Abstract
There is a high prevalence of depression in Germany and all over the world. Maternal depressive symptoms during pregnancy have been shown in some studies to be associated with an increased risk of preterm birth and low birth weight. The influence of maternal depressive symptoms during pregnancy on preterm delivery and fetal birth weight was investigated in a prospective single-centre study. A sample of 273 healthy pregnant women was assessed for symptoms of antepartum depression. Symptoms were measured using the Edinburgh Postnatal Depression Scale (EPDS) and the Patient Health Questionnaire (PHQ). Symptoms of anxiety were assessed using the State/Trait Anxiety Inventory. Patients who scored above the cutoff were contacted by phone for a Structured Clinical Diagnostic interview. Neonatal measurements were obtained from the birth registry of the Department of Obstetrics. Baseline data were assessed with a self-styled data sheet. Prevalence of elevated depressive symptoms was 13.2 % when measured with the EPDS and 8.4 % with the PHQ. According to DSM-IV criteria, only four (EPDS) respective two (PHQ-D) of these patients could be diagnosed with a depressive disorder and ten (EPDS) respective seven (PHQ) with an anxiety disorder. There was no significant influence on preterm birth or birth weight. Maternal depressive symptoms are self-reported. Elevated subclinical symptoms of depression and anxiety during pregnancy are common. However, this study showed no evidence that these symptoms are associated with adverse pregnancy outcome.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Depression is a widespread and significant public health concern worldwide. In comparison to males, females demonstrate a higher incidence for major depression (Essau et al. 2010).
Pregnancy and puerperium are times of particular vulnerability, therefore many women exhibit symptoms of emotional distress during this period. Since hormonal changes increase the lifetime risk for affective disorders, the risk of depression is naturally higher in women during the child-bearing years (Fishell 2010; Bennett et al. 2004). According to a systematic literature review (Bennett et al. 2004), antenatal depression affects approximately 12 % of women with its highest prevalence in the second and third trimester of pregnancy (7.4 % first trimester, 12.8 % second trimester, and 12.0 % third trimester). Due to a wide variety of methodologies and instruments for the assessment of emotional distress or depressive/anxiety symptoms, the data on untreated maternal depression are heterogeneous. However, the following clinical implications have emerged for the postnatal and antenatal setting. Postpartum depression can negatively influence mother–child interactions (Reck et al. 2004) and supposedly disturb fetal neuroendocrine developmental pathways (Marcus et al. 2011). It can also affect early child growth (Tegethoff et al. 2010). Stewart described in his review that maternal postpartum depression may affect the mother’s ability to provide adequate nutritional care to her infant (Stewart 2007). Symptoms like fatigue, impaired concentration, psychomotor slowing, or feelings of hopelessness may lead to functional impairment affecting breastfeeding, hygiene, and healthcare-seeking behaviors.
In the antenatal setting, an overwhelming amount of data links untreated depression and anxiety during pregnancy to unfavorable outcomes for both the mother and the fetus (Marcus 2009; Bonari et al. 2004). Several studies suggest that depression and stress during pregnancy are associated with poor neonatal outcome, such as preterm birth or low birth weight (Kelly et al. 2002) by potentially rising the hypothalamic–pituitary–adrenal (HPA) activity (Diego et al. 2009). In a recent review by Alder et al. on the influence of depression and anxiety on obstetric and neonatal outcome, women suffering from depressive symptoms experienced adverse obstetric, fetal, and neonatal outcome (Alder et al. 2007). Diego et al. were able to demonstrate a significant association between maternal depression, elevated maternal cortisol levels, and restricted fetal growth. The hyperactivity of the HPA axis may serve as a theoretical basis to understand the obstetric outcome and the negative impact on child development (Strohle and Holsboer 2003). Prenatal stress or depression during pregnancy stimulates the release of stress hormones, such as cortisol or catecholamines. These biological changes might cause placental hypofusion and consequent restriction of oxygen and nutrients to the fetus leading to fetal growth restriction and increased risk of preterm birth (Grote et al. 2010). Lasting heightened cortisol levels after delivery in infants of distressed or anxious mothers are also shown to have a negative influence on the developing brain (Dawson et al. 2000) and thus on the infants emotional development: infants of mothers afflicted with an anxiety or depressive disorder in the prenatal period have been shown to have heightened cortisol levels, especially if mothers interact less sensitively with their infants (Kaplan et al. 2008; Field et al. 2010). In comparison to infants of healthy controls, infants of mothers with anxiety disorders behave differently in experiments providing stressful experiences for toddlers (i.e., less negative affect and less contact to strangers), as demonstrated by Kaitz et al. (2010). Davis et al. also demonstrated that elevated maternal cortisol at 30–32 weeks of gestation in mothers with elevated levels of perceived stress, anxiety, or depression was significantly associated with greater maternal report of infant negative reactivity (Davis et al. 2007). Liu et al. confirmed the association between severe depressed mood and low birth weight and showed that the methylation and plasticity of regulatory sequences might play an important role (Liu et al. 2012). Taken together, these studies show that biobehavioral adaptation, even that initiated in utero, is influenced by interactions with the social world supporting the model of fetal programming.
