Abstract
Objective
The aim of this study was to evaluate and compare early postoperative discomfort and wound healing outcomes between patients who underwent periodontal surgery with and without enamel matrix derivative (EMD), using retrospective questionnaires and postoperative clinical examinations.
Materials and methods
All enrolled patients filled out the self-report questionnaire after suture removal. The severity and duration of subjective pain and swelling were evaluated using visual analog scale (VAS) scores and wound healing parameters (dehiscence/fenestration, spontaneous bleeding, persistent swelling, and ulceration). Chi-squared tests, two-tailed independent t tests, analysis of variance, and multiple logistic regression analysis were performed to identify significant differences between the two groups (surgery with EMD and surgery without EMD).
Results
The severity of pain and swelling did not differ between patients who underwent surgery with and without EMD, but the durations of pain (P < 0.001) and swelling (P = 0.019) were significantly lower in patients who underwent surgery with EMD. Multivariate analysis with adjustment for confounding variables showed that wound healing outcomes including dehiscence/fenestration, spontaneous bleeding, and ulceration did not differ significantly between the two groups, and only persistent swelling showed significant differences (odds ratio 4.03, 95% CI 1.17–13.78; P = 0.026).
Conclusions
No difference was observed in the severity of early postoperative discomfort and wound healing outcomes between patients who underwent surgery with and without EMD, but shorter durations of postoperative pain and swelling were evident in patients who underwent surgery with EMD.
Clinical relevance
Adjunctive EMD used in periodontal surgery has clinical advantages for reducing the durations of postoperative pain and swelling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Postoperative discomfort is a complex emotional and sensory experience, and the process of soft and hard tissue wound healing is also multifactorial [1, 2]. Postoperative discomfort and delayed wound healing are known to be influenced by a combination of emotional, clinical, and iatrogenic factors such as stress, anxiety, previous experience, surgery method, surgery time, and the surgeon’s operative experience and skill [3,4,5]. Periodontal soft and hard tissue damage during surgical treatment and manipulation directly stimulates the nociceptor terminals in the peripheral tissue and releases inflammatory cytokines and chemokines, which are the major causes of early discomfort and delayed wound healing after periodontal surgery [5, 6].
Periodontal disease is a representative oral inflammatory disease and is a known risk factor for various systemic diseases [7,8,9]. Patients with periodontal diseases must be treated with antimicrobial, non-surgical, surgical, or combined approaches; most patients with severe periodontal disease require surgical treatment [10]. Proper wound healing and a reduction in the severity and duration of postoperative discomfort play a crucial role in increasing patient compliance, facilitating long-term supportive periodontal therapy, and obtaining positive treatment outcomes and prognosis of chronic periodontal disease progression [11]. Therefore, various periodontal surgical techniques and procedures are gradually being developed to minimize discomfort and periodontal tissue damage as much as possible [12, 13].
Periodontal surgery with adjunctive enamel matrix derivative (EMD) is a conventional treatment modality for periodontal regeneration, mainly for deep intrabony defects and gingival recession [14, 15]. EMD induces and promotes new attachment and regeneration, including the adhesion, migration, proliferation, and differentiation of cells in periodontal tissues [16, 17]. In addition, amelogenin, a major component of EMD, exerts synergistic antibacterial effects on periodontal pathogenic bacteria, such as Porphyromonas gingivalis and Prevotella intermedia, and facilitates soft and hard tissue wound healing [18, 19].
Although adjunctive EMD used in periodontal surgery improves soft and hard tissue wound healing and reduces postoperative discomfort, such as pain and swelling, few clinical trials have evaluated its clinical effectiveness [20, 21]. A double-masked, split-mouth, placebo-controlled, randomized study demonstrated the positive effect of topically applied EMD on the early periodontal soft tissue wound healing, according to the proportion of a visual analog scale (VAS) score reported by patients [20]. Tonetti et al. found that soft tissue densities in subjects treated with EMD were significantly higher than those in the control group within 6 weeks postoperatively [21]. These findings indicate an earlier gain in soft tissue density following the application of EMD and an enhancement in both subjective and objective outcomes.
The purpose of this study was to evaluate and compare early postoperative discomfort and wound healing outcomes between patients who underwent periodontal surgery with and without EMD, using retrospective questionnaires and postoperative clinical examinations.
