Abstract
Long-term responses of terrestrial ecosystems to the combined effects of warming and elevated CO2 (eCO2) will likely be regulated by N availability. The stock of soil N determines availability for organisms, but also influences loss to the atmosphere or groundwater. eCO2 and warming can elicit changes in soil N via direct effects on microbial and plant activity, or indirectly, via soil moisture. Detangling the interplay of direct- and moisture-mediated impacts on soil N and the role of organisms in controlling soil N will improve predictions of ecosystem-level responses. We followed individual soil N pools over two growing seasons in a semiarid temperate grassland, at the Prairie Heating and CO2 Enrichment experiment. We evaluated relationships of N pools with environmental factors and explored the role of plants by assessing plant biomass, plant N, and plant inputs to soil. We also assessed N forms in plots with and without vegetation to remove plant-mediated effects. Our study demonstrated that the effects of warming and eCO2 are highly dependent on individual N form and on year. In this water-constrained grassland, eCO2, warming and their combination appear to impact soil N pools through a complex combination of direct- and moisture-mediated effects. eCO2 decreased NO3 − but had neutral to positive effects on NH4 + and dissolved organic N (DON), particularly in a wet year. Warming increased NO3 − availability due to a combination of indirect drying and direct temperature-driven effects. Warming also increased DON only in vegetated plots, suggesting plant mediation. Our results suggest that impacts of combined eCO2 and warming are not always equivalent for plant and soil pools; although warming can help offset the decrease in NO3 − availability for plants under eCO2, the NO3 − pool in soil is mainly driven by the negative effects of eCO2.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Understanding the combined impacts of elevated atmospheric CO2 (eCO2) and warming on terrestrial ecosystems remains a major objective of global change studies (Pendall and others 2004; Heimann and Reichstein 2008). Long-term responses of terrestrial ecosystems to these global changes will likely be regulated by the responses of nitrogen (N) cycling and N availability (Luo and others 2006). N availability has been demonstrated to control responses of ecosystem productivity (Reich and others 2006; Langley and Megonigal 2010; Niu and others 2010), decomposition (Hu and others 2001), and carbon (C) sequestration (Rastetter and others 2005; Luo 2007; Hungate and others 2009) to global change factors.
The soil N stock is an important component of N availability. It determines the pools available for uptake by organisms as well as for loss to the atmosphere or groundwater. eCO2 and ecosystem warming can influence soil N pools via multiple processes. For example, eCO2 can decrease mineral N by increasing microbial immobilization (de Graaff and others 2006) or plant N uptake (Jin and Evans 2010) but it can also, concomitantly, increase N mineralization (Ebersberger and others 2003; Phillips and others 2011). Warming tends to enhance N turnover and availability in soil through direct impacts on microbial activity and mineralization (Rustad and others 2001; Melillo and others 2002), but it can decrease the mineral N pool through enhanced plant uptake (Larsen and others 2010). Moreover, soil N stocks and the processes regulating them are highly sensitive to soil water status which, in turn, can be strongly altered by both eCO2 and warming. eCO2 can modify plant–water relations and often increases soil water availability (Morgan and others 2004; Morgan and others 2011), whereas warming, with its desiccating effects, can have the opposite impact (Harte and others 1995; Dermody and others 2007). Understanding the interplay of direct- and moisture-mediated impacts of eCO2 and warming on N availability in real-world systems will improve predictive understanding of ecosystem-level responses.
Very few field experiments have addressed the combined effects of eCO2 and warming on N dynamics. In a temperate grassland Hovenden and others (2008) found that eCO2 decreased soil N availability but that warming ameliorated this effect; although the effect of CO2 was attributed to enhanced soil moisture, the effect of warming was ascribed to temperature stimulation of mineralization. In contrast, Larsen and others (2010) observed no effects of eCO2, warming or their combination on leached N in a temperate heathland. Combined impacts of eCO2 and warming on N availability are probably highly system-dependent. Nonetheless, it is likely that the effects of eCO2 and warming are more strongly mediated by soil moisture in systems where water availability constrains biological activity. For semiarid grasslands, models predict strong effects of eCO2 and warming on soil moisture which will likely impact N availability, in addition to microbial decomposition and plant productivity (Pepper and others 2005; Parton and others 2007).
Parton and others (2007) modeled the effects of eCO2 and warming in a semiarid temperate grassland in Wyoming, USA, at the Prairie Heating and CO2 Enrichment (PHACE) experiment, where the present study took place. They predicted that eCO2 would increase net soil N release due to increased mineralization with greater soil moisture and that warming could have positive or negative effects on N mineralization depending on water availability. In a field study, Dijkstra and others (2010) corroborated the importance of soil moisture on eCO2 effects at PHACE, but in contrast to the predictions, eCO2 was found to decrease the soil inorganic N pool. Despite its desiccating effects, warming did not affect microbial N uptake but it increased the mineral N pool and plant N uptake (Dijkstra and others 2010). The contrast between predicted and observed outcomes demonstrates the need for a closer understanding of the environmental controls on individual soil N pools in this grassland. Specifically, strong seasonal fluctuations in moisture and temperature are likely to influence the impacts of eCO2 and warming on plant and microbial driven processes responsible for the availability of different soil N forms (Beier and others 2008; Sardans and others 2008). However, Dijkstra and others’ (2010) results were based on one point-in-time (late spring) and seasonally integrated total mineral N measurements. Another key unknown is the relative importance of microbial and plant responses in determining how soil N pools will behave in a warmer, CO2 enriched atmosphere. For this, experimentally removing plant-mediated processes could provide useful insights.
