Abstract
Purpose
Hemangiomas are benign vascular anomalies characterized by benign proliferation of blood vessels. There are no well-defined criteria for the diagnosis and treatment of oral capillary hemangioma (OCH). The objective of this study is to report a safe, effective, and low-cost protocol for diagnosis and treatment of OCH.
Materials and methods
Eight patients were diagnosed with OCH, through two physical examination maneuvers—the diascopy and head lowering maneuver with abdominal compression (HLMAC). The treatment performed was sclerotherapy with ethanolamine oleate, weekly, until the disappearance of the lesion. No local anesthetics were administered prior to the sclerosing agent injection.
Results
All cases showed a complete remission of the lesions after sclerotherapy. There were no visible scars or compromise esthetics or normal function, and there was no evidence of recurrence. Only one patient had superficial ulceration, which remained asymptomatic and healed without specific treatment.
Conclusions
This protocol was effective in the diagnosis of OCH through two physical examination maneuvers with low-cost and easy implementation, the diascopy and the HLMAC. The low concentration associated with a reduced amount of the sclerosing agent was responsible for the excellent results in the cases treated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The hemangioma is a vascular anomaly characterized by benign proliferation of blood vessels and is rated by many authors as a benign vascular neoplasm [1–12]. Hemangioma is a clinical term to designate a benign vascular neoplasm or vascular malformation of endothelial origin. They may be classified by the type of blood vessels involved in capillary, juvenile, cavernous, and arteriovenous hemangioma [4]. The most common type is the capillary hemangioma, and it occurs as a small localized lesion and has the less-aggressive clinical behavior. Moreover, cavernous hemangioma is the most aggressive type of hemangiomas and generates large and infiltrated lesions [12].
The etiology of hemangioma has not been fully elucidated, but two hypotheses are more accepted. The first suggests that hemangiomas originate from placental trophoblast embolism, which originates from a household embryonic cell called trophectoderm. This theory is based on similarities at the expression of endothelial markers and transcriptomes between hemangiomas and placental trophoblasts. Trophoblasts have high angiogenic potential and promote angiogenesis in the placenta through the production of angiogenic and vasoactive substances [13].
The second theory is related to endothelial progenitor cells, which have a potential to clonally duplicate in a precise milieu of cytokines and estrogen [13]. These cells are present in the bloodstream and have great ability to differentiate and proliferate. The endothelial progenitor cells not only play an important role in the repair of damaged blood vessels but also are associated with vascular pathologies [14].
Hemangiomas have a predilection for the head and neck [4], although they may be present in one or more body parts, such as limbs, bladder, genital area, and the central nervous system [15]. They are more common in women and children, being considered the most common tumor in newborns [5]. In some cases, the lesions tend to be self-limiting and may resolve spontaneously without any therapeutic intervention [9].
In specific situations, hemangiomas can become symptomatic and may need therapeutic intervention, for example, in the presence of ulcers, pain, bleeding, secondary infections, and tissue deformations [1].
The diagnosis of hemangiomas, in most cases, can be achieved by clinical examination of the lesion [3]. The treatment may be carried out without requiring prior diagnostic confirmation, such as histological and imaging examination [7, 16, 17]. However, hemangiomas may present clinical features similar to those of other oral lesions such as racial melanosis, oral melanoma, melanosis induced by tobacco and drugs [18], retention cysts, mucoceles, Kaposi's sarcoma, and pyogenic granuloma [3]. Due to the similarity of clinical findings, diagnostic errors may occur. For this reason, other diagnostic methods should be done besides clinical appearance, because the correct choice of the therapy lies in the accurate diagnosis of the lesion [3].
If there is a need for treatment, a wide variety of therapeutic methods can be used, of which one can refer the surgical excision, sclerotherapy, surgery associated with sclerotherapy, embolization, laser therapy, cryotherapy, and corticosteroid therapy [7, 11]. Each method has advantages and disadvantages, and so far, there is no treatment that is widely accepted and is considered the gold standard [19, 20].
