Abstract
Precipitation variability has been predicted to increase in a global warmer climate, and is expected to greatly affect plant growth, interspecies interactions, plant community composition, and other ecosystem processes. Although previous studies have investigated the effect of intra-annual rainfall variability on plant growth and ecosystem dynamics, the impacts of interannual rainfall variability remain understudied. This paper uses satellite data and develops a new mechanistic model to investigate the response of tree–grass composition to increasing interannual rainfall variability in arid to sub-humid ecosystems along the Kalahari Transect in Southern Africa. Both satellite data and model results show that increasing interannual rainfall fluctuations favor deep-rooted trees over shallow-rooted grasses in drier environments (that is, mean annual rainfall, MAP < 900–1000 mm) but favor grasses over trees in wetter environments (that is, MAP > 900–1000 mm). Trees have a competitive advantage over grasses in dry environments because their generally deeper root systems allow them to have exclusive access to the increased deep soil water resources expected to occur in wet years as a result of the stronger interannual rainfall fluctuations. In relatively wet environments, grasses are favored because of their high growth rate that allows them to take advantage of the window of opportunity existing in years with above average precipitation and thus increase fire-induced tree mortality. Thus, under increasing interannual rainfall fluctuations both direct effects on soil water availability and indirect effects mediated by tree–grass interactions and fire dynamics are expected to play an important role in determining changes in plant community composition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change studies predict an increase in precipitation variability at intra-annual, interannual, and decadal time scales (Easterling and others 2000; Fischer and others 2013; IPCC 2013). Such changes could result from a variety of mechanisms, including an increase of water-holding capacity in a warmer atmosphere (Kharin and others 2007; Liu and others 2013) and changes in atmospheric circulation (for example, intensification of El Niño Southern Oscillation) (Easterling and others 2000; Levis and others 2011). Increasing evidence indicates that precipitation variability is an important driver of ecological processes, including plant growth, interspecies interactions and community composition, primary productivity, and carbon and water fluxes (Knapp and others 2008; Reichstein and others 2013; Holmgren and others 2013; Zeppel and others 2014).
Previous studies have investigated the effects of intra-annual rainfall variability on plant and ecosystem processes (D’Odorico and others 2003; Knapp and others 2008; Kulmatiski and Beard 2013a; Zeppel and others 2014). Studies on the effect of interannual rainfall variability on soil moisture dynamics and plant water stress have also attracted some attention (Ridolfi and others 2000; D’Odorico and others 2000), but the impacts of interannual rainfall fluctuations on species composition and ecosystem productivity remain less investigated. Indeed, research in this field is strongly limited by the availability of long-term field observational data and technical capability to measure all relevant variables (Fatichi and Ivanov 2014). Long-term field campaigns have seldom addressed the impacts of interannual rainfall variability on ecosystem dynamics (Munson and others 2012; Collins and Xia 2015; but see Knapp and others 2002). Indeed, in field experiments with no rainfall manipulations, changes in rainfall variability may often be associated with changes in mean rainfall, thus making it difficult to differentiate the effects of changes in rainfall variability from those of changes in mean rainfall (Hsu and others 2012; Reyer and others 2013). To overcome these limitations, process-based models—possibly integrated with field or satellite data—could offer alternative approaches to gain a mechanistic understanding of the ecological impacts of trends in rainfall variability (Reyer and others 2013; Fatichi and Ivanov 2014).
Some studies have shown that interannual rainfall variability could affect the interannual fluctuations of vegetation productivity (Knapp and others 2002; Fatichi and Ivanov 2014; Hsu and Adler 2014). It has been argued that a nonlinear (“concave down”) relationship between mean annual precipitation and net primary productivity (ANPP) (also known as Jensen’s inequality) could lead to a negative response of mean ANPP in grasslands to increasing interannual rainfall variability (Hsu and others 2012). Using satellite data, Holmgren and others (2013) have reported a consistent positive response of tree cover to increasing rainfall variability in the dry tropics, but mixed responses of tree cover to interannual rainfall variability in the wet tropics (that is, neutral in Africa, positive in South America, or negative in Australia). Expanding an earlier spatially explicit savanna model, some studies have reported either a positive or an insignificant response of shrub cover (that is, G. flava) to increasing interannual rainfall fluctuations in southern Kalahari savannas (mean annual rainfall, 200 mm < MAP < 700 mm), a response that could depend also on seed availability (Tews and others 2004, 2006). Liedloff and Cook (2007) used a model of tree population dynamics to show a negative response of tree cover to increasing interannual rainfall variability in North Australian savannas (MAP ≈ 1000 mm). These studies shed light on the response of tree or grass alone to increasing interannual rainfall variability, whereas the effect on species composition remains poorly understood.
How a functional group (that is, trees) responds to an increase in interannual rainfall variability depends on the trade-off between wet and dry years (Holmgren and others 2013) as well as on its competitive relationship with other functional groups (grasses) and associated disturbance regime (that is, fires) (Williams and Albertson 2006; Reyer and others 2013; Zeppel and others 2014). A dynamic soil moisture model has been used to show the effect of grass dynamics on tree response to increasing interannual rainfall variability (Scanlon and Albertson 2003). Fernandez-Illescas and Rodriguez-Iturbe (2003) developed a hierarchical competition–colonization model in which dynamic water stress affects the tree–grass competition–colonization relationship; these authors used this model to investigate the impact of interannual rainfall fluctuations on tree–grass associations. Both of these models, however, ignored the roles of fires. Indeed, grasses typically have relatively high growth rates especially in wet environments (Teuling and others 2010; Collins and others 2012; Xu and others 2015) and thus may more quickly take up soil water or other resources, thereby more effectively competing with trees in wet years. The competitive advantage of grasses in wet years could enhance fire occurrences, thereby increasing tree mortality (Bond 2008; Ratajczak and others 2014); as a result, the increase in interannual rainfall variability is expected to favor grasses at the expenses of trees.
To our knowledge, only few studies have modeled the impact of interannual rainfall fluctuations in savannas accounting for both water competition and fires (Williams and Albertson 2006). Key to the understanding of vegetation response to changes in rainfall variability in water-limited ecosystems is an adequate representation of the soil water balance (Porporato and others 2002). In fact, the increase in rainfall intensity (with the total rainfall amount remaining constant) has been found to lead to shrub encroachment because of increased drainage into the deeper soil layer where trees with deeper roots can have exclusive access to soil water (Kulmatiski and Beard 2013a). Recent studies suggest that the increase in interannual rainfall variability may also increase soil water availability in the deep soil, thereby favoring deep-rooted over shallow-rooted species (Sala and others 2015; Gherardi and Sala 2015a, b). It remains unclear how the trait-based trade-off (that is, higher growth rate but shallower roots in grasses than trees) affects the response of tree–grass associations to increasing interannual rainfall variability along a rainfall gradient.
