Abstract
We describe the development of genetic tools (electroporation, conjugation, vector for targeted gene replacement) for use in the psychrophile Psychrobacter arcticus 273-4 to test hypotheses about cold adaptation. Successful electroporation only occurred with nonstandard parameters, such as: electrocompetent cells freshly prepared from stationary-phase cultures, high field strengths (25 kV cm−1), long recovery times (16–24 h), and selection with low concentrations of antibiotics. Transformation frequencies were greatly affected by a methylation-dependent restriction barrier homologous to DpnI. The vector pJK100 (which was self-transmissible and contained a Pir-dependent R6K origin of replication) proved effective as a suicide plasmid that could be used to recombine mutations into the P. arcticus 273-4 genome. We used this vector for targeted replacement of dctT, the substrate-binding periplasmic subunit of a TRAP (tripartite ATP-independent periplasmic) transporter (which we have named dctTUF), as it was more highly expressed at cold temperatures. The replacement of dctT (with kan) decreased the rate of growth at low temperatures in mineral medium with glutamate, acetate, butyrate, and fumarate, but not with pyruvate suggesting that DctTUF participates in the transport of glutamate, acetate, butyrate, and fumarate at cold temperatures. This is the first report to demonstrate the creation of site-specific mutants in the genus Psychrobacter, their affect on low-temperature growth, and a substrate range for TAXI proteins of TRAP transporters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among the microorganisms that have been recovered and isolated from Siberian permafrost samples, Psychrobacter spp. have remarkable capabilities at subzero temperatures which identify them as potential model organisms for the study of low-temperature adaptations relevant to inhabiting permafrost (Vishnivetskaya et al. 2000; Bakermans et al. 2003, 2007). Psychrobacter arcticus 273-4 grows quickly at low temperatures, actively reproduces at −10°C, easily survives freeze-thaw cycles, and is tolerant to 12% NaCl (Ponder et al. 2005; Bakermans et al. 2006). Transcriptome and proteome analyses of low-temperature growth in Psychrobacter spp. are providing a wealth of data on low-temperature adaptation (Bakermans et al. 2007; Bergholz et al. 2008); however, without validation of a gene’s contribution to low-temperature growth, these data only suggest the involvement of identified genes in low-temperature growth. Validation of the functional relevance of identified genes to low-temperature growth can be achieved via phenotypic analysis of mutants. While the potential for developing a genetic system in Psychrobacter spp. was apparent from previous studies (Stretton et al. 1998; Tutino et al. 2000); we were limited in that no genetic system previously existed to create site-specific mutants in P. arcticus 273-4.
A successful genetic system could be used to probe the function of genes and proteins that are upregulated during low-temperature growth of P. arcticus 273-4 such as genes psyc0746 and psyc0748 which encode a TRAP (tripartite ATP-independent periplasmic) transporter. Proteomic studies revealed that the relative abundance of Psyc0746, the substrate-binding periplasmic subunit of this TRAP transporter, increased during growth at low temperatures; in rich media the relative abundance of Psyc0746 was fourfold greater at −4°C compared to 16°C (unpublished data, also see Bakermans et al. 2007). Transport systems may be important to low-temperature adaptation to counteract lower rates of diffusion and transport across the membrane, to transport compatible solutes, or to contend with low-nutrient conditions (Nedwell 1999; Welsh 2000).
TRAP transporters are secondary transporters driven by an electrochemical ion gradient and consist of three components: an extracytoplasmic solute receptor and two integral membrane proteins that are conserved between all TRAP transporters and can be present as a single fused protein (for a complete review see Kelly and Thomas 2001). While the substrate specificity of most TRAP transporters remains unknown, some TRAP transporters are known to transport glutamine, glutamate, C4-dicarboxylate, L-xylulose, mannitol, or taurine across the membrane (Kelly and Thomas 2001).
The TRAP transporter encoded by genes psyc0746 and psyc0748 of P. arcticus 273-4 consists of a putative operon containing three genes: an extracytoplasmic solute receptor, a protein of unknown function associated with some TRAP transporters (psyc0747), and a fusion of the integral membrane proteins (Fig. 1). We will refer to these genes as dctT, dctU, and dctF, respectively. The same gene arrangement is found in a TRAP transporter of Bacillus halodurans; and the extracytoplasmic solute receptor of P. arcticus 273-4 is more closely related to the TAXI (TRAP-associated extracytoplasmic immunogenic) protein of B. halodurans than to the model DctP protein of Rhodobacter capsulatus that transports C-4-dicarboxylate (Kelly and Thomas 2001). In addition, dctT contains a region with significant homology to the glutamate-binding domain IPR001311 (Mulder et al. 2005), suggesting that dctTUF may be involved in the transport of glutamate and related carboxylic acids such as acetate, butyrate, or fumarate. In contrast, pyruvate is not expected to bind DctT as pyruvate has a carbonyl group immediately adjacent to the carboxylic acid that would likely hinder access to the substrate-binding site. Currently, no experimental evidence exists to indicate the range of substrates transported by TRAP transporters with TAXI proteins.
