Abstract
The soil of the former lake Texcoco is an extreme environment localized in the valley of Mexico City, Mexico. It is highly saline and alkaline, where Na+, Cl−, HCO −3 and CO 2−3 are the predominant ions, with a pH ranging from 9.8 to 11.7 and electrolytic conductivities in saturation extracts from 22 to 150 dS m−1. Metagenomic DNA from the archaeal community was extracted directly from soil and used as template to amplify 16S ribosomal gene by PCR. PCR products were used to construct gene libraries. The ribosomal library showed that the archaeal diversity included Natronococcus sp., Natronolimnobius sp., Natronobacterium sp., Natrinema sp., Natronomonas sp., Halovivax sp., “Halalkalicoccus jeotgali” and novel clades within the family of Halobacteriaceae. Four clones could not be classified. It was found that the archaeal diversity in an alkaline-saline soil of the former lake Texcoco, Mexico, was low, but showed yet uncharacterized and unclassified species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The development of molecular biological techniques that do not depend upon culturing has proven effective to gain insight into the diversity and structure of microbial communities. In particular, the ability to recover and analyze 16S rRNA genes directly from environmental DNA provides a mean to investigate microbial populations without the need to culture them (Ward et al. 1990; Amann et al. 1995; Hugenholtz et al. 1998; Dojka et al. 2000).
The 16S rRNA gene analysis have been used to study diverse bacterial and archaeal communities in extreme environments with a wide range of salinity, pH and temperature. These environments are interesting to study microbial diversity, to identify novel microorganisms and to understand the functioning of an ecosystem (Lizama et al. 2001). Few alkaline saline soils have been investigated, but sediments of saline and alkaline lakes have been studied, e.g., Mono Lake in California (Humayoun et al. 2003), saline meromictic Lake Kaiike in Japan (Koizumi et al. 2004), soda lakes in Inner Mongolia, China (Ma et al. 2004), saline and alkaline lakes of Wadi el Natrun in the Libyan Desert, Egypt, (Grant et al. 2004), athalassohaline lakes of the Atacama Desert, Chile (Demergasso et al. 2004) and lakes in the Antarctic (Tindall 2004).
The soil of the former lake Texcoco located in the valley of Mexico City (Mexico) at an altitude of 2,240 m above sea level with a mean annual temperature of 16.7°C and annual precipitation of 705 mm is formed from volcanic ash deposited in situ in a lacustrine environment and covered recently by colluvial materials (Luna-Guido et al. 2000). The aquifer is near to the surface (80–150 cm). pH in undrained soil can range from 9.8 to 11.7, electrolytic conductivities (EC) in saturation extracts from 22 to 150 dS m−1, exchangeable sodium percentages from 76 to 98% and sodium adsorption ratio from 103 to 1,718 mM (Beltrán-Hernández et al. 1999). The soil texture is loamy to clayey, the structure is granular in the topsoil and prismatic in the subsoil and the organic matter contents range from 20 to 50 g kg−1 dry soil. Natural drainage is poor and roots are restricted by a compact ash layer 5–20 cm thick to depths of 16–40 cm (Luna-Guido et al. 2001). Dynamics of C and N have been investigated intensively, and although the bacterial populations in waters of the former Texcoco lake has been studied no information exists about the microorganisms in the soil of the former lake (Jan-Roblero et al. 2004; Conde et al. 2005; Beltrán-Hernández et al. 2007). The objective of this study was to investigate the diversity of archaea in the alkaline saline allophanic soil of the former lake Texcoco.
Material and methods
Site description and soil sampling
The sampling site is located at the former lake Texcoco (Northern Latitude 19°30′52″ Western Longitude 98°59′24″) in the state of Mexico, Mexico (http://www.inegi.gob.mx). Soil samples were collected in the season with low rainfall from a site with EC 159 dS m−1. An area of 1 m2 was delimited, the first 2 cm of soil were discarded, and the next 10 cm layer was sampled with a hand spade and taken to the laboratory in a black polyethylene bag. The samples were 5 mm sieved under aseptic conditions, divided in 250 g sub-samples and stored at −80°C until analyzed. The pHw (2:1) in the clayey soil was 11.75, water holding capacity 968 g kg−1 soil, organic C 14.9 g kg−1 soil, total N content 1.32 g N kg−1soil and with water content 268 g kg−1 upon sampling.
