK and MAP kinase via activation of Ras. Neuron 12, 1207–1221.
Sgambato, V., Pages, C., Rogard, M., Besson, M.J., and Caboche, J. (1998). Extracellular signal-regulated kinase (ERK) controls immediate early gene induction on corticostriatal stimulation. J. Neurosci. 18, 8814–8825.
Song, G.Y., Kang, J.S., Lee, S.Y., and Myung, C.S. (2007). Regionspecific reduction of Gβ4 expression and induction of the phosphorylation of PKB/Akt and ERK1/2 by aging in rat brain. Pharmacol. Res. 56, 295–302.
Stariha, R.L., Kikuchi, S., Siow, Y.L., Pelech, S.L., Kim, M., and Kim, S.U. (1997). Role of extracellular signal-regulated protein kinases 1 and 2 in oligodendroglial process extension. J. Neurochem. 68, 945–953.
Sugino, T., Nozaki, K., Takagi, Y., Hattori, I., Hashimoto, N., Moriguchi, T., and Nishida, E. (2000). Activation of mitogen-activated protein kinases after transient forebrain ischemia in gerbil hippocampus. J. Neurosci. 20, 4506–4514.
Suzuki, T., Okumura-Noiji, K., and Nishida, E. (1995). ERK2-type mitogen-activated protein kinase (MAPK) and its substrates in postsynaptic density fractions from the brain. Neurosci. Res. 22, 277–285.
Sweatt, J.D. (2001). The neuronal MAPK cascade: a biochemical signal integration system subserving synaptic plasticity and memory. J. Neurochem. 76, 1–10.
Tokalov, S.V., Gruener, S., Schindler, S., Iagunov, A.S., Baumann, M., and Abolmaali, N.D. (2007). A number of bone marrow mesenchymal stem cells but neither phenotype nor differentiation capacities changes with age of rats. Mol. Cells 24, 255–260.
Tsunemi, A., Utsuyama, M., Seidler, B.K., Kobayashi, S., and Hirokawa, K. (2005). Age-related decline of brain monoamines in mice is reversed to young level by Japanese herbal medicine. Neurochem. Res. 30, 75–81.
Wati, H., Kudo, K., Qiao, C., Kuroki, T., and Kanba, S. (2006). A decreased survival of proliferated cells in the hippocampus is associated with a decline in spatial memory in aged rats. Neurosci. Lett. 399, 171–174.
Xi, Z.Q., Wang, X.F., He, R.Q., Li, M.W., Liu, X.Z., Wang, L.Y., Zhu, X., Xiao, F., Sun, J.J., Li, J.M., et al. (2007). Extracellular signalregulated protein kinase in human intractable epilepsy. Eur. J. Neurol. 14, 865–872.
Yang, J., Yu, Y., and Duerksen-Hughes, P.J. (2003). Protein kinases and their involvement in the cellular responses to genotoxic stress. Mutat. Res. 543, 31–58.
Yao, H., Sadoshima, S., Ooboshi, H., Sato, Y., Uchimura, H., and Fujishima, M. (1991). Age-related vulnerability to cerebral ischemia in spontaneously hypertensive rats. Stroke 22, 1414–1418.
Yoo, K.Y., Hwang, I.K., Eum, W.S., Kim, D.W., Kwon, Y.G., Kang, T.C., Choi, S.Y., Kim, Y.S., and Won, M.H. (2006). Differential effects and changes of ceruloplasmin in the hippocampal CA1 region between adult and aged gerbils after transient cerebral ischemia. Neurosci. Res. 55, 134–141.
Zhen, X., Uryu, K., Cai, G., Johnson, G.P., and Friedman, E. (1999). Age-associated impairment in brain MAPK signal pathways and the effect of caloric restriction in Fischer 344 rats. J. Gerontol. A Biol. Sci. Med. Sci. 54, B539–B548.
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Lee, C.H., Yoo, KY., Park, O.K. et al. Phosphorylated extracellular signal-regulated kinase 1/2 immunoreactivity and its protein levels in the gerbil hippocampus during normal aging. Mol Cells 29, 373–378 (2010). https://doi.org/10.1007/s10059-010-0046-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-010-0046-7