The dctTUF TRAP transporter operon of Psychrobacter arcticus 273-4 and surrounding genome (gene names and numbers are shown, arrows indicate the direction of transcription). Putative ribosome-binding sites and promoters can be located upstream of dctT and dctF, but not upstream of dctU. DctT and dctU overlap by 23 bp; while dctU and dctF overlap by 29 bp. Psyc0745 is a possible lipoprotein; while Psyc0750 is a possible 4-carboxymuconolactone decarboxylase
Here, we describe the development of a genetic system to deliver vectors into and create site-specific mutants in P. arcticus 273-4. We have successfully utilized both electroporation and conjugation for the delivery of vectors into P. arcticus 273-4. The allelic exchange vector pJK100 (Marx and Lidstrom 2002; Denef et al. 2006) was used for targeted gene replacement in P. arcticus 273-4. Subsequently, we used these genetic tools to explore the function of dctTUF as relevant to low-temperature growth.
Materials and methods
Organisms and growth conditions
P. arcticus 273-4, Escherichia coli WM3064 (∆dapA1341::[erm pir(wt)]), E. coli DH5α, and E. coli GM2163 (dam−dcm−) were grown in Luria broth or agar with amendments (100 μg diaminopimelic acid ml−1, 25 μg kanamycin ml−1, 50 μg ampicillin ml−1, and/or 20 μg tetracycline ml−1) as appropriate. P. arcticus 273-4 cultures were incubated at 20–24°C, while E. coli cultures were incubated at 37°C. Plasmid pJK100, pRL412, and pJB864 were provided by J. Klappenbach (CME, MSU), C. P. Wolk (DOE PRL, MSU), and S. Valla (The Norwegian University of Science and Technology), respectively (Elhai and Wolk 1988; Blatny et al. 1997; Denef et al. 2006).
Purification of restriction enzyme psyc0143, a homolog of DpnI of Streptococcus pneumoniae R6, and restriction assay: see supplementary material
Electroporation
P. arcticus was grown in 10 ml LB at 22°C with shaking for at least 4 days to late stationary phase; cultures were cooled on ice for 10 min; cells were recovered by centrifugation at 10,000× g and 4°C for 10 min; cell pellets were washed three times with an equal volume of ice cold H2O with final resuspension in 0.2–1 ml ice cold H2O. Freshly prepared electrocompetent cells (40–100 μl) were mixed gently with 100–500 ng purified plasmid DNA in a 0.1 cm electroporation cuvette and shocked at 200 Ω, 25 μF, and 0.2–2.5 V in a BioRad Gene Pulser II; 960 μl of ice-cold SOC was added immediately. Subsequently, samples were transferred to 16 × 100 mm sterile test tubes, incubated with shaking for 4–24 h at 22°C, spread on plates of LB plus 5 to 50 μg ml−1 kanamycin, incubated at 22°C for 4 days, and counted. Background resistance to kanamycin was assessed by plating P. arcticus 273-4 on plates of LB plus 5–50 μg ml−1 kanamycin.
Construction of vectors
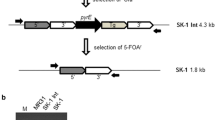
The regions upstream (using primers UF and UR) and downstream (using primers DF and DR) of dctT were PCR amplified using standard reaction conditions and a T a of 60°C (Table 1 for primer sequences and PCR product length and Fig. 2 for primer locations). PCR products were purified using a Qiagen QIAquick PCR Purification Kit (#28104) and ligated into the pGEM-T EASY Vector System (Promega Corp., Madison, WI, USA) creating vectors pGEM-Up and pGEM-Down. The presence of inserts in individual colonies was verified by PCR. Plasmid DNA was isolated using the Wizard Plus SV Minipreps DNA Purification System (Promega Corp.). pGEM-Up and pJK100 were digested with NcoI and NdeI (NE Biolabs, Ipswich, MA, USA). DNA fragments were separated by gel electrophoresis; appropriate bands were excised from the gel and purified using a Qiagen QIAquick Gel Purification Kit (#28704). DNA fragments were ligated together to create pJK101 with T4 DNA ligase (NE Biolabs) and electroporated into E. coli WM3064. The presence of inserts was verified by PCR. DNA from the plasmids pJK101 and pGEM-Down was isolated as above and digested with SacII and AgeI. As above, fragments were separated, purified, ligated together to create pJK102, and electroporated into E. coli WM3064. Presence of inserts was verified by PCR.
Creation of mutants
Aliquots (200 μl) of an overnight culture of E. coli WM3064 pJK102 were mixed with 200- and 800-μl aliquots of 2-day cultures of P. arcticus 273-4 (controls included 200-μl donor only, 200-μl recipient only, and 800-μl recipient only). Cells were mixed gently, collected by centrifugation at 4,000 rpm for 2 min, and supernatants decanted. Cell pellets were gently resuspended in 100 μl LB, spotted on LB plus 100 μg diaminopimelic acid ml−1 plates, and incubated for at least 20 h at room temperature. Subsequently, cells were resuspended in 1 ml PBS (8 g/l NaCl, 0.2 g/l KCl, 1.44 g/l Na2HPO4, 0.24 g/l KH2PO4, pH 7.4) and two serial 10−1 dilutions made. One hundred microliters of each sample and serial dilution were spread on LB plates plus 25 μg kanamycin ml−1 in duplicate and incubated at 20°C for 3–5 days. Transconjugants were screened by patch plating on both LB plus 25 μg kanamycin ml−1 and LB plus 20 μg tetracycline ml−1; kanR tetS transconjugants were selected for further analysis.
