Abstract
We identified eight Panamanian watersheds in which conversion from wet tropical forest to pastures differed and assessed the effects of degree of deforestation, and down-estuary transformations, on the suspended particulate matter discharged from the watersheds, entering, traversing through mangrove estuaries, and emerging into coastal waters. Deforested watersheds discharged larger concentrations of suspended particulate matter, with lower % C and N, higher mineral content, and heavier isotopic signatures into fresh reaches of estuaries. Down-estuary, sediment entrainment increased non-organic content of particulates, and watershed-derived imprints of deforestation on composition of particulate matter were mostly erased by within-estuary transformations. Isotopic signatures of C, N, and S in particulate matter demonstrated strong land-sea couplings, and indicated that the direction of the coupling was asymmetrical, with terrestrial and estuarine sources delivering particulate materials to coastal waters and sediments. Mangrove estuaries therefore both act as powerful modulators of human activities on land, while also exporting particulate materials to sea.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The widely reported deforestation of tropical watersheds (Wassenaer and others 2007; Scanlon and others 2007; Downing and others 1999) is a major agent of ecosystem change in tropical latitudes, with regional- and global-scale effects on climate (Gash and others 1996; IPCC 2007; Davin and Noblet-Ducoudre 2010), regional precipitation and water supply (Bosch and Hewlett 1982; Vorosmarty and others 2000; Sun and others 2006; Scanlon and others 2007) and carbon sequestration (Wolf and others 2011).
Deforestation may alter discharges of water and nutrients from watersheds. Where plant biomass is lower, there is less transpiration, and lower surface area for evaporative loss (Lewis and others 1999; Lewis 2002), which favor greater water discharge. Lower biomass may also have varied effects on interception, sequestering, and transport of sediments and nutrients down hydrological gradients to receiving waters (Williams and Melack 1997; Williams and others 1997; Cleveland and others 1999; Neill and others 2001; Zhang and others 2001; Bruijnzeel 2004; Nosetto and others 2005; Li and others 2007; Bahn and others 2010).
Discharges of materials from deforested watersheds could have further consequences because tropical streams in most circumstances grade into mangrove-lined estuaries, and these ecosystems could themselves be affected. The notion of wetlands, including mangrove estuaries, as components of coastal landscapes essential in the coupling of land and sea, whereby energy-rich exports of terrestrial and estuarine materials support marine food webs has been widely argued, with diverse results from mass balance and stable isotope studies (Jennerjahn and Ittekkot 2002; Dittmar and others 2006; Kristensen and others 2008). If indeed there are mechanisms that couple terrestrial watersheds to streams and estuaries, and to coastal waters, deforestation of tropical forests could have consequences for the receiving down-gradient ecosystems.
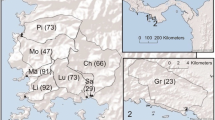
To understand land–sea couplings and possible effects of deforestation of tropical watersheds on down-gradient mangrove estuaries and adjacent coastal waters, we investigated the fate of materials transported and transformed as they coursed through coupled land–estuary–coastal ecosystems in the Pacific coast of Panama. By initial surveys, we followed the lead of earlier papers (Martinelli and others 1999; Ralison and others 2008), and identified eight watershed-mangrove estuaries whose watersheds were subject to different degrees of land use, in our case, conversion from forest to pastures (Figure 1) by targeted artisanal-level burning, and where pastures were maintained by machete-based removal of invasive or re-growing woody plants. These eight watershed–estuary systems constituted a landscape-level experimental setting in which we could assess consequences of different degrees of terrestrial watershed deforestation on land–sea coupling. Further details of land covers and deforestation were included in Valiela and others (2012, 2013, and in press) and additional features of the watersheds are added in Table 1.
Distribution of the watersheds included in this study [Pi: Rio Pixvae, Mo: Rio de la Mona, Ma: Rio Manglarito, Li: Rio Limon, Lu: Rio Luis, Sa: Rio Salmonete, and Ch: Rio Chamuscado, all in panel 1, and Gr: Rio Grande, in panel 2]. The numbers in parentheses indicate the % of the watershed area in each watershed that was forested. Location of the study areas in the broader Panama region is shown in the inset on top right.
Previous studies on the Pacific coast of Panama revealed that inter-annual increases in rainfall associated with La Niña severely altered salinity regimes (Valiela and others 2012), and altered fate and transport of dissolved nutrients. Nutrient retention within-watersheds was high, and forested watersheds discharged larger concentrations of dissolved inorganic nitrogen to streams than pasture-dominated watersheds. The imprint conferred by degree of deforestation on export of dissolved materials exported was detectable in fresh reaches of the streams, but was erased by active down-estuary biogeochemical transformations (Valiela and others 2013). In spite of the substantial transformations, and net interception within watersheds and estuaries, the export of dissolved nutrients to adjacent coastal sea from mangrove estuaries was still significant because of the extremely nutrient depauperate condition of the coastal waters of the region.
In this paper, we focus on particulate materials in transit from the watersheds to the streams and down the estuarine gradient to sea. The fate of particulates is of interest because deforestation of some watersheds increased downstream transport of suspended particulate matter (Martinelli and others 1999; Houser and others 2006) by erosion of soils, stream banks, stream beds, and transport of plant matter, and hence could increase consequent transport of particles out of watersheds and out to sea (Maréchal and others 2009). To evaluate effects of deforestation, we compared measurements taken from the eight Panamanian stream–estuary systems whose watersheds suffered different degrees of deforestation. We report concentrations, fates, and export of particulates in water, and assess sources and transformations by measuring concentration, % nitrogen, % carbon, and stable isotopic signatures of suspended material. To document down-estuary changes we sampled the water column of each stream–estuary along the entire range from fresh to seawater within estuaries and beyond the mouth of the estuaries. To capture possible inter-annual or seasonal variation, we repeated the sampling at the end of the dry and wet seasons, during 2009–2012.
Methods
Study Sites
The watershed-estuaries included in this study (Figure 1) are located in the Veraguas Province of Panama, and discharge into the Pacific Ocean. We selected the eight coupled watershed–estuary systems (rios) because they offered a range of conversions from forest to pasture land covers, with forest cover ranging from 23 to 92%. Terrain on the watersheds was generally steep, and largely first order streams carried mostly baseline freshwater discharge (with surface runoff after large rainfall events) down-gradient through mangrove estuaries, and eventually to the Pacific. Details of geological setting, precipitation, and other information were provided in Valiela and others (2012, 2013). Some of that information, plus added details, is summarized in Table 1.
Water Sampling
To determine concentrations of suspended particulate matter (SPM) in water that just exited watersheds and entered freshwater streams, moved through estuaries and out to sea, we sampled water during ebbing tides at six stations spaced between the fresh reaches and the mouth of each of the eight estuaries. This set of stations captured the fresh to seawater salinity gradient within each estuary (Valiela and others 2012). During each sampling trip, location of stations 1–6 was adjusted to make sure the sampling captured the changing gradient from freshwater to full salinity and nutrients. The gradient shifted location owing to differences in tidal state and stream flows from one sampling trip to the next. Locations of the many sampling stations in each of the rios are indicated in Figures SI2–SI9 in supplementary information.
Within-estuary sampling was done at one depth because during ebb tides water columns were mostly shallow (<1.5 m) and well mixed vertically. Some vertical stratification was present, but was spatially highly variable because of un-even bathymetry, fast down-gradient flow, and a large tidal excursion (well over 4 m for most tides). To assess what happened to the SPM once estuarine water emerged off the estuaries, we followed the ebb plume in each estuary and sampled at three distances away from the estuary discharge (stations 7–9), which, very roughly, were located about 50, 100, and 300 m off each estuary mouth.
The within- and outside-estuary sampling in stations 1–9 was repeated during periods of 3 weeks each during March 2009, December 2009, March 2010, December 2010, April 2011, and January 2012. This sampling schedule insured data were collected during the end of dry season (generally January–April) and the end of the wet season (generally May–December), across a span of 4 years.
