Abstract
Objective
To examine the contribution of premorbid function, duration of untreated psychosis (DUP), age of onset, severity of symptoms at presentation, and number of subsequent hospitalisations to the outcome of early onset schizophrenia (EOS; onset before 17th birthday).
Method
Twenty-three EOS patients (mean age at onset 15.16 ± 1.39 years) were re-assessed after a mean interval of 4 ± 1.08 years. At baseline and follow-up clinical diagnoses were confirmed using the Structured Clinical Interview for DSM-IV Axis I Disorders and symptoms were assessed with the Positive and Negative Syndrome Scale. Premorbid function, as measured with the Premorbid Adjustment Scale, age of onset and DUP were assessed at baseline only. Outcome was evaluated using the Social Adaptation Self-Evaluation Scale (SASS) and the Global Assessment of Functioning (GAF) Scale.
Results
Mean DUP was 2.95 ± 3.59 months and mean total PAS score was 6.65 ± 3.02. They had an average of 2.09 ± 1.44 hospitalisations and their mean SASS and GAF scores were 37.27 ± 6.5 and 54.19 ± 18.99, respectively. Poor childhood premorbid function and the severity of negative symptoms at baseline were correlated with worse SASS and GAF scores. No other significant associations were found.
Conclusions
Poor childhood function is the most significant predictor of outcome in EOS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Schizophrenia in childhood and adolescence (Early onset schizophrenia; EOS) lies in the same diagnostic continuum with the adult onset form [5] and shows high diagnostic stability over time [24, 35]. EOS (defined herein as schizophrenia with onset before the 17th birthday) typically runs a chronic course with while the majority (72–74%) requiring long-term psychiatric treatment [13, 28, 35]. Social function is severely impaired; patients are often unable to sustain close relationships outside their immediate family and remain financially dependent either on parents or public assistance [16, 28].
A large body of literature has focused on potential predictors and modifiers of outcome in schizophrenia. The factors identified are not always consistent across studies but premorbid function, age of onset, duration of untreated psychosis (DUP) and severity of presenting symptoms are amongst the most reliable [1, 20, 39].
EOS patients have worse premorbid function than adult onset cases [36, 37, 40] and consequently the contribution of premorbid function to outcome may be of greater magnitude for such patients. The few studies to have examined this have found that premorbid deficits are indeed predictive of poor outcome [5, 35]. With regards to age of onset, Werry et al. [41] suggested that onset of schizophrenia below the age of 12 is indeed predictive of particularly poor outcome while onset between 11 and 17 years had only a minor predictive validity. The severity of symptoms at presentation [35] and their persistence [28] have also been associated with poor outcome in EOS cases. The effect of DUP on outcome is a major current focus in research and service delivery in adult onset schizophrenia [31] but it has not been examined in EOS despite possibly being the only modifiable predictor.
The aim of this study was to examine relative contribution of premorbid function, age of onset, DUP, severity of clinical symptoms at presentation, and number of subsequent hospitalisations to psychosocial outcome in EOS. Defining and measuring outcome in mental health is more difficult than in other health areas and at present there is no agreed optimal definition [4]. Here we focus on psychosocial rather than clinical outcomes as they are more meaningful indicators of patients’ overall function [4, 42]. Furthermore, we used the Social Adaptation Self-Evaluation Scale (SASS) [8] as the main outcome measure in accordance with current views which emphasise the subjective perspective in any definition of outcome [42]. We hypothesised that given the increased premorbid deviance in EOS patients, premorbid function would be a more significant predictor of outcome in this group compared to the other potential predictors. We tested this hypothesis in our sample of 40 patients from the Maudsley Early Onset Schizophrenia study. Details of the study and the sample have been published previously [19, 26, 27, 32, 33, 40]. All patients were assessed at the beginning of the study, and 23 (57.4%) were re-evaluated after a mean interval of approximately 4 years.