This paper aims to present a simple, safe, low-cost, and minimally invasive protocol for the treatment of oral capillary hemangiomas (OCH), ranging from diagnosis to treatment, using physical examination maneuvers and a treatment with sclerotherapy. This protocol was easy to implement and was used effectively in eight cases OCH. The diagnosis was established by clinical examination associated with diascopy and head lowering maneuver with abdominal compression (HLMAC). The treatment protocol followed a sclerotherapy protocol of ethanolamine oleate using a 1:4 dilution with distilled water.
Materials and methods
This was a retrospective study that included eight patients from the Department of Oral Pathology and Oral and Maxillofacial Surgery, School of Dentistry, University Center of Pará, who underwent to the same diagnostic criteria that included anamnesis, physical examination of the lesion, and two maneuvers: diascopy and HLMAC. These two maneuvers were performed to observe the physical changes which occurred in the lesion after its enforcement. This study was approved by the ethics committee of the Institute of Health Sciences at University Center of Pará (number 184.068).
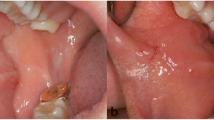
The diascopy was performed with a glass slide used in the laboratory for histological study. The mucosa was dried, applied with a gradually increasing pressure, and was held with the thumb on the glass sheet on the hemangioma. Changes in volume and staining were observed. A significant reduction on the intensity of the coloration or discoloration by total lesion was considered positive. After the diagnostic procedure, retention of shape, size, and color intensity was considered negative (Fig. 1).
Diascopy demonstration. OCH located at the lower lip (a). Positioning of the glass slide on the lesion (b). A finger pressure is exerted on the slide. As the pressure increases, the lesion decreases in size and staining intensity (c) until the complete disappearance thereof (d). After removal of the slide, the OCH returns to its initial shape and staining (e). Clinical appearance of the lip after 3 weeks and two sessions of sclerotherapy. The hemangioma disappeared, and mucosa shows characteristics of normality (f)
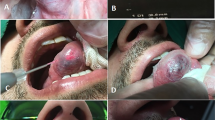
The HLMAC was performed in the dental chair that was adjusted to an angle of approximately 90°. The lesion was inspected and then the patient was oriented to lower his head as much as possible toward his legs and squeeze his abdomen. This position was maintained for about 60 s, and the lesion was inspected again. An increase in volume and/or staining intensity of the lesion was considered positive. If the lesion did not suffer changes and its original characteristics were preserved, the result of the maneuver was considered negative (Fig. 2).
HLMAC demonstration. OCH located in the cheek mucosa (a). The dental chair was positioned at an angle close to 90°, and the patient was instructed to lower his head toward his legs, compressing the abdomen—HLMAC (b). After the maneuver, the lesion showed a slight increase in volume and an obvious color change from light red to purplish (c). Clinical appearance in the cheek mucosa after 3 weeks and two sessions of sclerotherapy (d)
After having the clinical diagnosis of OCH, the patients were submitted to sessions of sclerotherapy with ethanolamine oleate (50 mg/ml). Each session consisted of a single injection of the solution with a total volume variable according to the size of the lesion. The solution of ethanolamine oleate was diluted 1:4 in distilled water without previous anesthesia. The procedure of sclerotherapy was performed weekly until the clinical disappearance of the lesion. After clinical remission of the lesions, the patients were followed up for a total period of 12 months, with scheduled returns on the 1st, 3rd, 6th, and 12th month.
Results
All patients presented remission of the lesion, and no prior local anesthesia was needed. The patients were subjected to sclerotherapy sessions weekly, requiring one to three sessions for clinical remission. Most patients were treated with one or two sessions; only one patient needed a third session, who presented the largest lesion (18 mm). Table 1 shows the characteristics of the treatment for each patient.