Material and methods
Patients
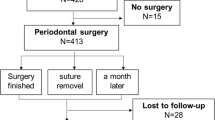
A total of 365 patients who underwent comprehensive periodontal surgery were recruited from the Department of Periodontology at Daejeon Dental Hospital, Wonkwang University from January 2017 to December 2017, and only one surgical site (premolar and molar region, including single or multiple defects) per patient was enrolled. The following inclusion criteria were applied: (1) age ≥ 20 years, (2) male or female with/without smoking history, (3) being healthy or having well-controlled systemic conditions or illnesses, (4) severe chronic periodontitis (surgical site with pocket probing depth [PPD] > 5 and clinical attachment loss [CAL] > 3 mm), (5) completed pre-surgical treatment (scaling and root planning) at the surgical site, and (6) surgical site including two-/three-wall intrabony defect and/or class I/II furcation involvement. The following exclusion criteria were established: (1) being a pregnant or breastfeeding female; (2) etiological periodontal surgery completed within 6 months at the same site; (3) surgical site including one-wall intrabony defect and/or class III furcation involvement; (4) horizontal and/or vertical defects with full probing depth and apical involvement including those with a hopeless prognosis; (5) signs of clinical infection including fistulae, suppuration, fever, severe pain, and swelling at the surgical wound site; and (6) periodontal surgery including gingivectomy, hard and soft tissue augmentation, and peri-implant treatment.
The study was approved by the Institutional Review Board (IRB) of Daejeon Dental Hospital, Wonkwang University (approval no. W1807/001-001). All enrolled patients provided written fully informed consent in accordance with the IRB guidelines, and the study was conducted in accordance with the Declaration of Helsinki and the Guidelines on Good Clinical Practice [22]. Because this study was designed as a retrospective case-control study, it was written according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE).
Study design
This retrospective case-control study was conducted using G*Power software, version 3.01 (Franz Faul, Christian-Albrechts-Universität Kiel, Kiel, Germany). To detect the medium effect size of Cohen’s f = 0.5, with α = 0.05 and power β = 0.8, 128 participants (each sample group size = 64) were required [23]. Out of 365 patients who underwent surgery with and without EMD, 118 were excluded for the following reasons: periodontal surgery completed within 6 months at the same site (n = 11), full probing depth and apical involvement (n = 40), signs of clinical infection (n = 6), and periodontal surgery including gingivectomy, hard and soft tissue augmentation, and peri-implant treatment (n = 61). Finally, from the 247 remaining eligible patients, 184 were assigned to two sex-matched groups (patients who underwent surgery with EMD [n = 92] and without EMD [n = 92]) using the Statistical Analysis System (version 9.4, SAS Institute, Cary, NC, USA) (Fig. 1).
Surgical procedure
All periodontal surgery was performed by one board-certified periodontist (JHL). Patients were administered an antibiotic (netilmicin 50 mg/2 mL) and analgesic (diclofenac 90 mg/2 mL) injection 30 min before periodontal surgery and provided with the following postoperative medication and mouthwash to be used for 5 days after periodontal surgery: antibiotics (amoxicillin 500 mg tid), analgesics (ibuprofen 200 mg tid), and antimicrobial rinse (GUM Activital, Sunstar, Osaka, Japan). The local anesthetic used was 2% lidocaine HCl with 1:100,000 epinephrine (Yuhan, Seoul, South Korea), and the full-thickness flap was elevated minimally to expose horizontal and/or vertical bone loss by using #12, 15, and 15c blades and an Orban knife (Hu-Friedy, Chicago, IL, USA). Granulated tissues were removed, and the exposed root surface was carefully debrided by hand (standard and mini Gracey curettes; Hu-Friedy) and using ultrasonic instruments (KaVo, Biberach, Germany). Patients who underwent surgery with EMD were only subjected to root conditioning with tetracycline (Chong-Kun-Dang, Seoul, South Korea) at a concentration of 50 mg/mL; the solution was applied to the exposed root surfaces and defects for 2 min using cotton pledgets. Subsequently, EMD (Straumann® Emdogain, Straumann, Basel, Switzerland) was applied according to the manufacturer’s instructions, as follows. The opened flap was repositioned, and tension-free primary flap closure was achieved using modified horizontal mattress and interrupted sutures with a non-absorbable polytetrafluoroethylene monofilament (Biotex®, Purgo, Seongnam, South Korea). After 2 weeks, sutures were removed, and all patients were instructed to use soft tooth cleaning in the surgical area. Routine clinical follow-up of prophylaxis were scheduled at 3, 6, and 12 months.