Water Quality and SPM Measurements
Methods and results regarding nutrient concentrations in the samples collected were discussed in Valiela and others (2013). Salinity, oxygen, and temperature were measured in all stations with an YSI 85-10 unit. Water samples to measure SPM and chlorophyll were collected in 20 l carboys and kept cool during transportation to the lab. Water was first passed through a 210-μm mesh to remove zooplankton that would have compromised stable isotope measurements of SPM. SPM samples were then obtained by filtering 0.5–1.5 L of water through pre-ashed and pre-weighed 0.7 mm GF/F filters. Filters were rinsed, dried in a drying oven for 2 days at 60°C and then reweighed. SPM concentrations (in mg l−1) were calculated as (final filter weight-original filter weight)/volume of water filtered. What we refer to as suspended particulate matter or SPM here can also be thought of as seston of less than 210 μm in diameter.
To measure chlorophyll a we filtered water through a 47-mm GF/F filter until clogged using a vacuum pump. The volumes varied between 0.8 and 3 l. The filters were then frozen until we were ready for analysis. In our Woods Hole laboratories, the filters were thawed and sonicated in 90% buffered acetone to lyse the cells, and chlorophyll was measured after 12 h of incubation, on a Turner Designs 10AU fluorometer for fluorescence before and after acidification. Chlorophyll a concentrations were calculated as
where F i is the slope of the calibration curve, R b-sam is the reading of the sample before being acidified, R a-sam is the reading of the sample after being acidified, r is the ratio of the fluorescence values of a pure chl a solution/acidified pure chl a solution, V acetone is the volume of the extract (ml), V sample is the volume of water filtered (ml).
Isotopic and Elemental Analysis
N, C, and S stable isotope and elemental analyses of SPM were performed by mass spectrometry at The Ecosystems Center, Marine Biological Laboratory, Woods Hole, MA. Solid samples were analyzed for δ15N, δ13C, and δ34S using an Europa 20-20 continuous-flow isotope ratio mass spectrometer interfaced with an Europa ANCA-SL elemental analyzer. The analytical precision, based on replicate analyses of isotopically homogeneous NIST Standard reference materials, was ± 0.1 ‰ for δ 15N and δ 13C measurements, ±0.3‰ for δ 34S, and about ±1% for %N, %C, and %S measurements. Stable isotope ratios were reported as per mil (‰) using delta notation determined as
where R was 15N/14N, 13C/12C, or 34S/32S. To aid interpretation of sources, composition, and transport of SPM, we determined N, C, and S contents of SPM samples analyzed for isotopic signatures. To constrain costs of isotopic and elemental analysis for sulfur, a subset of samples of SPM, collected during 2009 and 2010, were selected for analysis. These were randomly chosen from the larger set of samples of SPM, from estuaries within the entire range of % forest cover on their watershed.
To compare the isotopic signatures of SPM to those of the various potential sources of suspended materials, we first collected surface sediments within each estuary by taking 1-cm deep, 5-cm diameter cores in the stations down-estuary, and measured stable isotope and elemental contents as in the case of SPM samples. Second, we also sampled large particulate organic matter (POM) being transported down-estuary by installing a 30-cm diameter plankton net near the mouth of estuaries during ebbing tide. Third, to compare SPM values to those of mangrove trees, we used stable isotopic ratios and elemental values in mangrove tree leaves sampled within the same estuaries, reported in Valiela and others (in press). Fourth, we also reviewed published data on tropical terrestrial trees, soils, and coastal sediments as additional comparisons.
Statistical Analyses
SPM concentrations measured during our samplings varied substantially. In this paper we highlight effects of degree of watershed deforestation and down-estuary gradients, and what took place within the mangrove estuaries, and outside the estuaries. We acknowledge that variation in amount and quality of SPM can be influenced by inter-annual, seasonal, tidal elevation, meteorological differences, as well as watershed differences in topography, soils, and vegetation. We did not try to partition and evaluate all sources of variation, but instead focused on assessing whether the effects of watershed deforestation and estuarine gradient—scaled by salinity—were robust enough to emerge through the large variation in SPM concentrations in samples collected within estuaries or outside estuaries.
Comparisons of the eight different estuaries, sampled on five different dates, and with nine stations located from the fresh to marine salinity ranges proved rather cumbersome. To make comparisons practical and more accessible, we stratified the eight watershed–estuaries into three groups, representing high (91–92%, Limon and Manglarito, Figure 1), intermediate (47–76%, Chamuscado, Luis, de la Mona, and Pixvae, Figure 1), and low (23–29%, Grande and Salmonete, Figure 1) forest cover on the surface of contributing watersheds (see also Table 1).
We applied two types of analysis. To first document the link of SPM to both forest cover and salinity down-estuary, while simultaneously showing the entire data sets, we regressed SPM variables versus salinity for each of the three forest cover groups, and then compared the regressions among the forest groups, by either ANCOVA or, where slopes differed, by an equivalent procedure involving t tests of intercepts and slopes among regression equations. These analyses were separately applied to measurements taken within estuaries, and outside estuaries. Second, to then partition the effects of forest cover, salinity, and sampling trip (which included seasonal and inter-annual variation), and test their significance versus the residual from remaining sources of variation, we used a random factor three-way ANOVA with unequal replication. We used a random factors model because initial examination showed that high residual variation made effects of nested structure of sampling and repeated measures undetectable. Statistical analyses were run in R software.
Below we first document the down-estuary course of SPM concentrations and assess the effect of watershed deforestation. Then, we sort out some of the components that made up the SPM that we measured, and end with a consideration of the sources, connections, and transport of particulate materials in these coupled adjoined watershed–stream–mangrove estuary–coastal water ecosystems.
Results and Discussion
Effects of Deforestation and Down-Estuary Gradients on Amount of SPM
Within-Estuary Effects
In spite of substantial variability in concentration of SPM measured within estuaries, water emerging from deforested watersheds contained significantly larger concentrations of SPM compared to water derived from watersheds with greater forest cover (Figure 2). The contrasts were evident in regressions of SPM and down-estuary salinity, which revealed three significant features (Figure 2 left):
Concentrations of suspended particulate matter in water samples collected inside and outside estuaries draining watersheds shown in Figure 1, plotted versus the salinity of the water. Data from the estuaries pooled into groups with 23–29, 47–73, and 91–92% forest cover on contributing watersheds. Box below left panel shows statistics for samples collected within estuaries; **: highly significant regressions; regressions followed by different letter (a or b) differed significantly from each other. Box below right panel shows mean ± S.E. only; there were no significant regressions in samples collected outside estuaries.
-
(1)
water emerging from deforested watersheds had higher concentrations of particles to fresh reaches of receiving estuaries: concentrations of SPM delivered to fresh reaches of the estuaries (evaluated as the intercept of the regressions, where salinity = 0) were three times larger in watersheds with greater conversion to pastures (5.3 mg l−1) than in more forested watersheds (1.8 mg l−1);
-
(2)
there was net entrainment of particles as water moved down-estuary: SPM concentrations increased downstream, from fresh reaches to estuary mouth, by more than one order of magnitude (Figure 2 left); and
-
(3)
larger concentrations of SPM were present and transported within estuaries with deforested watersheds: mean concentrations of SPM within estuaries with deforested watersheds were more than twice the amount found in estuaries draining well-forested watersheds (Figure 2 left).
Partition of sources of variation by ANOVA confirmed the above results: watershed forest cover, salinity, and “sampling time” (a variable that included seasonal and inter-annual variation) all significantly influenced SPM (Table 2). Sampling time interacted significantly with both forest cover and salinity (Table 2). Slopes of forest cover group versus salinity relationships (Figure 2 left) did not differ, justifying application of ANCOVA to that analysis. The significant “sampling time” effect points to major seasonal and inter-annual contrasts. We have reported on rainfall effects (Valiela and others 2012), but initial examination of per sampling time effects on SPM did not reveal discernible patterns readily assignable to specific weather or seasonal conditions, so here we simply concluded that variation across time was notable but was, for present purposes, separable from the effects of forest cover and salinity, the focus of this analysis.