Some patients reported a burning sensation at the site during administration. Analgesics were prescribed for patients if they had pain, but no patient reported the need for postoperative use. Only one patient developed a superficial ulceration, which remained asymptomatic and healed without specific treatment. There were no visible scars or impairment of normal function and esthetics. There was no evidence of recurrence in any of the lesions (Fig. 3).
Sclerotherapy procedure demonstration. OCH located on the lower lip with about 12 mm of extension (a). A solution of ethanolamine oleate (50 mg/ml) was diluted in a ratio of 1:4 with distilled water and prepared into a 1-ml syringe with an insulin needle. Approximately 0.3 ml of the solution was injected intralesionally (b). After 1 week, a reduction in the intensity of staining could be observed (c). After 3 weeks and two sessions of sclerotherapy, the lesion disappeared and the mucosa regained its normal appearance (d)
Discussion
Hemangioma is one of the most common vascular anomalies in the head and neck, which may need a specific treatment when it does not have a spontaneous regression [4]. Despite its high frequency, there is no one established protocol that serves as a reference for diagnosis and treatment of OCH.
The diagnosis of OCH can be clinical [3]; however, two physical examination maneuvers were demonstrated. Together, they provide important information and reinforce the clinical diagnosis: the diascopy and the HLMAC.
The diascopy is to compress a glass plate or blade over the lesion with finger pressure [2, 4]. The pressure exerted by the glass on the hemangioma compresses the blood vessels that it forms, causing the extravasation of the blood to the periphery vessels. The result of this maneuver causes a temporarily color change of the lesion, changing an important clinical feature of the lesion which is its usual reddish or purplish color, and after the maneuver, it resembles the color of the adjacent normal mucosa. After removing the pressure, the blood returns to the inside of this hemangioma and regains its original appearance.
The HLMAC is based on the principle of blood redistribution to other body parts, including the head [21]. Thus, when the head is lowered, there is an accumulation of blood in the head. This is due to the absence of valves in the venous system of the head and neck [22], which is naturally aided by gravity in an upright posture, but during the execution of this maneuver, the head is near to the horizontal plane. This combination of factors promotes more blood supply to the OCH located in the head [23], which its clinical manifestation may present an increase in the volume and/or intensity of the red or purple color, or both of the OCH.
The association of diascopy with HLMAC is extremely useful in clinical diagnosis, with diascopy being an important alternative since it is a noninvasive procedure and because of its low cost. The histopathological methods and imaging tests have higher costs and cause local trauma [7, 16, 17], which are considerable disadvantages when compared with these simple maneuvers.
Positive results in these two maneuvers, associated with a clinical feature suggestive of OCH, were safe enough for clinical diagnosis and allowed for an indication of an appropriate treatment for each case. These simple maneuvers can help in the differential diagnosis of other pigmented lesions such as nevus, malignant melanoma, Peutz–Jeghers syndrome, Addison's disease, amalgam tattoo, and melanosis induce by drugs [18].
Once the clinical diagnosis of OCH is established, it becomes necessary to decide which treatment should be indicated. A wide variety of therapeutic methods can be used, many of these shows a satisfactory final result [3, 4, 24]. Thus, it becomes difficult to determine which method could be considered the gold standard, since each has its advantages and disadvantages, and most of them have their limitations when compared with each other [9].
The therapeutic modalities used in the treatment of hemangiomas present a risk of serious complications, which must be analyzed before each indication [9]. Among these, surgical treatment is indicated in cases that compromise the esthetic or interfere with the function of the region involved [25, 26]. Also, it is closely related to the possibility of complications such as bleeding, scarring, organ, and tissue dysfunction [1, 14, 27, 28]. Surgical treatment has a high rate of recurrence and exacerbation of lesion in the postoperative period [29].
