Abstract
Replicated evidence indicates that perinatal complications are associated with increased markers of oxidative stress and with mental health problems in children. However, there are fewer reports on the impact of perinatal complications in later phases of development. We aimed to investigate the estimated effects of perinatal complications on levels of lipid peroxidation and on psychopathology in children and adolescents. The study is part of the High Risk Cohort Study for Psychiatric Disorders; the population was composed by 554 students, 6–14 years of age. Serum levels of malondialdehyde, a product of lipid peroxidation, were measured by the TBARS method. A household interview with parents and caregivers was conducted and included inquiries about perinatal history, the Child Behavior Checklist (CBCL), and parent’s evaluation, using the Mini International Psychiatric Interview (MINI). We created a cumulative risk index, conceptualized as each individual’s cumulative exposure to perinatal complications. Results indicate that perinatal complications were associated with higher levels of TBARS. After adjusting for age, gender, socio-economic status, CBCL total problems score, parental psychopathology, and childhood maltreatment, children exposed to 3 or more perinatal complications had an 26.9% (95% CI 9.9%, 46.6%) increase in TBARS levels, relative to the unexposed group. Exploratory mediation analysis indicated that TBARS levels partially mediated the association between perinatal complications and externalizing problems. In conclusion, an adverse intrauterine and/or early life environment, as proxied by the cumulative exposure to perinatal complications, was independently associated with higher levels of lipid peroxidation in children and adolescents.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The framework of developmental origins of health and disease, or Barker hypothesis, postulates that an adverse intrauterine and/or early life environment have a major impact on late-life chronic diseases [1, 2]. Indeed, perinatal complications, including, but not limited to, preterm birth, low birth weight, maternal diabetes, eclampsia, and exposure to alcohol or tobacco, have been consistently associated with substantial long-term morbidity and mortality [2–4]. Conceptually, the enduring effects of perinatal events are thought to be a result of developmental plasticity, defined as adaptive modifications to a particular environment. Multiple biological systems, such as energy metabolism, inflammation, and stress response, may mediate these responses [2, 5, 6].
Converging evidence indicates that perinatal complications are often associated with an increased production of reactive oxygen species (ROS) and/or impairment in the antioxidant defenses, resulting in oxidative damage in both the mother and the fetus [7–11]. Increased oxidant molecules (i.e., ONOO−) and markers of lipid peroxidation [i.e., thiobarbituric acid-reactive substance (TBARS)], as well as those of decreased antioxidant mediators have been reported in preterm neonates with intrauterine growth restriction [7, 11, 12]. Nonetheless, there are no reports to this date of the impact of perinatal complications on markers of oxidative stress in later phases of neurodevelopment. Moreover, multiple other factors commonly associated with perinatal complications, such as psychopathology and childhood maltreatment, have been independently associated with oxidative imbalances and could act as underlying common denominators, but their role has not been directly evaluated so far.
This study aimed to investigate the estimated effects of perinatal complications on levels of the lipid peroxidation marker TBARS. We also aimed to assess the impact of the associated variables age, gender, socio-economic status (SES), childhood maltreatment, emotional and behavioral problems, and parental psychopathology. An exploratory aim was to assess if TBARS levels mediated the association between perinatal complications and childhood psychopathology. We used data from the High Risk Cohort Study for Psychiatric Disorders (HRC Study), a large, community study, which was enriched for individual and familial psychopathology. Perinatal complications were analyzed using a cumulative risk model, which is the model with the most consistent empirical support [4, 13–16]. We hypothesized that cumulative exposure to perinatal complications would have an effect on TBARS levels; a secondary hypothesis was that TBARS levels would serve as an intermediate phenotype, mediating the relationship between perinatal complications and psychopathology.
Methods
Participants
The sample is part of the HRC Study, which included multiple investigative steps as previously described [17]. To select this study population, we used a three-stage design: first, we assessed child symptoms and family history of psychiatric disorders in a screening interview, wherein 9937 interviews (the biological mother in 88% of them) using the Family History Survey (FHS) were conducted. In the second stage, a random subsample (aimed to be representative from the community, n = 958) and a high-risk subsample (a sample with children with increased risk for mental disorders, n = 1554) were selected for further evaluation. For subjects in the random-selection stratum, a simple randomization procedure was used. The high-risk stratum was composed of children that screened positively for any of one of the five targeted domains (attention-deficit/hyperactivity disorder, anxiety, obsessive–compulsive disorder, psychosis, and learning disorders), which were selected to generate a sample with diverse psychopathology. Among those, children with higher number of family members affected were prioritized. The oversampling procedure was used to select a sample with higher family rates of mental disorders, and, therefore, to enhance the power to identify developmental trajectories and underlying pathophysiological processes.
A subsample composed of 750 children was randomly selected for blood collection. We were able to collect and process a valid blood sample from 554 children; of those, 286 (51.6%) were from the high-risk stratum. Main reasons for lack of success in blood collection were: caregiver refused the participation in this step of the study; children refused to collect blood and technical complications during blood processing procedures.
