Abstract
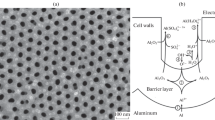
The present work was conducted to explore the growth mechanism of anodic oxide films on pure aluminium in aqueous acidic and alkaline solutions by using a.c. impedance spectroscopy and a beam deflection technique. From the analyses of a.c impedance data, it was found that the reciprocal capacitance of anodic oxide film on pure aluminium increased linearly with increasing film formation potential in both acidic and alkaline solutions, indicating a linear increase in the film thickness with film formation potential. However, as the film formation potential increased, the resistance of anodic oxide film decreased in acidic solution, while it increased in alkaline solution. From the measurements of the deflection, the deflection was observed to move towards only a compressive direction with time in acidic solution, but it showed a transition in the direction of movement from compressive to tensile in alkaline solution. Based upon the above experimental results, it is suggested that the movement of oxygen vacancy through the oxide film contributes to the growth of anodic oxide film on pure aluminium in acidic solution, but the movement of both aluminium vacancy and oxygen vacancy accounts for that oxide film growth in alkaline solution.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 12 August 1997 / Accepted: 9 October 1997
Rights and permissions
About this article
Cite this article
Moon, SM., Pyun Su-II, . Growth mechanism of anodic oxide films on pure aluminium in aqueous acidic and alkaline solutions. J Solid State Electrochem 2, 156–161 (1998). https://doi.org/10.1007/s100080050081
Issue Date:
DOI: https://doi.org/10.1007/s100080050081