The amounts of SPM we report from samples taken within the Panama estuaries fell within a lower range compared to reports from other tropical coastal environments (Table 3) and compared to European temperate latitude estuaries [1–1,000 mg SPM l−1, from Middelburg and Herman (2007)]. SPM in tropical estuaries seems to be extremely variable (Table 3). In general, water transparency in the Panama estuaries we studied was high, and the estuary floor was almost always visible during the occasions we sampled. We note, however, that our sampling did not extend through the entire rain season, so we probably missed short-term higher discharge events, when turbidity was likely much higher.
Outside-Estuary Effects
Samples of SPM from water collected in stations off the mouth of the estuaries were also variable (Figure 2 top right). Mean SPM concentrations sampled outside estuaries were similar to the bulk of the data obtained in within-estuary samples of high salinity (Figure 2 left). In spite of the variation, the mean SPM for samples outside the estuaries still retained a trace of effect of watershed deforestation, with larger SPM values associated with discharges from estuaries with deforested watersheds (Figure 2 right). Most samples of SPM collected outside the estuaries resembled values recorded at high salinities inside the estuaries, with some notable exceptions. Samples with salinity lower than 22 were from a December 2010 sampling that followed unusually high wet season rainfall that substantially freshened and diluted concentrations (Valiela and others 2012). Some samples collected outside the estuaries showed lower SPM, probably a measure of particle sinking toward the sea floor after the water mass emerged from the estuaries. Below, we will return to this likely directional flux from estuaries to coastal sediments. SPM measured outside the Panama estuaries fell within the lower range of values measured elsewhere in the tropics (Table 3).
Effects on composition of SPM
To evaluate the effect of deforestation and down-estuary transport on organic and inorganic composition of SPM, we plotted carbon, nitrogen, mineral content, and chlorophyll of SPM versus the % of the watershed that was deforested, for samples collected within salinity ranges of less than 5, 5–25, and greater than 25 inside the estuaries (Figure 3).
Rows: % carbon, nitrogen, and mineral content, and chlorophyll concentrations in suspended particulate matter in water samples collected inside estuaries. All variables plotted versus the % of the watershed area covered by forests. Columns data for each row variable, stratified into salinity groups (<5, 5–25, and >25‰) to show down-estuary transitions.
Carbon Content of SPM
Deforestation on the contributing watersheds significantly lowered the carbon content of SPM derived from watersheds (Figure 3, top panels; Table 1). % C in SPM discharged from watersheds with 91–92% forest cover into fresh reaches of receiving estuaries was more than seven times larger than in SPM discharged from watersheds with forest covers of 23–29% (Figure 3; and regression intercepts in Table 2).
The forest cover imprint on % C diminished down-estuary (Figure 3; Table 2, compare slopes of the regressions). By the time salinity in the estuary water column exceeded 25, %C in SPM in all estuaries converged to about 1% C, regardless of the forest cover on the contributing watershed (Figure 3).
These results suggest that, first, the lability of organic matter in SPM, as well as the amounts of SPM, likely diminished with deforestation. Second, the watershed imprints conveyed by degree of forest cover were erased during down-estuary transit of the particulate materials.
Nitrogen Content and C/N of SPM
The %N in SPM collected within fresh reaches depended on forest cover on the contributing watershed (Table 1). %N was significantly higher in SPM released into fresh reaches from the most forested watersheds (Figure 3, second row of panels, and note >6-fold difference in intercepts in Table 2). The imprint of forest cover, however, was soon erased during transit down-estuary, and %N in SPM converged toward lower values characteristic of the most deforested watersheds.
C/N values within SPM were relatively unchanging in these estuaries, remaining near about 10 throughout the salinity gradient to sea. A ratio of 10 is fairly N-rich, compared to the 17 most suitable to support heterotrophic animal activity (Valiela 1995, p. 208) so that, whatever the amount of SPM exported from the estuaries, these exports were likely to subsidize consumers in receiving coastal food webs. The range of C/N values we recorded in Panama SPM are within those reported for inside and emerging from other tropical estuaries [3.3–22, Robertson and others (1993); 7.3–9.3, Gonneea and others (2004); 13–14.2, Prasad and Ramanathan (2009)]
Mineral Content of SPM
SPM emerging from deforested watersheds held much larger fractions of mineral matter than SPM from forested watersheds. To assess mineral content of SPM, we used our measurements of the % carbon data of SPM, assumed that, on average, carbon made up 45% of organic matter, and calculated the % organic content for each sample of SPM, and then, by difference, obtained the % mineral content as the remainder of SPM weights. Seventy to 99% of SPM was mineral matter, with the lower values only found in fresh reaches of streams receiving inputs from well-forested watersheds (Figure 3; Table 2, note 12-fold difference in intercepts with zero salinity).
Mineral content of SPM increased during down-estuary transit, from 70 to 98% in fresh reaches, to 94–98% at intermediate salinities, to 98–99% mineral content at full salinity (Figure 3). This trend may be the joint effect of degradation of labile organic matter derived from forested watersheds plus entrainment of sediment during transit down-estuary.
Chlorophyll in SPM
Chlorophyll concentrations were low in the fresher reaches of the estuaries, most likely because groundwater seeping into streams obviously lacks chlorophyll, and these fast-flowing streams allowed too short a time for much cell division (Figure 3 bottom row of panels). Concentrations increased somewhat down-estuary, with an indication that there was more growth of cells within water columns of estuaries with less-forested watersheds (Figure 3, note significant slopes in saltier reaches of the estuaries). This response may be a result of greater light availability in streams where the riparian forest canopy is less dense. The response does not seem related to nutrient supply, because in waters with salinity greater than 5, available N/P in water varied, but averaged around 16:1, the critical Redfield ratio (Valiela and others 2013). Deegan and others (2011) argued that light may play a similar role affecting algal nitrogen uptake and use in Amazonian streams surrounded by pasture dominated vegetation. An additional feature to note in the Panama data was the contrast in mean chlorophyll concentrations emerging into coastal waters from the estuaries with different forest cover on contributing watersheds (Table 3): larger amounts of chlorophyll were associated with passage through estuaries with more deforested watersheds (Table 3). The concentrations of chlorophyll emerging from the mouth of the estuaries (in water with higher salinity) were modest (Figure 3 bottom row), but similar to the lower range that others found elsewhere in the tropics (Robertson and others 1993; Bouillon and others 2007; Ralison and others 2008). Nevertheless, the exports of chlorophyll constituted a significant addition of cells to the receiving coastal water column, because concentrations of chlorophyll leaving the estuaries were higher than chlorophyll concentrations measured in surface water 1–6 km off estuary mouths (Table 3). Robertson and others (1993) found similar contrasts, with 0.3–5.1 μg chlorophyll l−1 measured within estuaries, and only 0.3–0.7 μg chlorophyll l−1 outside estuaries. Deforested watersheds therefore made greater contributions of chlorophyll-bearing cells that could then either grow or were consumed in near-shore food webs. Deforestation increased land-to-sea connectivity and subsidies furnished by terrestrial to marine ecosystems.
To approximate the contribution of chlorophyll to total SPM mass, we used a mean ratio of cell weight to chlorophyll of 70 (calculated from data in MacIntyre and others 2002; Li and others 2010; Xiu and Chai 2012; Wang and others 2013; Harrison and others 1997). The calculated mass of phytoplankton ranged 0.07–0.56 mg chlorophyll l−1, expressed as % of SPM mass (which we estimated as 1–30% organic matter, estimated from %C/0.45, Figure 3). The contribution of chlorophyll was therefore quite small compared to the mass of SPM (0.7–40 mg SPM l−1, from Figure 2). Chlorophyll therefore ranges between 0.014 and 0.1% of the SPM. The minor contribution of chlorophyll to tropical estuarine SPM was also found by Robertson and others (1993), Machás and Santos (1999), and Bouillon and others (2007) (Table 4).