DNA extraction and purification
DNA was extracted from soil with a modified method based on direct cell lysis (Guo et al. 1997). A 5 g soil sample was added to a 50 ml Oak Ridge tube containing 5 ml 0.12 M sodium phosphate buffer (PB buffer pH 8.0), vortexed for 1 min, incubated at room temperature for 10 min and centrifuged at 7,700×g for 10 min. The supernatant was decanted and 5 ml PB buffer was added to the soil pellet. The procedure of vortexing, incubation and centrifugation was repeated once. The soil pellet was re-suspended in 8 ml lysis solution I (0.15 M NaCl, 0.1 M EDTA, pH 8.0, 10 mg lysozyme ml−1), mixed and incubated at 37°C for 1 h with occasional gentle mixing. A 8 ml lysis solution II (0.1 M NaCl, 0.5 M Tris–HCl, pH 8.0, 12% SDS) was added. The soil suspension was passed through two cycles of freezing at −40°C for 20 min and thawing at 65°C for 20 min and then centrifuged at 7,700×g for 10 min. The supernatant was mixed with 2.7 ml 5 M NaCl and 2.1 ml 10% TRITON-X100 in 0.7 M NaCl and incubated for 10 min at 65°C. An equal volume of CHCl3:isoamyl alcohol (24:1) was added, mixed and centrifuged for 5 min at 3,000×g. The supernatant was transferred to a clean tube. An equal volume of 13% PEG (polyethylene glycol [8,000 MW] dissolved in 1.6 M NaCl) was added to the supernatant, incubated on ice for 30 min and centrifuged at 12,000×g and 4°C for 10 min. The supernatant was decanted. The pellet was washed with 5 ml 70% cold ethanol and air-dried. The pellet of crude DNA extracts was re-suspended in 500 μl deionized H2O. Two volumes of ethanol were added to the supernatant, mixed and centrifuged at 13,000×g and 4°C for 30 min. The pellet was washed with 500 μl 70% cold ethanol and air-dried. The DNA was recovered in 500 μl of bi-distilled water. The DNA yield was quantified by the use of a UV Transilluminator 2000 (Gel Doc 2000, BIO-RAD Laboratories Inc., Carlsbad, CA) after 0.8% (w/v) agarose gel electrophoresis, and staining with ethidium bromide solution (0.5 μg ml−1). The 1-kb DNA ladder (Invitrogen Life Technologies Carlsbad, CA) was used as molecular size marker and compared with DNA. The DNA extract was stored at −20°C until required for PCR amplification.
PCR amplification, construction of 16S rRNA library and 16S rDNA sequencing
Purified DNA was used as template for the amplification of archaeal 16S rRNA genes via PCR. The reaction mixture (25 μl) contained 100 ng of genomic DNA; the appropriate primers at 0.5 μM each; dATP, dCTP, dCTP and dTT, each at a concentration of 200 μM; 2.5 mM MgCl; and 1 U of Taq DNA polymerase in the PCR buffer provided by the manufacturer (Invitrogen Life Technologies, Sao Paulo, Brazil). The archaeal specific primers were 25 F (5′-CYG GTT GAT CCT GCC RG-3′) and 1,492 R (5′-GGT TAC CTT GTT ACG ACT T-3′) (Dojka et al. 1998). Amplification conditions included a denaturation step for 12 min at 94°C followed by 25 cycles consisting of 1 min at 94°C, 50°C, 2 min at 72°C and 7.5 min at 72°C. The amplification was done with a Touchgene Gradient FTGRAD2D (TECHNE DUXFORT, Cambridge, UK). The expected size of the fragment amplified from the 16S rRNA gene was approximately 1,440 bp.
PCR products of 16S rRNA genes were cloned directly into the vector pCR®2.1-TOPO® by using the TOPO TA Cloning Kit (Invitrogen Life Technologies Carlsbad, CA). The positive clones were detected by the appearance of white colonies in LB plates containing 40 mg ml−1 of X-Gal (Invitrogen Life Technologies Carlsbad, CA) and 50 μg ml−1 ampicillin. Recombinant plasmids were isolated from overnight cultures by alkaline lysis (Sambrook et al. 1989) and a restriction analysis with EcoRI to detect the insertion was performed. The 16S rRNA gene sequences were obtained with an 3730X DNA Analyzer (Applied Biosystems, Foster City, CA) using M13 primers.