Analysis of mutants by PCR
PCR was performed as above at an annealing temperature of 61°C (Table 1 for sequences and Fig. 2 for primer locations). PCR products were visualized by agarose gel electrophoresis and staining with ethidium bromide.
RT-PCR analysis of expression of dctTUF operon: see supplementary material
Phenotype analysis
Strains were grown in mineral medium (MM) with 20 mM acetate, butyrate, fumarate, glutamate, or pyruvate added. MM contained 50 mM MOPS (pH 7), 1 mM K2HPO4, 5 mM NH4Cl, 0.5% sea salt, 1× Wolfe’s Mineral Solution and 1× Wolfe’s Vitamins (Atlas 1993). Cultures were incubated in triplicate or quadruplicate with shaking at 4 or 22°C. Optical density at 600 nm was monitored over time using a Spectronic 20D+ spectrophotometer. Growth rates at exponential phase were calculated by standard methods. Pairwise comparisons of mutant versus wildtype growth rates were tested for statistical significance (P < 0.025) using the Student’s t test.
Membrane fluidity
Strains were grown in MM as above at 22 or 4°C; when the optical density at 600 nm reached an average of 0.205 (range 0.17–0.27) cells were fixed with formaldehyde (0.5% final concentration) and stored at their growth temperature until assayed. Fixed cells (200 μl) were aliquoted into black, polystyrene flat-bottom 96-well plates. The membrane probes 6-dodecanoyl-2-dimethylaminoaphthalene (laurdan) and 1,3-diphenyl-1,3,5-hexatriene (DPH) were used. Either laurdan (1.4 × 10−6 g added from a stock solution in methanol) or DPH (2.1 × 10−6 g added from a stock solution in ethanol) was added to samples and incubated for 1 h at room temperature in the dark. Assays were performed on 6 to 12 replicates; each replicate consisted of sample with and without probe (scattering control). Fluorescence intensity was measured at the emission wavelength (440 nm for laurdan and 430 nm for DPH) with excitation at the appropriate wavelength (355 nm for laurdan and 360 nm for DPH) using a Spectramax M5 fluorometer (Molecular Devices) at room temperature. Anisotropy (r) was calculated as follows: r = (I V − GI H)/(I V + 2GI H), where I V and I H are the fluorescence intensities determined at vertical and horizontal orientations of the emission polarizer when the excitation polarizer is set in the vertical position and G is a correction factor for dissymmetry associated with the horizontal and vertical positions of the polarizers (Harris et al. 2002).
Results
Development of genetic tools to probe gene function in Psychrobacter
Restriction barrier
Analysis of the genome sequence of P. arcticus 273-4 revealed the presence of psyc0143 which was 61% identical and 77% similar at the amino acid sequence level to DpnI of S. pneumoniae R6. The G + C content of psyc0143 and dpnI (36.6 and 32.5%, respectively) suggests a relatively recent horizontal gene transfer event. Subsequently, Psyc0143 was partially purified from P. arcticus 273-4 and its ability to digest methylated DNA was confirmed (Fig. 1 in Electronic supplementary material).
Replication of plasmids in Psychrobacter
We sought to identify a plasmid (and origin of replication) that could not replicate in Psychrobacter for use as a delivery vector. The narrow host-range plasmids pRL1062a (p15a origin) and pRK2013 (ColE1 origin) were introduced into P. arcticus 273-4 by conjugation (cells were mixed in equal amounts and mated at room temperature for 1–2 days) and found to replicate (Figurski and Helinski 1979; Ditta et al. 1980; Cohen et al. 1998). Replication of plasmids in P. arcticus 273-4 was confirmed via purification and digestion with diagnostic restriction enzymes (Fig. 2 in Electronic supplementary material). Subsequently, we were able to use the vector pJK100 (see below).
In addition, we sought to identify a plasmid that could replicate in Psychrobacter for use as an expression vector and to test electroporation protocols. We chose the plasmid pRL412 because it has a broad host-range (RSF1010) origin and a kanamycin-resistance gene which is expressed in other species of Psychrobacter (Elhai and Wolk 1988; Stretton et al. 1998). Replication of plasmid pRL412 in P. arcticus 273-4 was confirmed as above (Fig. 2 in Electronic supplementary material). Subsequently, pRL412 was used in the development of an electroporation protocol.