This study uses satellite data and develops a new mechanistic model to investigate the response of tree–grass associations to increasing interannual rainfall variability. We apply this model to the Kalahari Transect in Southern Africa, a region that exhibits relatively homogenous soils (sand) along a rainfall gradient, and therefore provides a good setting to investigate changes in plant community composition associated with different rainfall regimes without confounding effects associated with soil heterogeneity (Koch and others 1995; Wang and others 2007). This model couples a soil water balance with vegetation dynamics and accounts for fire-induced disturbance and competition between trees and grasses in access to soil water. We particularly investigate the following: (1) how trees or grasses alone and tree–grass associations respond to increasing interannual rainfall variability along the rainfall gradient; (2) the mechanisms underlying the response of these plant functional groups to increasing interannual rainfall fluctuations.
Methods
We develop a model to investigate the response of trees or grasses alone and tree–grass associations to increasing interannual rainfall variability along a rainfall gradient. For simplicity, the term “tree” is used here to include all forms of woody plants (that is, trees and shrubs). The model simulates soil moisture dynamics in two soil layers and accounts for flows between them due to drainage. The model simulates the biomass dynamics of trees and grasses as a logistic growth coupled with the soil water balance and accounts for biomass loss from senescence, litter fall, and disturbance (fires). Trees are assumed to have roots in both layers, while grasses are assumed to have roots only in the shallow soil layer (van Wijk and Rodriguez-Iturbe 2002; Van Langevelde and others 2003; Kulmatiski and Beard 2013b; Yu and D’Odorico 2015a, b). Thus, trees and grasses are assumed to compete for soil water resources in the shallow soil layer, whereas trees can exclusively access the soil water resources in the deep soil layer. Although in some savannas grasses could have roots as deep as trees (Sankaran and others 2004; Beckage and others 2009), here we consider the case in which tree roots span a much deeper soil column than grasses (Walter 1971; Kulmatiski and Beard 2013a, b; Holdo and Nippert 2015). An increase in interannual rainfall variability changes the soil moisture profile in the root zone and thus is expected to affect the interactions between trees and grasses. Rainfall and soil moisture dynamics are represented as stochastic processes, as explained below (Rodriguez-Iturbe and others 1999); interannual rainfall fluctuations are accounted for by a two-parameter gamma distribution (D’Odorico and others 2000; Porporato and others 2006). Deterministic laws are used to express the interactions and dynamics of trees and grasses, without accounting for the randomness of demographic growth in trees (Higgins and others 2000; Gardner 2006), and the stochastic nature of fire (D’Odorico and others 2006). The model does not explicitly represent the spatial interactions among trees and grasses (Jeltsch and others 1996; van Wijk and Rodriguez-Iturbe 2002) and thus uses a lumped approach to simulate dynamics of trees and grasses.
Water Balance
Soil moisture dynamics in the two soil layers are modeled at the landscape scale by two coupled equations:
and
where the subscripts 1 and 2 refer to the shallow and deep soil layer, respectively; n is the soil porosity, Z 1 and Z 2 the soil layer thickness (mm), S 1 and S 2 the relative soil moisture (0 < S 1, S 2 ≤ 1), P the rate of rainfall infiltration into the top soil layer (mm d−1), ET1 and T 2 the soil moisture losses from each soil layer due to evapotranspiration (mm d−1), and D 1 and D 2 are the drainage rates (mm d−1). Runoff occurs when the surface layer is saturated (that is, S 1 = 1). Calculations of ET1, T 2, D 1, and D 2 are presented in Online Appendix. The soil water balance is interpreted at the daily time scale because no diurnal fluctuations in ET are accounted for; the equations, however, are solved numerically using a smaller (hourly) time step because rainfall and soil water resources are pulsed in time.
Landscape Dynamics of Trees and Grasses
The dynamics of grass biomass (V g, kg m−2) and tree biomass (W l, kg m−2) at the landscape scale are expressed as a logistic growth:
where the \( m_{\text{G}} V_{\text{g}} \) and m W W l mortality terms account for biomass loss from senescence and litter fall by grasses and trees, respectively; kηW l is a mortality term accounting for fire, and g g and g w are the growth coefficients for grasses and trees, respectively; V 1 and V 2 are the vegetation carrying capacities contributed by soil moisture in the shallow and deep soil layer (kg m−2), and γ is the fraction of woody plant biomass relying on the shallow soil layer. Thus, γ is a term expressing the belowground competition of trees on grasses for soil water in the shallow soil layer. Observations in several savanna ecosystems indicate that grasses rapidly recover after fires (Russell-Smith and others 2001); therefore, no fire-induced mortality is included in the grass dynamics equation. Plant biomass loss from senescence and litter fall is modeled as a process that removes a proportion of biomass every year at the end of the growing season (Yu and D’Odorico 2015a). In agreement with other studies (Walker and Noy-Meir 1982; van Langevelde and others 2003), fires are modeled as a process that continuously removes the biomass of woody plants; the fire frequency (η) depends on grass biomass (van Wilgen and others 2000) (Table 1). Satellite data report the values of tree cover (%); for data-model inter-comparisons, tree biomass simulated in this study is converted to tree cover (F c) following the framework by Caylor and others (2006) and Yu and D’Odorico (2015a). This approach represents the landscape as a mosaic of “canopy” and “between canopy” areas resulting from a two-dimensional Poisson distribution of individual woody plants (with circular footprint). In this case, F c is a function of the number of woody plant individuals per unit area (i w, individuals, m−2) and the average canopy radius of an individual woody plant (u c, m), expressed as
The biomass of woody plants at the landscape scale (kg m−2) can be expressed as
where W p is the biomass of an individual woody plant canopy (kg per individual). Rearranging Eqs. (5) and (6), we obtain
Details for calculating F c are provided in Caylor and others (2006) and Yu and D’Odorico (2015a). Table 1 presents symbols, descriptions, and values of all the variables used in this study. The dynamics of grass and tree biomass are solved on a daily time scale.