Molecular identification and phylogenetic analysis
All sequences obtained in this work were checked for chimeras using the CHIMERA-CHECK online analysis program of the RDP-II database (Cole et al. 2003). The partial sequences were then subjected to a BLAST search (Altschul et al. 1997) and RDP Analysis Tools of Ribosomal Database Project-II Release 9 (http://rdp.cme.msu.edu/index.jsp) to determine taxonomic hierarchy of the sequences. Multiple alignment analyses were performed with CLUSTAL X (Thompson et al. 1997) selecting related sequences from the NCBI Taxonomy Homepage (TaxBrowser) and Ribosomal Database Project-II databases. Only common 16S rRNA regions and parsimony informative sites were included in the analysis. All alignment gaps were treated as missing data. The transversion/transition weighting using the Tamura-Nei model (Tamura and Nei 1993) and the number of bases substitution between each pair of sequences was estimated using program MEGA v. 2.1 (Kumar et al. 2001). The phylogenetic trees were constructed using the Neighbor-joining method and Tamura-Nei model of distance analysis and 500 Bootstrap replications were assessed to support internal branches. Sequences that differed by less than 3% were considered to belong to the same phylotype (Stackebrandt and Goebel 1994). Sequences with similitude percentages below 95% were assigned to the closest family (Rossello-Mora and Amann 2001).
Nucleotide sequence accession numbers
The sequences were deposited in the GenBank database and assigned the accession numbers EF690556–EF690640.
Results and discussion
The total genomic DNA was extracted directly from the soil and amplified using the archeal 16S rRNA primers to investigate diversity of archaea. A fragment of approximately 1,430 nucleotides (5–1,435 region Natronomonas pharaonis D87971 numbering) was amplified and a 16S rRNA library was constructed. A total of 85 clones were obtained and sequenced. The phylogenetic analysis was done with obtained sequences and related ones obtained from the Genbank database. The phylogenetic tree was constructed using 1,317 nucleotides. The 16S rRNA gene sequence similarity percentages, microbial group affiliations and identified phylotypes are shown in Table 1.
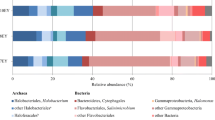
Eighty-one clones were affiliated with the Halobacteriaceae family belonging to Euryarchaeota phylum (Table 1, Fig. 1). The members of the family Halobacteriaceae are clustered in haloalkaliphilic (Natro group) and extreme halophilic (Kamekura and Dyall-Smith 1995). Eleven clones were closely related with the genera Natronococcus and Natronobacterium belonging to the haloalkaliphilic group (Table 1). Growth of the identified species is possible in these extreme soil conditions, i.e. alkalinity ranging between 9.8 and 11. “Natronococcus yunnanense”, Natronococcus amylolyticus, “Natronobacterium innermongoliae”, Natronococcus sp. and Natronobacterium sp. might be the dominant species of the archaea community in this habitat. These species were isolated previously from a soda lake in Kenya, i.e. Lake Magadi, and from a soda lake in Inner Mongolia (China), i.e. lake Hamatai (Kanal et al. 1995; Desmarais et al. 1997). They are obligate alkaliphilic and grow at pH between 7.8 and 10, a salinity of 8–30%, chemoorganotrophic and obligatory aerobic. “Natronococcus yunnanense” is a novel archaeon isolated from a salt mine in Yunnan, China (Report in NCBI taxonomy ID:405223). It appears that they can thus survive in even more extreme conditions than found in the soil of the former lake Texcoco. Natronococcus amylolyticus is capable of reducing nitrate to nitrite under aerobic conditions (Kanal et al. 1995).