Self-reported questionnaire and clinical examination
All enrolled patients filled out the self-report questionnaire after suture removal. The severity of subjective pain and swelling during the 2 weeks after periodontal surgery was evaluated using a 100-mm VAS score, ranging from 0 points (no pain and swelling) to 10 points (worst pain and swelling imaginable). The durations of subjective pain and swelling were also investigated. During suture removal and postoperative clinical examination, wound healing parameters including dehiscence/fenestration, spontaneous bleeding, persistent swelling (swelling was still observed 2 weeks after periodontal surgery), and ulceration were evaluated by one investigator (JHL) using a dental mirror, explorer, and community periodontal index probe (0.5 mm in diameter).
Statistical analysis
Each patient was considered as statistical unit. Chi-squared tests, two-tailed independent t tests, and one-way analysis of variance were used to identify significant differences in categorical and continuous sociodemographic (sex, age, residence area, and smoking and drinking status), clinical (surgical arch, defects, PPD, CAL, recession, full-mouth bleeding on probing score [FMBS], full-mouth plaque score [FMPS], and operation time), and comorbid (hypertension and diabetes mellitus) characteristics of patients who underwent surgery with and without EMD. The odds ratios (ORs) and 95% confidence intervals (CIs) were calculated in the patients who underwent periodontal surgery with and without EMD using multiple logistic regression models while adjusting for sociodemographic, clinical, and comorbid factors. In all assessments performed in this study, a P value < 0.05 was considered statistically significant.
Results
Baseline characteristics of patients
The 184 patients included based on the inclusion and exclusion criteria comprised 102 (55.4%) men and 82 (44.6%) women with a mean age of 49.9 years (range, 22 to 75 years). No statistically significant difference was observed in sociodemographic, clinical, or comorbidity factors between patients who underwent surgery with and without EMD. Those aged 60 to 69 years (n = 36) accounted for 19.6% of the eligible patients, 162 patients (88.0%) resided in urban areas, 152 were non-smokers, and 105 drank alcohol 0–1 times/week. Periodontal flap surgery was performed almost equally for the maxilla (n = 97, 52.7%) and the mandible (n = 87, 47.3%). Baseline periodontal defects (number of patients with the most severe intrabony defects and/or furcation involvements) and clinical parameters (including average PPD, average CAL, average recession, full-mouth bleeding on probing score, and full-mouth plaque score) at the surgical site were not significantly different between the two groups. Operation times for patients who underwent surgery with and without EMD were 40.5 ± 15.1 min (mean ± standard deviation) and 43.3 ± 10.9 min, respectively, and no statistically significant difference was observed between the groups. Hypertension and diabetes mellitus also showed no statistically significant difference between the groups (Table 1).
Postoperative discomfort
Severity of pain (VAS value difference [Df] = 0.18, 95% CI = − 0.53–0.97; P = 0.614) and swelling (Df = 0.32, 95% CI = − 0.34–1.00; P = 0.341) did not differ between patients who underwent surgery with and without EMD, but the durations of pain (Df = 1.09, 95% CI = 0.51–1.68; P < 0.001) and swelling (Df = 0.54, 95% CI = 0.08–0.99; P = 0.019) were significantly lower in patients who underwent surgery with EMD than in those who underwent surgery without EMD (Fig. 2).
Comparison of early postoperative discomfort between patients who underwent surgery with and without enamel matrix derivative during the 2 weeks after periodontal surgery. (a, b The severity of subjective pain (evaluated using a 100-mm visual analog scale) and swelling, respectively. c, d The durations of subjective pain and swelling, respectively. Statistically significant differences between the two groups were determined using a two-tailed independent t test (*P < 0.001; **P < 0.05)
Wound healing outcomes
Among the patients who underwent surgery with EMD, dehiscence/fenestration occurred in 7 patients (7.6%), spontaneous bleeding occurred in 13 patients (14.1%), persistent swelling occurred in 5 patients (5.4%), and ulceration occurred in 5 patients (5.4%). Among patients who underwent surgery without EMD, dehiscence/fenestration occurred in 9 patients (9.8%), spontaneous bleeding occurred in 19 patients (20.7%), persistent swelling occurred in 15 patients (16.3%), and ulceration occurred in 7 patients (7.6%), respectively. No statistically significant differences were observed in any clinical complications between the two groups, except persistent swelling which had a higher frequency in patients who underwent surgery without EMD (P = 0.006) (Table 2).