Measurements
General risk factors
Questions about risk factors were drawn based on a literature review about known major risk factors for mental disorders [17]. We assessed demographical factors (e.g., socio-economic status) and perinatal factors (e.g., gestational age at birth, exposure to tobacco or alcohol intra-utero, gestational infections, and clinical conditions). The questions about child maltreatment were selected to represent four categories of child maltreatment that have been distinguished in the literature: (a) physical abuse; (b) neglect; (c) emotional maltreatment; and (d) sexual abuse. Similar questions are frequently used in other survey research [18, 19] and are close to the constructs evaluated by other instruments that assess child maltreatment more comprehensively, such as the Childhood Trauma Questionnaire [20].
Perinatal problems index composition
We created a cumulative risk index, conceptualized as each individual’s cumulative exposure to a set of indicators of perinatal complications, according to the previous studies [4, 14, 15]. Definitions and descriptive statistics of risk factors indicators are depicted in Table 1. Each indicator was weighted equally and summed. Individuals’ exposure was categorized in four groups: (1) unexposed; (2) exposed to 1 perinatal complication; (3) exposed to 2 perinatal complications; and (4) exposed to 3 or more perinatal complications.
Child behavior checklist (CBCL)
The CBCL is a parent-report questionnaire that assesses various behavioral and emotional problems. It has been one of the most widely used standardized measures for evaluating maladaptive behavior and emotional complications in individuals with ages between 4 and 18 [21, 22]. For this study, we used the CBCL total problems score, as well as the internalizing and externalizing problems subscales.
Parental psychopathology
During the household interview, the respondent (biological mother in 91.5% of individuals) answered the Mini International Psychiatric Interview (MINI 6.0). DSM-IV algorithms were used to generate five diagnostic groups of parental psychopathology: major depressive disorder, bipolar disorder, anxiety disorders (including panic, generalized anxiety disorder, social anxiety disorder, or agoraphobia), substance use disorders (i.e. any alcohol/drug abuse or dependence), and psychotic disorders. We created a dichotomous variable indicating the presence of any parental diagnosis, defined by the presence of any of the aforementioned diagnostic categories at the time of the assessment.
Blood samples collection and TBARS assessment
Whole blood samples were obtained from all children. All samples were collected between 10:00 am and 04:00 pm. After collection, the blood was allowed to clot by leaving it undisturbed at room temperature. Serum was extracted after blood had been processed at 1000–2000×g for 10 min in a refrigerated centrifuge and was kept at −80 °C, without addition of antioxidants, for between 3 and 6 months until further analyzed. Serum levels of malondialdehyde (MDA), a product of lipid peroxidation, were measured by the TBARS method [23]. Results were expressed in micrometers of MDA. The inter- and intra-assay coefficients of variation were 5.9 and 7.4%, respectively.
Statistical analyses
For statistical analysis, the SPSS software for Windows (version 17.0) was used. For the comparison of the demographic and clinical data, the independent samples t test was used for quantitative variables; the Chi-square test was used for categorical variables. Generalized linear models were used to assess the relationship between TBARS levels and perinatal complications, after adjustment for age, gender, emotional and behavioral problems, parental psychopathology, and childhood maltreatment. As the outcome (i.e., TBARS levels) had a positively skewed distribution (many low values and few higher values), we used a model with a gamma distribution and log-link function. Due to the non-linearity of the model, the estimated β coefficients created by the model were transformed into rate ratio (RR) estimates.
For the exploratory mediation analysis, we used the Process SPSS macro developed by Preacher and Hayes [24]. This macro uses a bootstrapping resampling strategy to evaluate significance of the model and effects of mediators; for this analysis, we used 5000 bootstrap samples. A 95% confidence interval that does not include 0 was used as the criterion for indirect effects significance. Given that our cross-sectional data cannot establish temporal precedence between TBARS and children’s psychopathology, we chose, for the purposes of this study, to model TBARS as the mediator and children’s psychopathology as the outcome.
Results
Sample characteristics
The mean sample age was 10.03, SD ±1.47 (males 9.93 SD ±1.92, females 10.13 SD ±1.88); the age range was 6–14. The gender ratio was 53.4% male. There was no difference in age, gender, and SES between the perinatal complications exposure groups. There was a significant group effect on CBCL total problems (F 3,548 = 5.010; p = 0.002), internalizing (F 3,548 = 5.722; p = 0.001), and externalizing scores (F 3,548 = 3.790; p = 0.010). There were significant associations between the cumulative exposure to perinatal complications with parental psychopathology (at the time of the assessment) (χ2 = 33.261, p < 0.001) and with childhood maltreatment (χ2 = 13.823, p = 0.003) (Table 2).