Our goals were to investigate the interplay of direct- and moisture-mediated impacts of warming and eCO2 on soil N pools, and to explore the roles of plants and microbes in mediating those impacts. We examined the seasonal variation in the effects of free air CO2 enrichment, warming (with ceramic heaters), and their combination on soil N pools in a semiarid temperate grassland throughout two growing seasons. We followed individual pools of extractable nitrate (NO3 −), ammonium (NH4 +), and organic N (dissolved organic N, DON) in soil in intact native prairie and assessed the influence of soil moisture and temperature on these pools. To explore how plants mediated soil N pools, we assessed plant biomass, plant N, and plant-derived dissolved organic C (DOC). We monitored all N forms in plots with vegetation and without vegetation so as to remove plant-mediated effects.
Methods
Study Site
The PHACE experiment is located at the USDA-ARS High Plains Grassland Research Station (1930 m.a.s.l.), about 15 km west of Cheyenne, WY, USA (41°11′N, 104°54′W). The ecosystem is a northern mixed grass prairie with a plant community dominated by the C4 grass Bouteloua gracilis (H.B.K) Lag. and two C3 grasses, Hesperostipa comata Trin and Rupr. and Pascopyrum smithii (Rydb.). About 20% of the vegetation is composed of sedges and forbs. Annual precipitation is 384 mm, with approximately 60% falling during the growing season, which extends from late March through September. Mean air temperatures are 17.5°C in summer and −2.5°C in winter, with average maximum July temperature of 27°C. Soils at the experimental site are Mollisols (fine-loamy, mesic Aridic Argiustoll, mixed Ascalon and Altvan series), with a pH of 7.0. Average organic soil C content is 1.9% (SD = 0.27) at 0–5 cm and 1.5% (SD = 0.25) at 5–15 cm.
Experimental Setting and Environmental Monitoring
The experiment exposes plots to a full factorial combination of CO2 and temperature (ambient CO2 and ambient temperature: ct; ambient CO2 and elevated temperature: cT; elevated CO2 and ambient temperature: Ct; elevated CO2 and elevated temperature: CT) with five replications. It uses Free Air CO2 Enrichment technology (Miglietta and others 2001) installed in 3.4 m diameter rings to produce an atmosphere with a CO2 concentration of 600 ± 40 ppmv in the elevated CO2 treatments during the growing season. Infrared heaters (Kimball and others 2008) attached to a frame 1.5 m above the ground increase canopy temperature by 1.5°C during daytime and 3°C at night all year round. CO2 enrichment began at the start of the growing season (early April) in 2006 and heating began at the start of the growing season in 2007. To permanently remove plant effects, an approximately 0.4 m−2 area in each plot was sprayed with a broad spectrum systemic herbicide (glyphosate). Although glyphosate contains N, very small quantities were used, and previous research suggests that it does not increase N availability (Damin and others 2010). Prior to the first application, the selected area was isolated from the rest of the plot by burying metallic sheets down to 30 cm. Spraying took place in the first week of June 2008 and localized, minor applications were done occasionally over the rest of the growing season to kill any new or sprouting plants. Dead standing vegetation was left intact to avoid disturbance associated with hand pulling. During the following growing season (2009) there was minimal standing plant material and little plant emergence. Any new seedlings were carefully hand-pulled weekly to fortnightly.
In the intact portion of each experimental plot (referred to as native prairie) soil volumetric water content (5–15 cm depth) was monitored hourly with an Envirosmart sensor (Sentek Sensor Technologies, Stepney, Australia) during the entire year in 2008 and 2009 and with a 10 HS soil moisture sensor (0–15 cm) and associated Em50 data logger (Decagon Devices Inc., Pullman, WA, USA) starting in August of 2008. Soil water content was monitored independently in the portion treated with herbicide (referred to as plant-free plots) using a 10 HS sensor only. To obtain comparable measures of soil moisture content in both plant-free and native prairie plots for both growing seasons, we used the linear relationship (R 2 = 0.82; P < 0.0001) between measurements of the Sentek sensor and the 10 HS sensor in the native plot collected over a period of 450 days to convert the measurements of the 10 HS sensor in the plant-free plots. Soil temperature was measured hourly at 3 cm using thermocouples only in native plots.
Soil Sampling and Processing
Soils were sampled five times during each of the two growing seasons. Soil samples were obtained by collecting and compositing two 0–5 cm deep, 1.5 cm diameter cores from each of the native and plant-free plots. Collection in the native prairie plots started in the second week of April in 2008 and in the last week of June in plant-free plots—3 weeks after the first herbicide application, when all plants had died. The final collection in 2008 and 2009 occurred in the first week of October, when most of the vegetation had senesced. Soil samples were kept refrigerated and processed within 24 h. Soils were passed through a 2-mm sieve and further picked by hand to remove visible plant material and then shaken in 0.05 M K2SO4 (1:2 soil to solution ratio) for 1 h. Extracts were filtered through Whatman No. 1 paper and frozen until analysis. Subsamples were taken to determine gravimetric moisture content. Extracts were analyzed for NO3 − and NH4 + on a flow injection analyzer (QuickChem FIA+; Lachat Instruments, Milwaukee, WI, USA) and for total N on a Total Organic Carbon analyzer with an N measuring unit (Shimadzu TOC-VCPN; Shimadzu Scientific Instruments, Wood Dale, IL, USA). Concentrations of N in extracts were converted to g m−2 using bulk density values previously obtained for each plot. DON was determined by subtracting total mineral N (NO3 − and NH4 +) from total N (Melillo and others 2002).