As indicated in Fig. 3, in a multivariate analysis of confounding variables that were considered to affect wound healing outcomes, including sex, age, smoking status, and surgical arch and segment, dehiscence/fenestration (OR = 0.64, 95% CI = 0.15–2.59; P = 0.533), spontaneous bleeding (OR = 1.24, 95% CI = 0.47–3.27; P = 0.661), and ulceration (OR = 1.21, 95% CI = 0.25–5.82; P = 0.805) did not differ significantly between patients who underwent surgery with and without EMD, and only persistent swelling was clearly significant (OR = 4.03, 95% CI = 1.17–13.78; P = 0.026).
Comparison of early wound healing outcomes between patients who underwent surgery with and without enamel matrix derivative in multivariate logistic regression analysis adjusted for sociodemographic (sex, age, residence area, and smoking and drinking status), clinical (surgical arch and segment, and operation time), and comorbidity (hypertension and diabetes mellitus) factors. Data are odds ratios (95% confidence intervals) and P values (*P < 0.05)
Discussion
From the perspective of uncomplicated and rapid soft tissue wound healing after tissue damage during surgery, new capillary ingrowth and revascularization are crucial prerequisites for the early formation of granulation tissue, supply of nutrients and oxygen, growth factors, and pro- and anti-inflammatory cytokines and chemokines including interleukin (IL)-1β, IL-6, and tumor necrosis factor α [24]. Conversely, defective or absent angiogenesis may lead to delayed wound healing, such as dehiscence/fenestration, spontaneous bleeding, persistent swelling, and ulceration, and also increase the severity and duration of postoperative discomfort [25, 26].
Little research has been conducted on the perception of postoperative discomfort associated with periodontal regeneration, and discomfort following periodontal surgery has not yet been clearly established. Postoperative discomfort is most likely to occur as soon as the local anesthetic effect disappears; the maximum severity of pain and swelling occurs at 24 to 48 h, and most of the pain and swelling reduces or is eliminated within 1 to 2 weeks after periodontal surgery [5, 27]. Early reduction in postoperative discomfort, within 1–2 weeks, is considered to be the main cause of retention and stabilization of blood clots, which protects the periosteum and denuded tissue and promotes revascularization and angiogenesis [28].
This study showed that the durations of pain (Df = 1.09, 95% CI = 0.51–1.68; P < 0.001) and swelling (Df = 0.54, 95% CI = 0.08–0.99; P = 0.019) were significantly lower in patients who underwent surgery with EMD than in those who underwent surgery without EMD. In a multivariate analysis of confounding variables that were considered affecting wound healing outcomes, including sex, age, smoking, and surgical arch and segment, only persistent swelling for more than 2 weeks was clearly significant (OR = 4.03, 95% CI = 1.07–13.78; P = 0.026) [29,30,31].
We also compared the duration and severity of early postoperative discomfort according to sex and age (between those aged 20–49 years and 50–79 years) but did not find any significant difference between patients who underwent surgery with and without EMD (data not shown). These results are in contrast to previous findings, which indicated that females and younger people have lower thresholds of pain and higher pain sensitivity than males and elderly people [32]. Generally, because the severity of pain after periodontal surgery is not so severe as to exceed the threshold, no significant difference is considered to exist according to sex or age. None of the enrolled patients who underwent surgery with and without EMD showed critical or infectious signs such as fistulae, suppuration, or persisting severe pain and swelling. These findings are consistent with previous studies showing that the overall incidence of postoperative pain and infection is low and the severity is also mild [11, 33].
EMD has been used widely in clinical practice without severe side effects, but its biological and underlying mechanisms are still not fully understood [34]. EMD, which interacts with periodontal ligament mesenchymal stem cells, has been suggested to increase intracellular adenosine 3′,5′-monophosphate (cyclic AMP) levels and autocrine production, which initiates regeneration processes including osteogenesis, dentinogenesis, and cementogenesis [35]. Yuan et al. reported that EMD stimulates angiogenic factor production of periodontal ligament progenitors in animal models, thereby directly and/or indirectly promoting angiogenesis, thereby improving induced wound healing after periodontal surgery [36]. Schlueter et al. also hypothesized that EMD stimulates microvascular endothelial cell proliferation and chemotaxis and thereby further promotes wound healing and tissue remodeling [37].