Methods
Subjects
Baseline
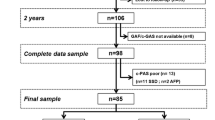
Forty adolescents (20 males) fulfilling criteria for schizophrenia according to the Diagnostic and Statistical Manual of Mental Disorders: Fourth Edition [3], were recruited from the adolescent services of the South London and Maudsley NHS Trust. Their mean age at assessment was 15.58 ± 2.27 years, their mean age of onset was 14.10 ± 2.10 years and their mean DUP was 2.9 ± 3.71 months. Only eight patients had a DUP longer than 8 months (range 8–12 months). Exclusion criteria for participants at baseline included: (a) current neurological disorder or a family history of any inherited neurological disorder, (b) history of any head injury resulting in loss of consciousness, (c) alcohol or substance abuse in the preceding 6 months, (d) any comorbid DSM-IV Axis 1 disorder. Written informed consent was obtained from all participants over the age of 16 and from the parents of those under the age of 16.
Follow-up
After a mean interval of 4.00 ± 1.08 years all patients in the initial sample were contacted for follow-up assessment. Twenty-three patients agreed to participate, 10 refused and 7 were not contactable. The majority of those who refused (n = 7) had moved to other parts of the country and were unwilling to travel, one was in a medium secure unit, one did not wish contact with psychiatric services and one did not wish to participate in our research.
Assessment
Diagnostic assessments were conducted at baseline and follow-up by qualified child and adolescent psychiatrists via personal interview using the Structured Clinical Interview for DSM-IV Axis I Disorders [14]. For patients under 16 years of age the interview was supplemented by the KID-SCID [34]. Additional information was obtained from patients’ medical records, parents, and treating physicians. Age of onset of schizophrenia was defined as the age when positive symptoms, either delusions or hallucinations first manifested. DUP was defined as the time interval between age of onset and age at initiation of antipsychotic treatment. Calculation of the number of hospitalisations during the follow-up period and information about medication was obtained from the patients, their parents and medical notes. Psychopathology at baseline and follow-up was rated with the Positive and Negative Syndrome Scale (PANSS) [25].
Premorbid functioning was assessed only at baseline using the Premorbid Adjustment Scale (PAS) [10] which has been used in other studies of EOS patients [37, 40]. The PAS measures premorbid function in age-appropriate areas: childhood (up to 11 years), early adolescence (12–15 years), late adolescence (16–18 years) and Adulthood (19 years and over). It specifically evaluates the functioning level in areas of social interaction, peer relationships, scholastic performance, and adaptation and interests by using an 8-point scale (0–7) of increasing deviance with precise anchor points. Items referring to adulthood were not included as they were not relevant to this age cohort.
Psychosocial outcome was evaluated only at follow-up using the Social Adaptation Self-Evaluation Scale (SASS) [8] and the Global Assessment of Functioning (GAF) [3]. The SASS is a 21-item, self-rated scale covering work and leisure, family relationships, social role satisfaction, intellectual interests and patients’ self perception. Each answer was rated on a 4-point scale (0–3). The SASS total score was used as the main psychosocial outcome measure while the GAF which is observer-based and more widely used, was included in order to verify the external validity of the SASS ratings.
Statistical analysis
Patients who did not participate at the follow-up were compared to those who did in terms of age of onset DUP, gender, parental socio-economic status and PANSS and PAS total scores at baseline using χ2 and Student’s t-test for categorical and continuous variables, respectively.
The total SASS score was the main outcome variable of the study. We examined the total childhood and adolescent PAS score, DUP, age of onset, the total positive and total negative PANSS score at baseline and total number of hospitalisations in the follow-up period as potential predictors of outcome. Normality of the distribution of each of these variables was confirmed using one sample Kolmogorov–Smirnov tests. Subsequently parametric Pearson’s correlations were carried out to examine the relationship of each of the predictor variables with the main outcome variable (total SASS score). Because all the correlations were hypothesis driven we did not adjust the threshold for statistical significance. In addition, in order to explore the external validity of any findings using the SASS we also examined the correlations of the same variables with the GAF total score.
Results
Patients who participated in the follow-up study did not differ from those who did not in gender distribution (Pearson’s χ2 = 0.51, df = 1, P = 0.82), PANSS and PAS mean total scores at baseline (t = −1.17, df = 33, P = 0.25 and t = −0.51, df = 33, P = 0.63, respectively), parental socio-economic status (Pearson’s χ2 = 6.62, df = 4, P = 0.15), DUP (t = 0.96, df = 33, P = 0.94) and age of onset (t = 1.92, df = 33, P = 0.63).
