Abstract
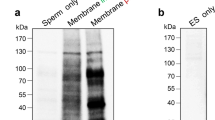
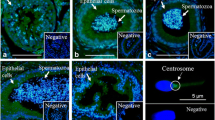
During epididymal transit, mammalian sperm acquire selected proteins secreted by the epididymis. We previously showed that a disintegrin and metalloprotease (ADAM) 7 is expressed specifically in the epididymis and transferred to the sperm surface during epididymal transit. Here, we show that mouse ADAM7 secreted to the epididymal lumen is associated with membranous vesicles known as epididymosomes. Furthermore, we found that ADAM7 can be transferred directly from epididymal vesicles to sperm and that it is an integral plasma membrane protein in sperm. Thus, our study provides new information regarding the unique mode of secretion and interaction of ADAM7 during the epididymis-to-sperm transfer process.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arienti, G., Carlini, E., De Cosmo, A.M., Di Profio, P., and Palmerini, C.A. (1998). Prostasome-like particles in stallion semen. Biol. Reprod. 59, 309–313.
Aumuller, G., Renneberg, H., Schiemann, P.J., Wilhelm, B., Seitz, J., Konrad, L., and Wennemuth, G. (1997). The role of apocrine released proteins in the post-testicular regulation of human sperm function. Adv. Exp. Med. Biol. 424, 193–219.
Aumuller, G., Wilhelm, B., and Seitz, J. (1999). Apocrine secretion—fact or artifact? Ann. Anat. 181, 437–446.
Brown, D.A., and London, E. (2000). Structure and function of sphingolipid- and cholesterol-rich membrane rafts. J. Biol. Chem. 275, 17221–17224.
Cho, C., Bunch, D.O., Faure, J.E., Goulding, E.H., Eddy, E.M., Primakoff, P., and Myles, D.G. (1998). Fertilization defects in sperm from mice lacking fertilin beta. Science 281, 1857–1859.
Cho, C. (2005). Mammalian ADAMs with testis-specific or -predominant expression. In The ADAM Family of Proteases, Vol. 4 in the Proteases in Biology and Disease series, N.M. Hoopers, and U. Lendekel, eds., (Netherlands: Springer), pp. 239–259.
Cooper, T.G. (1998). Interactions between epididymal secretions and spermatozoa. J. Reprod. Fertil. Suppl. 53, 119–136.
Cornwall, G.A., and Hsia, N. (1997). ADAM7, a member of the ADAM (a disintegrin and metalloprotease) gene family is specifically expressed in the mouse anterior pituitary and epididymis. Endocrinology 138, 4262–4272.
Edidin, M. (2003). The state of lipid rafts: from model membranes to cells. Annu. Rev. Biophys. Biomol. Struct. 32, 257–283.
Edwards, D.R., Handsley, M.M., and Pennington, C.J. (2008). The ADAM metalloproteinases. Mol. Aspects Med. 29, 258–289.
Frenette, G., and Sullivan, R. (2001). Prostasome-like particles are involved in the transfer of P25b from the bovine epididymal fluid to the sperm surface. Mol. Reprod. Dev. 59, 115–121.
Frenette, G., Lessard, C., and Sullivan, R. (2002). Selected proteins of “prostasome-like particles” from epididymal cauda fluid are transferred to epididymal caput spermatozoa in bull. Biol. Reprod. 67, 308–313.
Frenette, G., Lessard, C., Madore, E., Fortier, M.A., and Sullivan, R. (2003). Aldose reductase and macrophage migration inhibitory factor are associated with epididymosomes and spermatozoa in the bovine epididymis. Biol. Reprod. 69, 1586–1592.
Frenette, G., Legare, C., Saez, F., and Sullivan, R. (2005). Macrophage migration inhibitory factor in the human epididymis and semen. Mol. Hum. Reprod. 11, 575–582.
Frenette, G., Thabet, M., and Sullivan, R. (2006). Polyol pathway in human epididymis and semen. J. Androl. 27, 233–239.
Frohlich, O., and Young, L.G. (1996). Molecular cloning and characterization of EPI-1, the major protein in chimpanzee (Pan troglodytes) cauda epididymal fluid. Biol. Reprod. 54, 857–864.
Gatti, J.L., Metayer, S., Belghazi, M., Dacheux, F., and Dacheux, J.L. (2005). Identification, proteomic profiling, and origin of ram epididymal fluid exosome-like vesicles. Biol. Reprod. 72, 1452–1465.
Gesase, A.P., and Satoh, Y. (2003). Apocrine secretory mechanism: recent findings and unresolved problems. Histol. Histopathol. 18, 597–608.
Hermo, L., and Jacks, D. (2002). Nature’s ingenuity: bypassing the classical secretory route via apocrine secretion. Mol. Reprod. Dev. 63, 394–410.
Jin, E.J., Choi, Y.A., Sonn, J.K., and Kang, S.S. (2007). Suppression of ADAM 10-induced Delta-1 shedding inhibits cell proliferation during the chondro-inhibitory action of TGF-beta3. Mol. Cells 23, 139–147.