Perinatal complications and TBARS levels
TBARS median level was 14.66 μM of MDA (interquartile range 9.94, 22.81). There was no correlation between TBARS levels and age (r = 0.001, p = 0.998) or gender (p = 0.274). We examined the impact of perinatal complications on TBARS levels on an unadjusted model and on a model adjusted for age, gender, SES, emotional and behavioral problems, parental psychopathology, and childhood maltreatment. Exposure to three or more perinatal complications was significantly associated with increased TBARS levels. Although slightly attenuated, this effect persisted significant in the model that included the covariates. Relative to the unexposed group, the group exposed to three or more perinatal complications was associated with a 28.8% increase in the TBARS levels in the unadjusted model, and with a 26.9% increase after adjusting for age, gender, SES, CBCL total problems score, parental psychopathology, and childhood maltreatment (Table 3).
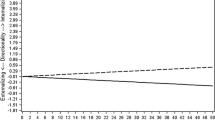
Pairwise comparison between groups suggested a stronger effect with cumulative exposure, insofar as the adjusted TBARS mean level for the group exposed to 3 or more perinatal complications (21.70 μM of MDA, 95% CI 18.36, 25.03) was significantly higher than the adjusted mean of the unexposed group (17.10 μM of MDA, 95% CI 14.50, 19.70, p = 0.010), and the adjusted mean of the group exposed to 1 perinatal complications (17.86 μM of MDA, 95% CI: 15.25; 20.47, p = 0.045). The difference between the group exposed to three or more perinatal complications and the group exposed to two was not significant (18.97 μM of MDA, 95% CI 16.15, 21.80, p = 0.471). Age and CBCL total problems score were included as covariates and all p values were adjusted with Bonferroni correction (Fig. 1).
Exploratory mediation analysis
Correlation analyses indicated that TBARS levels were positively correlated with externalizing problems (r = 0.103, p = 0.035), but not with internalizing (r = 0.071, p = 0.104) or total problems scores (r = 0.080, p = 0.063). The association between TBARS and externalizing problems remained significant after adjustment for age, gender, SES, perinatal complications, parental psychopathology, and childhood maltreatment (RR = 1.006, 95% CI 1.001, 1.011, p = 0.028).
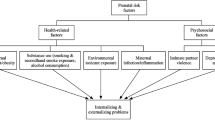
We then tested if serum TBARS levels mediated the association between perinatal complications and externalizing problems. Results showed that, after adjustment for age, gender, and SES, serum TBARS levels partially mediated the association between perinatal complications and CBCL externalizing problems score (indirect effect = 0.136, 95% CI 0.015, 0.353), accounting for approximately 14.2% of the total effect of perinatal complications on externalizing problems. Unstandardized coefficients for relations among the independent, mediating, and dependent variables are displayed in Fig. 2. The overall model was significant (F 5,523 = 2.320, R 2 = 0.022, p = 0.042).
Unstandardized regression coefficients from a bootstrap-mediation analysis, which indicated that TBARS levels partially mediated the relationship between perinatal complications and CBCL externalizing problems score. Adjusted for age, gender, and socio-economic status. *p < 0.05, **p < 0.001. TBARS Thiobarbituric acid-reactive substance, CBCL child behavior checklist
Sensitivity analyses
Twenty-four children (4.3%) in our sample were using psychotropic medications for mental health problems. There was no association between use of psychotropic medications and TBARS levels (p = 0.090). Sensitivity analyses were performed to investigate if the removal of these individuals would change the main results. After removing children using a psychotropic medication, there was still a significant effect of perinatal complications on CBCL total problems (F 3,524 = 5.147; p = 0.002), internalizing (F 3,524 = 4.694; p = 0.003), and externalizing scores (F 3,524 = 3.764; p = 0.011). Exposure to three or more perinatal complications also remained significantly associated with higher TBARS levels, relative to the unexposed group and adjusted for age, gender, SES, CBCL total problems score, parental psychopathology, and childhood maltreatment (RR 1.211, 95% CI 1.041; 1.409, p = 0.010). Pairwise comparison between groups continued to suggest a stronger effect with cumulative exposure, the adjusted TBARS mean level for the group exposed to three or more perinatal complications (20.58 μM of MDA, 95% CI 17.48, 24.24) was significantly higher than the adjusted mean of the unexposed group (16.99 μM of MDA, 95% CI 14.71, 19.62, p = 0.017), and the adjusted mean of the group exposed to 1 perinatal complications (17.61 μM of MDA, 95% CI: 15.34; 20.22, p = 0.045). Finally, the mediation analysis continued to show that TBARS partially mediated the association between perinatal complications and CBCL externalizing problems score (indirect effect = 0.288, 95% CI: 0.026; 0.744), accounting for approximately 12.8% of the total effect.
Discussion
Using data from a large, community sample of children and adolescents, we report that perinatal complications were associated with higher levels of TBARS. This effect was independent of multiple confounding factors. As expected, children exposed to cumulative perinatal complications had: (1) an increased risk of having a parent diagnosed with a mental illness at the time of assessment; (2) higher reported frequency of childhood maltreatment; and (3) higher levels of emotional and behavioral problems. Nonetheless, further analysis indicated that the inclusion of these factors as covariates did not substantially change the relationship between perinatal problems and TBARS levels.