The vascular endothelial growth factor (VEGF) family, including angiotensin II, insulin-like growth factor, epidermal growth factor, platelet-derived growth factor, and transforming growth factors (TGF) α and β, plays a critical role in increasing and promoting angiogenesis [38, 39]. Van der Pauw et al. reported that when EMD was applied to periodontal soft tissue, the production of VEGF, particularly TGF-β1, increased 3-fold. Schlueter et al. also showed that the secretion of TGF-β1 by periodontal ligament cells also increased 2.5-fold to 3.8-fold [37, 40]. Therefore, a reduction in the duration of pain and swelling and improved early wound healing outcomes after adjunctive application of EMD are considered to be closely related to improvement in the production and secretion of TGF-β1 and sequentially promoting angiogenesis and revascularization.
This study had some limitations. First, posthoc sample size calculations were performed and efforts were made to reduce the selection bias by enrolling an equal number of sex-matched patients, but the present study could not be free from the fundamental limitation of potential inherent bias. Due to the retrospective nature of this study, intraexaminer and interexaminer reliability and repeatability could not be assessed. Therefore, results may be subjectively biased and should be interpreted cautiously. Second, the sample included only patients who underwent periodontal surgery without bone grafting. Further research is necessary to evaluate the postoperative discomfort and wound healing outcomes including implant treatment and soft tissue augmentation with bone grafting. The other major limitation was the potential for memory bias in patients with pain and swelling. Because the memory of discomfort is highly subjective and not always reliable, it is considered to be more circumspect, and caution is required in interpreting the results.
Despite the limitations, this study revealed that no significant difference was present in the severity of early postoperative discomfort and wound healing outcomes between patients who underwent surgery with and without EMD, but that the durations of postoperative pain and swelling were shorter in patients who underwent surgery with EMD, which is a clear clinical advantage. Additional prospective clinical trials with large samples of patients who underwent surgery with EMD are therefore recommended to elucidate and clarify the positive effects of EMD, such as reduced discomfort and accelerated wound healing.
References
Fardal O, McCulloch CA (2012) Impact of anxiety on pain perception associated with periodontal and implant surgery in a private practice. J Periodontol 83:1079–1085. https://doi.org/10.1902/jop.2011.110562
Kim S, Lee YJ, Lee S, Moon HS, Chung MK (2013) Assessment of pain and anxiety following surgical placement of dental implants. Int J Oral Maxillofac Implants 28:531–535. https://doi.org/10.11607/jomi.2713
Griffin TJ, Cheung WS, Zavras AI, Damoulis PD (2006) Postoperative complications following gingival augmentation procedures. J Periodontol 77:2070–2079. https://doi.org/10.1902/jop.2006.050296
Weisensee W, Scheer M, Muller L, Rothamel D, Kistler F, Bayer G et al (2012) Impact of anxiety parameters on prospective and experienced pain intensity in implant surgery. Implant Dent 21:502–506. https://doi.org/10.1097/ID.0b013e3182703a44
Mei CC, Lee FY, Yeh HC (2016) Assessment of pain perception following periodontal and implant surgeries. J Clin Periodontol 43:1151–1159. https://doi.org/10.1111/jcpe.12618
Oswal S, Ravindra S, Sinha A, Manjunath S (2014) Antibiotics in periodontal surgeries: a prospective randomised cross over clinical trial. J Indian Soc Periodontol 18:570–574. https://doi.org/10.4103/0972-124X.142443
Lee JH, Lee JS, Park JY, Choi JK, Kim DW, Kim YT, Choi SH (2015) Association of lifestyle-related comorbidities with periodontitis: a Nationwide cohort study in Korea. Medicine (Baltimore) 94:e1567. https://doi.org/10.1097/MD.0000000000001567