In a current meta-analysis Grote et al. showed that depression during pregnancy regardless of the type of measurement is associated with a modest but statistically significant risk of preterm birth and low birth weight (Grote et al. 2010). Intrauterine growth restriction and preterm delivery are serious pregnancy complications with increasing pattern over the last decades constituting the principle risk factors for neonatal morbidity and mortality (Saigal and Doyle 2008). Low birth weight neonates are also at increased risk of coronary heart disease, stroke, type 2 diabetes, obesity, and some cancers in adulthood, therefore understanding their cause is a major research priority.
In spite of evident data on the perinatological implications of depressive disorders during pregnancy, it is still a matter difficult to assess and address in clinical routine. Patients highly suspected of depression are to be referred to a psychiatrist to provide a reliable axis I diagnosis. Although it may have a significant influence on the obstetric outcome, among limiting factors in the evaluation of patients at risk are those who rather represent the subtreshold spectrum of depressive symptoms that are insufficient for an axis I diagnosis. Alder et al. (2007) showed that an Edinburgh Postnatal Depression Scale (EPDS) score above 10 at 35–36 weeks of gestation was associated with substantially more sick leave, medical consults, pregnancy complications, and hospital admissions. Liu et al. observed that severe maternal depressed mood was associated with a 3-fold increase in the risk of low birth weight potentially mediated by a 2.4 % higher methylation level in infants than those born to nondepressed women (Liu et al. 2012).
Hence, aiming to address this issue in Germany the aim of our study was to prospectively assess the association of subclinical depressive symptoms above established cutoffs in a German sample in relation to premature delivery and lower birth weight in concordance to studies including pregnant women with symptomatic evaluable on an axis I diagnosis of depression.
Methods
Procedure
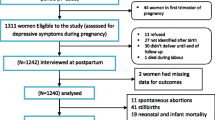
Patients were invited to participate in their late second or third trimester of pregnancy (>24 weeks) by trained female interviewers while attending prenatal care in the Department of Obstetrics of the University of Heidelberg Medical Centre between January 2007 and January 2010. Women younger than 18 years, bearing multiplets, with serious medical conditions or in a gestational age of lower than 24 weeks as well as the ones speaking inadequate German were excluded from the study. After informed consent, matching subjects received a set of questionnaires containing the German versions of the EPDS (Cox et al. 1987; Bergant et al. 1998), the Patient-Health Questionnaire (PHQ-D, Spitzer et al. 1999), the State/Trait Anxiety Inventory (STAI; Spielberger et al. 1970), as well as a demographic and medical data questionnaire to be completed at home and returned via mail or upon the next visit. Data regarding infant birth weight and time of delivery were obtained from the birth register of the Department of Obstetrics of the University of Heidelberg medical centre. At 6 weeks postpartum, the patients received another set of questionnaires by mail including questions about the delivery. This way patients delivering in other medical centers could be included. In patients scoring above the cutoff of the EPDS or suspected of being depressed by the gynaecologist, a structured clinical phone interview for DSM-IV disorders (SCID) (Wittchen et al. 1997) to determine psychiatric disorders was conducted.