Steroid therapy acts to reduce blood flow in the lesion due to nonspecific inhibitory effect on angiogenesis and the action by contraction of pre-capillary sphincters; however, this mechanism has not yet been fully clarified [11]. Corticosteroids administered orally have adverse effects such as cushingoid, irritability, hypertension, gastrointestinal disorders, reduced growth rate and the head circumference, and weight gain [30]. After drug interruption, the rebound effect can occur, leading to recurrence of the lesion. Application of intralesional corticosteroids presents side effects such as atrophy and pigmentary changes of the skin, vision impairment, skin necrosis, and trophic changes in the skin [11].
Interferon alpha is indicated for the treatment of hemangiomas when they do not respond to corticotherapy. It acts as an inhibitor of endothelial proliferation, although its side effects are superimposed on its therapeutic effects, such as the neurological disorder (spastic diplegia), fever, malaise, diarrhea, neutropenia, anemia, and elevated transaminases [11].
Laser therapy is widely used as a therapeutic modality of hemangioma. Laser therapy provides the excision of the lesion with low degree of bleeding. The reduction of bleeding is caused by obliteration of blood vessels caused by the treatment; however, this surgical procedure can cause tissue necrosis, tissue atrophy, hyperpigmentation (transient or permanent), and scars, in addition to its high cost and high recurrence rate [31–35].
Cryotherapy has been indicated as a treatment for minor injuries but is rarely used as a therapeutic protocol for hemangiomas. It is a procedure that can be quite painful and can result in tissue atrophy, scarring, and hyperpigmentation. Cryotherapy also has a high cost because it requires specific equipment [1, 2, 36].
Embolization and radiotherapy are normally indicated for cases of intraosseous hemangiomas and/or large lesions, in which there is presence of blood vessels with large diameter [37]. These procedures are associated with very important side effects [10, 27].
The use of sclerosing agents in the treatment of vascular abnormalities has been practised for a long time [38, 39]. It is considered conservative, effective, safe, and inexpensive [5, 6, 38, 40], being the more suitable for treatment of minor lesions [4]. The sclerosing agents most widely used are ethanolamine oleate, sodium morrhuate, polidocanol, sodium tetradecyl sulfate, absolute ethanol, sodium psylliate, quinidine urethane, hypertonic saline, and ethibloc [8, 15, 35, 39]. These substances may exhibit local and systemic complications potentially serious, such as anaphylactic shock and allergic reactions after administration of sodium morrhuate [41] and tetradecyl sulfate [42].
The polidocanol produces a reduced sclerosing effect on endothelium in comparison with ethanolamine oleate [43], with less effective results. The absolute ethanol offers high risk of complications and can cause tissue damage in case of leakage of the solution [29, 44]. There are reports that the substance ethibloc caused pain, severe inflammation, and leakage, with consequent need for surgical drainage [45].
Ethanolamine oleate primarily causes irritation in the endothelial intimal layer of the vein, producing an inflammatory response culminating in fibrosis of the vessel wall and possibly vein occlusion. Ethanolamine can diffuse through the venous wall, producing an extravascular inflammatory reaction and thus may induce venous sclerosis. The components of ethanolamine oleate have different properties. Oleic acid may have procoagulant activity through the release of Hageman factor. However, the component of ethanolamine can inhibit the formation of the fibrin clot by chelation of calcium [8]. Ethanolamine oleate may have adverse effects when used in the treatment of esophageal varices, such as anaphylactic shock [46], renal failure, hemolytic anemia, hemoglobinuria, pleural effusion, pneumonia, infiltration, edema, pyrexia, and transient pain in the chest [47]. These conditions are typically self-resolutive and resolve spontaneously over a period of 48 h [48]. There are reports of generalized urticaria that occurred after sclerotherapy for varicose veins with ethanolamine oleate in doses larger than 12 ml per session, but this reaction can be controlled with antihistamines [47].
