Abstract.
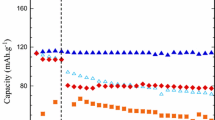
The effect of LiBF4 on the low-temperature performance of a Li-ion cell was studied by using a 1:1:1 (wt) EC/DMC/DEC mixed solvent. The results show that the LiBF4-based electrolyte has a 2- to 3-fold lower ionic conductivity and shows rather higher freezing temperature compared with a LiPF6-based electrolyte. Owing to electrolyte freezing, cycling performance of the Li-ion cell using LiBF4 was significantly decreased when the temperature fell below –20 °C. However, impedance data show that at –20 °C the LiBF4 cell has lower charge-transfer resistance than the LiPF6 cell. In spite of the relatively lower conductivity of the LiBF4-based electrolyte, the cell based on it shows slightly lower polarization and higher capacity in the liquid temperature range (above –20 °C) of the electrolyte. This fact reveals that ionic conductivity of the electrolytes is not a limitation to the low-temperature performance of the Li-ion cell. Therefore, LiBF4 may be a good salt for the low-temperature electrolyte of a Li-ion cell if a solvent system that is of low freezing temperature, high solubility to LiBF4, and good compatibility with a graphite anode can be formulated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Zhang, .S., Xu, .K. & Jow, .T. Low-temperature performance of Li-ion cells with a LiBF4-based electrolyte. J Solid State Electrochem 7, 147–151 (2003). https://doi.org/10.1007/s10008-002-0300-9
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10008-002-0300-9