Soil extracts from native plots were analyzed for DOC with a Total Organic Carbon analyzer (see above) after treating with 1 M H3PO4 (1 μl per 10 ml of extract) to remove carbonates. Extracts from samples collected at the biomass peak (late July in each year) in the eCO2 plots were further processed to determine their isotopic composition. As recent plant C, since the start of the eCO2 treatment, is depleted in 13C because of fumigation of the plots with fuel-derived CO2, it is possible to estimate the fraction of DOC derived from recent plant inputs in the eCO2 plots via a mass balance as:
where %DOCp is the fraction of DOC derived from recent plant inputs; δ13C DOC is the δ13C of the K2SO4 extract; δ13Cs is the δ13C of bulk soil and δ13Cp is the δ13C of the plant biomass. δ13C DOC was measured on a dried aliquot of the extract (60°C) from each plot. δ13Cs was obtained from bulk soil after carbonate removal as described in Carrillo and others (2011). δ13Cp was obtained from aboveground plant biomass samples (see below). δ13C of the aboveground plant biomass was chosen as the end-member for plant inputs over the root biomass, because roots in this system have an average turnover time of about 6 years (Milchunas and others 2005). The δ13Cp represents the C that has been fixed and translocated to the roots over the 2007, 2008, and 2009 growing seasons (thus termed recent plant inputs). All measurements of δ13C were done using an elemental analyzer connected to a mass spectrometer (20–20 Stable Isotope Analyzer; Europa Scientific, Cheshire, UK). The calculated fraction of recent plant-derived C was applied to the total pool of DOC at the respective collection time.
Plant Biomass and Plant N Content
In late July of each year, near the time of peak aboveground biomass in this system, and at the time of the third soil collection in the native prairie plots, standing aboveground plant biomass and fine root biomass were assessed. Aboveground biomass was obtained by clipping an area of 0.75 m2 in each plot. Biomass was dried at 60°C, weighed and ground to be analyzed for C, δ13C, and N using an elemental analyzer interfaced to mass spectrometer (Europa Scientific, Cheshire, UK). Fine roots (<1 mm) were handpicked from soil samples derived from three composited soil cores (15 cm depth; 3 cm diameter) and weighed.
Statistical Analyses
Repeated measures two-factor ANOVA was used to determine the effect of eCO2, warming and time on soil moisture and temperature. To assess treatment effects on the concentrations of N forms in each year, we used repeated measures split-plot two-factor ANOVA, with eCO2 and warming as whole plot factors, vegetation presence/absence as the split-plot and sampling time as the repeated measure. We subsequently performed analyses for plant-free and native plots separately. DOC in native plots was analyzed in each year with two-factor repeated measures ANOVA with eCO2 and warming as factors and time as the repeated measure. ANOVAs were performed with JMP (version 7.0; SAS Institute, Cary, N.C. USA) except for the split-plot repeated measures ANOVA which was carried out using PROC MIXED with heterogeneous autoregressive covariance structure in SAS (version 9.2; SAS Institute, Cary, N.C. USA). We evaluated relationships between N pools and moisture, temperature and plant biomass by selecting the function that best matched the behavior of the plotted data, and which explained the greatest variation (R 2 values) and was also statistically significant (P < 0.05) using the linear and nonlinear regressions tools in SigmaPlot (version 10.0 Systat Software Inc., San Jose, CA, USA). We evaluated these relationships using soil moisture and temperature means for 5, 10, 15, 20, and 30 days prior to soil collection. Although the different intervals led to qualitatively similar results, the 15-day (2 weeks) period provided the strongest relationships, indicating its relevance for the studied variables, and thus we present the values and relationships for this period. We used two-way ANOVA to evaluate treatment effects on aboveground biomass N pool. Significant effects and relationships are reported at P < 0.05 unless otherwise stated.
Results
Soil Moisture and Temperature
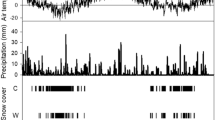
Average daily soil temperatures (3 cm depth) in the growing seasons were about 2.5°C in early April and reached an average maximum of 26°C in mid to late July (Figure 1A, B). By the end of the growing season temperatures declined to an average of 9°C. Warming increased soil temperature in native plots by 2.7°C on average in 2008 and by 1.8°C in 2009 (P < 0.0001 and P = 0.006, respectively). Temperature was not measured separately in plant-free plots during this study period. However, during the growing season of 2011 continuous measurements at 3 cm in a subset of plots indicated that native plots averaged 1°C warmer than plant-free plots in ambient and eCO2 treatments, and were 0.9°C cooler than plant-free plots in the warmed treatment (data not shown). In 2008, three periods of increasing soil moisture alternated with periods of decrease, including a protracted dry period in the middle of the growing season. In 2009 soil moisture was relatively high and stable until early July, after which it declined and remained low. Soil water content (5–15 cm depth) was more variable in native than plant-free plots mainly due to the accentuation of drying in vegetated plots, likely due to transpirational losses (Figure 1C, D). In native plots warming reduced soil water content by 2% v/v on average (P = 0.015 in 2008, P = 0.012 in 2009). The decrease in soil moisture with warming was smaller in plant-free plots. In 2008 water content decreased by 1.2% v/v. In 2009 warming decreased water content in the first half of the season (by 1% v/v on average) when water content was high, but the effect disappeared around mid-July, when soil moisture started decreasing (Warming × Time, P = 0.03). A greater decrease in water content with warming in native plots suggests that soil drying with warming was partly due to enhanced plant water uptake and/or transpiration. Soil water content was higher in native plots under eCO2 in both years. In 2008, water content was 2.3% v/v greater on average (P = 0.005) in eCO2 plots. In 2009, the magnitude of the increase was dependent on time (CO2 × Time, P = 0.03). As expected, there were no effects of elevated CO2 on soil moisture in plant-free plots (Figure 1E, F).