Terrestrial and Estuarine Influences on Stable Isotope Values of SPM
Isotopic Comparisons Within Panama Estuaries
To confirm effects of deforestation of land cover, and of down-estuary transformations on SPM, we plotted δ13C, δ15N, and δ34S of SPM versus the % forest cover on the watersheds, for samples collected within salinities below 5, 5–25, and above 25 (Figure 4). δ13C of SPM were significantly enriched in fresher and intermediate reaches of estuaries receiving inputs from more deforested watersheds, compared to SPM sampled from estuaries with more forested watersheds (Figure 4). The terrestrial land cover imprint on δ13C of SPM was erased down-estuary, and in saltier reaches of estuaries, values of δ13C became enriched. δ15N of SPM were also enriched in fresher reaches of estuaries receiving inputs from deforested watersheds (Figure 4). As in the case of δ13C, terrestrial imprint was lost in intermediate and salty reaches. Nitrogen isotopic signatures were variable and remained between 3 and 7 though transit to the sea. δ34S of SPM varied, with no evidence of effect of watershed forest cover, but a suggestion of substantial enrichment down-estuary (Figure 4).
To discern possible sources and processes generating the trends of Figure 4, we tried two approaches. First, we plotted isotopic signatures of SPM versus signatures of likely source materials (mangrove sediment, mangrove leaves, and large particulate matter) (Figure SI 1). Within the Panama estuaries, δ13C of SPM were similar to those of mangrove estuary sediments, were heavier than those of mangrove leaves, and lighter than those of large particulate matter (POM) (Figure SI 1). Isotopic C signatures of SPM therefore could readily have originated from erosion of sediments [which may make up 70–100% of SPM (Figure 3)], plus a mix of leaf and other plant fragments [leaves tend to have lighter carbon signatures than woody parts (Martinelli and others 1998; Ometto and others 2006; Nardoto and others 2008)]. δ15N of SPM were heavier than those of sediment and mangroves, and generally lighter than those of POM (Figure SI). These results imply that SPM contained nitrogen remaining after some microbial reworking had preferentially removed the lighter isotope of N during transit in the water column.
Second, to make more evident the spatial distribution of the potential sources of SPM, and its fate during transit through and out of estuaries, we plotted the stable isotopic signatures of C, N, and S in SPM versus salinity, and added, for comparison, the ranges in isotopic values, culled from the literature and our own data, for a variety of potential sources (Figure 5). This depiction shows changes during down-estuary trajectories, compares the effects of deforestation, and shows possible links of signatures in SPM to potential sources.
Carbon, nitrogen, and sulfur stable isotope values of suspended particulate matter (samples from all stations), plotted versus salinity to reveal the down- and outside-estuary spatial gradients. The shaded bars show the ranges of values for different potential sources of the SPM; degree of shading indicates likely position of source contribution along the gradient from fresh to coastal waters. The ranges of isotopic values for the various sources obtained from our own and published data: for tropical tree leaves: Cordell and Sandquist 2008, Courty and others 2011, Domingues and others 2007, Jennerjahn and others 2004, Martinelli and others 1998, Martinelli and others 1999, Muzuka 1999, Nardoto and others 2008, Norstrom and others 2012, Ometto and others 2006, Pessenda and others 2004, Peterson and Howarth 1987, Szpak and others 2013, Tamooh and others 2012; for tropical soils: Bouillon and others 2007, Desjardins and others 2006, Gao and others 2012, Nardoto and others 2008, Ometto and others 2006, Alkhatib and others 2007, Alongi and others 1989, Bouillon and others 2002, Bouillon and others 2008, Gonneea and others 2004, Loneragan and others 1997, Okada and Sadaki 1998, Prasad and Ramanathan 2009, Ralison and others 2008, Tue and others 2012, Wooller and others 2004; for mangrove sediments: Alkhatib and others 2007, Alongi and others 1989, Alongi and others 2004, Bouillon and others 2002, Bouillon and others 2007, Canfield and others 1998, Gao and others 2012, Jennerjahn and Ittekkot 2002, Jennerjahn and others 2004, Ku and others 1999, Loneragan and others 1997, Norstrom and others 2012, Pereira and others 2010, Pessenda and others 2004, Pessenda and others 2012 (only recent samples), Prasad and Ramanathan 2009, Ralison and others 2008, Ranjan and others 2011, Tamooh and others 2012, Tue and others 2012a; for coastal SPM: Alkhatib and others 2007, Bouillon and others 2002, Bouillon and others 2007, Bouillon and others 2008, Bouillon and others 2007a, Cifuentes and others 1996, Dunn and others 2008, Gonneea and others 2004, Jennerjahn and others 2004, Kitheka and others 2005, Loneragan and others 1997, Martinelli and others 1999, Martinelli and others 2012, Maya and others 2011, Miranda and others 2009, Prasad and Ramanathan 2009, Ralison and others 2008, Robertson and others 1993, Tamooh and others 2012, Tue and others 2012a; for coastal sediments: Alkhatib and others 2007, Alongi and others 1989, Barros and others 2010, Baumgart and others 2010, Bouillon and others 2002, Boning and others 2004, Bruchert and Pratt 1999, Canfield and others 1998, Dunn and others 2008, Gao and others 2012, Harji and others 2010, Jennerjahn and Ittekkot 2002, Jennerjahn and others 2004, Mazumdar and others 2012, Miranda and others 2009, Muzuka 1999, Norstrom and others 2012, Pereira and others 2010, Peterson and Howarth 1987, Prasad and Ramanathan 2009, Ralison and others 2008, Robinson and others 2012, Sampaio and others 2010, Strauss and others 2012, Tamooh and others 2012, Tue and others 2012a, Wijsman and others 2001, Zhu and others 2013.
Judging from δ13C values, the SPM that entered the fresh reaches of the streams were a mix of material derived from terrestrial vegetation and soils. δ13C values of SPM, regardless of the degree of forest cover on the corresponding watersheds, resembled signatures characteristic of tropical trees and tropical soils. We could have expected some influence of C4 grasses growing on the pastures, but such sources were not in evidence in SPM collected at the fresh reaches of the estuaries (Figure 5). Even in watersheds with only 23% forest cover, the effect of the C4 grasses did not dominate, a result also found by Deegan and others (2011). Whatever trend to heavier carbon isotope signatures may be present, we could not attribute the shift entirely to C4 grasses, as there were algae and C3 aquatic plants present in the fresh reaches that might have contributed carbon.
Figure 5 includes tropical soil data from sites supporting largely C3 vegetation, even though vegetative covers in certain of our watersheds likely held an undetermined portion of C4 plants. Pasture and forest soils are isotopically distinguishable within certain specific inland sites (Bernoux and others 1998; Yonekura and others 2012), but the sorting out of C3 and C4 carbon in watersheds with wet tropical soils is complicated by a number of issues (Powers and Veldcamp (2005). Soils mostly developed under long-term forest cover, carbon added by forest and grassland sources decay and survive at different rates, and tropical pastures often are a combination of C4 and C3 grasses. In addition, in many tropical pastures there are often remnant C3 trees left within or near pastures. Moreover, there is often a riparian edge in many tropical streams, populated with C3 plants, where interception of particulate materials from pastures inland might take place. Because of these complications, it is not surprising to not find a clear-cut C4 imprint in our isotopic data for particulate materials entering fresh reaches of estuaries, and so we did not include data from C4 pasture soils in Figure 5.
The link of stream SPM and tropical trees was rapidly lost as salinity increased (Figure 5 top). The erasure of the terrestrial imprint seemed likely to be a result of the order-of-magnitude increase in total SPM as salinities increased, visible in Figure 2, where the added material, contributed by a mix of mangrove sediment and mangrove biomass, overwhelmed the terrestrial signatures. The dominance of mangrove material extended through the salinity gradient. At higher salinities near the mouth of the estuaries and beyond, δ13C of SPM became further enriched (and resembled what the literature reports as coastal SPM in many coastal systems). Finally, the δ13C of some of the SPM in the saltiest part of the gradient matched the δ13C reported for coastal sediments. The pattern therefore seems to be that terrestrial inputs to upper reaches of streams were distinctive in the relatively clear waters of fresh reaches, but were overwhelmed by larger amounts of particles derived from the mangrove estuary as water became saltier. Once SPM left the estuaries, additional enrichment took place, and carbon signatures of SPM then shifted to levels near those of coastal surface sediments.