Different from the study by Williams and Albertson (2006), V 1 and V 2 are here expressed as a function of soil moisture and soil layer thickness (Yu and D’Odorico 2015a):
where C 0 (kg m−2 mm−1) is a coefficient converting the soil water resources into vegetation carrying capacity. In contrast to other studies which related vegetation carrying capacity to annual rainfall (for example, Williams and Albertson 2006), this approach relates carrying capacity to soil moisture availability, which depends on soil texture, rainfall variability, rainfall gradient, and interspecies interactions (Knapp and others 2008; Reyer and others 2013; Zeppel and others 2014); thus, this framework allows us to explicitly investigate the impacts of increasing interannual rainfall variability with a detailed process-based approach. Growth rate is an important parameter in affecting tree–grass dynamics under interannual rainfall fluctuations. Previous studies imposed a constant growth rate in trees and grasses (Walker and Noy-Meir 1982; Anderies and others 2002) or expressed the growth rate as a function of annual rainfall (Williams and Albertson 2006). However, rainfall and soil water resources are pulsed in time, particularly in arid environments (Schwinning and Sala 2004; Knapp and others 2008; Collins and others 2014); thus, an appropriate temporal resolution needs to be used to capture pulsed rainfall fluctuations (Reyer and others 2013). Here we express g g and g w as a function of the fraction of time during the growing season in which grasses and trees are not water stressed (that is, with soil moisture exceeding the vegetation-specific value of soil moisture above which plants experience unstressed transpiration and photosynthesis):
where t g and t w are the number of hours grasses and trees do not experience water stress, respectively; T is the duration of the growing season (in h), and g gmax and g wmax are the maximum growth rates of grasses and trees when they remain unstressed during the whole growing season. Soil moisture (and the occurrence of water stress) evolves as a function of the accumulated biomass and therefore cannot be determined a priori before having simulated vegetation dynamics during the growing season. Thus, in this model t g/T and t w/T refer to the previous growing season. This approach allows us to capture the widely documented legacy effect of trees and grasses in the response to rainfall fluctuations (Sherry and others 2008; Sala and others 2012; Anderegg and others 2015). We note that growth rate was not determined by instantaneous soil moisture. In fact, the ability of plants to photosynthesize and grow (that is, the growth rate) also depends on traits such as specific leaf area and leaf phenology (Tomlinson and others 2013); plants with a low specific leaf area and/or senescent leaves could have a low photosynthetic rate and thus have a low growth rate even when soil moisture is high as a result of a rainfall pulse. Investment in these key traits for plant photosynthesis has energetic cost (DeWitt and others 1998; Kishida and Nishimura 2006). The energetic costs associated with developing these traits require carbon capture (and therefore soil moisture availability) over longer time scales (that is, one growing season).
Numerical Simulations
Three sets of numerical simulations were conducted, each addressing a specific objective. The first set of numerical simulations is to investigate the response of trees alone to increasing interannual rainfall variability along a rainfall gradient. For this purpose, Eqs. (1), (2), (4), (7), (8), (9), and (11) were used; grass biomass (G l) and the fire frequency (η) in Eq. (4) are taken to be zero because in this case there is no grass and thus no fire occurrence. The second set of numerical simulations is to investigate the response of grass alone to increase in interannual rainfall variability along a rainfall gradient. To this end, Eqs. (1), (3), (8), and (10) were used; tree biomass (W l) in Eq. (3) is taken to be zero since there are no trees in this case. For the response of tree–grass associations in the third set of numerical simulations, Eqs. (1)–(11) were used.
Precipitation (R) is modeled as a sequence of intermittent rainfall events using a marked Poisson process of storm occurrences with average rainfall frequency, λ (events per day). Each storm is modeled as an exponentially distributed random depth with mean, h (mm per event) (Rodriguez-Iturbe and others 1999). This stochastic rainfall model fits observed daily rainfall data during growing seasons quite well, but underestimates the interannual rainfall variability when applied to all years combined (Porporato and others 2006). To account for interannual rainfall fluctuations, we follow the approach by D’Odorico and others (2000) and Porporato and others (2006), in which interannual fluctuations in the average rainfall frequency (λ) and the average rainfall depth (h) can be assumed to be independent; interannual rainfall fluctuations can be represented by a two-parameter gamma distribution. Because the model is parameterized along the Kalahari Transect in Southern Africa where h is relatively uniform (Porporato and others 2003; Caylor and others 2005; Bhattachan and others 2012) (see section “Study site: the Kalahari Transect in Southern Africa” for details), the two-parameter gamma distribution is used to determine the interannual fluctuations only in λ, while h is kept constant. In this case, we have a two-parameter gamma distribution (\( g_{\lambda } \left( \lambda \right) \)):
where b λ is the scale parameter, and a λ is the shape parameter of this distribution; both parameters can be expressed as a function of the mean and standard deviation of λ. This approach reasonably represents the interannual rainfall fluctuations along Kalahari Transect in Southern Africa and elsewhere (D’Odorico and others 2000; Porporato and others 2006). Soil moisture in the shallow (S 1) and deep (S 2) soil layers is then quantified using Eqs. (1)–(2), which drives the dynamics of grasses and trees according to Eqs. (3)–(4). Each of the three sets of numerical simulations was run for 200 years and was repeated 100 times. Then the response of tree or grass alone and tree–grass associations to interannual rainfall fluctuations is averaged over simulation years and averaged across ensemble simulations.
Study Site: The Kalahari Transect in Southern Africa
The Kalahari Transect in Southern Africa is one of the International Geosphere-Biosphere Programme (IGBP) transects. It spans a north–south mean annual rainfall gradient from Angola and Zambia, through Botswana, into South Africa. Various studies based on field campaigns and model simulations have investigated various aspects of the ecology, ecohydrology, and biogeochemistry of savanna ecosystems along this transect (Porporato and others 2003; Caylor and others 2005; Bhattachan and others 2012). It remains unclear, however, how an increase in interannual rainfall variability would affect plant community composition in this region.
The duration of the rainy/growing season varies along the transect but it lasts roughly 210 days from October to May and accounts for about 95% of mean annual rainfall (MAP) (Bhattachan and others 2012). Across the Kalahari rainfall gradient, the average rainfall frequency (λ) typically spans from 0.1 to 0.5 d−1, whereas mean rainfall depth (h) is relatively uniform, h = 10 mm per event (Porporato and others 2003; Caylor and others 2005; Bhattachan and others 2012). Thus, numerical experiments in this study are mainly parameterized with two rainfall regimes corresponding to dry (λ = 0.2 d−1 and h = 10 mm per event) and wet (λ = 0.4 d−1 and h = 10 mm per event) environments. As noted above, the two-parameter gamma distribution [Eq. (12)] is used to account for interannual fluctuations in λ. Note that the average total precipitation over the growing season remains constant with changes in interannual fluctuations in λ and thus allows us to differentiate the effects of interannual rainfall fluctuations from those of changes in mean precipitation.