Soil temperature at 3 cm in soil under native prairie vegetation (A, B) and soil moisture at 5–15 cm in native prairie (C, D) and plant-free soil (E, F) in the 2008 and 2009 growing seasons. ct ambient CO2, unwarmed; cT ambient CO2, warmed; Ct elevated CO2, unwarmed; CT elevated CO2, warmed plot. Dotted lines indicate times when soil samples were collected for N analysis. Each data point represents a weekly average of hourly logged data for five replicate plots. Error bars indicate SE.
Extractable Inorganic and Organic Soil N Pools
The concentrations of NH4 +, NO3 −, and DON were highly dependent on the presence/absence of vegetation and on time (Figures 2, 3, 4; Table 1). Notably, the pool of NO3 − in plant-free plots was on average sixfold greater than that measured in native plots, a condition likely resulting from the absence of plant N uptake, but to which an after-effect of the method of vegetation removal could have contributed. eCO2 had significant effects on all N forms. In 2009 eCO2 consistently increased NH4 + in native plots (Figure 2). Although, no effects of eCO2 were expected in plant-free plots, we observed higher NH4 + on some sampling dates, which we interpret as a result of legacy effects of the plant residue following the herbicide application in 2008. eCO2 had a negative effect on NO3 − in native plots but not in plant-free plots (Figure 3; Table 1), and it increased DON in native plots in 2009 but did not have consistent effects in 2008 or in plant-free plots (Figure 4; Table 1). Warming did not have any clear effects on NH4 +, but it increased NO3 − in plant-free plots in both years (significant Warming × Vegetation interactions, Table 1; Figure 3), and it increased DON in 2008 in native plots (Table 1; Figure 4).
Pools of extractable NO3 − in 0–5 cm soil in native prairie and plant-free plots over the 2008 and 2009 growing seasons. ct ambient CO2, unwarmed; cT ambient CO2, warmed; Ct elevated CO2, unwarmed; CT elevated CO2, warmed plot. Data represent averages of five replicates. Error bars indicate SE. Note different Y axes scales in native and plant-free plots.
In plant-free plots, regardless of CO2 and warming treatments and across sampling times, the concentration of NO3 − was negatively related to soil water content and positively related to soil temperature (Figure 5A). The NH4 + pool was considerably less variable than NO3 − and showed a negative exponential relationship with moisture but no relationship with temperature (Figure 5B). DON was highest at intermediate soil water content but showed no relationship with temperature (Figure 5C). In native plots none of the three pools were related to temperature or moisture (data not shown). Plant biomass (assessed as the sum of aboveground and fine root biomass at the time of peak biomass in native plots) did not show a relationship with NO3 − in soil at the time of plant harvest in any of the years studied (data not shown). Both NH4 + and DON were positively correlated with plant biomass in 2009 (Figure 6), whereas no significant relationship was observed in 2008 (data not shown).
Relationships between soil moisture and temperature with the pools of extractable NH4 +, NO3 −, and dissolved organic N in plant-free soil. Each data point represents the average of 20 plots (all treatments and replicates) from each of the sampling dates in 2008 and 2009. Values of soil moisture and temperature are averages for the 15 days previous to soil collection. Error bars represent SE.
Relationships between plant biomass and the pool of NH4 + and dissolved organic N in 0–5 cm soil in native prairie plots in late July, near the peak of plant biomass in 2009. ct ambient CO2, unwarmed; cT ambient CO2, warmed; Ct elevated CO2, unwarmed; CT elevated CO2, warmed plots. Data points represent individual plots.
Plant N Pools
N pool sizes in 2008 were previously reported in Dijkstra and others (2010) and we present the 2009 data here (Table 2). Aboveground biomass N increased with warming in 2008 but not in 2009. eCO2 did not significantly impact the aboveground N pool in any of the years studied. N pools were overall greater in 2009 due to high productivity in this greater-than-average precipitation year (Morgan and others 2011).
Dissolved Organic C
DOC was lowest early in the growing seasons and tended to increase as the season progressed (Figure 7A, B). eCO2 increased the pools of DOC in 2009 whereas warming increased them in 2008 (Figure 7A, B). Based on isotopic partitioning of DOC at the biomass peak sampling date (late July) in the eCO2 plots, on average, approximately 30% of DOC was derived from recent plant C inputs. In 2008, there was a significant positive effect of warming on total DOC and recent plant-derived C (t test, P < 0.05) so that recent plant inputs amounted to 45% of the DOC (Figure 7A).
Pools of dissolved organic C in native prairie plots over the 2008 (A) and 2009 (B) growing seasons. ct ambient CO2, unwarmed; cT ambient CO2, warmed; Ct elevated CO2, unwarmed; CT elevated CO2, warmed plots. Bars represent the contributions of recent plant-derived C to the total DOC pool in late July at the peak biomass sampling date in each year (based on C isotopic partitioning in plots with elevated CO2). Data represent averages of five replicates. Error bars indicate SE.