Instruments
Edinburgh postnatal depressive scale
The EPDS is a ten-item self-rating scale, scored from 0 to 3 (normal response 0 and severe response 3) that has been validated in the detection of post- and antepartum depression in numerous studies around the world (Matthey et al. 2006). The total score can range from 0 to 30, with higher scores being indicative for increasing distress. Answers are based on the psychical state over the past 7 days. Originally developed as a screening instrument for the postpartum period, the EPDS is a simple and short questionnaire that can also be used during pregnancy (Cox et al. 1996). The scale is sensitive to changes in severity of depression and has been shown to have a sensitivity and specificity of 91 and 95 % in predicting depressive disorders in mothers and only marginally lower in fathers (Matthey et al. 2001). The recommended cutoff scores include >12 points for minor depression and >14 points for major depression (Bergant et al. 1998).
Patient health questionnaire
The PHQ was developed based on the Primary Care Evaluation of Mental Disorders (PRIME-MD) as a self-rating questionnaire that can assess the most common psychiatric disorders according to the DSM IV. Several extended versions exist, however, the shorter version used in this study (PHQ-9) contains nine items to assess symptoms of depression over the four weeks preceding the test. Questions are scored from 0 to 3 points, while the most sensitive cutoff score for major depression values at 10 points shows a sensitivity and specificity of 98 and 80 % in a German sample. It showed an excellent internal consistency (Cronbach’s Alpha, 0.88) (Löwe et al. 2004).
State/Trait Anxiety Inventory
This STAI has been used to measure anxiety related to evaluation apprehension (Spielberger et al. 1970). The STAI differentiates between the temporary condition of “state anxiety” and the more general and long-standing quality of “trait anxiety”. The state scale evaluates feelings of apprehension, tension, nervousness, and worry. Scores on the state scale increase in response to physical danger and psychological stress. The state portion of the STAI (STAI-S) was administered to participants, and items were scored from 1 to 4, with higher numbers corresponding to greater agreement. Several studies have demonstrated that the STAI has adequate concurrent validity and internal consistency (r = 0.83) (Meades and Ayers 2011). It takes about 5 min to complete. The recommended cutoff score for high anxiety is 48. The scale has been used in several studies with pregnant women (Giardinelli et al. 2012).
Structured clinical interview for DSM-IV disorders
All women in the study who scored above the cutoff of the EPDS were contacted for a phone version of the SCID to determine depression or anxiety diagnosis and to screen out other disorders. The SCID was administered by a trained female research associate. All depressed pregnant women were not taking antidepressants and were not receiving other treatments for depression.
Participants
Finally, N = 273 women with singleton pregnancies were included in the analyses. Of these, 79.9 % were married, 15.8 % were in a partnership, and 2.6 % were single as shown in Table 1; 47.3 % had a college degree, 20.9 % had a high-school qualification, 24.9 % had a high secondary school qualification, 5.1 % had a low secondary school qualification, and only 0.7 % had no school-leaving qualification (Table 2). Of n = 3 women, no data concerning education or social status were available. Mean age during this period was M = 32.8 years (SD = 4.6 years); 39.9 % beard boys, and of 17.9 % no data concerning infant sex were available. Mean gestational age at time of prepartal assessment was the 32nd week of pregnancy (M = 31.82 weeks, SD = 4.20 weeks).
Only n = 1 woman could be diagnosed a Major Depression Episode following DSM-IV criteria. N = 4 women fulfilled criteria for a minor depression. The rest of the sample (n = 268) was free of DSM-IV depression diagnoses. N = 5 women could be diagnosed with a generalized anxiety disorder and n = 8 patients with an Anxiety Disorder NOS. Distribution of other DSM-IV disorders is shown in Table 3.
Statistical analysis
For all the analyses conducted in this study we used the Statistical Package for Social Sciences (IBM SPSS v. 20.0). Estimators of effect size and power-analyses have been conducted using G-Power (v. 3.1.2.; Faul et al. 2007, 2009; Bortz 2005). Parametric testing strategy was used, since the linear model proved to be robust against violations of premises or even adaptable relative to their extent. Prior to main analyses the distributions of relevant variables were analyzed. In a second step, Pearson correlations between maternal depressiveness and infant outcome variables were examined. Finally, a multiple linear regression was performed to explore the magnitude of influence of each theoretically relevant variable in relation to another predicting infant birth weight.