Electroporation
The conditions required for successful electroporation of vector pRL412 into P. arcticus 273-4 were surprisingly exacting (Fig. 3). Late-log to stationary-phase cells and field strengths of 25 kV cm−1 yielded the best electroporation efficiencies. In addition, cells needed at least 16 h for maximal recovery from electroporation and expression of antibiotic resistance prior to spreading on plates containing minimal concentrations of antibiotic (5 μg kanamycin ml−1). Because the doubling time of P. arcticus 273-4 in exponential phase in rich media at 22°C is ~4 h, the dramatic increase in transformants from 4 to 16 h of recovery time was not attributed to growth of the cells. Furthermore, only freshly prepared electrocompetent cells could be transformed. No transformants were obtained from electrocompetent cells which were frozen in 10% glycerol and stored at −80°C; although electrocompetent cells successfully survived freezing and thawing as determined by plating on non-selective media (average number of surviving cells was 1.2 × 1010 cfu ml−1).
Electroporation of pRL412 into Psychrobacter arcticus 273-4. a Electroporation efficiency (transformants per recipients) versus growth phase and field strength. Experimental conditions: 500 μg of pRL412, 24 h recovery time, 5 μg kanamycin ml−1 in selective media. OD600 of mid-log, late-log, stationary, and late stationary cultures was 0.98, 1.48, 1.72, and 1.64, respectively. b Survival of electroporated cells in recovery medium over time with counts on selective (transformants) and non-selective (total) media. Experimental conditions: field strength of 25 kV cm−1, electrocompetent late-stationary phase cells, 100 μg of pRL412, and 5 μg kanamycin ml−1 in selective media
Allelic exchange vector
We used the allelic exchange vector pJK100 which has a pir-dependent origin of replication to ensure that the vector would not replicate within P. arcticus 273-4 (Denef et al. 2006). Frequency of successful conjugation and insertion into the chromosome ranged from 5 × 10−6 to 4 × 10−7. A high percentage (53 to 93%) of the kanr tets transconjugants tested contained one or both of the genome/vector junctions indicating that single or double recombination had occurred. Many Kanr Tets transconjugants (~80%) contained the tetracycline-resistance gene indicating that only a single recombination event had occurred and that tetracycline resistance was not well expressed in P. arcticus 273-4.
Role of TRAP family transporter in growth at low temperatures
The substrate-binding subunit of a TRAP transporter, dctT, was targeted for deletion because preliminary studies demonstrated that this protein was upregulated during growth at low temperatures (Bakermans et al. 2007).
Construction of mutant
Successful construction of the replacement vector pJK102 was verified by sequencing. When Kanr Tets transconjugants were tested by PCR for the kanamycin gene, the upstream genome/vector junction, the downstream genome/vector junction, and the tetracycline gene, only two ∆dctT mutants (numbers 10 and 19) contained all of the appropriate PCR products demonstrating that successful replacement of the wildtype gene had occurred via double recombination with subsequent loss of the vector backbone (Fig. 4). While a complement for ∆dctT was constructed by PCR amplification of the dctTUF operon and insertion into pRL412 (data not shown), the complement could not be electroporated into either P. arcticus 273-4 ∆dctT or wildtype P. arcticus 273-4. However, reverse transcriptase-PCR analysis of RNA from actively growing cultures detected both dctU and dctF in P. arcticus 273-4 ∆dctT during growth on acetate at 22 and 4°C; while dctT, dctU and dctF were not detected during growth of the mutant or wildytpe strains on pyruvate (Fig. 3 in Electronic supplementary material).
PCR analysis of Psychrobacter arcticus 273-4 ΔdctT. PCR amplification was performed with the following primer pairs: Indel, insertion/deletion site; GUF/kanR, the upstream genome/vector junction; GDR/kanF, downstream genome/vector junction; and tet, tetracycline-resistance gene. Samples included: w, wildtype P. arcticus 273-4; Δ, P. arcticus 273-4 ΔdctT ; v, vector pCB102 only; and -, PCR reagents only. Size markers, M, included both 100 bp and 1 Kb ladder from New England Biolabs
Phenotype analysis
The growth rate of P. arcticus 273-4 ∆dctT grown on glutamate was significantly lower than wildtype (P < 0.025), regardless of growth temperature (Fig. 5). The growth rate of P. arcticus 273-4 ∆dctT grown on acetate, butyrate, or fumarate was significantly lower than wildtype (P < 0.025) only at 4°C. At 22°C, growth rates of ∆dctT were 79, 96, or 72% of the wildtype rates when grown on acetate, butyrate, or fumarate, respectively; while at 4°C, growth rates of mutants were 32, 15, or 12% of the wildtype rates, respectively. In contrast, the growth rate of ∆dctT did not vary significantly from that of wildtype when grown with pyruvate, regardless of temperature, suggesting that DctTUF is specifically involved in the transport of glutamate, acetate, butyrate, and fumarate. Final optical densities of wildtype and mutant cultures were similar, although specific to carbon source, at 22°C but not at 4°C (mutant cultures had lower final optical densities than wildtype cultures). In addition, final optical densities of wildtype cultures at 22 and 4°C were similar.