Discussion
Our study demonstrated strong effects of both warming and eCO2 on N pools, but no interactions between these global changes. Warming and CO2 effects were highly dependent on the individual N form studied. The responses of soil N pools to eCO2, warming and their combination resulted from a combination of direct (microbial and plant-mediated) and indirect (moisture-mediated) effects. These results suggest that although biomass responses in this water-constrained ecosystem are primarily mediated by moisture availability (Morgan and others 2011), biogeochemical responses are more complex.
eCO2 Effects
We observed that eCO2 decreased soil NO3 − throughout the growing season—although it had neutral to positive effects on NH4 + (see below). eCO2 could have decreased soil NO3 − via increased plant N uptake (Jin and Evans 2010; Larsen and others 2010). Community aggregated plant-shoot N pools measured at the time of peak biomass are a good proxy for ecosystem level plant N uptake in this system as peak biomass represents the great majority of net annual primary productivity (Morgan and others 2011). Plant-shoot N pools in 2008 (Dijkstra and others 2010) and in 2009 (this study, Table 2) did not increase with eCO2, suggesting that greater plant N uptake was not the sole factor driving decreased soil NO3 − and that microbially driven processes may have played a role in this response. We found consistently smaller NO3 − pools under wetter conditions (Figure 5A), suggesting that increased soil moisture with eCO2 could have contributed to lower NO3 − concentrations. Increased microbial N immobilization under eCO2 (Barnard and others 2006a; Lagomarsino and others 2008; Bloor and others 2009) could have resulted from increased soil water content with eCO2 enhancing microbial activity and N demand. Consistent with this, in a one point-in-time 15N assay at PHACE, eCO2 increased microbial NO3 − immobilization (Dijkstra and others 2010). Another factor that could have contributed to the decrease in the NO3 − pool with greater water content under eCO2 is a decrease in nitrification due to less aerobic conditions with greater water content, which has been found in several studies (Niklaus and others 2001; Barnard and others 2006b; Lagomarsino and others 2008; Rutting and others 2010). Although higher water availability with eCO2 can also increase leaching and denitrification (Korner and Arnone 1992; Baggs and others 2003), the magnitude of those processes in this type of grassland is probably small relative to the observed decrease in NO3 − (Mosier and others 2008; Xu and others 2008). N2O emissions were less than 2 mg N m−2 during the growing season at the PHACE experiment (Dijkstra and others, unpublished), suggesting that the N cycle in general is rather tight at this site with little loss to the atmosphere.
In 2009 eCO2 consistently increased both DON and NH4 + concentrations in native plots. Although we did not measure NH4 + production directly, an increase in NH4 + is consistent with an increase in gross N mineralization observed in other grasslands (Hungate and others 1997; Ebersberger and others 2003; Rutting and others 2010). The fact that DON, partly a product of decomposition of SOM and a source of NH4 + (Schimel and Bennett 2004) increased concomitantly with NH4 + is also in accord with the possibility that eCO2 increased production of NH4 +. Increased gross N mineralization with eCO2 has been attributed to increased soil moisture (Hungate and others 1997; Ebersberger and others 2003; Dijkstra and others 2008), which we observed under eCO2. Greater plant C inputs to soil under eCO2 increasing the mining of SOM for N by microbes have also been associated with increased gross N mineralization (de Graaff and others 2009; Rutting and others 2010). In 2009 eCO2 increased DOC (composed of ca. 30% recent plant inputs, Figure 7B) and there were positive relationships of both NH4 + and DON with plant biomass, which is a proxy for C input to soil in this system (Carrillo and others 2011). Thus, either enhanced moisture or increased plant C inputs or a combination of both may be responsible for the increases in NH4 + and DON with eCO2,
Parton and others (2007) predicted that eCO2 would increase net release of N in soil at PHACE. Our study suggests that the effects of eCO2 on N in soil were highly dependent on the N form studied and on the year. In both years, and in contrast to predictions, eCO2 had a negative effect on the NO3 − pool. However, eCO2 increased NH4 + and, as consequence, total mineral soil N in 2009, according to predictions. Yet, the increase in NH4 + occurred only in the year with substantially higher than average precipitation (453 mm in 2009; 375 mm in 2008; 384 mm historic average (NOAA 1994)), suggesting that the predicted increase in N may only occur in wet years. This poses the important question of how dependent the effect of eCO2 on N pools will be on precipitation regimes.
Warming Effects
Warming commonly increases accumulation of soil mineral N (Rustad and others 2001). However, despite the positive and consistent effects of warming on NO3 − in plant-free plots, in native plots NO3 − only increased with warming in one of the ten sampling times. However, aboveground N pools were greater in warmed plots in 2008 (Dijkstra and others 2010) and greater under warming + eCO2 compared to eCO2 plots in 2009 (Table 2) which suggests that the extra soil NO3 − generated by warming was not present in the soil in native plots because it was taken up by plants. We hypothesize that if the vegetation in this grassland is able to take up the extra NO3 − produced under warming across most of the growing season, as our results suggest, increased NO3 − losses to leaching or denitrification associated with warming (Ineson and others 1998; Schmidt and others 2004; Patil and others 2010), may not be important. The consistent increase in NO3 − in both years observed in the absence of plants could have resulted, at least in part, from the drying effect of warming reducing microbial NO3 − immobilization (Allison and Treseder 2008). The strong negative relationship between NO3 − and soil water content in plant-free plots (Figure 5A), supports this hypothesis. The overall positive relationship we found between NO3 − and soil temperature suggests that a direct effect of warming on soil temperature (Figure 5B) may also have contributed to NO3 − increase. Several studies have attributed higher NO3 − concentrations to temperature-driven increases in nitrification (Wang and others 2006; Sardans and others 2008; Malchair and others 2010).
Our results suggest that in this semiarid grassland, warming-induced drying is more important than direct effects of temperature in determining responses of NH4 + to warming. Gross N mineralization tends to increase with temperature (Peterjohn and others 1994; Rustad and others 2001) and thus NH4 + would also be expected to increase with warming. However, NH4 + did not show a consistent response to the warming treatment or an overall relationship with temperature in either plant-free or native plots, suggesting no direct temperature effects. This is consistent with observations by Hovenden and others (2008) in a Tasmanian grassland. Rather, the exponential decrease of NH4 + with soil moisture (Figure 5B), suggests that the NH4 + pool can increase with soil drying during periods of low moisture content (for example, July 2008, Figures 1C and 2).