The pattern for δ15N of SPM seemed similar to that of δ13C, but somewhat more variable and less marked (Figure 5 middle panel). There were a number of relatively low values of δ15N in SPM released into fresh reaches, which suggests the influence of nitrogen fixation (by symbiosis in legumes, orchids, bromeliads, cyanobacteria on tree leaves, and free-living bacteria). There were no other evident effects of degree of deforestation on isotopic signatures of SPM. In studies in much deeper coastal environments, Robinson and others (2012) reviewed the complex possibilities that sharply alter δ15N of nitrogen within sediments, once SPM was deposited. There may be no such processes operating in the shallow systems we sampled, because the range of δ15N of SPM we measured overlap comfortably with reported values for coastal surface sediments. This lack of further fractionation merits further study.
The emergent pattern revealed by the carbon and nitrogen isotopic data is that the down-estuary gradients in SPM indeed revealed land-sea linkages in this Panamanian coastal zone. SPM signatures made evident the effects of terrestrial and mid-estuary mangrove sources, of active transformation within the estuaries, and, showed that after transit and export, the isotopic signatures of SPM that managed to be exported from estuaries resembled those characteristic of coastal surface sediments. So far, therefore, the isotopic data demonstrated that SPM transport links land, estuary, and coastal sediments, but do not unambiguously tell us the direction of fluxes: the δ13C gradient, for example, could have merely resulted from a passive mixing of terrestrial and marine sources.
The down-estuary pattern of the δ34S signatures (Figure 5 bottom) significantly adds to our interpretation of the fate of land- and mangrove-derived SPM. In fresh reaches, δ34S of SPM resembled those of terrestrial trees. As water became saltier, the isotopic signatures of SPM spanned a rather variable mix that might have been from mangrove leaves or sediments. The resulting exported SPM, however, converged on higher values (around 15‰), quite similar to values reported for SPM from a variety of coastal environments (Figure 5 bottom). This resemblance points to some common, likely terrestrial-estuarine, sources for SPM found in many coastal sites.
The novel aspect that the sulfur isotope data add is that the exported SPM was, however, notably enriched compared to signatures characteristic of coastal surface sediments (by about 10‰, a significant contrast, Figure 5 bottom). The major point here is that sulfur signatures of SPM measured in our samples, and in those from many other tropical coastal waters, differ from those characteristic of coastal sediments (Figure 5). Coastal sea floor sulfur signatures are usually dominated by reduced sulfide (FeS, FeS2, or elemental S). This contrast points out that the likely direction of transport, in these Panamanian ecosystems, and probably others elsewhere, is likely to be from land and estuarine toward the coastal seas. If we were, instead, dealing with a passive mixing of estuarine and coastal sediments, we might not find what we found: a large gap in isotopic values between SPM that just emerged from estuaries, and isotopic values on surface sediments under near-shore waters. The estuarine SPM thus seem most likely to flux out of the estuaries, and sink to the coastal sea floor (recall SPM data collected outside estuaries in Figure 2 right). These results suggest that it is the land, and more so, the mangrove estuaries, that export particulate materials to coastal waters. The direction of the ecosystem-level couplings of watershed-mangrove-coastal waters by suspended particulate matter is asymmetrical, with terrestrial and mangrove exports dominating over marine sources.
The results we report on exports confirm the significant exports from estuarine and wetland ecosystems to coastal waters, reported for tropical (Jennerjahn and Ittekkot 2002; Dittmar and others 2006) as well as temperate systems (Valiela and Teal 1979). These papers used a variety of data to make such inferences. In the data of Figure 5 we show a first demonstration of the detailed trajectory of materials that show the mechanisms involved through the couplings of land, estuary and sea, and confirm the suspected exports toward marine ecosystems. Many references conclude that there are significant exports of terrestrial material to coastal waters. Some ambitious efforts are cast at rather large spatial scales, even global estimates (Milliman and others 2008; Mayorga and others 2010). These large-scale efforts focus on mass river transport, and elide over details such as the effects of watershed land covers, or the relative inputs via groundwater versus surface runoff, details that might be important to understand mechanisms that govern land–sea coupling in adjoined watershed–mangrove estuary–coastal water ecosystems. For example, in an earlier paper (Valiela and others 2013) we showed that exports of dissolved inorganic nitrogen from the estuaries studied here differed significantly depending on the land cover on the contributing watersheds. As it turns out, from the results of the present paper, particulate matter exports seem, in contrast, less affected by watershed land covers, at least in the Panama systems. Such contrasts might be important in assessing large-scale budgets.
In more general terms, there is no doubt that mangrove estuaries play a significant function in such land-sea couplings. First, results from this paper and Valiela and others (2013), and those cited elsewhere (Kristensen and others 2008; Breithaupt and others 2012, among others) make the point that there is rather substantial transformations, interception, and sequestering of watershed-derived dissolved and particulate materials within mangrove estuaries. Second, our results, and those of others (Jennerjahn and Ittekkot 2002; Dittmar and others 2006) also suggest that, in spite of the within-mangrove estuary retention and interception, there are exports of particulate matter from the coupled watershed-mangrove ecosystems to adjoined coastal waters, exports that compared to the depauperate condition of the receiving tropical waters are likely to be biologically meaningful. The joint, countering effects of within-estuary interception and the significant export, means that the biogeochemical functioning of these coastal wetland systems is even more quantitatively important than we might have known by just considering exports. Losses of mangrove estuaries, among the largest affecting any environment (Valiela and others 2001) might result in large—and, depending on the trajectories of land cover on the watersheds and rainfall regimes, unpredictable—shifts in sequestration and transfer of land-derived exports to receiving coastal waters throughout the tropics.
References
Alkhatib M, Jennerjahn TC, Samiaji J. 2007. Biogeochemistry of the Dumai River estuary, Sumatra, Indonesia, a tropical black-water river. Limnol Oceanogr 52:2410–17.
Alongi DM, Boto KG, Tirendi F. 1989. Effect of exported mangrove litter on bacterial productivity and dissolved organic carbon fluxes in adjacent tropical nearshore sediments. Marine Ecol Prog Ser 56:133–44.
Alongi DM, Sasekumar A, Chong VC, Pfitzner J, Trott LA, Tirendi F, Dixon P, Brunskill GJ. 2004. Sediment accumulation and organic material flux in a managed mangrove ecosystem: estimates of land—ocean—atmosphere exchange in peninsular Malaysia. Marine Geol 208:383–402.
Bahn M, Reichstein M, Davidson EA et al. 2010. Soil respiration at mean annual temperature predicts annual total across vegetation types and biomes. Biogeosciences 7:2147–57.
Barros GV, Martinelli LA, Oliveria Novais TM, Ometto JPHB, Zuppi GM. 2010. Stable isotopes of bulk organic matter to trace carbon and nitrogen dynamics in an estuarine ecosystem in Babitonga Bay (Santa Catarina, Brazil). Sci Total Environ 408:2226–32.
Baumgart A, Jennerjahn T, Mohtadi M, Hebbeln D. 2010. Distribution and burial of organic carbon in sediments from the Indian Ocean upwelling region off Java and Sumatra, Indonesia. Deep-Sea Res I 57:458–67.
Bernoux M, Cerri CC, Neill C, de Moraes JFL. 1998. The use of stable carbon isotopes for estimating soil organic matter turnover rates. Geoderma 82:43–58.
Boning P, Brumsack HJ, Bottcher ME, Schnetger B, Kriete C, Kallmeyer J, Borchers SL. 2004. Geochemistry of Peruvian near-surface sediments. Geochim Cosmochim Acta 68:4429–51.
Bosch JM, Hewlett JD. 1982. A review of catchment experiments to determine the effects of vegetation changes on water yield and evapotranspiration. J Hydrol 55:3–23.
Bouillon S, Raman AV, Dauby P, Dehairs F. 2002. Carbon and nitrogen stable isotope ratios of subtidal benthic invertebrates in an estuarine mangrove ecosystem (Andhra Pradesh, India). Estuar Coast Shelf Sci 54:901–13.
Bouillon S, Dehairs F, Velimirov B, Abril G, Borges AV. 2007a. Dynamics of organic and inorganic carbon across contiguous mangrove and seagrass systems (Gazi Bay, Kenya). J Geophys Res 112:G02018. doi:10.1029/2006JG000325.