Phylogenetic relationship between the 16S rRNA sequences of the clones from the library of PCR products corresponding to Archaea from alkaline-saline soil of the former lake Texcoco. The tree was constructed with sequences related to members of the family Halobacteriaceae by using the Neighbor-joining algorithm. Methanospirillum hungatei M60880 was used as the outgroup. Numbers before branch points represent percentages of bootstrap resampling based on 500 trees. Bootstrap values below 50% are not presented. The scale bar represents the expected number of substitutions averaged over all sites analyzed
Other haloalkaliphilic archaea identified were affiliated to two genera outside the Natro group, i.e. Natronolimnobius and Halalkalicoccus and only two species were identified, Natronolimnobius innermongolicus and “Halalkalicoccus jeotgali”. Natronolimnobius innermongolicus was isolated from a soda lake in Inner Mongolia (China) (Xue et al. 2005; Itoh et al. 2005). It is an aerobic chemoorganotrophic archaeon and grows at pH 9.0–9.5 and 15–20% NaCl. “Halalkalicoccus jeotgali” is a novel species isolated from saline shrimp (Report in NCBI taxonomy ID:413810).
A 9% of the clones were closely related (95.21–96.27% similarity) to the genera Natrinema, Halovivax and Natronomonas. The members of these genera are extreme halophilic and neutrophilic archaea and require high concentration of sodium to grow. Some of them can grow at pH 8–8.6 and the majority of the species have been isolated from hypersaline environments, such as saline lakes (Castillo et al. 2006), salterns (Nuttall and Dyall-Smith 1993) and salt crystals taken from the Salado formation in southeastern New Mexico (Vreeland et al. 2002). The members of these genera are strictly aerobic, chemoorganotrophic, grow in presence of glucose, fructose, lactose, urea, casaminoacids, organic acids, glycerol, etc. Some species of genus Natrinema can reduce nitrate to nitrite and nitrite to unknown end product (McGenity et al. 1998) while Natronomonas pharaonis can reduce nitrate and thiosulfate.
In this study, 70% of the clones showed less than 95% sequence similarity, making it difficult to affiliate them to a genus or species, but they could represent a novel taxon of the family of Halobacteriaceae. These clones form defined clusters in the phylogenetic tree (Fig. 1). The clusters III and IV were related to haloalkaliphilic archaea nearby Halorubrum alkaliphilum and “Halalkalicoccus jeotgali” (Hezayen et al. 2002). The clusters I, II, V and VI were distantly related to Halobiforma haloterrestris, Halovivax asiaticus, Halosimplex carlsbadense, Halobacterium noricense and Natronomonas pharaonis and could be novel extreme halophilic members of this family of archaea.
In previous studies archaea affiliated to members of genera Natronobacterium and Natronococcus were also isolated from an alkaliphilic soda lake (Duckworth et al. 1996). Another similar study, analyzed samples from a solar saltern in Spain and from an alkaline lake in Nevada (USA) and from a small pond of a slag heap of a potassium mine in Germany (Ochsenreiter et al. 2002). Only two 16S rRNA sequences were not related to haloarchaea. In our investigation, we found that the TX4CA 37 16S rRNA clone was related with clones from a methanogenic environment while the TX4CA 67–69 clones were related to uncultured crenarchaeote, apparently, not related to the indigenous haloalkalophilic community of soil of the former lake Texcoco.
The archaeal diversity was low in soil of the former lake Texcoco. Jiang et al. (2007) showed that an increase in soil salinity was accompanied by an increase in haloarchaeal diversity and a corresponding decrease in other archaeal groups and the overall archaeal diversity decreased. They found that with increased salinity the relative abundance of the Crenarchaeota decreased, but that of the Euryarchaeota increased. The Crenarchaeota phylum are generally absent in high salinity environments (Cytryn et al. 2000; Benlloch et al. 2002; Demergasso et al. 2004; Maturrano et al. 2006; Jiang et al. 2007) although Walsh et al. (2005) detected the presence of crenarchaeotal sequences in a soil with 13% salinity.
On the base of the phylogenetic affiliation of the identified archaea it is possible to predict some physiological characteristics. The archaea found in this alkaline-saline ecosystem are extreme halophilic and aerobic organisms with the capacity to grow in neutral and alkaline conditions while others are alkaline tolerant (Fig. 2). Some have the ability to reduce nitrate to nitrite under aerobic conditions with unknown end-products. In previous studies investigating dynamics of N in soil of Texcoco, large concentrations of NO −2 were detected under aerobic conditions when an easily decomposable C substrate was added (Vega-Jarquin et al. 2003; Conde et al. 2005). The production of NO −2 under these extreme alkaline saline conditions might be the product of these archaea.