Adverse reactions associated with the administration of ethanolamine oleate are related to the amount of the applied dose; it is being considered the amount of (0.4 ml/kg) the maximum safe dosage per patient [16, 29, 44]. In this study, the treatment of choice was sclerotherapy with ethanolamine oleate in small amounts (0.1 to 0.5 ml) and reduced concentration (1:4 distilled water). When using this amount and dilution, the results were satisfactory, with a good margin of safety regarding the recommended dosage. The solution of ethanolamine oleate was diluted in a ratio of 1:4 with distilled water and administered intralesionally, weekly until disappearance thereof. We opted for dilution with distilled water, and this did not result in great discomfort to patients who merely reported a burning sensation at the site during administration. The postoperative period was painless, and despite analgesics are being prescribed in cases of painful events, their use was not necessary.
Only in one case transient appearance of an ulcer was observed, but in this case, the patient had a lesion on the lip associated with actinic cheilitis. The ulcer healed without requiring any special treatment and without any residual complication.
The authors present a protocol of treatment for patients with OCH, in which diagnostic maneuvers and a therapeutic method are low cost and easy to implement. The protocol used included clinical examinations of the lesion and is associated with physical examination maneuvers, the diascopy and HLMAC. The clinical appearance of OCH associated with positive results in at least one of the maneuvers was sufficiently reliable for the diagnosis and treatment of the lesions. The cases were treated with sclerotherapy with ethanolamine oleate with efficient outcome in this study. The protocol used was effective in the diagnosis of OCH with low cost and easy implementation, as it does not require invasive tests, expensive equipment, or even materials for its execution. The low concentration associated with a reduced amount of the sclerosing agent was responsible for the excellent results in cases treated. This protocol has been effective in the treatment with minimal side effects.
References
Johann AC, Aguiar MC, do Carmo MA, Gomez RS, Castro WH, Mesquita RA (2005) Sclerotherapy of benign oral vascular lesion with ethanolamine oleate: an open clinical trial with 30 lesions. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 100:579–84
Prado BN, Santos AF, Oliveira JMM, Vaccarezza GF, Freitas CF (2011) Uso de oleato de etanolamina para hemangiomas da cavidade bucal: um estudo de cinco años. Rev Odontol Univ Cid Sao Paulo 23:42–5 [in Portuguese]
Pedron IG, Carnaval TG, Loureiro CCS, Utumi ER, Magalhães JCA, Adde CA (2008) Opção terapêutica de hemangioma labial. Rev Inst Ciênc Saúde 26:477–81 [in Portuguese]
Assis GM, Silva RP, Moraes PH, Amaral JIQ, Germano AR (2009) Hemangioma de língua: relato de caso. Rev Cir Traumatol Buco-Maxilo-fac 9(2):59–66 [in Portuguese]
Selim H, Selim A, Khachemoune A, Metwally SA (2007) Use of sclerosing agent in the management of oral and perioral hemangiomas: Review and case reports. Med Sci Monit 13(9):CS114–119
Zanettini I, Zanettini RM, Gollo G (2005) Escleroterapia como alternativa de tratamento de lesões vasculares bucais. Clin Pesq Odontol 2(2):119–126 [in Portuguese]