Bouillon S, Middelburg JJ, Deharis F, Borges AV, Abril G, Flindt MR, Ulomi S, Kristensen E. 2007b. Importance of intertidal sediment processes and porewater exchange on the water column biogeochemistry in a pristine mangrove creek (Ras Dege, Tanzania). Biogeosciences 4:311–22.
Bouillon S, Connolly RM, Lee SY. 2008. Organic matter exchange and cycling in mangrove ecosystems: recent insights from stable isotope studies. J Sea Res 59:44–58.
Breithaupt JL, Smoak JM, Smith TJ, Sanders CJ Hoare A. 2012. Organic carbon burial rates in mangrove sediments: strengthening the global budget. Glob Biogeochem Cycles 26: GB3011. doi:10.1029/2012GB004375.
Bruchert V, Pratt LM. 1999. Stable sulfur isotopic evidence for historical changes of sulfur cycling in estuarine sediments from Northern Florida. Aquat Geochem 5:249–68.
Bruijnzeel LA. 2004. Hydrological functions of tropical forests: not seeing the soil for the trees? Agric Ecosyst Environ 104:185–228.
Canfield DE, Boudreau BP, Mucci A, Gundersen JK. 1998. The early diagenetic formation of organic sulfur in the sediments of Mangrove Lake, Bermuda. Geochim Cosmochim Acta 62:767–81.
Cifuentes LA, Coffin RB, Solorzano L, Cardenas W, Espinoza J, Twilley RR. 1996. Isotopic and elemental variations of carbon and nitrogen in a mangrove estuary. Estuar Coast Shelf Sci 43:781–800.
Cleveland CC, Townsend AR, Schimel DS et al. 1999. Global patterns of terrestrial biological nitrogen (N2) fixation in natural systems. Glob Biogeochem Cycles 13:623–45.
Cordell S, Sandquist DR. 2008. The impact of an invasive African bunchgrass (Pennisetum setaceum) on water availability and productivity of canopy trees within a tropical dry forest in Hawaii. Funct Ecol 22:1008–17.
Courty PE, Walder F, Boller T, Ineichen K, Wiemken A, Rousteau A, Selosse MA. 2011. Carbon and nitrogen metabolism in mycorrhizal networks and mycoheterotrophic plants of tropical forest: a stable isotope analysis. Plant Physiol 156:952–61.
Davin EL, Noblet-Ducoudre N. 2010. Climatic impact of global-scale deforestation: radiative versus non-radiative processes. J Climatol 23:97–112.
Deegan LA, Neill C, Haupert CL, Ballester MVR, Krusche AV, Victoria RL, Thomas SM, de Moor E. 2011. Amazon deforestation alters small stream structure, nitrogen biogeochemistry and connectivity to larger rivers. Biogeochemistry 105:53–74.
Desjardins T, Folgarait PJ, Pando-Bahuon A, Girardin C, Lavelle P. 2006. Soil organic matter dynamics along a rice chronosequence in north-eastern Argentina: evidence from natural 13C abundance and particle size fractionation. Soil Biol Biogeochem 38:2753–61.
Dittmar T, Hertkorn N, Kattner G, Lara RJ. 2006. Mangroves a major source of dissolved organic carbon to the oceans. Glob Biogeochem Cycles 20:GB1012. doi:10.1029/2005GB002570.
Domingues TF, Martinelli LA, Ehleringer JR. 2007. Ecophysiological traits of plant functional groups in forest and pasture ecosystems from eastern Amazonia, Brazil. Plant Ecol 193:101–12.
Downing JA, McClain M, Twilley R et al. 1999. The impact of accelerating land-use change on the N-cycle of tropical aquatic ecosystems: current conditions and projected changes. Biogeochemistry 46:109–48.
Dunn RJK, Welsh DT, Teasdale PR, Lee SY, Lemckert CJ, Meziane T. 2008. Investigating the distribution and sources of organic matter in surface sediment of Coombabah Lake (Australia) using elemental, isotopic and fatty acid biomarkers. Cont Shelf Res 28:2535–49.
Gao X, Yang Y, Wang C. 2012. Geochemistry of organic carbon and nitrogen in surface sediments of coastal Bohai Bay inferred from their ratios and stable isotopic signatures. Mar Pollut Bull 64:1148–55.
Gash JHC, Nobre CA, Roberts JM et al. 1996. Amazonian deforestation and climate. Chichester, UK: Wiley.
Gonneea ME, Paytan A, Herrera-Silveira JA. 2004. Tracing organic mater sources and carbon burial in mangrove sediments over the past 160 years. Estuar Coast Shelf Sci 61:211–27.
Harji RR, Bhosle NB, Garg A, Sawant SS, Venkat K. 2010. Sources of organic matter and microbial community structure in the sediments of the Visakhapatnam harbor, east coast of India. Chem Geol 276:309–17.
Harrison PJ, Khan N, Yin K, Saleem M, Bano N, Nisa M, Ahmed SI, Rizvi N, Azam F. 1997. Nutrient and phytoplankton dynamics in two mangrove tidal creeks of the Indus River delta, Pakistan. Mar Ecol Progr Ser 157:13–19.
Houser JN, Mulholland PJ, Maloney KO. 2006. Upland disturbance affects headwater stream nutrients and suspended sediments during baseflow and stormflow. J Environ Qual 35:352–65.
IPCC. 2007. Climate Change 2007: Mitigation. Contribution of working group III to the fourth report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, UK.
Jennerjahn TC, Ittekkot V. 2002. Relevance of mangroves for the production and deposition of organic matter along tropical continental margins. Naturwissenschaften 89:23–30.
Jennerjahn TC, Ittekkot V, Klopper S, Adi S, Nugroho SP, Sudiana N, Yusmal A, Prihartanto , Gaye-Haake B. 2004. Biogeochemistry of a tropical river affected by human activities in its catchment: Brantas River estuary and coastal waters of Madura Strait, Java. Indonesia. Estuar Coast Shelf Sci 60:503–14.
Kitheka JU, Obiero M, Nthenge P. 2005. River discharge sediment transport and exchange in the Tana Estuary, Kenya. Estuar Coast Shelf Sci 63:455–68.
Kristensen E, Bouillon S, Dittmar T, Marchand C. 2008. Organic carbon dynamics in mangrove ecosystems: a review. Aquat Bot 89:201–19.
Ku TCW, Walter LM, Coleman ML, Blake RE, Martini AM. 1999. Coupling between sulfur recycling and syndepositional carbonate dissolution: evidence from oxygen and sulfur isotope composition of pore water sulfate, South Florida Platform, USA. Geochim Cosmochim Acta 63:2529–46.
Lewis WM. 2002. Yield of nitrogen from minimally disturbed watersheds of the United States. Biogeochemistry 57–58:375–85.
Lewis WM, Melack JM, McDowell WH et al. 1999. Nitrogen yields from undisturbed watersheds in the Americas. Biogeochemistry 46:149–62.
Li KY, Coe MT, Ramankutty N et al. 2007. Modeling the hydrological impact of land-use change in West Africa. J Hydrol 337:258–68.
Li QP, Franks PJS, Landry MR, Goericke R, Taylor AG. 2010. Modeling phytoplankton growth rates and chlorophyll to carbon ratios in California coastal and pelagic ecosystems. J Geophys Res. doi:10.1029/2009JG001111.
Loneragan NR, Bunn SE, Kellaway DM. 1997. Are mangroves and seagrasses sources of organic carbon for penaeid prawns in a tropical Australian estuary? A multiple stable-isotope study. Mar Biol 130:289–300.
Machás R, Santos R. 1999. Sources of organic matter in Ria Formosa revealed by stable isotope analysis. Acta Oecol 20:463–9.
MacIntyre HL, Kana TM, Anning T, Geider RJ. 2002. Photoacclimation of photosynthesis irradiance response curves and photosynthetic pigments in microalgae and cyanobacteria. J Phycol 38:17–38.
Maréchal JC, Varma MRR, Riotte J et al. 2009. Indirect and direct recharges in a tropical forested watershed: Mule Hole, India. J Hydrol 364:272–84.