In conclusion, mostly halophilic and haloalkalophilic archaea were identified in the alkaline-saline soil of the former lake Texcoco. This environment could be a source of novel species belonging to new archaeal lineages of the family of the Halobacteriaceae with still unknown physiological characteristics.
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl Acids Res 25:3389–3402
Amann RI, Ludwig W, Schleifer KH (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59:143–169
Beltrán-Hernández RI, Coss-Muñoz E, Luna-Guido ML, Mercado-García F, Siebe C, Dendooven L (1999) Carbon and nitrogen dynamics in alkaline saline soil of the former Lake Texcoco (Mexico) as affected by application of sewage sludge. Eur J Soil Sci 50:601–608
Beltrán-Hernández RI, Luna-Guido ML, Dendooven L (2007) Emission of carbon dioxide and dynamics of inorganic N in a gradient of alkaline saline soils of the former lake Texcoco. Appl Soil Ecol 35:390–403
Benlloch S, Lopez-Lopez A, Casamayor EO, Ovreas L, Goddard V, Daae FL, Smerdon G, Massana R, Joint I, Thingstad F, Pedros-Alio C, Rodriguez-Valera F (2002) Prokaryotic genetic diversity throughout the salinity gradient of a coastal solar saltern. Environ Microbiol 4:349–360
Castillo AM, Gutierrez MC, Kamekura M, Ma Y, Cowan DA, Jones BE, Grant WD, Ventosa A (2006) Halovivax asiaticus gen. nov., sp. nov., a novel extremely halophilic archaeon isolated from Inner Mongolia, China. Int J Syst Evol Micr 56:765–770
Cole JR, Chai B, Marsh TL, Farris RJ, Wang Q, Kulam SA, Chandra S, McGarrell DM, T. Schmidt M, Garrity GM, Tiedje JM (2003) The Ribosomal Database Project (RDP-II): previewing a new autoaligner that allows regular updates and the new prokaryotic taxonomy. Nucl Acids Res 31:442–443
Conde E, Cardenas M, Ponce-Mendoza A, Luna-Guido ML, Cruz-Mondragon C, Dendooven L (2005) The impacts of inorganic nitrogen application on mineralization of 14C-labelled maize and glucose, and on priming effect in saline alkaline soil. Soil Biol Biochem 37:681–691
Cytryn E, Minz D, Oremland RS, Cohen Y (2000) Distribution and diversity of archaea corresponding to the limnological cycle of a hypersaline stratified lake (Solar Lake, Sinai, Egypt). Appl Environ Microbiol 66:3269–3276
Demergasso C, Casamayor EO, Chong G, Galleguillos P, Escudero L, Pedros-Alio C (2004) Distribution of prokaryotic genetic diversity in athalassohaline lakes of the Atacama Desert, Northern Chile. FEMS Microbiol Ecol 48:57–69
Desmarais D, Jablonski PE, Fedarko NS, Roberts MF (1997) 2-Sulfotrehalose, a novel osmolyte in haloalkaliphilic archaea. J Bacteriol 179:3146–3153
Dojka MA, Harris JK, Pace NR (2000) Expanding the known diversity and environmental distribution of an uncultured phylogenetic division of Bacteria. Appl Environ Microbiol 66:1617–1621
Dojka MA, Hugenholtz P, Haack SK, Pace NR (1998) Microbial diversity in a hydrocarbon- and chlorinated-solvent-contaminated aquifer undergoing intrinsic bioremediation. Appl Environ Microbiol 64:3869–3877
Duckworth AW, Grant WD, Jones BE, van Steenbergen R (1996) Phylogenetic diversity of soda lake alkaliphiles. FEMS Microbiol Ecol 19:181–191
Feng J, Zhou P, Zhou Y-G, Liu S-J, Warren-Rhodes K (2005) Halorubrum alkaliphilum sp. nov., a novel haloalkaliphile isolated from a soda lake in Xinjiang, China. Int J Syst Evol Microbiol 55:149–152
Grant S, Sorokin DY, Grant WD, Jones BE, Heaphy S (2004) A phylogenetic analysis of Wadi el Natrun soda lake cellulase enrichment cultures and identification of cellulase genes from these cultures. Extremophiles 8:421–429