Notably, warming had a strong positive effect on DON concentrations in 2008, but only in vegetated plots, thus suggesting plant mediation of this response. It is unclear if the increase in DON was due to an increase in SOM decomposition or to greater root inputs and/or turnover. However, DOC concentrations also increased with warming in 2008 and this was due to a greater amount of recent plant-derived C being incorporated into DOC (Figure 7A). This indicates that greater plant inputs and/or turnover with warming (Fitter and others 1999; Wan and others 2005) could have contributed to increased DON pools.
Combined Effects of eCO2 and Warming
When warming and eCO2 treatments were combined (CT treatment), the effects observed on both NH4 + and DON were consistent with their effects as single factors (cT and Ct, respectively). However, for NO3 −, the combination of eCO2 and warming in native prairie plots produced soil concentrations that were not different from those under eCO2 alone, which were lower than under ambient conditions. Yet, we determined that warming consistently increased NO3 − concentrations in plant-free plots. Together, these observations suggest that in the native plots the positive effect of warming under eCO2 was not manifested in the soil pool because the extra NO3 − generated was utilized by plants. Plant N pools are consistent with this interpretation, as under eCO2 + warming shoot N pools in 2008 and 2009 were equivalent to those under the control treatment, even though they were lower under eCO2 alone. Hence, our results suggest that impacts of combined eCO2 and warming are not always equivalent for plant and soil pools, as even though warming can offset the decrease in NO3 − availability for plants grown under eCO2, the response of NO3 − concentrations in soil can still be dominated by the negative effect of eCO2.
In summary, despite the high variability in moisture status across the growing season, we found that in this grassland the effects of eCO2 on the pools of extractable N forms were mostly consistent over time. We found that the responses of soil pools to eCO2 were highly contrasting for different N forms. eCO2 had neutral to positive effects on DON and NH4 +. The latter occurred only in a wetter than average year and were likely due to a combination of greater soil moisture and greater plant inputs under eCO2. In contrast, eCO2 consistently decreased NO3 − possibly due to moisture-mediated effects on microbial activity. We conclude that the effect of eCO2 on the total pool of soil mineral N is dependent on the balance between the contrasting responses of NO3 − and NH4 +; under average precipitation conditions the eCO2 effect will tend to be dominated by a negative effect on NO3 −, but in wet conditions by positive effects on NH4 +. Warming increased DON and our results suggested that plants mediated this response. We found that warming increased NO3 − likely due to a combination of indirect drying impacts and direct temperature-driven effects. Finally, our results suggest that impacts of combined eCO2 and warming are not always equivalent for plant and soil pools; whereas warming can help offset the decrease in NO3 − availability for plants under eCO2, the NO3 − pool in soil is mainly driven by the negative effects of eCO2.
References
Allison SD, Treseder KK. 2008. Warming and drying suppress microbial activity and carbon cycling in boreal forest soils. Glob Change Biol 14:2898–909.
Baggs EM, Richter M, Cadisch G, Hartwig UA. 2003. Denitrification in grass swards is increased under elevated atmospheric CO2. Soil Biol Biochem 35:729–32.
Barnard R, Barthes L, Leadley PW. 2006a. Short-term uptake of N-15 by a grass and soil micro-organisms after long-term exposure to elevated CO2. Plant Soil 280:91–9.
Barnard R, Le Roux X, Hungate BA, Cleland EE, Blankinship JC, Barthes L, Leadley PW. 2006b. Several components of global change alter nitrifying and denitrifying activities in an annual grassland. Funct Ecol 20:557–64.
Beier C, Emmett BA, Penuelas J, Schmidt IK, Tietema A, Estiarte M, Gundersen P, Llorens L, Riis-Nielsen T, Sowerby A, Gorissen A. 2008. Carbon and nitrogen cycles in European ecosystems respond differently to global warming. Sci Total Environ 407:692–7.
Bloor JMG, Niboyet A, Leadley PW, Barthes L. 2009. CO2 and inorganic N supply modify competition for N between co-occurring grass plants, tree seedlings and soil microorganisms. Soil Biol Biochem 41:544–52.
Carrillo Y, Pendall E, Dijkstra FA, Morgan JA, Newcomb JM. 2011. Response of soil organic matter pools to elevated CO2 and warming in a semi-arid grassland. Plant Soil 347:339–50.
Damin V, Trivelin PCO, Franco HCJ, Barbosa TG. 2010. Nitrogen 15N loss in the soil-plant system after herbicide application on Pennisetum glaucum. Plant Soil 328:245–52.
de Graaff MA, van Groenigen KJ, Six J, Hungate B, van Kessel C. 2006. Interactions between plant growth and soil nutrient cycling under elevated CO2: a meta-analysis. Glob Change Biol 12:2077–91.
de Graaff MA, Van Kessel C, Six J. 2009. Rhizodeposition-induced decomposition increases N availability to wild and cultivated wheat genotypes under elevated CO2. Soil Biol Biochem 41:1094–103.
Dermody O, Weltzin JF, Engel EC, Allen P, Norby RJ. 2007. How do elevated CO2, warming, and reduced precipitation interact to affect soil moisture and LAI in an old field ecosystem? Plant Soil 301:255–66.
Dijkstra FA, Pendall E, Mosier AR, King JY, Milchunas DG, Morgan JA. 2008. Long-term enhancement of N availability and plant growth under elevated CO2 in a semi-arid grassland. Funct Ecol 22:975–82.
Dijkstra FA, Blumenthal D, Morgan JA, Pendall E, Carrillo Y, Follett RF. 2010. Contrasting effects of elevated CO2 and warming on nitrogen cycling in a semiarid grassland. New Phytol 187:426–37.