Our results provide support for the hypothesis that the early life programing (i.e., during pregnancy and early life years) of a biological system (i.e., oxidative regulation) may have further impact on the development of the organism, including brain development. Converging findings have indicated that the early life adverse environments affect markers of inflammation [25], hypothalamic–pituitary–adrenal (HPA) axis activity [26, 27] and insulin homeostasis [28]. Interestingly, a recent study, using a similar construct of perinatal insults, documented an association between perinatal complications and markers of accelerated aging (i.e., telomere length and perceived age) in individuals at age 38 years [4]. Oxidative stress has a critical role in aging processes through a pleiotropic effect on multiple cellular mechanisms [29, 30]; our results indicate that some of these processes may be already active, and detectable, in children and adolescents.
Several mechanisms have been postulated to explain the long-term effects of perinatal complication on oxidative stress. Evidence from animal models suggests that a key consequence of the early life adversity may be the reprogramming of mitochondrial function, leading to increased production of ROS [31, 32]. This impairment may be underlied by mitochondrial DNA mutations [33]. Epigenetic modifications and changes in gene expression may also be involved, with studies showing, for example, a decreased expression of the antioxidant enzyme catalase in newborns with intrauterine growth restriction [2, 11]. Moreover, there is evidence that prenatal/obstetric complications (e.g., gestational diabetes and preterm birth) are associated with decreased antioxidant defenses, including the activities of the enzymes glutathione peroxidase and superoxide dismutase [34–36]. Interestingly, it has been reported that abnormalities in the regulation of oxidative stress are risk factors for the development of perinatal complications [36–38]; for example, altered concentrations of the ROS plasma 8-isoprostane and urinary 8-hydroxydeoxyguanosine were associated with higher risk of preeclampsia and low birth weight, respectively [37, 38]. Conversely, a separate study indicated that higher levels of antioxidants were protective of hypoxic ischemic brain injury in asphyxiated newborns [39], indicating that the presence of perinatal complication may reflect the presence of inherited inefficiencies in oxidative stress regulation. Nonetheless, there is scarce evidence regarding the role of these mechanisms on later-life oxidative balance and they should be the focus of additional investigation.
Furthermore, our exploratory analysis indicated that TBARS levels partially mediated the relationship between perinatal complications and CBCL externalizing problems score. Our results suggest that variations in lipid peroxidation could be a component of the neurobiological pathway linking the early life adverse environments to an emotional and behavioral phenotype. Notwithstanding, our mediational models indicated partial mediation; therefore, the effects of perinatal complications were not exclusively through TBARS levels. There is consistent evidence that oxidative stress is prominently involved in mental disorders pathogenesis, with multiple studies documenting abnormal levels of oxidative stress markers, including, but not limited to TBARS, in children and adults with disparate mental disorders [40–43]. There are reports that plasma markers of oxidative stress are increased, relative to healthy controls, in individuals with externalizing problems (i.e., children and adolescents with attention-deficit hyperactivity disorder; adults with intermittent explosive disorder and/or aggressive behavior) [44, 45]. It has been shown that lipid peroxidation is associated with white matter damage, which putatively could disrupt the neural circuits involved in the regulation of aggressive/confrontational behavior [46, 47]. However, there is a little evidence on the specific effects of TBARS on different pathophysiological pathways. Importantly, this was a cross-sectional study and no definitive conclusions about causality can be drawn. For the purposes of this study, we modeled TBARS levels as a causative factor of psychopathology. However, the direction of the relationship between oxidative stress and psychopathology remains unclear, as there is paucity of longitudinal studies. There is evidence that increased oxidative stress may precede the onset of mental health problems [48–50] and, conversely, may also predict loss of brain volume [51], supporting the validity of our presumed model. Nonetheless, evidence has also indicated that depressive symptoms may induce an abnormal oxidative status, suggesting that the relationship between oxidative stress and psychopathology is, in fact, bidirectional [52].