Because experimental measurement of the growth coefficients, g gmax and g wmax, are not available, their values are estimated, relying on other studies (Anderies and others 2002; Williams and Albertson 2006) and verifying that model simulations are in good agreement—in terms of vegetation composition (that is, relative/absolute magnitude of tree and grass biomass)—with experimental observations (Sankaran and others 2005; Bhattachan and others 2012) and satellite data (see below for details). Values of other parameters are from previous studies and are summarized in Table 1. The sensitivity of this model is investigated with respect to changes in g gmax, g wmax, and plant sensitivity to fire (Online Appendix). To investigate the generality of these results, the response of tree–grass composition to increased interannual rainfall variability is also investigated in less sandy soil (sandy loam).
Satellite Data
Daily Tropical Rainfall Measuring Mission (TRMM) rainfall data (3B42_daily product) were used to characterize interannual variability in rainfall over the period 1998-2014 (Huffman and others 2007). These data provide daily estimates of rainfall rates at 0.25° × 0.25° resolution. To avoid potential issues with tallying rainfall over the calendar year, we defined annual rainfall as cumulative rainfall from August 1 to July 31 each year, corresponding to the dry season over the study area. For each pixel, we calculated mean annual precipitation (MAP) and the standard deviation of annual precipitation (σAP) as the average and standard deviation of yearly rainfall, respectively.
Woody plant cover was characterized using the tree cover product Moderate Resolution Imaging Spectroradiometer (MODIS) MOD44 collection 5 version 1 vegetation continuous field product (Hansen and others 2003). The MODIS tree cover algorithm estimating tree cover with regression trees is available globally at 250 m resolution on a yearly basis. We resampled tree cover to the native 0.25° × 0.25° native TRMM resolution by averaging the value of MODIS pixels whose centroid was within each larger TRMM pixel, omitting missing data and pixels over water bodies. We then averaged tree cover across all available years of data (2000–2013).
Grasses favor fires and therefore the presence of grass cover increases the average proportion of burned areas (Bond 2008; Ratajczak and others 2014). Therefore, we use the burned area as a proxy for grass biomass and investigate grass (that is, burned area) response to changes in interannual rainfall variability. The MODIS MOD45A1 collection 5 version 1 burned area product was acquired for all months (Roy and others 2008). This product estimates whether each 250 m pixel has burned in a given month and estimates the day that the burn occurred if a fire was sensed. We resampled burned area to the 0.25° × 0.25° by calculating the percentage of MODIS pixels within each TRMM pixel that had burned in a given year. We then averaged the annual percent burned across 14 full years of available data starting in April 2000 to achieve a long-term of proportion burned in each pixel.
Results
Impacts of Interannual Rainfall Variability on Tree–Grass Associations: Satellite Data
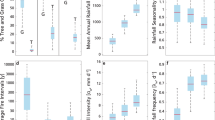
Satellite data show a positive response of tree cover to increasing interannual rainfall variability in dry environments (that is, mean annual precipitation, MAP < 900–1000 mm) but a negative response of tree cover in wet environments (MAP > 900–1000 mm) (Figure 1A). The average proportion of burned areas (that is, grass biomass) increases with increasing interannual rainfall variability in wet environments (that is, MAP > 1000–1100 mm), in contrast to what happens in dry environments (that is, MAP < 1000–1100 mm) (Figure 1B).
Modeling Impacts of Interannual Rainfall Variability on Tree or Grass Alone
The modeling results show that tree cover/biomass alone has a positive response, but grass biomass alone has a negative response to increasing interannual rainfall variability in dry environments (that is, λ = 0.2 event d−1, h = 10 mm per event) (Figure 2A); this pattern is explained by the increase in drainage (D 1) beneath the root zone of grasses on wet years (Figure 3A). D 1 increases and thus soil evaporation decreases, while drainage (D 2) beneath the root zone of trees remains negligible (Figure 3B), thereby leading to a positive response of trees. Conversely, in wet environments (that is, λ = 0.4 event d−1, h = 10 mm per event), both grass and tree biomass alone have a negative response to the increase in interannual rainfall variability (Figure 2B) because of the increase in drainage beneath the root zones of both grasses (D 1) and trees (D 2) (Figure 3A, B). Changes in runoff are negligible because of the coarse texture of Kalahari sands.
The modeled average grass biomass (V g) and tree cover (F c) alone or in associations for the case of sandy soil as affected by interannual rainfall variability (standard deviation of λ, δ) in dry (A λ = 0.2 event d−1, h = 10 mm per event) and wet (B λ = 0.4 event d−1, h = 10 mm per event) environments.
A Drainage (D1) from the shallow soil layer to the deep soil layer for the case of grass alone as affected by soil texture (sand and sandy loam) and interannual rainfall variability (standard deviation of λ, δ) in dry (λ = 0.2 event d−1, h = 10 mm per event) and wet environments (λ = 0.4 event d−1, h = 10 mm per event). (B) D1 and drainage (D2) from the deep soil layer (that is, from beneath the root zone of trees) for the case of trees alone in sandy soil as affected by interannual rainfall variability (standard deviation of λ,) in dry and wet environments.
Modeling Impacts of Interannual Rainfall Variability on Tree–Grass Associations
The modeling results show that in dry environments (that is, λ = 0.2 event d−1, h = 10 mm per event) tree–grass associations exhibit a substantial increase in tree biomass and cover, and a decrease in grass biomass with increasing interannual rainfall variability (Figure 2A), in agreement with our results based on satellite data (Figure 1). Grasses in tree–grass associations substantially decrease with increasing interannual rainfall variability because of increased competition from trees (Eq. 3; Figure S1). In wet environments (that is, λ = 0.4 event d−1, h = 10 mm per event), grass biomass in tree–grass associations increases, while tree cover in tree–grass associations decreases (Figure 2B), in agreement with satellite data (Figure 1B). This pattern of grass in tree–grass associations, which is opposite to the results obtained in the case of grasses alone (Figure 2B), could be explained by the high growth rate of grasses (Figure S2) and their ability to take advantage of increased pulses in shallow soil moisture with increasing interannual rainfall variability (Eq. 3; Figure S3). The increase in grass biomass enhances the fire regime, thereby increasing fire-induced tree mortality (Eq. 4) and thus reducing competition from trees (Eq. 3). Sensitivity tests indicate that the increase in grass growth rate and/or fire-induced tree mortality increase the competitive advantage of grasses (Figures S4, S5). Overall, the increase in interannual rainfall variability increases the total biomass in tree–grass associations in dry environments, whereas it decreases the total biomass in wet environments (Figure S6).