Results
Preliminary analyses
Mean EPDS score was M = 6.26 (SD = 5.07). 86.1 % scored below the cutoff of 12; 13.2 % scored above the cutoff. Of n = 2 women, no data concerning the EPDS were available. Mean PHQ-D score was M = 5.19 (SD = 3.50); 91.2 % scored below the cutoff of 10; 8.4 % scored above the cutoff. Of n = 1 woman no data concerning PHQ was available. Only n = 5 women were given a DSM-IV Depression diagnosis. Thus, No χ 2 test aiming the concordance between the EPDS cutoff scale respective the PHQ-D cutoff scale (depressive vs. nondepressive) and the DSM-IV diagnostic (depressive vs. nondepressive) could be run, because at least 25 % of the cells would contain less than n = 5 frequencies. But the data in Tables 4 and 5 claim for a low specificity of the EPDS cutoff of 12 respective the PHQ-D cutoff of 10 in our sample. The data in Tables 6 and 7 show that n = 10 (EPDS) and 7 (PHQ) women who scored above the cutoff were diagnosed with an anxiety disorder according to DSM-IV criteria.
Mean STAI-T score was M = 34.15 (SD = 9.79). Mean STAI-S score was M = 35.73 (SD = 10.44). Of n = 6 women, no STAI-S data were available. Mean gestation age was M = 39.4 weeks (SD = 1.78 weeks); 9.9 % delivered their child prematurely, before the end of the 37th week. Mean birth weight was M = 3,324.14 g (SD = 529.89 g). Of n = 52 women, no data concerning infant birth were available due to delivery in other medical centers and not returning the completed data sheet. Because of scale-specific amounts of missing values, the listwise valid number of cases n varies dependent on the data subsets used for the particular test.
Main analyses depressive symptoms
There was no significant correlation between the EPDS scores and gestation age (r = −0.01, p = 0.85, n = 271) or birth weight (r = −0.05, p = 0.44, n = 219). Furthermore, the PHQ scores did not correlate significantly neither with gestational age (r = −0.03, p = 0.67, n = 272) nor with infant birth weight (r = −0.02, p = 0.73, n = 220). Post hoc computed power for large (ρ = 0.5) and medium-sized effects (ρ = 0.3) with α = 0.05 ranges from 1-β = 0.9994 to 1-β = 0.9999. So the probability to mistakenly decide in favor of the H 0 for large- and medium-sized effects is approximately zero. Post-hoc computed power for small effects of ρ = 0.1 (Cohen 1988) with α = 0.05 ranges from 1-β = 0.3186 to 1-β = 0.3814. So the probability to mistakenly decide in favor of the H 0 for small effects is approximately over 40 %; a reliable decision whether there is no small association between depressiveness and birth weight cannot be made.
Regression analyses
Conducting a stepwise linear multiple regression with birth weight as dependent variable and EPDS scores, PHQ scores, gestation age, maternal age, infant sex, STAI-T and STAI-S scores as predictors, variables were selected, if the multiple correlation R significantly changes with p ≤ 0.05. The stepwise strategy was used to separate the empirical important variables in this data set from the empirical unimportant ones out of the theoretical important variable pool. The final model (R 2 = .41, F 7, 208 = 48.57, p ≤ 0.01) simply contains week of gestation (β = 0.62, t = 11.68, p ≤ 0.01) infant sex (β = 0.13, t = 2.49, p ≤ 0.01) and maternal age (β = 0.11, t = 2.05, p ≤ 0.05) as significant predictors. Correction for shrinkage following Olkin and Pratt (1958) adjusts R 2 leading to an explanation of variance of 39.03 %, which according to Cohen (1988) is interpretable as a large effect. Structure coefficients indicate “gestation age” to have the largest contribution to the explanation of variance (c gw = 0.96, c sex = 0.22, and c age = 0.14). The usefulness of “gestation age” can be interpreted as large in accordance to Cohen’s conventions from 1988 (U gw = 0.38). The usefulness of “infant sex” can be interpreted as small (U sex = 0.02). The usefulness of “maternal age” can be interpreted as small (U age = 0.01). Multicollinearity seems improbable, because VIF is around VIF = 1 for all variables.