Demographic and clinical characteristics of follow-up sample
Thirteen male and 10 female patients were reassessed at mean ± SD age of 19.36 ± 4.21 years and the diagnosis of schizophrenia was confirmed in all of them. Their mean age of onset was 15.16 ± 1.39 years, their mean DUP was 2.95 ± 3.59 months and their mean PAS score was 6.65 ± 3.02. On average, they had 2.09 ± 1.44 hospital admissions during the follow-up period. At baseline, their mean PANSS positive, negative and general psychopathology symptom scores were 13.76 ± 4.26, 17.52 ± 8.49 and 27.47 ± 8.40, respectively. At follow-up, their mean PANSS positive, negative and general psychopathology symptom scores were 14.06 ± 6.27, 15.94 ± 8.38 and 35.33 ± 11.94, respectively. The majority was on atypical antipsychotics and 8.7% were on conventional antipsychotics. The mean SASS and GAF scores were 37.27 ± 6.5 and 54.19 ± 18.99, respectively. The total SASS and GAF scores were not significantly correlated (Pearson’s r = 0.12, P = 0.31). Eighteen still lived with their family, four lived in independent and one in supported accommodation.
Total SASS score correlated negatively with total PAS scores in childhood (Pearson’s r = −0.46) and adolescence (Pearson’s r = −0.36). The correlation with the total PAS childhood score was significant (P = 0.03) but that with the total PAS score in adolescence was not (P = 0.08). The total SASS score correlated negatively with the PANSS positive (Pearson’s r = −0.34, P = 0.10) and negative symptom score (Pearson’s r = −0.45, P = 0.04) but this reached statistical significance only for the latter. No significant correlations were found between total SASS score and age of onset of schizophrenia (Pearson’s r = −0.24, P = 0.18), DUP (Pearson’s r = −0.30, P = 0.23) or number of hospital admissions (Pearson’s r = −0.14, P = 0.28).
Figure 1 shows the correlation between total SASS and total PAS childhood scores. Despite the lack of correlation between SASS and GAF scores, their pattern of correlations with the predictor variables was very similar with one exception; GAF scores did not show a significant correlation with negative PANSS symptom scores (Pearson’s r = −0.24, P = 0.17). Otherwise, the total GAF score was also significantly negatively correlated total childhood and adolescent PAS scores (Pearson’s r = −0.44, P = 0.03 and Pearson’s r = −0.61, P = 0.003, respectively). Correlations with positive symptom scores (Pearson’s r = −0.33, P = 0.09), DUP (Pearson’s r = −0.22, P = 0.36), age of onset (Pearson’s r = −0.11, P = 0.33) and number of hospitalisations (Pearson’s r = −0.32, P = 0.08) were not significant (Fig. 2).
Discussion
Premorbid function and severity of negative symptoms at baseline were found to be predictive of 4-year psychosocial outcome in EOS. The impact of childhood premorbid function on psychosocial outcome appeared more robust as it was found to correlate with two independent measures of outcome, the SASS and the GAF. Premorbid function in adolescence correlated negatively with the total scores of both scales but this correlation was statistically significant only for the GAF. Age of onset, DUP and number of admissions during the follow-up period were not predictive of social outcome.
The lack of a significant association between adolescent PAS scores and SASS maybe due to lack of power as the correlation approached statistical significance. GAF scores were however significantly negatively correlated with PAS scores in adolescence. In a previous study, Vourdas et al. [40] compared PAS scores from the baseline EOS sample to those of patients with adult onset schizophrenia and found increased premorbid deviance in the EOS sample particularly during the adolescent period. It is possible that the increased deviance in adolescent PAS scores in EOS may actually reflect prodromal deterioration since the assessment of adolescent premorbid function was performed very closely to their illness onset when the boundaries between the premorbid and prodromal stages of schizophrenia are often unclear. Therefore, childhood PAS scores may be a more robust measure of true premorbid function and hence more predictive of social outcome. It is also important to note that in the study by Vourdas et al. childhood (and not adolescent) premorbid dysfunction was predictive of age of onset in EOS suggesting a close association between childhood premorbid function and illness course. The results of this study add to the large body of literature on the negative impact of premorbid function on the psychosocial outcome of schizophrenia and suggest the poor premorbid adjustment is a predictor of poor outcome regardless of age of onset of schizophrenia.