Membrane fluidity
The membrane fluidity of the mutant ∆dctT did not vary significantly from that of the wildtype under all conditions tested (temperature, carbon source, laurdan, or DPH; Fig. 6) except when grown on glutamate. The membrane fluidity of both wildtype and mutant strains was higher at 22°C than at 4°C when grown on glutamate.
Membrane fluidity of wildtype (solid) and mutant (patterned) cultures grown on different carbon sources (x-axis) at 22 or 4°C. The average of at least 6, and not more than 12, samples is shown; error bars indicate standard deviation. Lower anisotropy values indicate more rotation of the probe and imply a more fluid membrane compared to higher anisotropy values
Discussion
We are studying the psychrophile P. arcticus 273-4 as a model for cold adaptation in terrestrial ecosystems. In this study, we have described the development of several genetic tools for use in Psychrobacter, as well as the use of these tools to probe the function of a TRAP transporter as relevant to low-temperature growth of P. arcticus 273-4.
Development of genetic tools to probe gene function in Psychrobacter
The potential for developing a genetic system in P. arcticus 273-4 was apparent from a previous study examining reporter gene expression in marine bacteria (Stretton et al. 1998). This study successfully demonstrated that a mini-Tn10-kan-gfp was capable of inserting into the chromosome of Psychrobacter sp. strain SW5H. While expression of Gfp was possible from the npt-2 promoter in Psychrobacter sp. strain SW5H, Gfp was not expressed from the lac promoter indicating that expression of selective markers in Psychrobacter will depend greatly on recognition of the promoter. In addition, limited information exists about what types of plasmids replicate in Psychrobacter. An RSF1010-based plasmid was maintained weakly in Psychrobacter sp. strain SW5H, while cryptic plasmids have been discovered in Psychrobacter sp. TA144 and P. cryohalolentis K5 (Stretton et al. 1998; Tutino et al. 2000).
Unfortunately, when developing genetic systems in novel organisms, there are many unforeseen technical challenges that must be overcome. In P. arcticus 273-4, the following difficulties were discovered and surmounted: a methylation-dependent restriction system, maintenance of vectors, and narrow electroporation parameters. Successful procedures were also identified and developed, including: conjugation and an allelic exchange vector; while the susceptibility of P. arcticus 273-4 to many antibiotics had been demonstrated previously (Ponder et al. 2005).
The presence of a methylation-dependent restriction enzyme homologous to the type II restriction endonuclease DpnI in P. arcticus 273-4 was confirmed. Methylation-dependent restriction systems present a significant barrier to the introduction of foreign DNA. The propagation of vectors for electroporation in E. coli GM2163 circumvented the restriction barrier (as did conjugation of vectors into Psychrobacter from E. coli regardless of methylation status).
We were surprised that plasmids containing the colE1 and p15a origins of replication, which are narrow host-range origins specific to the family Enterobacteriaceae, were maintained in P. arcticus 273-4 (Kues and Stahl 1989). Recent work has demonstrated that plasmids with the P15a origin are also maintained in Shewanella putrefaciens and Haemophilus influenza (Chandler 1991; Myers and Myers 1997), suggesting a broader host range for p15a plasmids within selected orders of the class Gammaproteobacteria.
The electroporation protocol developed for P. arcticus 273-4 contained many nonstandard parameters, such as: the use of stationary-phase cells, high field strengths, long recovery times, and selection on very low concentrations of antibiotics. For most organisms, early- to mid-log phase cells provide the highest transformation efficiencies (Calvin and Hanawalt 1988; Tatebe et al. 1995; Liang and Lee 1998); however, late-log to stationary-phase cells yielded the best results in Psychrobacter. High field strengths (≥18 kV/cm) are commonly used to electroporate Clostridium and Bacillus spp. and are attributed to the rigid structure and composition of the Gram-positive cell wall (Whyte and Inniss 1994; Xue et al. 1999; Ito and Nagane 2001; Tyurin et al. 2004). The high field strengths required for successful electroporation of Psychrobacter suggest that the cell wall and outer membrane may be particularly rigid during stationary phase. The long recovery time required for successful electroporation in Psychrobacter may also be indicative of the relatively slow growth rate of the organism. Alternatively, long recovery times may allow for the adequate expression of antibiotic-resistance genes and have been found to increase electroporation efficiency in Zymomonas and Lactobacillus (Wei et al. 1995; Liang and Lee 1998). Most interestingly, electrocompetent Psychrobacter cells could not be stored frozen, they had to be freshly prepared and used. Electroporation relies on low temperatures of 0 to 4°C to keep microorganisms inactive as they are being manipulated; however, P. arcticus 273-4 is able to actively grow at these temperatures. Many of the standard electroporation procedures that did not work with Psychrobacter could have enhanced the activity of Psychrobacter at low temperatures (such as preparing electrocompetent cells from exponentially growing cells).