Lee JH, Oh JY, Youk TM, Jeong SN, Kim YT, Choi SH (2017) Association between periodontal disease and non-communicable diseases: a 12-year longitudinal health-examinee cohort study in South Korea. Medicine (Baltimore) 96:e7398. https://doi.org/10.1097/MD.0000000000007398
Lee JH, Choi JK, Jeong SN, Choi SH (2018) Charlson comorbidity index as a predictor of periodontal disease in elderly participants. J Periodontal Implant Sci 48:92–102. https://doi.org/10.5051/jpis.2018.48.2.92
Smiley CJ, Tracy SL, Abt E, Michalowicz BS, John MT, Gunsolley J, Cobb CM, Rossmann J, Harrel SK, Forrest JL, Hujoel PP, Noraian KW, Greenwell H, Frantsve-Hawley J, Estrich C, Hanson N (2015) Systematic review and meta-analysis on the nonsurgical treatment of chronic periodontitis by means of scaling and root planning with or without adjuncts. J Am Dent Assoc 146:508–524 e505. https://doi.org/10.1016/j.adaj.2015.01.028
Eli I, Schwartz-Arad D, Baht R, Ben-Tuvim H (2003) Effect of anxiety on the experience of pain in implant insertion. Clin Oral Implants Res 14:115–118
Cortellini P, Tonetti MS (2009) Improved wound stability with a modified minimally invasive surgical technique in the regenerative treatment of isolated interdental intrabony defects. J Clin Periodontol 36:157–163. https://doi.org/10.1111/j.1600-051X.2008.01352.x
Cortellini P (2012) Minimally invasive surgical techniques in periodontal regeneration. J Evid Based Dent Pract 12:89–100. https://doi.org/10.1016/S1532-3382(12)70021-0
Heden G, Wennstrom JL (2006) Five-year follow-up of regenerative periodontal therapy with enamel matrix derivative at sites with angular bone defects. J Periodontol 77:295–301. https://doi.org/10.1902/jop.2006.050071
Sculean A, Chiantella GC, Arweiler NB, Becker J, Schwarz F, Stavropoulos A (2008) Five-year clinical and histologic results following treatment of human intrabony defects with an enamel matrix derivative combined with a natural bone mineral. Int J Periodontics Restorative Dent 28:153–161
Izumi Y, Aoki A, Yamada Y, Kobayashi H, Iwata T, Akizuki T, Suda T, Nakamura S, Wara-Aswapati N, Ueda M, Ishikawa I (2011) Current and future periodontal tissue engineering. Periodontol 56:166–187. https://doi.org/10.1111/j.1600-0757.2010.00366.x
Miron RJ, Sculean A, Cochran DL, Froum S, Zucchelli G, Nemcovsky C, Donos N, Lyngstadaas SP, Deschner J, Dard M, Stavropoulos A, Zhang Y, Trombelli L, Kasaj A, Shirakata Y, Cortellini P, Tonetti M, Rasperini G, Jepsen S, Bosshardt DD (2016) Twenty years of enamel matrix derivative: the past, the present and the future. J Clin Periodontol 43:668–683. https://doi.org/10.1111/jcpe.12546
Bosshardt DD (2008) Biological mediators and periodontal regeneration: a review of enamel matrix proteins at the cellular and molecular levels. J Clin Periodontol 35:87–105. https://doi.org/10.1111/j.1600-051X.2008.01264.x
Isehed C, Svenson B, Lundberg P, Holmlund A (2018) Surgical treatment of peri-implantitis using enamel matrix derivative, an RCT: 3- and 5-year follow-up. J Clin Periodontol 45:744–753. https://doi.org/10.1111/jcpe.12894
Wennstrom JL, Lindhe J (2002) Some effects of enamel matrix proteins on wound healing in the dento-gingival region. J Clin Periodontol 29:9–14
Tonetti MS, Fourmousis I, Suvan J, Cortellini P, Bragger U, Lang NP, on behalf of the European Research Group on Periodontology (ERGOPERIO) (2004) Healing, post-operative morbidity and patient perception of outcomes following regenerative therapy of deep intrabony defects. J Clin Periodontol 31:1092–1098. https://doi.org/10.1111/j.1600-051X.2004.00615.x
Morris K (2013) Revising the Declaration of Helsinki. Lancet 381:1889–1890
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Rincon JC, Haase HR, Bartold PM (2003) Effect of Emdogain on human periodontal fibroblasts in an in vitro wound-healing model. J Periodontal Res 38:290–295
Tonnesen MG, Feng X, Clark RA (2000) Angiogenesis in wound healing. J Investig Dermatol Symp Proc 5:40–46. https://doi.org/10.1046/j.1087-0024.2000.00014.x