The level of negative symptoms at baseline as measured by the PANSS score was also significantly negatively correlated with SASS scores at follow-up. This is in line with previous reports of an independent association of negative symptoms with psychosocial functioning [28, 35] in EOS and adult onset schizophrenia [17, 22].
In our study age of onset was not predictive of psychosocial outcome. This may be because of the restricted range of this variable in our sample (from 12 to 17 years) but also supports findings by Werry et al. [41] that within the early onset group small differences in age of onset are less important determinants of outcome. Perhaps within EOS only extremely early onsets (before the age of 12) may be of further predictive significance [13].
DUP was not associated with outcome as measured by SASS and GAF. This lack of an association between DUP and outcome has been reported in several studies [6, 11, 12, 21, 23, 38]. Those that do usually find an association with poor outcome for patients with particularly long DUP (6 months or more) [2, 7, 9, 18, 29, 30] DUP was short in this study and similar to that reported in other studies from countries with public health care systems [15].
Number of hospitalisations had no predictive power although its correlation with social adaptation was negative as expected. To our knowledge no other studies have looked at this variable as a predictor of outcome in EOS. The number of hospital admissions in this sample ranged from 0 to 5; the limited range of this variable may have reduced its predictive power. However, it is more likely that hospital admissions are not a particularly useful predictor of outcome in this age group for several reasons. EOS patients live with their families and therefore they have a greater support network in the community than do adult patients. In addition in the current climate of community-based delivery of mental health services, hospitalisation is becoming a relatively uncommon occurrence.
The sample size in this study is very small. Although a lower attrition rate would have been ideal it is reassuring that there were no significant differences between those patients who participated in the follow-up compared to those who did not. The study reports on a relatively rare variant of schizophrenia (early onset schizophrenia) which is however on a neurobiological continuum with adult onset cases. Therefore, the expected number of participating patients is by necessity small. Outcome in this study was defined quite narrowly in terms of patients’ scores in a single scale (the SASS), which is a self-evaluation scale. However, the focus of the study was to assess patients’ social outcome in terms of their own perceptions The GAF scores were used to examine external validity. The correlation between the two scales was low suggesting that they tap into different aspects of outcome. However, the pattern of their correlations with possible predictors was remarkably similar. This also indicates that to a large extent several facets of outcome are inter-correlated and therefore identification of predictors may be relatively independent of the approach used to define outcome.
The association between premorbid function and outcome in schizophrenia suggests that childhood dysfunction maybe an early manifestation of the disease processes that ultimately may lead to full blown psychosis. Childhood dysfunction however is characterised by non-specific difficulties and cannot be used to predict whether an individual may develop schizophrenia or not.