Guo C, Sun W, Harsh JB, Ogram A (1997) Hybridization analysis of microbial DNA from fuel oil-contaminated and noncontaminated soil. Microb Ecol 34:178–187
Hezayen FF, Tindall BJ, Steinbuchel A, Rehm BHA (2002) Characterization of a novel halophilic archaeon, Halobiforma haloterrestris gen. nov., sp. nov. and transfer of Natronobacterium nitratireducens to Halobiforma nitratireducens comb. nov. Int J Syst Evol Micr 52:2271–2280
Hugenholtz P, Goebel BM, Pace NR (1998) Impact of culture-independent studies on the emerging phylogenetic view of bacterial diversity. J Bacteriol 180:4765–4774
Humayoun SB, Bano N, Hollibaugh JT (2003) Depth distribution of microbial diversity in mono lake, a meromictic soda lake in California. Appl Environ Microbiol 69:1030–1042
Itoh T, Yamaguchi T, Zhou P, Takashina T (2005) Natronolimnobius baerhuensis gen. nov., sp. nov. and Natronolimnobius innermongolicus sp. nov., novel haloalkaliphilic archaea isolated from soda lakes in Inner Mongolia, China. Extremophiles 9:111–116
Jan Roblero J, Magos X, Hernández-Rodríguez C, Le Borgne S (2004) Phylogenic analysis of bacterial populations in waters of the former Texcoco Lake, Mexico. Can J Microbial 50:1049–1059
Jiang H, Dong H, Yu B, Liu X, Li Y, Ji S, Zhang CL (2007) Microbial response to salinity change in Lake Chaka, a hypersaline lake on Tibetan plateau. Environ Microbiol 9:2603–2621
Kamekura M, Dyall-Smith ML (1995) Taxonomy of the family halobacteriaceae and the description of two new genera Halorubrobacterium and Natrialba. J Gen Appl Microbiol 41:333–350
Kamekura M, Dyall-Smith ML, Upasani V, Ventosa A, Kates M (1997) Diversity of alkaliphilic halobacteria: proposals for transfer of Natronobacterium vacuolatum, Natronobacterium magadii, and Natronobacterium pharaonis to Halorubrum, Natrialba, and Natronomonas gen. nov., respectively, as Halorubrum vacuolatum comb. nov., Natrialba magadii comb. nov., and Natronomonas pharaonis comb. nov., respectively. Int J Syst Bacteriol 47:853–857
Kanal H, Kobayashi T, Aono R, Kudo T (1995) Natronococcus amylolyticus sp. nov., a haloalkaliphilic archaeon. Int J Syst Bacteriol 45:762–766
Koizumi Y, Kojima H, Oguri K, Kitazato H, Fukui M (2004) Vertical and temporal shifts in microbial communities in the water column and sediment of saline meromictic Lake Kaiike (Japan), as determined by a 16S rRNA-based analysis and related to physicochemical gradients. Environ Microbiol 6:622–637
Kumar S, Tamura K, Jakobsen I, Nei M (2001) MEGA2: molecular evolutionary genetics analysis software. Arizona State University, Tempe
Lizama C, Monteoliva-Sanchez M, Prado B, Ramos-Cormenzana A, Weckesser J, Campos V (2001) Taxonomic study of extreme halophilic archaea isolated from the “Salar de Atacama”, Chile. Syst Appl Microbiol 24:464–474
Luna-Guido ML, Beltrán-Hernández RI, Dendooven L (2001) Dynamics of 14C-labelled glucose in alkaline saline soil. Soil Biol Biochem 33:707–719
Luna-Guido ML, Beltrán-Hernández RI, Solís-Ceballos NA, Hernández-Chávez N, Mercado-García F, Catt JA, Olalde-Portugal V, Dendooven1 L (2000) Chemical and biological characteristics of alkaline saline soils from the former Lake Texcoco as affected by artificial drainage. Biol Fert Soils 32:102–108
Ma Y, Zhang W, Xue Y, Zhou P, Ventosa A, Grant W (2004) Bacterial diversity of the Inner Mongolian Baer Soda Lake as revealed by 16S rRNA gene sequence analyses. Extremophiles 8:45–51
