Abstract
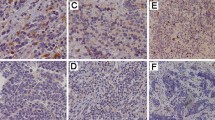
Livin is a recently identified member of the Inhibitor-of-Apoptosis protein (IAP) family of antiapoptosis proteins, and expression has been reported in melanoma and some types of carcinoma. We evaluated livin expression in paraffin-embedded tumor tissue from 68 patients with neuroblastoma (NB) and 7 NB cell lines by immunoperoxidase using an anti-livin monoclonal antibody. Eighteen (26.5%) of the 68 NB tumor tissues showed high livin expression, 36 (53%) showed low-intermediate expression, and 14 (20.5%) were negative. Similarly, 4 NB cell lines showed high livin expression, and 3 showed intermediate expression. In primary NB tissue, livin was observed mainly in tumor neuropil, an extension of tumor cell cytoplasm, and the cytoplasm itself. By reverse transcriptase–polymerase chain reaction, livin expression was confirmed in all 7 NB lines and in frozen tissue from 1 of 3 primary tumors examined to date, in agreement with immunohistochemical data; both livin α and β isoforms were detected. In the NB cases, we further analyzed the correlation between livin expression and clinical and biological features with established prognostic significance (i.e., age at diagnosis, stage, histology, and MYCN oncogene status), and patients’ outcome. Livin expression alone did not appear to have an effect on survival; however, patients with high livin expression and amplified MYCN had significantly decreased survival compared with patients lacking both markers or with either of these markers alone. These results suggest that (a) livin is expressed in primary and cultured neuroblastoma cells and (b) high livin expression may identify a subset of neuroblastoma patients with a particularly poor prognosis among those with MYCN amplified tumors.



Similar content being viewed by others
References
Maris JM, Matthay KK. Molecular biology of neuroblastoma. J Clin Oncol 1999;17:2264–2279
Brodeus GM. Neuroblastoma: biological insights into a clinical enigma. Nat Rev 2003;3:203–216
Look AT, Hayes FA, Nitsclike R, et al. Clinical relevance of tumor cell ploidy and N-myc gene amplification in, childhood neuroblastoma: a Pediatric Oncology Group Study. J Clin Oncol 1991; 9:581–591
Seeger RC, Brodeur GM, Sather H, et al. Association of multiple copies of the N-myc oncogene with rapid progression of neuroblastomas. N Engl J Med 1985;313:1111–1116
Shimada H, Chatten J, Newton WA Jr, et al. Histopathologic prognostic factors in neuroblastic tumors: definition of subtypes of ganglioneuroblastoma and an age-linked classification of neuroblastomas. J Natl Cancer Inst 1984, 73:405–416
Nitsclike R, Smith EI, Shochat S, et al. Localized neuroblastoma treated by surgery: a Pediatric Oncology Group study. J Clin Oncol 1988;6:1271–1279
Alvarado CS, London WB, Look AT, et al. Natural history and biology of stage A neuroblastoma: a Pediatric Oncology Group study. J Pediatr Hematol Oncol 2000;22:197–205
Perez CA, Matthay KK, Atkinson JB et al. Biologic variables in the outcome of stages I and II neuroblastoma treated with surgery as primary therapy: A Children’s Cancer Group study. J Clin Oncol 2000;18:18–26
Kawa K, Ohnuma N, Kaneko M, et al. Long-term survivors of advanced neuroblastoma with MYCN amplification: a report of 19 patients surviving disease-free for the more than 66 months. J Clin Oncol 1999;10:3216–3220
LaCasse EC, Baird S, Komeluk RG, et al. The inhibitors of apoptosis (IAPs) and their emerging role in cancer. Oncogene 1998;17:3247–3259
Clem RJ, Duckett CS. The IAP genes; unique arbitrators of cell death. Trends Biochem Sci 1998;23:159–162
Deveraux QL, Stennicke HR, Salvesen GS, et al. Endogenous inhibitors of caspases. J Clin Immunol 1999;19:388–398
Lin JH. KIAP, a novel member of the inhibitor of apoptosis protein family. Biochem Biophys Res Commun 2000;279:820–831
Ashhab Y, Alian A, Polliack A, et al. Two splicing variants of livin with different biological properties, and tissue distribution pattern. FEBS Lett 2001;495:56–60
Kasof K, Gomes BC. Livin, a novel inhibitor-of-apoptosis protein family member. J Biol Chem 2000;276:3238–3246
Vucic D, Stennicke HR, Pisabarro MT, et al. ML-IAP, a novel inhibitor of apoptosis preferentially expressed in melanomas. Curr Biol 2000;10:1359–1366
Brodeur GM, Pritchard J, Berthold F, et al. Revisions of the international criteria for neuroblastoma diagnosis, staging and response to treatment. J Clin Oncol 1993;11:1466–1477
Seeger RC, Brodeur GM, Sather H, et al. Association of multiple copies of the N-myc oncogene with rapid progression of neuroblastomas. N Engl J Med 1985;313:1111–1116
Shapiro DN, Valentine MB, Rowe ST, et al. Detection of N-myc gene amplification by fluorescence in-situ hybridization. Diagnostic utility for neuroblastoma. Am J Pathol 1993;142:1339–1346
Gazzaniga P, Gradilone A, Guilliani L, et al. Expression and prognostic significance of Livin, Survivin and other apoptosis-related genes in the progression of superficial bladder cancer. Ann Oncol 2003;4:85–90
Nachmias B, Ashhab Y, Bucholtz V, et al. Caspase-mediated cleavage converts livin from an anti-apoptotic to pro-apoptotic factor: implications for drug-resistant melanoma. Cancer Res 2003;63:6340–6349
Sanna M, Correia JS, Ducrey O, et al. IAP suppression of apoptosis involves distinct mechanisms: the TAK1/JNK1 signaling cascade and caspase inhibition. Mol Cell Biol 2001;22:1754–1766
Cole MD, McMahon SB. The Myc oncoprotein: a critical evaluation of transactivation and target gene regulation. Oncogene 1999;18:2916–2924
Fulda S, Lutz W, Schwab M, Debatin KM. MycN sensitizes neuroblastoma cells for drug-induced apoptosis. Oncogene 1999;18:1479–1486
Acknowledgments
This study was supported by a grant from CURE Childhood Cancer, Inc. We thank Dr. Nao Ikegaki (Emory University) for critical reading of this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Harry W. Findley and Carlos S. Alvarado contributed equally to this study.
Rights and permissions
About this article
Cite this article
Kim, DK., Alvarado, C.S., Abramowsky, C.R. et al. Expression of Inhibitor-of-Apoptosis Protein (IAP) Livin by Neuroblastoma Cells: Correlation with Prognostic Factors and Outcome. Pediatr Dev Pathol 8, 621–629 (2005). https://doi.org/10.1007/s10024-005-4108-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10024-005-4108-3