There are limitations to this study that should be noted. First, we used only one marker of oxidative stress (i.e. TBARS). Oxidative balance has numerous components, including multiple markers of ROS-induced damage, as well as antioxidant molecules. The use of a single marker, albeit useful, does not fully account for the complexity of this system. As there are no longitudinal studies evaluating the levels of TBARS in children, there are questions about its stability over time. We collected the samples in a relatively narrow period of the day; however, it is possible that the TBARS levels were affected by temporal and contextual factors. Moreover, whereas TBARS measurements have widely been used in clinical studies and we used validated and well-established methods, there are questions about its value as a biomarker, mainly due to concerns about the reliability of the assay; therefore, interpretation and comparisons of reported values should carefully consider the technical aspects of the measurement [53]. Our risk factors assessment was mostly retrospective. There is evidence that individuals with more psychopathology tend to report exposure to risk factors more frequently [54]. To assess childhood maltreatment, we used selected questions from the Childhood Trauma Questionnaire, but we did not use a fully validated instrument, and questions were answered by the parents, which can be susceptible to response bias. There are also limitations to our perinatal complications index. There are several other important determinants of environmental risk factors impact, such as the extent and the timing of exposure, which, in our cumulative composite of exposures, were only partially incorporated and were not directly and fully assessed. Conversely, the use of a risk index that incorporated multiple different events incurs in a loss of sensitivity. It is likely that, for example, the impact of prenatal alcohol exposure is different from the impact of gestational diabetes. Our process of sample selection was designed to increase the prevalence of psychopathology and likely also increased the prevalence of risk factors, when compared with the general population [17]. As in any study with selected populations, results cannot be generalized to populations with different distributions, as well as for different age groups. Our study included a wide age range (6–14). Although we statistically controlled for age and did not find any age-related effect, it is possible that other age-related variations in experience and/or biology (e.g. pubertal stage) were not captured by the statistical control. Finally, we did not account for other possible confounders, such as body mass index and sleep quality/quantity. For example, medical comorbidities are not only highly prevalent in children with mental health problems [55], but they also have been consistently documented to impact oxidative stress markers’ levels and could have had confounding, as well as putative mediating effects.
Nonetheless, this study has several strengths. We used data from a large, community-based sample, enriched for the presence of psychopathology and familial risk. We used a standardized assessment, which included structured clinical interviews, as well as detailed information about multiple risk factors. Our perinatal complications index included prenatal and neonatal complications, as it has previously been suggested [56, 57], and had a robust association with a developmental outcome (i.e., psychopathology), reinforcing its validity.
In conclusion, we present evidence that cumulative exposure to perinatal complications is independently associated with higher levels of lipid peroxidation in children and adolescents. Our findings offer insights on possible mechanisms underlying adverse intrauterine and/or the early life environment and future expression of emotional and behavior problems in children and adolescents and thereby possible targets for preventive interventions.
References
Barker DJ (2007) The origins of the developmental origins theory. J Intern Med 261:412–417
Gluckman PD, Hanson MA, Cooper C, Thornburg KL (2008) Effect of in utero and early-life conditions on adult health and disease. N Engl J Med 359:61–73
D’Onofrio BM, Class QA, Rickert ME, Larsson H, Langstrom N, Lichtenstein P (2013) Preterm birth and mortality and morbidity: a population-based quasi-experimental study. JAMA Psychiatry 70:1231–1240
Shalev I, Caspi A, Ambler A, Belsky DW, Chapple S, Cohen HJ, Israel S, Poulton R, Ramrakha S, Rivera CD, Sugden K, Williams B, Wolke D, Moffitt TE (2014) Perinatal complications and aging indicators by midlife. Pediatrics 134:e1315–e1323
Bouret S, Levin BE, Ozanne SE (2015) Gene-environment interactions controlling energy and glucose homeostasis and the developmental origins of obesity. Physiol Rev 95:47–82
deRosset L, Strutz KL (2015) Developmental origins of chronic inflammation: a review of the relationship between birth weight and C-reactive protein. Ann Epidemiol 25:539–543