Martinelli LA, Almeida S, Brown IF, Moreira MZ, Victoria RL, Sternberg LSL, Ferreira CAC, Thomas WW. 1998. Stable carbon isotope ratio of three leaves, boles and fine litter in a tropical forest in Rondonia, Brazil. Oecologia 114:170–9.
Martinelli LA, Piccolo MC, Townsend AR, Vitousek PM, Cuevas E et al. 1999. Nitrogen stable isotopic composition of leaves and soil: tropical versus temperate forests. Biogeochemistry 46:45–65.
Martinelli LA, Ballester MV, Krusche AV, Victoria RL, de Camargo PB, Bernardes M, Ometto JPHB. 2012. Landcover changes and δ13C composition of riverine particulate organic matter in the Piracicaba river basion (Southeast Region of Brazil). Limnol Oceanogr 44:1826–33.
Maya MV, Soares MA, Agnihotri R, Pratihary AK, Karapurkar S, Naik H, Naqvi SWA. 2011. Variations in some environmental characteristics including C and N stable isotopic composition of suspended organic matter in the Mandovi estuary. Environ Monit Assess 175:501–17.
Mayorga E, Seitzinger SP, Harrison JA, Dumont E, Beusen AHW, Bouwman AF, Fekete BM, Kroeze C, van Drecht G. 2010. Global nutrient export from watersheds 2 (NEWS 2): model development and implementation. Environ Model Software 25:837–53.
Mazumdar A, Peketi A, Joao H, Dewangan P, Borole DV, Kocherla M. 2012. Sulfidization in a shallow coastal depositional setting: diagenetic and paleoclimatic implications. Chem Geol 322–323:68–78.
Middelburg JJ, Herman PJM. 2007. Organic matter processing in tidal estuaries. Mar Chem 106:127–47.
Milliman JD, Farnsworth KL, Jones PD, Xu KH, Smith LC. 2008. Climatic and anthropogenic factors affecting river discharge to the global ocean, 1951–2000. Glob Planet Chang 62:187–94.
Miranda MCDC, Rossetti DDF, Pessenda LCR. 2009. Quaternary paleoenvironments and relative sea-level changes in Marajo Island (Northern Brazil): facies δ13C, δ15N and C/N. Palaeogeogr Palaeoclimatol Palaeoecol 282:19–31.
Muzuka AN. 1999. Isotopic compositions of tropical East African flora and their potential as source indicators of organic matter in coastal marine sediments. J Afr Earth Sci 28:757–66.
Nardoto GB, Ometto JPHB, Ehleringer JR, Higuchi N, da Cunha Bustamante MM, Martinelli LA. 2008. Understanding the influences of spatial patterns of N availability within the Brazilian Amazon forest. Ecosystems 11:1234–46.
Neill C, Deegan LA, Thomas SM et al. 2001. Deforestation for pasture alters nitrogen and phosphorus in small Amazonian streams. Ecol Appl 11:1817–28.
Norstrom E, Risberg J, Grondahl H, Holmgren K, Snowball I, Mugabe JA, Sitoe SR. 2012. Coastal paleo-environment and sea-level change at Macassa Bay, southern Mozambique, since c 6600 cal BP. Quat Int 260:153–63.
Nosetto MD, Jobbágy EG, Paruelo JM. 2005. Land-use change and water losses: the case of grassland afforestation across a soil textural gradient in central Argentina. Glob Chang Biol 11:1101–17.
Okada N, Sadaki A. 1998. Sulfur isotopic composition of mangroves. Isot Environ Health Stud 34:61–5.
Ometto JPHB, Ehleringer JR, Domingues TF, Berry JA, Ishida FY, Mazzi E, Higuchi N, Flanagan LB, Nardoto GB, Martinelli LA. 2006. The stable carbon and nitrogen isotopic composition of vegetation in tropical forest of the Amazon basin. Biogeochemistry 79:251–74.
Pereira AA, van Hattum B, de Boer J, van Bodegom PM, Rezende CE, Salomons W. 2010. Trace elements and carbon and nitrogen stable isotopes in organisms from a tropical coastal lagoon. Arch Environ Contam Toxicol 59:464–77.
Pessenda LCR, AdS Ribeiro, Gouveia SEM, Aravena R, Boulet R, Bendassolli JA. 2004. Vegetation dynamics during the late Pleistocene in the Barreirinhas region, Maranhao State, northeastern Brazil, based on carbon isotopes in soil organic matter. Quat Res 62:183–93.
Pessenda LCR, Vidotto E, De Oliveria PE, Busco AA Jr, Cohen MCL, Fatima Rossetti D, Ricardi-Branco F, Bendassolli JA. 2012. Late quaternary vegetation and coastal environmental changes at Ilha do Cardoso mangrove, southeastern Brazil. Palaeogeogr Palaeoclimatol Palaeoecol 363–364:57–68.
Peterson BJ, Howarth RW. 1987. Sulfur, carbon, and nitrogen isotopes used to trace organic matter flow in the salt-marsh estuaries of Sapelo Island, Georgia. Limnol Oceanogr 32:1195–213.
Powers JS, Veldcamp E. 2005. Regional variation in soil carbon and δ13C in forests and pastures of northeastern Costa Rica. Biogeochemistry 72:315–36.
Prasad MBK, Ramanathan AL. 2009. Organic matter characterization in a tropical estuarine-mangrove ecosystem of India: preliminary assessment by using stable isotopes and lignin phenols. Estuar Coast Shelf Sci 84:617–24.
Ralison OH, Borges AV, Dehairs F, Middelburg JJ, Bouillon S. 2008. Carbon biogeochemistry of the Betsiboka estuary (north-western Madagascar). Org Geochem 39:1649–58.
Ranjan RK, Routh J, Ramanathan AL, Val Klump JV. 2011. Elemental and stable isotope records of organic matter input and its fate in the Pichavaram mangrove-estuarine sediments (Tamil Nadu, India). Mar Chem 126:163–72.
Rivera-Monroy VH, Day JW, Twilley RR, Vera-Herrera F, Coronado-Molina C. 1995. Flux of nitrogen and sediment in a fringe mangrove forest in Terminos Lagoon, Mexico. Estuar Coast Shelf Sci 40:139–60.
Robertson AI, Daniel PA, Dixon P, Alongi DM. 1993. Pelagic biological processes along a salinity gradient in the Fly delta and adjacent river plume (Papua New Guinea). Cont Shelf Res 13:205–24.
Robinson RS, Kienast M, Albuquerque AL, Altabet M, Contreras S, De Pol Holz R, Dubois N, Francois R, Galbraith E, Hsu TC, Ivanochko T, Jaccard S, Kao SJ, Kiefer T, Kienast S, Lehmann M, Martinez P, McCarthy M, Mobius J, Pedersen T, Quan TM, Ryabenko E, Schmittner A, Schneider R, Schneider-Mor A, Shigemitsu M, Sinclair D, Somes C, Studer A, Thunell R, Yang JY. 2012. A review of nitrogen isotopic alteration in marine sediments. Paleoceanography 27:PA4203. doi:10.1029/2012PA002321.
Sampaio L, Freitas R, Maguas C, Rodrigues A, Quintino V. 2010. Coastal sediments under the influence of multiple organic enrichment sources: an evaluation using carbon and nitrogen stable isotopes. Mar Pollut Bull 60:272–82.
Scanlon BR, Jolly I, Sophocleous M et al. 2007. Global impacts of conversions from natural to agricultural ecosystems on water resources: quantity versus quality. Water Resour Res 43:W03437. doi:10.1029/2006WR005486.
Smith J, Burford MA, Revill AT, Haese RR, Fortune J. 2012. Effects of nutrient loading on biogeochemical processes in tropical tidal creeks. Biogeochemistry 108:359–80.
Strauss H, Bast R, Cording A, Diekrup D, Fumann A, Garbe-Schonberg D, Lutter A, Oeser M, Rabe K, Reinke D, Teichert BMA, Westernstroer U. 2012. Sulphur diagenesis in the sediments of the Kiel Bight, SW Baltic Sea, as reflected by multiple stable sulphur isotopes. Isot Environ Health Stud 48:166–79.