Maturrano L, Santos F, Rossello-Mora R, Anton J (2006) Microbial diversity in Maras Salterns, a hypersaline environment in the peruvian andes. Appl Environ Microbiol 72:3887–3895
McGenity TJ, Gemmell RT, Grant WD (1998) Proposal of a new halobacterial genus Natrinema gen. nov., with two species Natrinema pellirubrum nom. nov. and Natrinema pallidum nom. nov. Int J Syst Bacteriol 48:1187–1196
Nuttall SD, Dyall-Smith ML (1993) Ch2, a novel halophilic archaeon from an Australian solar saltern. Int J Syst Bacteriol 43:729–734
Ochsenreiter T, Pfeifer F, Schleper C (2002) Diversity of Archaea in hypersaline environments characterized by molecular-phylogenetic and cultivation studies. Extremophiles 6:267–274
Rossello-Mora R, Amann R (2001) The species concept for prokaryotes. FEMS Microbiol Rev 25:39–67
Sambrook J, Fritseh EF, Maniatis T (1989) Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor Laboratory, New York, pp I.25–I.28
Stackebrandt E, Goebel BM (1994) Taxonomic note: a place for DNA–DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int J Syst Bacteriol 44:846–849
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol Biol Evol 10:512–526
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucl Acids Res 25:4876–4882
Tian X, Xu Y, Liu H, Zhou P (1997) New species of Natronobacterium. Acta Microbiol Sin 37:1–6
Tindall BJ (2004) Prokaryotic diversity in the antarctic: The Tip of the Iceberg. Microb Ecol 47:271–283
Vega-Jarquin C, Garcia-Mendoza M, Jablonowski N, Luna-Guido M, Dendooven L (2003) Rapid immobilization of applied nitrogen in saline–alkaline soils. Plant Soil 256:379–388
Vreeland R, Straight S, Krammes J, Dougherty K, Rosenzweig W, Kamekura M (2002) Halosimplex carlsbadense gen. nov., sp. nov., a unique halophilic archaeon, with three 16S rRNA genes, that grows only in defined medium with glycerol and acetate or pyruvate. Extremophiles 6:445–452
Walsh DA, Papke RT, Doolittle WF (2005) Archaeal diversity along a soil salinity gradient prone to disturbance. Environ Microbiol 7:1655–1666
Ward DM, Weller R, Bateson MM (1990) 16S rRNA sequences reveal numerous uncultured microorganisms in a natural community. Nature 345:63–65
Xue Y, Fan H, Ventosa A, Grant WD, Jones BE, Cowan DA, Ma Y (2005) Halalkalicoccus tibetensis gen. nov., sp. nov., representing a novel genus of haloalkaliphilic archaea. Int J Syst Evol Micr 55:2501–2505
Yang Y, Cui H-L, Zhou P-J, Liu SJ (2006) Halobacterium jilantaiense sp. nov., a halophilic archaeon isolated from a saline lake in Inner Mongolia, China. Int J Syst Evol Microbiol 56:2353–2355
Acknowledgments
This work was funded by “Secretaria de medio ambiente y recursos naturals” (SEMARNAT) project SEMARNAT-2004-C01-257 and “Consejo Nacional de Ciencia y Tecnología” (CONACYT) project SEP-2004-C01-479991.I.N. -G. received a postdoctoral grant from Cinvestav while C.V. -E. and R.A. -H. received grant-aided support from CONACYT.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. Horikoshi.
César Valenzuela-Encinas and Isabel Neria-González contributed equally to this publication.
Rights and permissions
About this article
Cite this article
Valenzuela-Encinas, C., Neria-González, I., Alcántara-Hernández, R.J. et al. Phylogenetic analysis of the archaeal community in an alkaline-saline soil of the former lake Texcoco (Mexico). Extremophiles 12, 247–254 (2008). https://doi.org/10.1007/s00792-007-0121-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-007-0121-y