Ebersberger D, Niklaus PA, Kandeler E. 2003. Long term CO2 enrichment stimulates N-mineralisation and enzyme activities in calcareous grassland. Soil Biol Biochem 35:965–72.
Fitter AH, Self GK, Brown TK, Bogie DS, Graves JD, Benham D, Ineson P. 1999. Root production and turnover in an upland grassland subjected to artificial soil warming respond to radiation flux and nutrients, not temperature. Oecologia 120:575–81.
Harte J, Torn MS, Chang FR, Feifarek B, Kinzig AP, Shaw R, Shen K. 1995. Global warming and soil microclimate -results from a meadow-warming experiment. Ecol Appl 5:132–50.
Heimann M, Reichstein M. 2008. Terrestrial ecosystem carbon dynamics and climate feedbacks. Nature 451:289–92.
Hovenden MJ, Newton PCD, Carran RA, Theobald P, Wills KE, Schoor JKV, Williams AL, Osanai Y. 2008. Warming prevents the elevated CO2-induced reduction in available soil nitrogen in a temperate, perennial grassland. Glob Change Biol 14:1018–24.
Hu S, Chapin FS, Firestone MK, Field CB, Chiariello NR. 2001. Nitrogen limitation of microbial decomposition in a grassland under elevated CO2. Nature 409:188–91.
Hungate BA, Chapin FS, Zhong H, Holland EA, Field CB. 1997. Stimulation of grassland nitrogen cycling under carbon dioxide enrichment. Oecologia 109:149–53.
Hungate BA, van Groenigen KJ, Six J, Jastrow JD, Lue YQ, de Graaff MA, van Kessel C, Osenberg CW. 2009. Assessing the effect of elevated carbon dioxide on soil carbon: a comparison of four meta-analyses. Glob Change Biol 15:2020–34.
Ineson P, Coward PA, Hartwig UA. 1998. Soil gas fluxes of N2O, CH4 and CO2 beneath Lolium perenne under elevated CO2: The Swiss free air carbon dioxide enrichment experiment. Plant Soil 198:89–95.
Jin VL, Evans RD. 2010. Elevated CO2 increases plant uptake of organic and inorganic N in the desert shrub Larrea tridentata. Oecologia 163:257–66.
Kimball BA, Conley MM, Wang S, Lin X, Luo C, Morgan J, Smith D. 2008. Infrared heater arrays for warming ecosystem field plots. Glob Change Biol 14:309–20.
Korner C, Arnone JA. 1992. Responses to elevated carbon-dioxide in artificial tropical ecosystems. Science 257:1672–5.
Lagomarsino A, Moscatelli MC, Hoosbeek MR, De Angelis P, Grego S. 2008. Assessment of soil nitrogen and phosphorous availability under elevated CO2 and N-fertilization in a short rotation poplar plantation. Plant Soil 308:131–47.
Langley JA, Megonigal JP. 2010. Ecosystem response to elevated CO2 levels limited by nitrogen-induced plant species shift. Nature 466:96–9.
Larsen KS, Andresen LC, Beier C, Jonasson S, Albert KR, Ambus PER, Arndal MF, Carter MS, Christensen S, Holmstrup M, Ibrom A, Kongstad J, Van Der Linden L, Maraldo K, Michelsen A, Mikkelsen TN, Pilegaard KIM, PriemÉ A, Ro-Poulsen H, Schmidt IK, Selsted MB, Stevnbak K. 2010. Reduced N cycling in response to elevated CO2, warming, and drought in a Danish heathland: synthesizing results of the CLIMAITE project after two years of treatments. Glob Change Biol 17:1884–99.
Luo YQ. 2007. Terrestrial carbon-cycle feedback to climate warming. Annu Rev Ecol Evol Syst 38:683–712.
Luo YQ, Hui DF, Zhang DQ. 2006. Elevated CO2 stimulates net accumulations of carbon and nitrogen in land ecosystems: a meta-analysis. Ecology 87:53–63.
Malchair S, De Boeck HJ, Lemmens C, Merckx R, Nijs I, Ceulemans R, Carnol M. 2010. Do climate warming and plant species richness affect potential nitrification, basal respiration and ammonia-oxidizing bacteria in experimental grasslands? Soil Biol Biochem 42:1944–51.
Melillo JM, Steudler PA, Aber JD, Newkirk K, Lux H, Bowles FP, Catricala C, Magill A, Ahrens T, Morrisseau S. 2002. Soil warming and carbon-cycle feedbacks to the climate system. Science 298:2173–6.
Miglietta F, Hoosbeek MR, Foot J, Gigon F, Hassinen A, Heijmans M, Peressotti A, Saarinen T, van Breemen N, Wallen B. 2001. Spatial and temporal performance of the MiniFACE (Free Air CO2 Enrichment) system on bog ecosystems in northern and central Europe. Environ Monit Assess 66:107–27.
Milchunas DG, Morgan JA, Mosier AR, LeCain DR. 2005. Root dynamics and demography in shortgrass steppe under elevated CO2, and comments on minirhizotron methodology. Glob Change Biol 11:1837–55.
Morgan JA, Pataki DE, Korner C, Clark H, Del Grosso SJ, Grunzweig JM, Knapp AK, Mosier AR, Newton PCD, Niklaus PA, Nippert JB, Nowak RS, Parton WJ, Polley HW, Shaw MR. 2004. Water relations in grassland and desert ecosystems exposed to elevated atmospheric CO2. Oecologia 140:11–25.
Morgan JA, LeCain DR, Pendall E, Blumenthal DM, Kimball BA, Carrillo Y, Williams DG, Heisler-White J, Dijkstra FA, West M. 2011. C4 grasses prosper as carbon dioxide eliminates desiccation in warmed semi-arid grassland. Nature 476:202–5.