References
Addington J, Addington D (2005) Patterns of premorbid functioning in first episode psychosis: relationship to 2-year outcome. Acta Psychiatr Scand 112:40–46
Altamura AC, Bassetti R, Sassella F, Salvadori D, Mundo E (2001) Duration of untreated psychosis as a predictor of outcome in first-episode schizophrenia: a retrospective study. Schizophr Res 52:29–36
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders (DSM-IV), 4th edn. American Psychiatric Association Press, Washington, DC
Andreasen NC, Carpenter WT Jr, Kane JM, Lasser RA, Marder SR, Weinberger DR (2005) Remission in schizophrenia: proposed criteria and rationale for consensus. Am J Psychiatry 162(3):441–449
Asarnow JR, Tompson MC, Goldstein MJ (1994) Childhood-onset schizophrenia: a follow-up study. Schizophr Bull 20:599–617
Barnes TRE, Hutton SB, Chapman MJ, Mutsatsa S, Puri BK, Joyce EM (2000) West London first-episode study of schizophrenia. Clinical correlates of duration of untreated psychosis. Br J Psychiatry 177:207–211
Black K, Peters L, Rui Q, Milliken H, Whitehorn D, Kopala LC (2001) Duration of untreated psychosis predicts treatment outcome in an early psychosis program. Schizophr Res 47:215–222
Bosc M, Dubini A, Polin V (1997) Development and validation of a social functioning scale, the Social Adaptation Self-evaluation Scale. Eur Neuropsychopharmacol 7(Suppl 1):S57–S70
Browne S, Clarke M, Gervin M, Waddington JL, Larkin C, O’Callaghan E (2000) Determinants of quality of life at first presentation with schizophrenia. Br J Psychiatry 176:173–176
Cannon-Spoor HE, Potkin SG, Wyatt KJ (1982) Measurement of premorbid adjustment in chronic schizophrenia. Schizophr Bull 8:470–484
Craig TJ, Bromet EJ, Fennig S, Tanenberg-Karant M, Lavelle J, Galambos N (2000) Is there an association between duration of untreated psychosis and 24-month clinical outcome in a first-admission series? Am J Psychiatry 157:60–66
de Haan L, van der Gaag M, Wolthaus J (2000) Duration of untreated psychosis and the long-term course of schizophrenia. Eur Psychiatry 15:264–267
Eggers C, Bunk D (1997) The long-term course of childhood-onset schizophrenia: a 42-year follow up. Schizophr Bull 23:105–117
First MB, Spitzer RL, Gibbon M, Williams JBW (1997) Structured clinical interview for DSM-IV Axis I disorders, research version, patient edition (SCID-I/P) Biometrics research. New York State Psychiatric Institute, New York
Friis S, Melle I, Larsen TK, Haahr U, Johannessen JO, Simonsen E, Opjordsmoen S, Vaglum P, McGlashan TH (2004) Does duration of untreated psychosis bias study samples of first-episode psychosis? Acta Psychiatr Scand 110:286–291
Gillberg IC, Hellgren L, Gillberg C (1993) Psychotic disorders diagnosed in adolescence. Outcome at age 30 years. J Child Psychol Psychiatry Allied Disciplines 34:1173–1185
Gupta S, Andreasen N, Arndt S, Flaum M, Hubbard W, Ziebell S (1997) The Iowa longitudinal study of recent onset psychosis: one-year follow-up of first episode patients. Schizophr Res 23:1–13
Haas GL, Garratt LS, Sweeney JA (1998) Delay to first antipsychotic medication in schizophrenia: impact on symptomatology and clinical course of illness. J Psychiatr Res 32:151–159
Hadjulis M, Pipe R, Frangou S (2004) Normal cerebral volume asymmetries in early onset schizophrenia. Biol Psychiatry 55:148–153
Häfner H, Nowotny B (1995) Epidemiology of early-onset schizophrenia. Eur Arch Psychiatry Clin Neurosci 245:80–92
Ho BC, Andreasen NC, Flaum M, Nopoulos P, Miller D (2000) Untreated initial psychosis: its relation to quality of life and symptom remission in first-episode schizophrenia. Am J Psychiatry 157:808–815
Ho BC, Nopoulos P, Flaum M, Arndt S, Andreasen NC (1998) Two-year outcome in first-episode schizophrenia: predictive value of symptoms for quality of life. Am J Psychiatry 155:1196–1201
Hoff AL, Sakuma M, Razi K, Heydebrand G, Csernansky JG, DeLisi LE (2000) Lack of association between duration of untreated illness and severity of cognitive and structural brain deficits at the first episode of schizophrenia. Am J Psychiatry 157:1824–1828