Apparently, antibiotic resistance is poorly expressed in P. arcticus 273-4, as evidenced by the low concentration of kanamycin required for selection of transformants and the inability to detect tetracycline resistance. Low concentrations of antibiotics are used to alleviate stress when antibiotic-resistance genes are poorly expressed (Wei et al. 1995). In addition, attempts to electroporate pJB864 (Ampr) into P. arcticus 273-4 failed—that is, no transformants were obtained—possibly because ampicillin resistance was not sufficiently expressed. Antibiotic-resistance genes may not be well expressed in Psychrobacter due to differences in promoter sequences, promoter recognition, and/or the RNA polymerase holoenzyme. Alternatively, antibiotic resistance may not be realized due to poor translation, poor protein stability or incompatibility of the protein with the host. Notably, all the vectors we have worked with successfully contained the nptII kanamycin-resistance genes from Tn5. It is possible that other antibiotic resistances (besides amp and tet) may be better expressed in Psychrobacter.
We were able to construct site-specific replacement mutants of P. arcticus 273-4 using the allelic exchange vector pJK100. For our purposes, the most useful features of this vector were the self-transmissibility and Pir-dependent origin of replication given the difficulty of electroporation and the variety of plasmids that were found to replicate in Psychrobacter, respectively. Subsequently, this vector has been used to create deletion mutants of dctT and several other genes (Bergholz et al. 2008). Because dctT is the first gene in an operon containing the TRAP transporter, we anticipated that the deletion of dctT was polar on the two downstream open reading frames; however, it was not.
Role of TRAP family transporter in growth at low temperatures in P. arcticus 273-4
The TRAP transporter encoded by dctTUF appears to be the primary transporter for glutamate and for acetate, butyrate, and fumarate at low temperatures. This is the first report to suggest a substrate range for a TAXI protein of a TRAP transporter. Like some TRAP transporters, DctTUF transports carboxylic acids. It remains to be determined whether or not the substrate range of DctT is representative of other TAXI family proteins.
To verify that the replacement of dctT had no adverse affect on the membrane, we measured the fluidity of the cytoplasmic membrane via fluorescence polarization (anisotropy) of two membrane probes: laurdan and DPH. Laurdan has been shown to localize near the polar head groups in the cytoplasmic membrane, and is therefore sensitive to changes at the water–lipid interface (Harris et al. 2002; Vanounou et al. 2002); whereas, DPH has been shown to localize within the core of the lipids, parallel to the phospholipid chains, of the cytoplasmic membrane and is sensitive to changes in saturation of the acyl chains (Aricha et al. 2004; Beney et al. 2004). Notably, the membrane fluidity did not vary with temperature (except when grown on glutamate) reflecting homeoviscous adaptation. Hence, diffusion of the growth substrate across the membrane was likely not enhanced at higher growth temperatures due to an increase in membrane fluidity.
The replacement of dctT with kan significantly decreased the efficiency of growth on acetate, butyrate, and fumarate at low temperature. Growth was minimally affected at high temperatures, suggesting that other transporters compensated for the loss of dctT. There are several putative transporters identified within the genome of P. arcticus 273-4 with unknown substrate specificity that could provide such functional redundancy and include: psyc0303 to 0306, ABC glutamate/aspartate transporter; psyc1653, glycolate/L-lactate transporter; psyc1836, sodium/proton:dicarboxylate symporter; psyc1963 to 1965, TRAP family transporter; psyc0854, periplamic substrate-binding subunit of a TRAP family transporter; and psyc1173, transport protein in catabolism of dicarboxylic acids (MFS superfamily).
These data suggest that isofunctional enzymes (isozymes) transport acetate, butyrate, and fumarate at higher temperatures while dctTUF is required to effectively transport these substrates at low temperatures. Organisms can employ isozymes as an adaptation to low temperatures by having separate enzymes with distinct thermal characteristics available for use at specific temperatures. While poikilothermic animals commonly use isozymes as an adaptation to temperature changes (Marcus 1977; Yamawaki and Tsukuda 1979; Lin and Somero 1995), few examples have been documented in bacteria (Ishii et al. 1987; He et al. 2001). Bacterial isozymes were first documented for isocitrate dehydrogenase of the psychrophile Colwellia maris (Ochiai et al. 1979, 1984; Ishii et al. 1987). Specialized low-temperature transporters may be required to compensate for transport proteins with narrow temperature optima or to counteract low rates of diffusion or to counter changes in the membrane.
As in poikilothermic animals, the use of isozymes by Psychrobacter spp. may be advantageous for thriving across a range of temperatures. In P. arcticus 273-4, putative isozymes have been identified in transcriptome experiments where the expression of one isozyme is increased at high temperatures and the expression of the second isozyme is increased at low temperatures (Bergholz et al. 2008). In addition, proteome experiments revealed the presence of an isozyme for the outer membrane receptor for ferric citrate transport, FecA, that was expressed only at subzero temperatures in P. cryohalolentis K5, which was also isolated from Siberian permafrost and is a close relative of P. arcticus 273-4 (Bakermans et al. 2007). The use of isozymes may be particularly useful to microorganisms that live in permafrost given that their initial habitat, prior to burial, is within the active layer of permafrost where temperatures fluctuate on a seasonal—and sometimes daily—basis around the freezing point of water. These results highlight the importance of maintaining transport systems for successful growth at low temperatures.