Folkman J, Shing Y (1992) Angiogenesis. J Biol Chem 267:10931–10934
Burkhardt R, Hammerle CH, Lang NP, Research Group on Oral Soft Tissue B, Wound H (2015) Self-reported pain perception of patients after mucosal graft harvesting in the palatal area. J Clin Periodontol 42:281–287. https://doi.org/10.1111/jcpe.12357
Weisel JW (2007) Structure of fibrin: impact on clot stability. J Thromb Haemost 5(Suppl 1):116–124. https://doi.org/10.1111/j.1538-7836.2007.02504.x
Canakci CF, Canakci V (2007) Pain experienced by patients undergoing different periodontal therapies. J Am Dent Assoc 138:1563–1573
Urban T, Wenzel A (2010) Discomfort experienced after immediate implant placement associated with three different regenerative techniques. Clin Oral Implants Res 21:1271–1277. https://doi.org/10.1111/j.1600-0501.2010.01943.x
Tan WC, Krishnaswamy G, Ong MM, Lang NP (2014) Patient-reported outcome measures after routine periodontal and implant surgical procedures. J Clin Periodontol 41:618–624. https://doi.org/10.1111/jcpe.12248
Canakci V, Canakci CF (2007) Pain levels in patients during periodontal probing and mechanical non-surgical therapy. Clin Oral Investig 11:377–383. https://doi.org/10.1007/s00784-007-0126-z
Matthews DC, McCulloch CA (1993) Evaluating patient perceptions as short-term outcomes of periodontal treatment: a comparison of surgical and non-surgical therapy. J Periodontol 64:990–997. https://doi.org/10.1902/jop.1993.64.10.990
Cattaneo V, Rota C, Silvestri M, Piacentini C, Forlino A, Gallanti A, Rasperini G, Cetta G (2003) Effect of enamel matrix derivative on human periodontal fibroblasts: proliferation, morphology and root surface colonization. An in vitro study. J Periodontal Res 38:568–574
Lyngstadaas SP, Lundberg E, Ekdahl H, Andersson C, Gestrelius S (2001) Autocrine growth factors in human periodontal ligament cells cultured on enamel matrix derivative. J Clin Periodontol 28:181–188
Yuan K, Chen CL, Lin MT (2003) Enamel matrix derivative exhibits angiogenic effect in vitro and in a murine model. J Clin Periodontol 30:732–738
Schlueter SR, Carnes DL, Cochran DL (2007) In vitro effects of enamel matrix derivative on microvascular cells. J Periodontol 78:141–151. https://doi.org/10.1902/jop.2007.060111
Iruela-Arispe ML, Sage EH (1993) Endothelial cells exhibiting angiogenesis in vitro proliferate in response to TGF-beta 1. J Cell Biochem 52:414–430. https://doi.org/10.1002/jcb.240520406
Guimaraes GF, de Araujo VC, Nery JC, Peruzzo DC, Soares AB (2015) Microvessel density evaluation of the effect of enamel matrix derivative on soft tissue after implant placement: a preliminary study. Int J Periodontics Restorative Dent 35:733–738. https://doi.org/10.11607/prd.2044
Van der Pauw MT, Van den Bos T, Everts V, Beertsen W (2000) Enamel matrix-derived protein stimulates attachment of periodontal ligament fibroblasts and enhances alkaline phosphatase activity and transforming growth factor beta1 release of periodontal ligament and gingival fibroblasts. J Periodontol 71:31–43. https://doi.org/10.1902/jop.2000.71.1.31
Funding
This research was supported by Wonkwang University in 2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Institutional Review Board (IRB) of Daejeon Dental Hospital, Wonkwang University (approval no. W1807/001-001).
Informed consent
All enrolled patients provided written fully informed consent in accordance with the IRB guidelines, and the study was conducted in accordance with the Declaration of Helsinki and the Guidelines on Good Clinical Practice.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, JH., Park, YS., Kim, YT. et al. Assessment of early discomfort and wound healing outcomes after periodontal surgery with and without enamel matrix derivative: an observational retrospective case-control study. Clin Oral Invest 24, 229–237 (2020). https://doi.org/10.1007/s00784-019-02941-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-02941-w