Mosier AR, Parton WJ, Martin RE, Valentine DW, Ojima DS, Schimel DS, Burke IC, Adair EC, Del Grosso SJ. 2008. Soil-atmosphere exchange of trace gases in the Colorado shortgrass steppe. In: Lauenroth WK, Burke IC, Eds. Ecology of the shortgrass steppe: a long-term perspective. New York: Oxford University Press. p 342–72.
Niklaus PA, Kandeler E, Leadley PW, Schmid B, Tscherko D, Korner C. 2001. A link between plant diversity, elevated CO2 and soil nitrate. Oecologia 127:540–8.
Niu SL, Sherry RA, Zhou XH, Wan SQ, Luo YQ. 2010. Nitrogen regulation of the climate-carbon feedback: evidence from a long-term global change experiment. Ecology 91:3261–73.
NOAA. 1994. Local climatological data, Cheyenne, Wyoming. National Climate Data Center, Ashville, NC.
Parton WJ, Morgan JA, Wang GM, Del Grosso S. 2007. Projected ecosystem impact of the Prairie Heating and CO2 Enrichment experiment. New Phytol 174:823–34.
Patil RH, Laegdsmand M, Olesen JE, Porter JR. 2010. Effect of soil warming and rainfall patterns on soil N cycling in Northern Europe. Agric Ecosyst Environ 139:195–205.
Pendall E, Bridgham S, Hanson PJ, Hungate B, Kicklighter DW, Johnson DW, Law BE, Luo YQ, Megonigal JP, Olsrud M, Ryan MG, Wan SQ. 2004. Below-ground process responses to elevated CO2 and temperature: a discussion of observations, measurement methods, and models. New Phytol 162:311–22.
Pepper DA, Del Grosso SJ, McMurtrie RE, Parton WJ. 2005. Simulated carbon sink response of shortgrass steppe, tallgrass prairie and forest ecosystems to rising [CO2], temperature and nitrogen input. Global Biogeochem Cycles 19:GB1004.
Peterjohn WT, Melillo JM, Steudler PA, Newkirk KM, Bowles FP, Aber JD. 1994. Responses of trace gas fluxes and N availability to experimentally elevated soil temperatures. Ecol Appl 4:617–25.
Phillips RP, Finzi AC, Bernhardt ES. 2011. Enhanced root exudation induces microbial feedbacks to N cycling in a pine forest under long-term CO2 fumigation. Ecol Lett 14:187–94.
Rastetter EB, Perakis SS, Shaver GR, Agren GI. 2005. Terrestrial C sequestration at elevated-CO2 and temperature: the role of dissolved organic N loss. Ecol Appl 15:71–86.
Reich PB, Hobbie SE, Lee T, Ellsworth DS, West JB, Tilman D, Knops JMH, Naeem S, Trost J. 2006. Nitrogen limitation constrains sustainability of ecosystem response to CO2. Nature 440:922–5.
Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AE, Cornelissen JHC, Gurevitch J. 2001. A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–62.
Rutting T, Clough TJ, Muller C, Lieffering M, Newton PCD. 2010. Ten years of elevated atmospheric carbon dioxide alters soil nitrogen transformations in a sheep-grazed pasture. Glob Change Biol 16:2530–42.
Sardans J, Penuelas J, Estiarte M, Prieto P. 2008. Warming and drought alter C and N concentration, allocation and accumulation in a Mediterranean shrubland. Glob Change Biol 14:2304–16.
Schimel JP, Bennett J. 2004. Nitrogen mineralization: challenges of a changing paradigm. Ecology 85:591–602.
Schmidt IK, Tietema A, Williams D, Gundersen P, Beier C, Emmett BA, Estiarte M. 2004. Soil solution chemistry and element fluxes in three European heathlands and their responses to warming and drought. Ecosystems 7:638–49.
Wan SQ, Hui DF, Wallace L, Luo YQ. 2005. Direct and indirect effects of experimental warming on ecosystem carbon processes in a tallgrass prairie. Global Biogeochemical Cycles 19:GB2014.
Wang CH, Wan SQ, Xing XR, Zhang L, Han XG. 2006. Temperature and soil moisture interactively affected soil net N mineralization in temperate grassland in Northern China. Soil Biol Biochem 38:1101–10.
Xu YQ, Wan SQ, Cheng WX, Li LH. 2008. Impacts of grazing intensity on denitrification and N2O production in a semi-arid grassland ecosystem. Biogeochemistry 88:103–15.
Acknowledgments
We thank Dan LeCain, David Smith, Erik Hardy, and Matthew Parsons for their technical assistance and Joanne Newcomb, Megan Steinweg, Hannah Munn, Courtney Ellis, Christine Rumsey, and Jennifer Bell, for assistance in the field and in the laboratory. This project was supported by a USDA-CSREES Soil Processes Program (Grant no. 2008-35107-18655), by the US Department of Energy’s Office of Science (BER) through the Terrestrial Ecosystem Science program and the Western Regional Center of the National Institute for Climatic Change Research at Northern Arizona University, by NSF (DEB# 1021559), and by the Australian Research Council (FT100100779).
Author information
Authors and Affiliations
Corresponding author
Additional information
Author Contributions
YC, FAD, EP, JAM, DMB designed the study and performed the research. YC analyzed the data and wrote the paper with contributions from FAD, EP, JAM, and DMB.
Rights and permissions
About this article
Cite this article
Carrillo, Y., Dijkstra, F.A., Pendall, E. et al. Controls over Soil Nitrogen Pools in a Semiarid Grassland Under Elevated CO2 and Warming. Ecosystems 15, 761–774 (2012). https://doi.org/10.1007/s10021-012-9544-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-012-9544-0