Abstract.
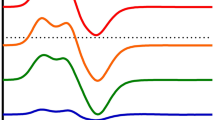
The reduction potentials of the metalloproteins pyruvate ferredoxin oxidoreductase (POR), ferredoxin, and hydrogenase isolated from hyperthermophilic Thermococcus celer (T opt=88 °C) were determined as a function of temperature from 10 to 85 °C. Square-wave voltammetry experiments were carried out on 15 µL samples directly at an unmodified "edge-polished" pyrolytic graphite electrode using MgCl2 as an electrode promoter. POR exhibited two voltammetric waves with peaks at –280 and –403 mV at room temperature, indicating multiple redox centers, and a single wave at –420 mV at 85 °C. These waves displayed different temperature-dependent peak positions and peak heights, indicating that these redox centers have different thermodynamic and kinetic properties. Ferredoxin displayed a single linear temperature-dependent voltammetric wave at –280 mV at room temperature and –327 mV at 85 °C. Hydrogenase displayed a single biphasic temperature-dependent voltammetric wave at –197 mV at room temperature and –211 mV at 85 °C. Thermodynamic parameters associated with electron transfer, namely standard enthalpies and entropies for the redox centers in the various proteins, are reported.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Smith, E., Odom, L., Awramko, J. et al. Direct electrochemical characterization of hyperthermophilic Thermococcus celer metalloenzymes involved in hydrogen production from pyruvate. J. Biol. Inorg. Chem. 6, 227–231 (2001). https://doi.org/10.1007/s007750000179
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s007750000179