References
Aricha B, Fishov I, Cohen Z, Sikron N, Pesakhov S, Khozin-Goldberg I, Dagan R, Porat N (2004) Differences in membrane fluidity and fatty acid composition between phenotypic variants of Streptococcus pneumoniae. J Bacteriol 186:4638–4644
Atlas RM (1993) Handbook of microbiological media. CRC Press, Boca Raton
Bakermans C, Ayala-del-Río HL, Ponder MA, Vishnivetskaya T, Gilichinsky D, Thomashow MF, Tiedje JM (2006) Psychrobacter cryohalolentis sp nov. and Psychrobacter arcticus sp. nov. isolated from Siberian permafrost. Int J Sys Evol Microbiol 56:1285–1291
Bakermans C, Tollaksen SL, Giometti CS, Wilkerson C, Tiedje JM, Thomashow MF (2007) Proteomic analysis of Psychrobacter cryohalolentis K5 during growth at subzero temperatures. Extremophiles 11:345–354
Bakermans C, Tsapin AI, Souza-Egipsy V, Gilichinsky DA, Nealson KH (2003) Reproduction and metabolism at–10°C of bacteria isolated from Siberian permafrost. Environ Microbiol 5:321–326
Beney L, Mille Y, Gervais P (2004) Death of Escherichia coli during rapid and severe dehydration is related to lipid phase transition. Appl Microbiol Biotech 65:457–464
Bergholz PW, Bakermans C, Tiedje JM (2008) Resource efficiency and molecular motion dominate the transcriptome response during growth below 0°C in Psychrobacter arcticus 273-4 (submitted)
Blatny J, Brautaset T, Winther-Larsen H, Karunakaran P, Valla S (1997) Improved broad-host-range RK2 vectors useful for high and low regulated gene expression levels in Gram-negative bacteria. Plasmid 38:35–51
Calvin NM, Hanawalt PC (1988) High-efficiency transformation of bacterial-cells by electroporation. J Bacteriol 170:2796–2801
Chandler MS (1991) New shuttle vectors for Haemophilus influenzae and Escherichia coli: P15A-derived plasmids replicate in H. influenzae Rd. Plasmid 25:221–224
Cohen MF, Meeks JC, Cai YA, Wolk CP (1998) Transposon mutagenesis of heterocyst-forming filamentous cyanobacteria. Methods Enzymol 297:3–17
Denef VJ, Klappenbach JA, Patrauchan MA, Florizone C, Rodrigues JLM, Tsoi TV, Verstraete W, Eltis LD, Tiedje JM (2006) Genetic and genomic insights into the role of benzoate-catabolic pathway redundancy in Burkholderia xenovorans LB400. Appl Environ Microbiol 72:585–595
Ditta G, Stanfield S, Corbin D, Helinski DR (1980) Broad host range DNA cloning system for Gram-negative bacteria: construction of a gene bank of Rhizobium meliloti. Proc Natl Acad Sci USA 77:7347–7351
Elhai J, Wolk CP (1988) A versatile class of positive-selection vectors based on the nonviability of palindrome-containing plasmids that allows cloning into long polylinkers. Gene 68:119–138
Figurski DH, Helinski DR (1979) Replication of an origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans. Proc Natl Acad Sci USA 76:1648–1652
Harris FM, Best KB, Bell JD (2002) Use of laurdan fluorescence intensity and polarization to distinguish between changes in membrane fluidity and phospholipid order. Biochim Biophys Acta Biomembr 1565:123–128
He HJ, Gordon R, Gow JA (2001) The effect of temperature on the fatty acids and isozymes of a psychrotrophic and two mesophilic species of Xenorhabdus, a bacterial symbiont of entomopathogenic nematodes. Can J Microbiol 47:382–391
Ishii A, Ochiai T, Imagawa S, Fukunaga N, Sasaki S, Minowa O, Mizuno Y, Shiokawa H (1987) Isozymes of isocitrate dehydrogenase from an obligately psychrophilic bacterium, Vibrio sp strain Abe-1—purification, and modulation of activities by growth-conditions. J Biochem 102:1489–1498
Ito M, Nagane M (2001) Improvement of the electro-transformation efficiency of facultatively alkaliphilic Bacillus pseudofirmus OF4 by high osmolarity and glycine treatment. Biosci Biotech Biochem 65:2773–2775
Kelly D, Thomas G (2001) The tripartite ATP-independent perimplasmic (TRAP) transporters of bacteria and archaea. FEMS Microb Rev 25:405–424
Kues U, Stahl U (1989) Replication of plasmids in Gram-negative bacteria. Microbiol Rev 53:491–516
Liang CC, Lee WC (1998) Characteristics and transformation of Zymomonas mobilis with plasmid pKT230 by electroporation. Bioprocess Eng 19:81–85
Lin JJ, Somero GN (1995) Temperature-dependent changes in expression of thermostable and thermolabile isozymes of cytosolic malate-dehydrogenase in the eurythermal goby fish Gillichthys mirabilis. Physiol Zool 68:114–128