Kim, T., Oh, J., Woo, J.M., Choi, E., Im, S.H., Yoo, Y.J., Kim, D.H., Nishimura, H., and Cho, C. (2006). Expression and relationship of male reproductive ADAMs in mouse. Biol. Reprod. 74, 744–750.
Kirchhoff, C., and Hale, G. (1996). Cell-to-cell transfer of glycosylphosphatidylinositol-anchored membrane proteins during sperm maturation. Mol. Hum. Reprod. 2, 177–184.
Kravets, F.G., Lee, J., Singh, B., Trocchia, A., Pentyala, S.N., and Khan, S.A. (2000). Prostasomes: current concepts. Prostate 43, 169–174.
Legare, C., Berube, B., Boue, F., Lefievre, L., Morales, C.R., El-Alfy, M., and Sullivan, R. (1999). Hamster sperm antigen P26h is a phosphatidylinositol-anchored protein. Mol. Reprod. Dev. 52, 225–233.
Lin, Y.C., Sun, G.H., Lee, Y.M., Guo, Y.W., and Liu, H.W. (2001). Cloning and characterization of a complementary DNA encoding a human epididymis-associated disintegrin and metalloprotease 7 protein. Biol. Reprod. 65, 944–950.
Liu, H.W., Lin, Y.C., Chao, C.F., Chang, S.Y., and Sun, G.H. (2000). GP-83 and GP-39, two glycoproteins secreted by human epididymis are conjugated to spermatozoa during maturation. Mol. Hum. Reprod. 6, 422–428.
Llorente, A., de Marco, M.C., and Alonso, M.A. (2004). Caveolin-1 and MAL are located on prostasomes secreted by the prostate cancer PC-3 cell line. J. Cell Sci. 117, 5343–5351.
Nishimura, H., Cho, C., Branciforte, D.R., Myles, D.G., and Primakoff, P. (2001). Analysis of loss of adhesive function in sperm lacking cyritestin or fertilin beta. Dev. Biol. 233, 204–213.
Nishimura, H., Kim, E., Nakanishi, T., and Baba, T. (2004). Possible function of the ADAM1a/ADAM2 Fertilin complex in the appearance of ADAM3 on the sperm surface. J. Biol. Chem. 279, 34957–34962.
Oh, J., Woo, J.M., Choi, E., Kim, T., Cho, B.N., Park, Z.Y., Kim, Y.C., Kim, D.H., and Cho, C. (2005). Molecular, biochemical, and cellular characterization of epididymal ADAMs, ADAM7 and ADAM28. Biochem. Biophys. Res. Commun. 331, 1374–1383.
Perry, A.C., Jones, R., Barker, P.J., and Hall, L. (1992). A mammalian epididymal protein with remarkable sequence similarity to snake venom haemorrhagic peptides. Biochem. J. 286, 671–675.
Primakoff, P., and Myles, D.G. (2000). The ADAM gene family: surface proteins with adhesion and protease activity. Trends Genet. 16, 83–87.
Rejraji, H., Vernet, P., and Drevet, J.R. (2002). GPX5 is present in the mouse caput and cauda epididymidis lumen at three different locations. Mol. Reprod. Dev. 63, 96–103.
Rejraji, H., Sion, B., Prensier, G., Carreras, M., Motta, C., Frenoux, J.M., Vericel, E., Grizard, G., Vernet, P., and Drevet, J.R. (2006). Lipid remodeling of murine epididymosomes and spermatozoa during epididymal maturation. Biol. Reprod. 74, 1104–1113.
Saez, F., Frenette, G., and Sullivan, R. (2003). Epididymosomes and prostasomes: their roles in posttesticular maturation of the sperm cells. J. Androl. 24, 149–154.
Seals, D.F., and Courtneidge, S.A. (2003). The ADAMs family of metalloproteases: multidomain proteins with multiple functions. Genes Dev. 17, 7–30.
Sullivan, R., Saez, F., Girouard, J., and Frenette, G. (2005). Role of exosomes in sperm maturation during the transit along the male reproductive tract. Blood Cells Mol. Dis. 35, 1–10.
Sun, G.H., Lin, Y.C., Guo, Y.W., Chang, S.Y., and Liu, H.W. (2000). Purification of GP-83, a glycoprotein secreted by the human epididymis and conjugated to mature spermatozoa. Mol. Hum. Reprod. 6, 429–434.
Udenfriend, S., and Kodukula, K. (1995) How glycosylphosphatidylinositol-anchored membrane proteins are made. Annu. Rev. Biochem. 64, 563–591.
Weininger, R.B., Fisher, S., Rifkin, J., and Bedford, J.M. (1982). Experimental studies on the passage of specific IgG to the lumen of the rabbit epididymis. J. Reprod. Fertil. 66, 251–258.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Oh, J.S., Han, C. & Cho, C. ADAM7 is associated with epididymosomes and integrated into sperm plasma membrane. Mol Cells 28, 441–446 (2009). https://doi.org/10.1007/s10059-009-0140-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-009-0140-x