Seo J, Utumi E, Zambon C, Pedron I, Rocha A (2009) Escleroterapia de hemangioma labial. Rev Odonto 17(34):106 [in Portuguese]
Wang L, Oliveira DT, Consolaro H, Perez F (1998) Tratamento de hemangioma bucal com agente esclerosante. ROBRAC 7(24):20–2 [in Portuguese]
Sarmiento ME, Espinosa IV, Quiñones LT, Arias AB (2008) Tratamiento quirúrgico de los hemangiomas faciales en niños. Rev Esp Cir Oral y Maxilofac 30(4):274–280 [in Spanish]
Marwah N, Agnihotri A, Dutta S (2006) Central hemangioma: an overview and case report. Pediatr Dent 28(5):460–6 [in Portuguese]
Hiraki PY, Goldenberg DC (2010) Diagnóstico e tratamento do hemangioma infantil. Rev Bras Cir Plast 25(2):388–97 [in Portuguese]
Neville BW, Damm DD, Allen CM, Bouquot JE (2009) Oral and maxillofacial pathology, 3rd edn. Elsevier, St. Louis
Eivazi B, Ardelean M, Bäumler W, Berlien HP, Cremer H, Elluru R, Koltai P, Olofsson J, Richter G, Schick B, Werner JA (2009) Update on hemangiomas and vascular malformations of the head and neck. Eur Arch Otorhinolaryngol 266:187–197
Pessôa BS (2011) Células endoteliais progenitoras: uma terapia possível? Rev Bras Cardiol 24(2):122–124 [in Portuguese]
Das BK, Hoque S (2008) Treatment of venous malformation with ethanolamine oleate. Asian J Surg 31(4):220–4
Mulliken JB, Glowacki J (1982) Hemangiomas and vascular malformations in infants and children: A classification based on endothelial characteristics. Plastic Reconstr Surg 69:412–22
Uehara S, Osuga K, Yoneda A, Oue T, Yamanaka H, Fukuzawa M (2009) Intralesional sclerotherapy for subcutaneous venous malformations in children. Pediatr Surg Int 25:709–713
Egg NSO, Castro CDLS, Rodrigues FN, Cury VF (2009) Melanose racial e outras lesões pigmentadas da cavidade bucal—revisão de literatura. Rev Perio 19(3):49–55 [in Portuguese]
Cardoso CL, Fernandes LMPSR, Rocha JF, Gonçales ES, Ferreira-Júnior O, Taveira LAA (2010) Abordagem cirúrgica de hemangioma intra-oral. Odontol Clin Cient 9(2):177–180 [in Portuguese]
Gontijo B, Silva CMR, Pereira LB (2003) Hemangioma da infância. An Bras Dermatol 78(6):651–673 [in Portuguese]
Hilz MJ, Ehmann EC, Pauli E, Baltadzhieva R, Koehn J, Moeller S, Defina P, Axelrod FB (2012) Combined counter-maneuvers accelerate recovery from orthostatic hypotension in familial dysautonomia. Acta Neurol Scand 126(3):162–70
DuBrul EL (1988) Sicher and DuBrul's oral anatomy, 8th edn. Ishiyaku EuroAmerica, St Louis
Cole F (1952) Head lowering in treatment of hypotension. J Am Med Assoc 150(4):273–4
Castro AL, Castro EVFL, Crivelini MM, Simonato LE (2007) Hemangioma capilar em língua: relato de caso. Rev Odonto Cien 22(56):186–189 [in Portuguese]
Lloret P (2004) Tratamiento médico de los hemangiomas. An Sist Sanit Navar 27(Suppl 1):81–92 [in Spanish]
Pagnocelli RM, Sant'Ana Filho M (1994) Tratamento do hemangioma capilar. Rev Fac Odontol Porto Alegre 35(1):2–5 [in Portuguese]
Kim JY, Kim DI, Do YS, Lee BB, Kim YW, Shin SW, Byun HS, Roh HG, Choo IW, Hyon WS, Shim JS, Choi JY (2006) Surgical treatment for congenital arteriovenous malformation: 10 years' experience. Eur J Vasc Endovasc Surg 32:101–106
Nishikawa M, Sakamoto K, Hidaka M, Yamashita A, Yamamoto G (2006) Venous malformation of the tongue in a child treated by sclerotherapy with ethanolamine oleate: A case report. J Pediatr Surg 41(3):599–600