Lee YS, Chou YH (2005) Antioxidant profiles in full term and preterm neonates. Chang Gung Med J 28:846–851
Howlader MZ, Parveen S, Tamanna S, Khan TA, Begum F (2009) Oxidative stress and antioxidant status in neonates born to pre-eclamptic mother. J Trop Pediatr 55:363–367
Bizon A, Milnerowicz-Nabzdyk E, Zalewska M, Zimmer M, Milnerowicz H (2011) Changes in pro/antioxidant balance in smoking and non-smoking pregnant women with intrauterine growth restriction. Reprod Toxicol 32:360–367
Ardalic D, Stefanovic A, Kotur-Stevuljevic J, Vujovic A, Spasic S, Spasojevic-Kaliomanvska V, Jelic-Ivanovic Z, Mandic-Markovic V, Mikovic Z, Cerovic N (2014) The influence of maternal smoking habits before pregnancy and antioxidative supplementation during pregnancy on oxidative stress status in a non-complicated pregnancy. Adv Clin Exp Med 23:575–583
Ferencz A, Orvos H, Hermesz E (2015) Major differences in the levels of redox status and antioxidant defence markers in the erythrocytes of pre- and full-term neonates with intrauterine growth restriction. Reprod Toxicol 53:10–14
Negi R, Pande D, Kumar A, Khanna RS, Khanna HD (2012) Evaluation of biomarkers of oxidative stress and antioxidant capacity in the cord blood of preterm low birth weight neonates. J Matern Fetal Neonatal Med 25:1338–1341
Stanton WR, McGee R, Silva PA (1991) Indices of perinatal complications, family background, child rearing, and health as predictors of early cognitive and motor development. Pediatrics 88:954–959
Flouri E, Kallis C (2007) Adverse life events and psychopathology and prosocial behavior in late adolescence: testing the timing, specificity, accumulation, gradient, and moderation of contextual risk. J Am Acad Child Adolesc Psychiatry 46:1651–1659
Gustafsson PE, San Sebastian M, Janlert U, Theorell T, Westerlund H, Hammarstrom A (2014) Life-course accumulation of neighborhood disadvantage and allostatic load: empirical integration of three social determinants of health frameworks. Am J Public Health 104:904–910
Lahat A, Van Lieshout RJ, Saigal S, Boyle MH, Schmidt LA (2015) Small for gestational age and poor fluid intelligence in childhood predict externalizing behaviors among young adults born at extremely low birth weight. Dev Psychopathol 27:181–188
Salum GA, Gadelha A, Pan PM, Moriyama TS, Graeff-Martins AS, Tamanaha AC, Alvarenga P, Krieger FV, Fleitlich-Bilyk B, Jackowski A, Sato JR, Brietzke E, Polanczyk GV, Brentani H, de Jesus Mari J, Do Rosario MC, Manfro GG, Bressan RA, Mercadante MT, Miguel EC, Rohde LA (2015) High risk cohort study for psychiatric disorders in childhood: rationale, design, methods and preliminary results. Int J Methods Psychiatr Res 24:58–73
Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H, McClay J, Mill J, Martin J, Braithwaite A, Poulton R (2003) Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 301:386–389
Kieling C, Hutz MH, Genro JP, Polanczyk GV, Anselmi L, Camey S, Hallal PC, Barros FC, Victora CG, Menezes AM, Rohde LA (2013) Gene-environment interaction in externalizing problems among adolescents: evidence from the Pelotas 1993 Birth Cohort Study. J Child Psychol Psychiatry 54:298–304
Bernstein DP, Ahluvalia T, Pogge D, Handelsman L (1997) Validity of the Childhood Trauma Questionnaire in an adolescent psychiatric population. J Am Acad Child Adolesc Psychiatry 36:340–348
Bordin IA, Rocha MM, Paula CS, Teixeira MC, Achenbach TM, Rescorla LA, Silvares EF (2013) Child behavior checklist (CBCL), Youth Self-Report (YSR) and Teacher’s Report Form (TRF): an overview of the development of the original and Brazilian versions. Cad Saude Publica 29:13–28
Ivanova MY, Dobrean A, Dopfner M, Erol N, Fombonne E, Fonseca AC, Frigerio A, Grietens H, Hannesdottir H, Kanbayashi Y, Lambert M, Achenbach TM, Larsson B, Leung P, Liu X, Minaei A, Mulatu MS, Novik TS, Oh KJ, Roussos A, Sawyer M, Simsek Z, Dumenci L, Steinhausen HC, Metzke CW, Wolanczyk T, Yang HJ, Zilber N, Zukauskiene R, Verhulst FC, Rescorla LA, Almqvist F, Weintraub S, Bilenberg N, Bird H, Chen WJ (2007) Testing the 8-syndrome structure of the child behavior checklist in 30 societies. J Clin Child Adolesc Psychol 36:405–417
Wills ED, Wilkinson AE (1966) Release of enzymes from lysosomes by irradiation and the relation of lipid peroxide formation to enzyme release. Biochem J 99:657–666
Preacher KJ, Hayes AF (2008) Asymptotic and resampling strategies for assessing and comparing indirect effects in multiple mediator models. Behav Res Methods 40:879–891
Slopen N, Loucks EB, Appleton AA, Kawachi I, Kubzansky LD, Non AL, Buka S, Gilman SE (2015) Early origins of inflammation: an examination of prenatal and childhood social adversity in a prospective cohort study. Psychoneuroendocrinology 51:403–413
Entringer S, Kumsta R, Hellhammer DH, Wadhwa PD, Wust S (2009) Prenatal exposure to maternal psychosocial stress and HPA axis regulation in young adults. Horm Behav 55:292–298