Generality and Synthesis of These Results
Model simulations show that the results shown above for tree–grass associations in sandy soil along the Kalahari Transect in Southern Africa are consistent with those obtained in the case of sandy loam. In fact, tree cover (grass biomass) in tree–grass associations increases (decreases) with increase in interannual rainfall fluctuations in dry environments (Figure 4A), whereas the opposite pattern is found in the case of wet environments (Figure 4B). Moreover, with a lower soil hydraulic conductivity and thus a lower drainage (D 2) beneath the root zone of trees (Figure 3A), the results obtained for sandy loam exhibit a higher rate of increase in tree cover and decrease in grass biomass with increasing interannual rainfall fluctuations in dry environments (Figure 4A). With an increased proportion of wet years, increase in interannual variation in precipitation provides windows of opportunity in soil moisture enrichment in the shallow and deep soil in wet years (Figure 5A); the response of tree–grass composition to increased interannual variation in precipitation would depend on their key traits in taking advantage of these windows of opportunity. Grasses typically have shallow roots that cannot take advantage of the water stored in the deep soil; however, grasses with a high growth rate could greatly benefit from soil moisture enhancement in the shallow soil, increase their biomass, and consequently generate fires and kill trees; trees typically have deep roots and can take advantage of deep soil moisture, while they typically have a low growth rate and thus are less competitive than grasses in getting access to shallow soil moisture. Thus, as synthesized in Figure 5B, the response of vegetation composition in savannas to an increase in interannual rainfall variability depends on the relative magnitude of the growth rates and root depths of grasses and trees. A higher ratio of grass to tree growth rate and a lower ratio of tree to grass root depth favors grasses in the response to increasing interannual rainfall variability, while the opposite pattern of growth rates and root depths favors trees.
The modeled average grass biomass (V g) and tree cover (F c) alone or in associations for the case of sandy loam as affected by interannual rainfall variability (standard deviation of λ) in dry (A λ = 0.2 event d−1, h = 10 mm per event) and wet (B λ = 0.4 event d−1, h = 10 mm per event) environments.
A A conceptual diagram showing that increased interannual rainfall fluctuations provide windows of opportunity in resource enrichment in the shallow and deep soil in wet years. The solid black line represents the case of normal rainfall, whereas the dash black line represents the case of increased interannual rainfall fluctuations. The red line represents the threshold of windows of opportunity in resource enrichment below which the growth rate of grasses is low and fires are rare and thus grasses would be outcompeted by trees. B A conceptual framework of the contingent responses of tree–grass composition to alterations in soil water dynamics resulting from increased interannual rainfall variability (Color figure online).
Discussion
Climate change studies predict an increase both in intra-annual and interannual rainfall variability (Easterling and others 2000; Fischer and others 2013; IPCC 2013). Although previous studies have investigated the ecological impacts of changes in intra-annual rainfall variability (Knapp and others 2008; Kulmatiski and Beard 2013a; Zeppel and others 2014), the effects of interannual rainfall variability on vegetation composition and ecosystem processes is not well understood, mostly because of limitations in long-term observational data (Fatichi and Ivanov 2014). This study uses satellite data and develops a new mechanistic model to investigate the response of tree/grass composition to increasing interannual rainfall variability along the Kalahari Transect in Southern Africa.
Satellite data show that increasing interannual rainfall variability favors trees over grasses in dry environments (that is, mean annual precipitation, MAP < 900–1000 mm) and disfavors trees over grasses in wet environments (that is, MAP > 900–1000 mm) (Figures 1B, 2B). In contrast to this study, Holmgren and others (2013) used the satellite data to show a neutral response of tree cover to increasing rainfall variability in wet tropics in Africa, possibly because of soil texture effects and disturbance (that is, grazing). Most of the other studies on this subject have used models as diagnostic tools to gain mechanistic understanding of ecosystem dynamics in response to increasing interannual rainfall fluctuations (Fernandez-Illescas and Rodriguez-Iturbe 2003; Tews and others 2004, 2006; Williams and Albertson 2006; Liedloff and Cook 2007). This study develops a new mechanistic model to clarify the role of tree–grass competition for soil water resources and fire-induced disturbance as determinants of savanna response to changes in interannual rainfall variability.
Previous studies have stressed the role of competition in the response of vegetation composition to increasing interannual rainfall variability (Scanlon and Albertson 2003; Fernandez-Illescas and Rodriguez-Iturbe 2003), whereas the effect of fires has been ignored (but see Williams and Albertson 2006). This new mechanistic model shows that in dry environments an increase in interannual rainfall variability leads to a reduction in grass competition and fire or an increase in competition from trees (Figure S3) that cause a higher rate of reduction in grass biomass and a higher rate of increase in tree cover than in the case of tree or grass alone (Figure 2A). The competitive advantage of trees results from deeper root systems than grasses, which allow trees to have exclusive access to increased deep soil water (on wet years) with increasing interannual rainfall variability. Extensive field studies in Africa have found deeper root systems in trees than grasses (Kulmatiski and Beard 2013a, b; Holdo and Nippert 2015), although in some savannas grasses could have roots as deep as trees (Sankaran and others 2004; Beckage and others 2009). Consistent with this study, Gherardi and Sala (2015a, b) show that in a 6-year field experiment an increase in interannual rainfall variability shifts species composition in favor of deep-rooted (that is, trees) over shallow-rooted (that is, grasses) species.
Interestingly, our study shows that increases in interannual rainfall variability in wet environments shift species composition favoring grasses over trees (Figures 1, 2). This is a novel finding because other studies in grasslands (Hsu and others 2012) have shown that grasses alone have a negative response to increasing interannual rainfall variability in wet environments (Figure 2). Grasses typically have a high growth rate especially in wet environments (Teuling and others 2010; Collins and others 2012; Xu and others 2015), consistent with the concept of the world-wide ‘fast–slow’ plant economics spectrum (Reich 2014); thus, grasses could quickly take advantage of the window of opportunity existing in years with above average precipitation. The high growth rate in grasses increases fire frequency and fire-induced tree mortality (Bond 2008; Ratajczak and others 2014), thereby leading to a reduction in tree competition with grasses for soil water, which further favors grass biomass. These results are in agreement with a general theory of invisibility in plant communities under fluctuating resources (Davis and others 2000). Based on field studies (Davis and Pelsor 2001; Corbin and D’Antonio 2004), this theory holds that fluctuations in resource availability provide windows of opportunity in resource enrichment, whereby species with a high growth rate could quickly take up resources, change the disturbance regime, and then invade or dominate the landscape. Consistent with these studies, our study shows that the way tree–grass composition responds to increased interannual variation in precipitation would depend on key traits of trees and grasses (that is, growth rate and root depth) that determine their ability to take advantage of the windows of opportunity offered by periods with higher soil moisture.
The encroachment of woody plants into grasslands has been widely documented in arid and semiarid environments in many regions of the world including southern Africa (Moleele and others 2002). The mechanisms typically invoked to explain this phenomenon involve exogenic drivers including overgrazing, fire suppression, increase in CO2 concentration, and long-term global changes in rainfall or temperature and endogenic positive feedbacks (D’Odorico and others 2012; Yu and D’Odorico 2014). This study shows that the increase in tree dominance in dry environments may also result from an increase in interannual rainfall variability.