Marcus NH (1977) Temperature induced isoenzyme variants in individuals of sea-urchin, Arbacia punctulata. Comp Biochem Physiol B Biochem Mol Biol 58:109–113
Marx CJ, Lidstrom ME (2002) Broad-host-range cre-lox system for antibiotic marker recycling in Gram-negative bacteria. Biotechniques 33:1062–1067
Mulder NJ, Apweiler R, Attwood TK, Bairoch A, Bateman A, Binns D, Bradley P, Bork P, Bucher P, Cerutti L, Copley R, Courcelle E, Das U, Durbin R, Fleischmann W, Gough J, Haft D, Harte N, Hulo N, Kahn D, Kanapin A, Krestyaninova M, Lonsdale D, Lopez R, Letunic I, Madera M, Maslen J, McDowall J, Mitchell A, Nikolskaya AN, Orchard S, Pagni M, Ponting CP, Quevillon E, Selengut J, Sigrist CJA, Silventoinen V, Studholme DJ, Vaughan R, Wu CH (2005) InterPro, progress and status in 2005. Nucl Acids Res 33:D201–D205
Myers C, Myers J (1997) Replication of plasmids with the p15A origin in Shewanella putrefaciens MR-1. Lett Appl Microbiol 24:221–225
Nedwell DB (1999) Effect of low temperature on microbial growth: lowered affinity for substrates limits growth at low temperature. FEMS Microb Ecol 30:101–111
Ochiai T, Fukunaga N, Sasaki S (1979) Purification and some properties of two NADP+-specific isocitrate dehydrogenases from an obligately psychrophilic marine bacterium, Vibrio sp. strain Abe-1. J Biochem 86:377–384
Ochiai T, Fukunaga N, Sasaki S (1984) Two structurally different NADP-specific isocitrate dehydrogenases in an obligately psychrophilic bacterium, Vibrio sp strain Abe-1. J Gen Appl Microbiol 30:479–487
Ponder MA, Gilmour SJ, Bergholz PW, Mindock CA, Hollingsworth R, Thomashow MF, Tiedje JM (2005) Characterization of potential stress responses in ancient Siberian permafrost psychroactive bacteria. FEMS Microbiol Ecol 53:103–115
Stretton S, Techkarnjanaruk S, McLennan AM, Goodman AE (1998) Use of green fluorescent protein to tag and investigate gene expression in marine bacteria. Appl Environ Microbiol 64:2554–2559
Tatebe W, Muraji M, Fujii T, Berg H (1995) Reexamination of electropermeabilization on yeast cells—dependence on growth-phase and ion concentration. Bioelectrochem Bioenerg 38:149–152
Tutino ML, Duilio A, Moretti MA, Sannia G, Marino G (2000) A rolling-circle plasmid from Psychrobacter sp. TA144: evidence for a novel Rep subfamily. Biochem Biophys Res Commun 274:488–495
Tyurin MV, Desai SG, Lynd LR (2004) Electrotransformation of Clostridium thermocellum. Appl Environ Microbiol 70:883–890
Vanounou S, Pines D, Pines E, Parola AH, Fishov I (2002) Coexistence of domains with distinct order and polarity in fluid bacterial membranes. Photochem Photobiol 76:1–11
Vishnivetskaya T, Kathariou S, McGrath J, Gilichinsky D, Tiedje JM (2000) Low-temperature recovery strategies for the isolation of bacteria from ancient permafrost sediments. Extremophiles 4:165–173
Wei MQ, Rush CM, Norman JM, Hafner LM, Epping RJ, Timms P (1995) An improved method for the transformation of Lactobacillus strains using electroporation. J Microbiol Meth 21:97–109
Welsh DT (2000) Ecological significance of compatible solute accumulation by micro-organisms: from single cells to global climate. FEMS Microbiol Rev 24:263–290
Whyte LG, Inniss WE (1994) Electroporation and its effect on the psychrotrophic bacterium Bacillus psychrophilus. Can J Microbiol 40:83–89
Xue GP, Johnson JS, Dalrymple BP (1999) High osmolarity improves the electro-transformation efficiency of the Gram-positive bacteria Bacillus subtilis and Bacillus licheniformis. J Microbiol Meth 34:183–191
Yamawaki H, Tsukuda H (1979) Significance of the variation in isozymes of liver lactate-dehydrogenase with thermal acclimation in goldfish—thermostability and temperature dependency. Comp Biochem Physiol B Biochem Mol Biol 62:89–93
Acknowledgments
This work was supported through membership in the NASA Astrobiology Institute. C. Bakermans was supported by a National Academy of Sciences National Research Council research associateship. We thank J. Klappenbach for providing pJK100 and E. coli WM3064.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Matsunaga.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bakermans, C., Sloup, R.E., Zarka, D.G. et al. Development and use of genetic system to identify genes required for efficient low-temperature growth of Psychrobacter arcticus 273-4. Extremophiles 13, 21–30 (2009). https://doi.org/10.1007/s00792-008-0193-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-008-0193-3