Bosch NM, Riese H, Reijneveld SA, Bakker MP, Verhulst FC, Ormel J, Oldehinkel AJ (2012) Timing matters: long term effects of adversities from prenatal period up to adolescence on adolescents’ cortisol stress response. The TRAILS study. Psychoneuroendocrinology 37:1439–1447
Entringer S, Wust S, Kumsta R, Layes IM, Nelson EL, Hellhammer DH, Wadhwa PD (2008) Prenatal psychosocial stress exposure is associated with insulin resistance in young adults. Am J Obstet Gynecol 199(498):e491–e497
Finkel D, Pedersen NL, Berg S, Johansson B (2000) Quantitative genetic analysis of biobehavioral markers of aging in Swedish studies of adult twins. J Aging Health 12:47–68
Vitale G, Salvioli S, Franceschi C (2013) Oxidative stress and the ageing endocrine system. Nat Rev Endocrinol 9:228–240
Selak MA, Storey BT, Peterside I, Simmons RA (2003) Impaired oxidative phosphorylation in skeletal muscle of intrauterine growth-retarded rats. Am J Physiol Endocrinol Metab 285:E130–E137
Peterside IE, Selak MA, Simmons RA (2003) Impaired oxidative phosphorylation in hepatic mitochondria in growth-retarded rats. Am J Physiol Endocrinol Metab 285:E1258–E1266
Simmons RA, Suponitsky-Kroyter I, Selak MA (2005) Progressive accumulation of mitochondrial DNA mutations and decline in mitochondrial function lead to beta-cell failure. J Biol Chem 280:28785–28791
Wender-Ozegowska E, Kozlik J, Biczysko R, Ozegowski S (2004) Changes of oxidative stress parameters in diabetic pregnancy. Free Radic Res 38:795–803
Falciglia HS, Johnson JR, Sullivan J, Hall CF, Miller JD, Riechmann GC, Falciglia GA (2003) Role of antioxidant nutrients and lipid peroxidation in premature infants with respiratory distress syndrome and bronchopulmonary dysplasia. Am J Perinatol 20:97–107
Turpin CA, Sakyi SA, Owiredu WK, Ephraim RK, Anto EO (2015) Association between adverse pregnancy outcome and imbalance in angiogenic regulators and oxidative stress biomarkers in gestational hypertension and preeclampsia. BMC Pregnancy Childbirth 15:189
Hsieh TT, Chen SF, Lo LM, Li MJ, Yeh YL, Hung TH (2012) The association between maternal oxidative stress at mid-gestation and subsequent pregnancy complications. Reprod Sci 19:505–512
Ferguson KK, McElrath TF, Chen YH, Loch-Caruso R, Mukherjee B, Meeker JD (2015) Repeated measures of urinary oxidative stress biomarkers during pregnancy and preterm birth. Am J Obstet Gynecol 212(208):e201–e208
Hussein MH, Daoud GA, Kakita H, Kato S, Goto T, Kamei M, Goto K, Nobata M, Ozaki Y, Ito T, Fukuda S, Kato I, Suzuki S, Sobajima H, Hara F, Hashimoto T, Togari H (2010) High cerebrospinal fluid antioxidants and interleukin 8 are protective of hypoxic brain damage in newborns. Free Radic Res 44:422–429
Brown NC, Andreazza AC, Young LT (2014) An updated meta-analysis of oxidative stress markers in bipolar disorder. Psychiatry Res 218:61–68
Black CN, Bot M, Scheffer PG, Cuijpers P, Penninx BW (2015) Is depression associated with increased oxidative stress? A systematic review and meta-analysis. Psychoneuroendocrinology 51:164–175
Flatow J, Buckley P, Miller BJ (2013) Meta-analysis of oxidative stress in schizophrenia. Biol Psychiatry 74:400–409
Cunha GR, Asevedo E, Mansur RB, Zugman A, Pan PM, Gadelha A, Belangero SI, Rizzo LB, Coelho R, Stertz L, Cogo-Moreira H, Grassi-Oliveira R, Teixeira AL, Kauer-Sant’Anna M, Mari JJ, Miguel EC, Bressan RA, Brietzke E (2015) Inflammation, neurotrophism and oxidative stress and childhood psychopathology in a large community sample. Acta Psychiatr Scand 133:122–132
Coccaro EF, Lee R, Gozal D (2016) Elevated plasma oxidative stress markers in individuals with intermittent explosive disorder and correlation with aggression in humans. Biol Psychiatry 79:127–135
Guney E, Cetin FH, Alisik M, Tunca H, Tas Torun Y, Iseri E, Isik Taner Y, Cayci B, Erel O (2015) Attention deficit hyperactivity disorder and oxidative stress: a short term follow up study. Psychiatry Res 229:310–317
Lin WM, Chen MH, Wang HC, Lu CH, Chen PC, Chen HL, Tsai NW, Su YJ, Li SH, Kung CT, Chiu TM, Weng HH, Lin WC (2014) Association between peripheral oxidative stress and white matter damage in acute traumatic brain injury. Biomed Res Int 2014:340936
Versace A, Andreazza AC, Young LT, Fournier JC, Almeida JR, Stiffler RS, Lockovich JC, Aslam HA, Pollock MH, Park H, Nimgaonkar VL, Kupfer DJ, Phillips ML (2014) Elevated serum measures of lipid peroxidation and abnormal prefrontal white matter in euthymic bipolar adults: toward peripheral biomarkers of bipolar disorder. Mol Psychiatry 19:200–208
Perez-Cornago A, Lopez-Legarrea P, de la Iglesia R, Lahortiga F, Martinez JA, Zulet MA (2014) Longitudinal relationship of diet and oxidative stress with depressive symptoms in patients with metabolic syndrome after following a weight loss treatment: the RESMENA project. Clin Nutr 33:1061–1067
Martinez-Cengotitabengoa M, Mico JA, Arango C, Castro-Fornieles J, Graell M, Paya B, Leza JC, Zorrilla I, Parellada M, Lopez MP, Baeza I, Moreno C, Rapado-Castro M, Gonzalez-Pinto A (2014) Basal low antioxidant capacity correlates with cognitive deficits in early onset psychosis. A 2-year follow-up study. Schizophr Res 156:23–29