Hyodoh H, Akiba H, Hyodoh K, Ezoe K, Yotsuyanagi T, Hareyama M (2009) Effects of blood flow control on clinical outcomes after ethanolamine oleate sclerotherapy for vascular malformations. Jpn J Radiol 27(8):297–302
Frieden IJ, Eichenfield LF, Esterly NB, Geronemus R, Mallory SB (1997) Guidelines of care for hemangiomas of infancy. J Am Acad Dermatol 37(4):631–7
Barak S, Katz J, Kaplan I (1991) The CO2 laser in surgery of vascular tumors of the oral cavity in children. ASDC J Dent Child 58(4):293–6
Volpe A, Boraks S, Genovese WJ (1999) Utilização do Nd:YAP laser como tratamento alternativo de hemangiomas na boca: relato de dois casos clínicos. Rev Odontol Univ Cid Sao Paulo 11(1):45–50 [in Portuguese]
Van Doorne L, Maeseneer MD, Stricker C, Vanrensbergen R, Stricker M (2002) Diagnosis and treatment of vascular lesions of the lip. Br J Oral Maxillofac Surg 40:497–503
Cohen BA (1987) Hemangioma in infancy and childhood. Pediatr Ann 16:17–26
Hong SK, Lee HJ, Seo JK, Lee D, Hwang SW, Sung HS (2010) Reactive vascular lesions treated using ethanolamine oleate sclerotherapy. Dermatol Surg 36:1148–1152
Carramaschi F, Ferreira MC, Goldenberg DC, Camargo CP, Faria JCM, Freitas JM (1991) Tratamento dos angiomas do lábio. Rev Hosp Clin Fac Med Sao Paulo 46(3):128–32 [in Portuguese]
Loureiro CC, Falchet PC, Gavranich J Jr, Lobo Leandro LF (2010) Embolization as the treatment for a life-threatening mandibular arteriovenous malformation. J Craniofac Surg 21(2):380–2
Robertson CS, Womack C, Robson K, Morris DL (1989) A study of the local toxicity of agents used for variceal injection sclerotherapy. HPB Surg 1(2):149–52
Holland GA (1939) Reactions from sodium morrhuate in the sclerosing of varicose veins. Can Med Assoc J 41(3):262–3
Meyer NE (1938) Monoethanolamine oleate—a new chemical for the obliteration of varicose veins. Am J Surg 40:628–9
Chin DC (1983) Treatment of maxillary hemangioma with a slerosing agent. Oral Surg Oral Med Oral Pathol 55(3):247–9
Baurmash H, Mandel L (1963) The nonsurgical treatment of hemangioma with Sotradecol. Oral Surg Oral Med Oral Pathol 16:777–82
Nishida R, Inoue R, Takimoto Y, Kita T (1999) A sclerosant with astringent properties developed in China for oesophageal varices: comparison with ethanolamine oleate and polidocanol. J Gastroenterol Hepatol 14:481–8
Hoque S, Das BK (2011) Treatment of venous malformations with ethanolamine oleate: A descriptive study of 83 cases. Pediatr Surg Int 27:527–531
Gelbert F, Enjolras O, Deffrenne D, Aynard A, Mounayer C, Merland JJ (2000) Percutaneous sclerotherapy for venous malformation of the lips: A retrospective study of 23 patients. Neuroradiology 42:692–6
Bordas JM, Feu F, Vilella A, Rodés J (1989) Anaphylactic reaction to ethanolamine oleate injection in sclerotherapy of esophageal varices. Endoscopy 21(1):50
Kiripolsky MG (2010) More on ethanolamine oleate as a vascular sclerosant. Dermatol Surg 36:1153–1154
Hughes RW Jr, Larson DE, Viggiano TR, Adson MA, van Heerden JA, Reeves CB (1982) Endoscopic variceal sclerosis: A one-year experience. Gastrointest Endosc 28(2):62–6
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
da Silva, W.B., Ribeiro, A.L.R., de Menezes, S.A.F. et al. Oral capillary hemangioma: A clinical protocol of diagnosis and treatment in adults. Oral Maxillofac Surg 18, 431–437 (2014). https://doi.org/10.1007/s10006-013-0436-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10006-013-0436-z