A number of studies have invoked fire–vegetation feedbacks to explain the existence of savannas in a wide range of rainfall conditions in southern Africa (Staver and others 2011a, b). The increase in grass biomass and thus fire frequency in wet environments found in this study indicates that interannual rainfall fluctuations may expand the range of environmental conditions in which savannas are stable. This idea is in agreement with the emerging view that the interplay between tree/grass growth rate and fires regimes governs savanna–forest transitions (Hoffmann and others 2012; Murphy and Bowman 2012).
The model developed in this study expressed vegetation capacity as a function of instantaneous soil moisture and thus allows for a process-based analysis of impacts of increasing interannual rainfall variability and/or soil texture on tree–grass composition. We also noted that an alternative modeling approach relating vegetation capacity to mean growing seasonal rainfall and root depth by trees and grasses did not change the general pattern found in this study (Figure S7). Moreover, we notice that this model does not account for other factors, including rainfall seasonality (Vico and others 2015) and plant life histories (that is, annuals or perennials; evergreen or deciduous) (Kos and others 2012), which play an important role in determining plant community composition in savannas. In fact, an increase in winter rainfall increases deep soil water and thus favors trees (deep-rooted plants) over grasses (shallow-rooted plants) (Brown and others 1997; Germino and Reinhardt 2014). The high interannual variability of rainy season duration favors deciduous trees over evergreen trees, which may affect the competition with grasses (Vico and others 2015). The legacy effect of trees and grasses attributed to water and/or carbohydrate storage, available seeds/meristems, and/or nutrient availability from litter decomposition in response to rainfall fluctuations are also expected to be affected by plant life histories (Sherry and others 2008; Sala and others 2012; Anderegg and others 2015).
Conclusions
This study uses satellite data and develops a new mechanistic model to assess the effects of increasing interannual rainfall variability on tree/grass composition along the Kalahari Transect in Southern Africa. Both satellite data and model results show that increasing interannual rainfall fluctuations favor deep-rooted trees over shallow-rooted grasses in drier environments (that is, mean annual rainfall, MAP < 900–1000 mm), whereas in wetter environments it favors grasses over trees (that is, MAP > 900–1000 mm). The relative magnitude of the growth rates and root depths of grasses and trees greatly affects the response of tree–grass composition in response to increasing interannual rainfall variability. When interpreting the response of each functional group (that is, trees or grasses), it is crucial to account for the direct effects of interannual rainfall variability on soil water availability and also the indirect effects mediated by tree–grass interactions.
References
Anderegg WRL, Schwalm C, Biondi F, Camarero JJ, Koch G, Litvak M, Ogle K, Shaw JD, Shevliakova E, Williams AP, Wolf A, Ziaco E, Pacala S. 2015. Pervasive drought legacies in forest ecosystems and their implications for carbon cycle models. Science 349:528–32.
Anderies JM, Janssen MA, Walker BH. 2002. Grazing management, resilience, and the dynamics of a fire-driven rangeland system. Ecosystems 5:23–44.
Beckage B, Platt WJ, Gross LJ. 2009. Vegetation, fire, and feedbacks: a disturbance-mediated model of savannas. Am Nat 174:805–18.
Bhattachan A, Tatlhego M, Dintwe K, O’Donnell FC, Caylor KK, Okin GS, Perrot DO, Ringrose S, D’Odorico P. 2012. Ecohydrologic controls on root biomass and depth in southern African savannas. PLoS ONE 7:e33996.
Bond WJ. 2008. What limits trees in C4 grasslands and savannas? Annu Rev Ecol Evol Syst 39:641–59.
Brown JH, Valone TJ, Curtin CG. 1997. Reorganization of an arid ecosystem in response to recent climate change. Proc Natl Acad Sci USA 94:9729–33.
Caylor KK, D’Odorico P, Rodriguez-Iturbe I. 2006. On the ecohydrology of structurally heterogeneous semiarid landscapes. Water Resour Res 42:W07424.
Caylor KK, Shugart HH, Rodriguez-Iturbe I. 2005. Tree canopy effects on simulated water stress in southern African savannas. Ecosystems 8:17–32.
Collins SL, Belnap J, Dahm CN et al. 2014. A multi-scale, hierarchical model of pulse dynamics in aridland ecosystems. Annu Rev Ecol Evol Syst 45:397–419.
Collins SL, Koerner SE, Plaut JA, Okie JG, Brese D, Calabrese LB, Carvajal A, Evansen RJ, Nonaka E. 2012. Stability of tallgrass prairie during a 19-year increase in growing season precipitation. Funct Ecol 26:1450–9.
Collins SL, Xia Y. 2015. Long-term dynamics and hotspots of change in a desert grassland plant community. Am Nat 185:E30–43.
Corbin JD, D’Antonio CM. 2004. Competition between native perennial and exotic annual grasses: implications for an historical invasion. Ecology 85:1273–83.
Davis MA, Grime JP, Thompson K. 2000. Fluctuating resources in plant communities: a general theory of invasibility. J Ecol 88:528–34.
Davis MA, Pelsor M. 2001. Experimental support for a resource-based mechanistic model of invasibility. Ecol Lett 4:421–8.
DeWitt TJ, Sih A, Wilson DS. 1998. Costs and limits of phenotypic plasticity. Trends Ecol Evol 13:77–81.
D’Odorico P, Ridolfi L, Porporato A, Rodriguez-Iturbe I. 2000. Preferential states of seasonal soil moisture: the impact of climate fluctuations. Water Resour Res 36:2209–19.
D’Odorico P, Laio F, Porporato A, Rodriguez-Iturbe I. 2003. Hydrologic controls of soil carbon and nitrogen cycles II. A case study. Adv Water Resour 26:59–70.
D’Odorico P, Laio F, Ridolfi L. 2006. A probabilistic analysis of fire induced tree-grass coexistence in savannas. Am Nat 167:E79–87.
D’Odorico P, Okin GS, Bestelmeyer BT. 2012. A synthetic review of feedbacks and drivers of shrub encroachment in arid grasslands. Ecohydrology 5:520–30.
Easterling DR, Meehl GA, Parmesan C, Changnon SA, Karl TR, Mearns LO. 2000. Climate extremes: observations, modeling, and impacts. Science 289:2068–74.
Fatichi S, Ivanov VY. 2014. Interannual variability of evapotranspiration and vegetation productivity. Water Resour Res 50:3275–94.
Fernandez-Illescas CP, Rodriguez-Iturbe I. 2003. Hydrologically driven hierarchical competition–colonization models: the impact of interannual climate fluctuations. Ecol Monogr 73:207–22.