Sun G, Zhou G, Zhang Z et al. 2006. Potential water yield reduction due to forestation across China. J Hydrol 328:548–58.
Szpak P, White CD, Longstaffe FJ, Millaire JF, Sanchez VFV. 2013. Carbon and nitrogen isotopic survey of northern Peruvian plants: baselines for paleodietary and paleoecological studies. PLoS ONE 8:e53763.
Tamooh F, Van den Meersche K, Meysman F, Marwick TR, Borges AV, Merckx R, Dehairs F, Schmidt S, Nyunja J, Bouillon S. 2012. Distribution and origin of suspended matter and organic carbon pools in the Tana River Basin, Kenya. Biogeosciences 9:2905–20.
Tue NT, Hamaoka H, Sogabe A, Quy TD, Nhuan MT, Omori K. 2012a. Food sources of macro-invertebrates in an important mangrove ecosystem of Vietnam determined by dual stable isotope signatures. J Sea Res 72:14–21.
Tue NT, Ngoc NT, Quy TD, Hamaoka H, Nhaun MT, Omori K. 2012b. A cross–system analysis of sedimentary organic carbon in the mangrove ecosystems of Xuan Thuy National Park. Vietnam J Sea Res 67:69–76.
Valiela I. 1995. Marine ecological processes. 2nd ed. New York: Springer.
Valiela I, Bowen JL, York JK. 2001. Mangrove forests: one of the world’s threatened major tropical environments. BioScience 51:807-15.
Valiela I, Teal JM. 1979. The nutrient budget of a salt marsh ecosystem. Nature 280:652–6.
Valiela I, Camilli L, Stone T, Giblin A, Crusius J, Fox S, Barth-Jensen C, Monteiro RO, Tucker J, Martinetto P, Harris C. 2012. Increased rainfall remarkably freshens estuarine and coastal waters on the Pacific coast of Panama: magnitude and likely effects on upwelling and nutrient supply. Glob Planet Chang. doi:10.1016/j.gloplacha.2012.05.006.
Valiela I, Barth-Jensen C, Stone T, Crusius J, Fox S, Bartholomew M. 2013. Deforestation of watersheds of Panama: nutrient retention and export to streams. Biogeochemistry. doi:10.007/s10533-013-9836-2.
Valiela I, Viana IG, Pascual J, Giblin A, Barth-Jensen C, Martinetto P, Otter M, Stone T, Tucker J, Bartholomew M. In press. External and local controls on land-sea controls on land-sea couplings assessed by stable isotopic signatures of mangrove producers in estuaries of Pacific Panama. Aquat Bot.
Vorosmarty CJ, Fekete BM, Meybeck M et al. 2000. Global system of rivers: its role in organizing continental land mass and defining land-to-ocean linkages. Glob Biogeochem Cycles 14:599–621.
Wang X, Murtugudde R, Hackert E, Maranon E. 2013. Phytoplankton carbon and chlorophyll distributions in the equatorial Pacific and Atlantic: a basin-scale comparative study. J Mar Syst 109–110:138–48.
Wassenaer T, Gerber P, Verburg PH et al. 2007. Projecting land use changes in the Neotropics: the geography of pasture expansion into forest. Glob Environ Chang 17:86–104.
Wijsman JWM, Middleburg JJ, Herman PMJ, Bottcher ME, Heip CHR. 2001. Sulfur and iron speciation in surface sediments along the northwester margin of the Black sea. Mar Chem 74:261–78.
Williams MR, Melack JM. 1997. Solute export from forested and partially deforested catchments in the central Amazon. Biogeochemistry 38:67–102.
Williams MR, Fisher TR, Melack JM. 1997. Chemical composition and deposition of rain in the central Amazon, Brazil. Atmosphere Environ 31:207–17.
Wolf S, Eugster W, Potvin C et al. 2011. Carbon sequestration potential of tropical pasture compared with afforestation in Panama. Glob Chang Biol 17:2763–80.
Wooller MJ, Behling H, Smallwood BJ, Fogel M. 2004. Mangrove ecosystem dynamics and elemental cycling at Twin Cays, Belize, during the Holocene. J Quat Sci 19:703–11.
Xiu P, Chai F. 2012. Spatial and temporal variability in phytoplankton carbon, chlorophyll, and nitrogen in the North Pacific. J Geophys Res 117:C11023. doi:10.1029/2012JC008067.
Yonekura Y, Ohta S, Kiyono Y, Aksa D, Morisada K, Tanaka N, Tayasu I. 2012. Dynamiocs of soil carbon following destruction of tropical rainforest and the subsequent establishment of Imperata grassland in Indonesian Borneo using stable carbon isotopes. Glob Chang Biol 18:2606–16.
Zhang L, Dawes WR, Walker GR. 2001. Response of mean annual evapotranspiration to vegetation changes at catchment scales. Water Resour Res 37:701–8.
Zhu MX, Shi XN, Yang GP, Hao XC. 2013. Formation and burial of pyrite and organic sulfur in mud sediments of the East China Sea inner shelf: constraints from solid-phase sulfur speciation and stable sulfur isotope. Cont Shelf Res 54:24–36.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author Contribution
IV and AG conceived the work, obtained the funding, drafted the paper, and guided all the field and analytic work; MB worked on statistical analyses and graphics; JT, CH, PM, and LC were involved in the field work and chemical analyses; MO did the mass spectrometry and elemental analysis; TS was responsible for the remote sensing work; all authors reviewed and edited the many versions of the text.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure SI 1
Sampling stations 1-9, occupied in sampling trips between 2009 to 2012, from Rio Limon, the watershed-estuary system with 92% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Quickbird imagery acquired April 5, 2003. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 9383 kb)
Figure SI 2
Sampling stations 1-9, occupied in sampling trips between 2009 to 2012, from Rio Manglarito, the watershed-estuary system with 91% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Quickbird imagery acquired April 5, 2003. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 8638 kb)
Figure SI 3
Sampling stations 1-9, occupied in sampling trips between 2009 to 2012, from Rio Pixvae, a watershed-estuary system with 73% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Quickbird imagery acquired April 5, 2003. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 8629 kb)
Figure SI 4
Sampling stations 1-9, occupied in sampling trips between 2009 to 2012, from Rio Luis, a watershed-estuary system with 73% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Quickbird imagery acquired April 5, 2003. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 9470 kb)
Figure SI 5
Sampling stations 1-9, occupied in sampling trips between 2009 to 2012, from Rio Chamuscado, the watershed-estuary system with 66% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Quickbird imagery acquired April 5, 2003. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 8940 kb)
Figure SI 6
Sampling stations 1-9, occupied in sampling trips during 2009 to 2012, from Rio de la Mona, the watershed-estuary system with 47% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Quickbird imagery acquired April 5, 2003. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 8658 kb)
Figure SI 7
Sampling stations 1-9, occupied in sampling trips between 2009 to 2012, from Rio Salmonete, the watershed-estuary system with 29% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Quickbird imagery acquired April 5, 2003. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 9430 kb)
Figure SI 8
Sampling stations 1-9, occupied during sampling trips between 2009 to 2012, from Rio Grande, the watershed-estuary system with 23% forest cover (Table 1). Sample locations varied to capture salinity gradient and exiting water; some points are overlapping, and do not show as separate samplings. Sampling points overlaid on Aster satellite imagery acquired February 7, 2006. Specific dates and latitude and longitude for each station are provided in Supplementary Information in Valiela and others (2013). (TIFF 5053 kb)
Figure SI 9
Comparisons of stable carbon and nitrogen isotopic signatures of suspended particulate matter (SPM) and potential sources, including sediments from mangrove estuaries, mangrove leaves, and large particulate matter (POM) collected from water ebbing the estuaries. The data shown stratified into three bins of % forest cover on the watersheds, and three bins of salinity. (TIFF 518 kb)
Rights and permissions
About this article
Cite this article
Valiela, I., Bartholomew, M., Giblin, A. et al. Watershed Deforestation and Down-Estuary Transformations Alter Sources, Transport, and Export of Suspended Particles in Panamanian Mangrove Estuaries. Ecosystems 17, 96–111 (2014). https://doi.org/10.1007/s10021-013-9709-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-013-9709-5