Perkins DO, Jeffries CD, Addington J, Bearden CE, Cadenhead KS, Cannon TD, Cornblatt BA, Mathalon DH, McGlashan TH, Seidman LJ, Tsuang MT, Walker EF, Woods SW, Heinssen R (2015) Towards a psychosis risk blood diagnostic for persons experiencing high-risk symptoms: preliminary results from the NAPLS project. Schizophr Bull 41:419–428
Fraguas D, Gonzalez-Pinto A, Mico JA, Reig S, Parellada M, Martinez-Cengotitabengoa M, Castro-Fornieles J, Rapado-Castro M, Baeza I, Janssen J, Desco M, Leza JC, Arango C (2012) Decreased glutathione levels predict loss of brain volume in children and adolescents with first-episode psychosis in a two-year longitudinal study. Schizophr Res 137:58–65
Black CN, Penninx BW, Bot M, Odegaard AO, Gross MD, Matthews KA, Jacobs DR Jr (2016) Oxidative stress, anti-oxidants and the cross-sectional and longitudinal association with depressive symptoms: results from the CARDIA study. Transl Psychiatry 6:e743
Khoubnasabjafari M, Ansarin K, Jouyban A (2015) Reliability of malondialdehyde as a biomarker of oxidative stress in psychological disorders. Bioimpacts 5:123–127
van Winkel R, van Nierop M, Myin-Germeys I, van Os J (2013) Childhood trauma as a cause of psychosis: linking genes, psychology, and biology. Can J Psychiatry 58:44–51
Jerrell JM, McIntyre RS, Tripathi A (2010) A cohort study of the prevalence and impact of comorbid medical conditions in pediatric bipolar disorder. J Clin Psychiatry 71:1518–1525
Stanton WR, McGee RO, Silva PA (1989) A longitudinal study of the interactive effects of perinatal complications and early family adversity on cognitive ability. Aust Paediatr J 25:130–133
Foley DL, Thacker LR 2nd, Aggen SH, Neale MC, Kendler KS (2001) Pregnancy and perinatal complications associated with risks for common psychiatric disorders in a population-based sample of female twins. Am J Med Genet 105:426–431
Acknowledgements
This work was supported by the following Brazilian government agencies: The National Council for Scientific and Technological Development (CNPq), the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and the São Paulo Research Foundation (FAPESP). The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Mansur has received support from FAPESP, Brazil, and fellowship funding from Lundbeck, Canada. Dr. Pan has received payment for development of educational material for Janssen-Cilag and AstraZeneca and has received meeting payment expenses from Eli Lilly and Co. Dr. Salum is supported by a CAPES/FAPERGS post-doctoral scholarship. Dr. Gadelha has received support from CNPq, Brazil, and has served on the speakers’ bureau and the advisory board for Janssen-Cilag. Dr. Kauer-Sant’Anna has received research grants from CNPq–INCT–TM, CNPq Universal, CAPES, SMRI, NARSAD, Astra–Zeneca, Eli Lilly and FIPE–HCPA. Dr. Bressan has received honoraria and/or consultation fees from AstraZeneca, Janssen-Cilag, Ache, and Lundbeck ,and has received research funding from Janssen-Cilag, Lundbeck, Novartis, and Roche. He has been a shareholder of Biomolecular Technology Ltd. He is affiliated with the Schizophrenia Program at Federal University of São Paulo, which has received unrestricted educational and research support from the following pharmaceutical companies in the last three years: Janssen-Cilag, Novartis, and Lundbeck. Dr. Brietzke has received support from CNPq, FAPESP, and CAPES, Brazil, as well as and from L’Oeal/UNESCO/Brazilian Academy of Sciences For Women in Science Fellowship and has served as an advisor of Janssen-Cilag. Dr. Grassi-Oliveira, Dr. Cunha, Dr. Asevedo, Dr. Zugman, Dr. Rios, Dr. Rizzo, Dr. Manfro, Dr. Belangero, Dr. Stertz, Dr. Levandowski, and Dr. Mari report no biomedical financial interests and potential conflicts of interests.
Ethical standards
Written informed consent was provided by all parents of participants, and verbal assent was obtained from all children. The study was approved by the Ethics Committee of the University of São Paulo (IORG0004884) and was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All families were invited for an appointment with a trained psychologist and social worker, in case they were interested in receiving the results of the study evaluation. All children identified as being under the need of care were referred for clinical evaluation. Situations involving serious risk of physical or psychological harm received special attention in accordance to competent authorities’ guidelines.
Rights and permissions
About this article
Cite this article
Mansur, R.B., Cunha, G.R., Asevedo, E. et al. Perinatal complications, lipid peroxidation, and mental health problems in a large community pediatric sample. Eur Child Adolesc Psychiatry 26, 521–529 (2017). https://doi.org/10.1007/s00787-016-0914-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-016-0914-6