Fischer EM, Beyerle U, Knutti R. 2013. Robust spatially aggregated projections of climate extremes. Nat Clim Chang 3:1033–8.
Gardner TA. 2006. Tree-grass coexistence in the Brazilian cerrado: demographic consequences of environmental instability. J Biogeogr 33:448–63.
Germino MJ, Reinhardt K. 2014. Desert shrub responses to experimental modification of precipitation seasonality and soil depth: relationship to the two-layer hypothesis and ecohydrological niche. J Ecol 102:989–97.
Gherardi LA, Sala OE. 2015a. Enhanced interannual precipitation variability increases plant functional diversity that in turn ameliorates negative impact on productivity. Ecol Lett 18:1293–300.
Gherardi LA, Sala OE. 2015b. Enhanced precipitation variability decreases grass- and increases shrub-productivity. Proc Natl Acad Sci USA 112:12735–40.
Hansen MC, DeFries RS, Townshend JRG, Carroll M, Dimiceli C, Sohlberg RA. 2003. Global percent tree cover at a spatial resolution of 500 meters: first results of the MODIS vegetation continuous fields algorithm. Earth Interact 7:1–15.
Higgins SI, Bond WJ, Trollope WSW. 2000. Fire, resprouting and variability: a recipe for tree-grass coexistence in savanna. J Ecol 88:213–29.
Hoffmann WA, Geiger EL, Gotsch S et al. 2012. Ecological thresholds at the savanna-forest boundary: how plant traits, resources and fire govern the distribution of tropical biomes. Ecol Lett 15:759–68.
Holdo RM, Nippert JB. 2015. Transpiration dynamics support resource partitioning in African savanna trees and grasses. Ecology 96:1466–72.
Holmgren M, Hirota M, Van Nes EH, Scheffer M. 2013. Effects of interannual climate variability on tropical tree cover. Nat Clim Chang 3:755–8.
Hsu JS, Adler PB. 2014. Anticipating changes in variability of grassland production due to increases in interannual precipitation variability. Ecosphere 5:art58.
Hsu JS, Powell J, Adler PB. 2012. Sensitivity of mean annual primary production to precipitation. Glob Chang Biol 18:2246–55.
Huffman GJ, Bolvin DT, Nelkin EJ et al. 2007. The TRMM multisatellite precipitation analysis. TMPA: quasi-global, multiyear, combined-sensor precipitation estimates at fine scales. J Hydrometeorol 8:38–55.
IPCC. 2013. Climate change 2013: the physical science basis. In: Stocker TF, Qin D, Plattner GK, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM, Eds Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Cambridge: Cambridge University Press.
Jeltsch F, Milton SJ, Dean WRJ, van Rooyen N. 1996. Tree spacing and coexistence in semiarid savannas. J Ecol 84:583–95.
Kharin VV, Zwiers FW, Zhang XB, Hegerl GC. 2007. Changes in temperature and precipitation extremes in the IPCC ensemble of global coupled model simulations. J Clim 20:1419–44.
Kishida O, Nishimura K. 2006. Flexible architecture of inducible morphological plasticity. J Anim Ecol 75:705–12.
Knapp AK, Beier C, Briske DD et al. 2008. Consequences of more extreme precipitation regimes for terrestrial ecosystems. Bioscience 58:811–21.
Knapp AK, Fay PA, Blair JM. 2002. Rainfall variability, carbon cycling, and plant species diversity in a mesic grassland. Science 298:2202–5.
Koch GW, Scholes RJ, Steffen WL, Vitousek PM, Walker BH. 1995. The IGBP terrestrial transects: science plan, Report No. 36. International geosphere-biosphere program, Stockholm.
Kos M, Baskin CC, Baskin JM. 2012. Relationship of kinds of seed dormancy with habitat and life history in the Southern Kalahari fl201. J Veg Sci 23:869–79.
Kulmatiski A, Beard KH. 2013a. Woody plant encroachment facilitated by increased precipitation intensity. Nat Clim Chang 3:833–7.
Kulmatiski A, Beard KH. 2013b. Root niche partitioning among grasses, saplings, and trees measured using a tracer technique. Oecologia 171:25–37.
Laio F, Porporato A, Ridolfi L, Rodriguez-Iturbe I. 2001. Plants in water-controlled ecosystems: active role in hydrologic processes and response to water stress. II. Probabilistic soil moisture dynamics. Adv Water Resour 24:707–23.
Lewis SL, Brando PM, Phillips OL, van der Heijden GMF, Nepstad D. 2011. The 2010 amazon drought. Science 331:554.
Liedloff AC, Cook GD. 2007. Modelling the effects of rainfall variability and fire on tree populations in an Australian tropical savanna with the FLAMES simulation model. Ecol Model 201:269–82.
Liu J, Wang B, Cane MA, Yim S-Y, Lee J-Y. 2013. Divergent global precipitation changes induced by natural versus anthropogenic forcing. Nature 493:656–9.
Moleele NM, Ringrose S, Matheson W, Vanderpost C. 2002. More woody plants? The status of bush encroachment in Botswana’s grazing areas. J Environ Manag 64:3–11.
Munson SM, Webb RH, Belnap J, Andrew Hubbard J, Swann DE, Rutman S. 2012. Forecasting climate change impacts to plant community composition in the Sonoran Desert region. Glob Chang Biol 18:1083–95.
Murphy BP, Bowman DMJS. 2012. What controls the distribution of tropical forest and savanna? Ecol Lett 15:748–58.
Porporato A, Laio F, Ridolfi L, Caylor KK, Rodriguez-Iturbe I. 2003. Soil moisture and plant stress dynamics along the Kalahari precipitation gradient. J Geophys Res 108(D3):4127.
Porporato A, D’Odorico P, Laio F, Ridolfi L, Rodriguez-Iturbe I. 2002. Ecohydrology of water-controlled ecosystems. Adv Water Resour 25:1335–48.
Porporato A, Vico G, Fay PA. 2006. Superstatistics of hydroclimatic fluctuations and interannual ecosystem productivity. Geophys Res Lett 33:L15402.
Roy DP, Boschetti L, Justice CO, Ju J. 2008. The collection 5 MODIS burned area product—global evaluation by comparison with the MODIS active fire product. Remote Sens Environ 112:3690–707.
Ratajczak Z, Nippert JB, Briggs JM, Blair JM. 2014. Fire dynamics distinguish grasslands, shrublands and woodlands as alternative attractors in the Central Great Plains of North America. J Ecol 102:1374–85.
Reich PB. 2014. The world-wide ‘fast-slow’ plant economics spectrum: a traits manifesto. J Ecol 102:275–301.
