Abstract
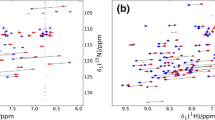
The availability of NOE constraints and of the relative solution structure of a paramagnetic protein permits the use of pseudocontact shifts as further structural constraints. We have developed a strategy based on: (1) determination of the χ tensor anisotropy parameters from the starting structure; (2) recalculation of a new structure by using NOE and pseudocontact shift constraints simultaneously; (3) redetermination of the χ tensor anisotropy parameters from the new structure, and so on until self-consistency. The system investigated is the cyanide derivative of a variant of the oxidized Saccharomyces cerevisiae iso-1-cytochrome c containing the Met80Ala mutation. The structure has been substantially refined. It is shown that the analysis of the deviation of the experimental pseudocontact shifts from those calculated using the starting structure may be unsound, as may the simple structure refinement based on the pseudocontact shift constraints only.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 11 July 1995 / Accepted: 30 October 1995
Rights and permissions
About this article
Cite this article
Banci, L., Bertini, I., Bren, K. et al. The use of pseudocontact shifts to refine solution structures of paramagnetic metalloproteins: Met80Ala cyano-cytochrome c as an example. JBIC 1, 117–126 (1996). https://doi.org/10.1007/s007750050030
Issue Date:
DOI: https://doi.org/10.1007/s007750050030