Hollis C (2000) Adult outcome of child- and adolescent-onset schizophrenia: diagnostic stability and predictive validity. Am J Psychiatry 157:1652–1659
Kay SR, Opler LA, Lindenmayer JP (1989) The Positive and Negative Syndrome Scale (PANSS): rationale and standardisation. Br J Psychiatry 155(Suppl 7):S59–S65
Kravariti E, Morris RG, Rabe-Hesketh S, Murray R, Frangou S (2003a) The Maudsley early onset schizophrenia study: cognitive function in adolescents with recent onset schizophrenia. Schizophr Res 61:137–148
Kravariti E, Morris RG, Rabe-Hesketh S, Murray R, Frangou S (2003b) The Maudsley early onset schizophrenia study: cognitive function in adolescent – onset schizophrenia. Schizophr Res 65:95–103
Lay B, Blanz B, Hartmann M, Schmidt MH (2000) The psychosocial outcome of adolescent onset schizophrenia: a 12-year follow-up. Schizophr Bull 26:801–816
Loebel AD, Lieberman JA, Alvir JM, Mayerhoff DI, Geisler SH, Szymanski SR (1992) Duration of psychosis and outcome in first-episode schizophrenia. Am J Psychiatry 149:1183–1188
Malla AK, Norman RM, Manchanda R, Ahmed MR, Scholten D, Harricharan R, Cortese L, Takhar J (2002) One year outcome in first episode psychosis: influence of DUP and other predictors. Schizophr Res 54:231–242
Marshall M, Lockwood A (2004) Early intervention for psychosis. Cochrane Database System Rev 2:CD004718
Matsumoto H, Simmons A, Williams S, Hadjulis M, Pipe R, Murray R, Frangou S (2001a) Superior temporal gyrus abnormalities in early-onset schizophrenia: similarities and differences with adult-onset schizophrenia. Am J Psychiatry 158:1299–1304
Matsumoto H, Simmons A, Williams S, Pipe R, Murray R, Frangou S (2001b) Structural magnetic imaging of the hippocampus in early onset schizophrenia. Biol Psychiatry 49:824–831
Matzner F, Silva R, Silvan M, Chowdhury M, Nastasi L (1997) Preliminary test–retest reliability of the KID-SCID. In: 42nd annual meeting of the American academy of child and adolescent psychiatry, New Orleans, USA
Maziade M, Bouchard S, Gingras N, Charron L, Cardinal A, Roy M-A, Gauthier B, Tremblay G, Cote S, Fournier C, Boutin P, Hamel M, Merette C, Martinez M (1996) Long term stability of diagnosis and symptom dimensions in a systematic sample of patients with onset of schizophrenia in childhood and early adolescence. II: positive/negative distinction and childhood predictors of adult outcome. Br J Psychiatry 169:371–378
McClellan J, Breiger D, McCurry C, Hlastala SA (2003) Premorbid functioning in early-onset psychotic disorders. J Am Acad Child Adolesc Psychiatry 42:666–672
Nicolson R, Lenane M, Singaracharlu S, Malaspina D, Giedd JN, Hamburger SD, Gochman P, Bedwell J, Thakar GK, Fernandez T, Wudarsky M, Hommer DW, Rapoport JL (2000) Premorbid speech and language impairments in childhood – onset schizophrenia: association with risk factors. Am J Psychiatry 157:794–800
Norman RMG, Townsend L, Malla AK (2001) Duration of untreated psychosis and cognitive functioning in first-episode patients. Br J Psychiatry 179:340–345
Robinson DG, Woerner MG, McMeniman M, Mendelowitz A, Bilder RM (2004) Symptomatic and functional recovery from a first episode of schizophrenia or schizoaffective disorder. Am J Psychiatry 161:473–479
Vourdas A, Pipe R, Corrigal R, Frangou S (2003) Increased developmental deviance and premorbid dysfunction in early onset schizophrenia. Schizophr Res 62:13–22
Werry JS, McClellan JM, Andrews LK, Ham M (1994) Clinical features and outcome of child and adolescent schizophrenia. Schizophr Bull 20:619–630
WHOQOL Group (1995) The World Health Organization Quality of Life Assessment (the WHOQOL): position paper from the World Health Organisation. Soc Sci Med 41:1403–1409
Acknowledgements
This study was supported by a research grant from the Stanley Medical Research Institute to Dr S. Frangou.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vyas, N.S., Hadjulis, M., Vourdas, A. et al. The Maudsley early onset schizophrenia study. Eur Child Adolesc Psychiatry 16, 465–470 (2007). https://doi.org/10.1007/s00787-007-0621-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-007-0621-4