Reichstein M, Bahn M, Ciais P et al. 2013. Climate extremes and the carbon cycle. Nature 500:287–95.
Reyer CPO, Leuzinger S, Rammig A et al. 2013. A plant’s perspective of extremes: terrestrial plant responses to changing climatic variability. Glob Chang Biol 19:75–89.
Ridolfi L, D’Odorico P, Porporato A, Rodriguez-Iturbe I. 2000. Impact of climate variability on the vegetation water stress. J Geophys Res 105(D14):18013–25.
Rodriguez-Iturbe I, D’odorico P, Porporato A, Ridolfi L. 1999. On the spatial and temporal links between vegetation, climate, and soil moisture. Water Resour Res 35:3709–22.
Russell-Smith J, Start T, Woinarski J. 2001. Effects of fire in the landscape. In: Dyer R, Jacklyn P, Partridge I, Russell-Smith J, Williams R, Eds. Savanna burning understanding and using fire in northern Australia. Darwin: Tropical Savannas CRC. p 29–49.
Sala OE, Gherardi LA, Reichmann L, Jobbagy E, Peters D. 2012. Legacies of precipitation fluctuations on primary production: theory and data synthesis. Philos Trans R Soc B 367:3135–44.
Sala O, Gherardi L, Peters DC. 2015. Enhanced precipitation variability effects on water losses and ecosystem functioning: differential response of arid and mesic regions. Clim Chang 131:213–27.
Sankaran M, Hanan NP, Scholes RJ et al. 2005. Determinants of woody cover in African savannas. Nature 438:846–9.
Sankaran M, Ratnam J, Hanan NP. 2004. Tree-grass coexistence in savannas revisited: insights from an examination of assumptions and mechanisms invoked in existing models. Ecol Lett 7:480–90.
Scanlon TM, Albertson JD. 2003. Inferred controls on tree/grass composition in a savanna ecosystem: combining 16 year normalized difference vegetation index data with a dynamic soil moisture model. Water Resour Res 39:1224.
Scholes RJ, Walker BH. 1993. An African savanna: synthesis of the Nylsvlei study. Cambridge: Cambridge University Press.
Schwinning S, Sala OE. 2004. Hierarchy of responses to resource pulses in arid and semi-arid ecosystems. Oecologia 2:211–20.
Sherry RA, Weng E, Arnone JA, Johnson DW, Schimel DS, Verburg PS, Wallace LL, Luo Y. 2008. Lagged effects of experimental warming and doubled precipitation on annual and seasonal aboveground biomass production in a tallgrass prairie. Glob Chang Biol 14:2923–36.
Staver AC, Archbald S, Levin AS. 2011a. The global extent and determinants of savanna and forest as alternative biome states. Science 334:230–2.
Staver AC, Archbald S, Levin AS. 2011b. Tree cover in sub-Saharan Africa: rainfall and fire constrain savanna and forest as alternative stable states. Ecology 92:1063–72.
Teuling AJ, Seneviratne SI, Stockli R et al. 2010. Contrasting response of European forest and grassland energy exchange to heatwaves. Nat Geosci 3:722–7.
Tews J, Esther A, Milton SJ, Jeltsch F. 2006. Linking a population model with an ecosystem model: assessing the impact of land use and climate change on Savanna shrub cover dynamics. Ecol Model 195:219–28.
Tews J, Schurr F, Jeltsch F. 2004. Seed dispersal by cattle may cause shrub encroachment of Grewia flava on southern Kalahari rangelands. Appl Veg Sci 7:89–102.
Ttomlinson KW, Poorter L, Sterck F, Borghetti F, Ward D, Biesand De, van Langevelde F. 2013. Leaf adaptations of evergreen and deciduous trees of semi-arid and humid savannas on three continents. J Ecol 101:430–40.
Van Langevelde F, Van De Vijver CA, Kumar L et al. 2003. Effects of fire and herbivory on the stability of savanna ecosystems. Ecology 84:337–50.
van Wijk MT, Rodriguez-Iturbe I. 2002. Tree-grass competition in space and time: insights from a simple cellular automata model based on ecohydrological dynamics. Water Resour Res 38:1811–15.
Van Wilgen BW, Biggs HC, O’Regan S, Mare N. 2000. A fire history of the savanna ecosystems in the Kruger National Park, South Africa between 1941 and 1996. S Afr J Sci 96:167–78.
Vico G, Thompson S, Manzoni S, Molini A et al. 2015. Climatic, ecophysiological and phenological controls on plant ecohydrological strategies in seasonally dry ecosystems. Ecohydrology 8:660–81.
Walker BH, Noy-Meir I. 1982. Aspects of stability and resilience of savanna ecosystems. In: Walker BH, Huntley BJ, Eds. Ecology of subtropical savannas. Berlin: Springer. p 556–90.
Walter H. 1971. Ecology of tropical and subtropical vegetation. Edinburgh: Oliver and Boyd.
Williams CA, Albertson JD. 2006. Dynamical effects of the statistical structure of annual rainfall on dryland vegetation. Glob Chang Biol 12:777–92.
Wang L, D’Odorico P, Macko S, Ringrose S, Coetzee S. 2007. Biogeochemistry of Kalahari sands. J Arid Environ 71:259–79.
Xu XT, Medvigya D, Rodriguez-Iturbeb I. 2015. Relation between rainfall intensity and savanna tree abundance explained by water use strategies. Proc Natl Acad Sci USA 112:12992–6.
Yu KL, D’Odorico P. 2014. An ecohydrological framework for grass displacement by woody plants in savannas. J Geophys Res 119:192–206.
Yu KL, D’Odorico P. 2015a. Hydraulic lift as a determinant of tree-grass coexistence on savannas. New Phytol 207:1038–51.
Yu KL, D’Odorico P. 2015b. Direct and indirect facilitation of plants with crassulacean acid metabolism CAM. Ecosystems 18:985–99.
Zeppel MJB, Wilks JV, Lewis JD. 2014. Impacts of extreme precipitation and seasonal changes in precipitation on plants. Biogeosciences 11:3083–93.
Acknowledgements
This research was funded through a fellowship from China Scholarship Council, a Grant from the VPR Office of the University of Virginia, and a Grant from the National Socio-Environmental Synthesis Center, NSF DBI-1052875. The authors would like to thank the initial discussion with Dr. Zak Ratajczak.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
KLY conceived and designed study, performed research, contributed new models, and wrote the article; MS performed analysis of satellite data; PD conceived and designed study, and wrote the article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, K., Saha, M.V. & D’Odorico, P. The Effects of Interannual Rainfall Variability on Tree–Grass Composition Along Kalahari Rainfall Gradient. Ecosystems 20, 975–988 (2017). https://doi.org/10.1007/s10021-016-0086-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-016-0086-8