Discussion
In this prospective single-centre study, we evaluated the association between maternal depressive symptoms during the late second and third trimester of pregnancy and adverse pregnancy outcome such as preterm delivery and lower fetal birth weight. The prevalence of antenatal depressive symptoms assessed with self rating scales above the cutoff was 8.4 (PHQ-D) and 13.2 % (EPDS). This is consistent with the prevalence described in a meta-analysis from Bennett et al. 2004 (prevalence, 7.2–12.8 %). To our knowledge comparable data from Germany does not exist. In only 4 (EPDS) respective 2 (PHQ-D) of 36 (23) patients who scored above the cutoff a DSM-IV diagnosis of a depressive disorder by conducting a structured clinical interview could be established. Apparently, the women who tested positive above the thresholds of the self rating measurements in our study present with a variety of subclinical symptoms of distress and seem to be a rather healthy sample. None of them received antidepressant therapy. Furthermore, in our sample Anxiety Disorders constitute the most common DSM-IV diagnosis. N = 10 (EPDS) and n = 7 (PHQ) patients who scored above the cutoff were diagnosed with an anxiety disorder. The EPDS is routinely administered but high scores are falsely interpreted as evidence for depressive illness only. This is concordant with recent literature. Grigoriadis et al. found evidence that the prevalence of anxiety disorders in the perinatal period may be higher than previously thought and that anxiety may be more prevalent than depression in certain populations (Grigoriadis et al. 2011). It also represents exactly what Salomon et al. argued for in their review (Salomon et al. 2001). Regarding the question if diagnosable unipolar depression and limited depressive symptoms are qualitatively distinct or only differ in degree they stated that analysis based on self-report measures are somewhat ambiguous in their implications, because elevated self-report scores may rather reflect various kinds of negative affect and psychological disorders than unipolar clinical depression.
The rate of preterm deliveries <37th week of gestation in this study was 9.9 % which is similar to the overall rate of preterm deliveries for Europe, including Germany (Goldenberg et al. 2008). Although we hypothesized that even subclinical antenatal depressive and anxiety symptoms could be associated with low gestational age at the time of delivery and/or with preterm birth, no significant influence on both of these outcomes could be found. Only gestational age seems to be a reliable predictor for infant birth weight. The multiple linear regression model revealed no further reliable suppressed effects.
The findings of the present study rather suggest that, given the relatively healthy and well educated sample, maternal subtreshold depressive symptoms during pregnancy do not influence neonatal outcome. Despite the limited representativeness, our data are in line with other study results. Andersson et al. 2004 could not find any association between maternal depression and adverse neonatal outcome in one of the largest studies conducted. The prevalence of depressive disorders diagnosed according to the version of PRIME-MD used in Andersson’s study was 11.6 % and after applying DSM IV criteria, major depression was diagnosed in 46 women (3.1 %) showing a comparably healthy sample.
Furthermore in another large study, by evaluating 8.052 women, Goedhart et al. showed that in a high risk group of pregnant women screened above the cutoff of self measurement scales for depressive symptoms they found no influence whatsoever on the risk of preterm birth only on the risk of SGA (Goedhart et al. 2010).
In contrast, several studies demonstrated a significant association between preterm birth and/or lower birth weight. As noted earlier, Fransson et al. investigated in a comparable Swedish study population that the presence of antenatal depressive symptoms above a cutoff score of 12 or higher on the EPDS increased the risk for preterm birth (OR, 1.56 (95 % CI), 1.03–2.35) (Fransson et al. 2011). Similarly, Diego et al. found that prenatal depression was associated with adverse perinatal outcomes including premature delivery and slower fetal growth rates (Diego et al. 2009). In contrast to our study, their study population consisted of 40 already as clinical depressed diagnosed women and 40 nondepressed women.
In a recent meta-analysis conducted by Grote et al., 29 studies in which an effect of depressive symptoms or unipolar depressive disorder on fetal outcome was assessed were analyzed together. Grote et al. showed that depression during pregnancy regardless the type of antenatal depression measurement used (categorical or continuous) is associated with a modest but statistically significant risk of PTB or SGA (Grote et al. 2010). They found evidence that the type of depression measurement moderated the strength of the association between antenatal depression and PTB and LBW. Considering the EPDS, continuous measures of depression showed an albeit weaker pattern.
Regarding the healthy sample in this study, the EPDS showed a relatively low specificity. The EPDS classified 36 participants at being at high risk of depression by using a cutoff score of 12. However, only four (11.1 %) of these women met diagnostic criteria for a depressive disorder and ten (27.8 %) for anxiety disorders. Previous studies have found sensitivity values for the EPDS ranging between 34 and 100 %, and specificity values from 49 to 100 %. The authors of a recent review of the sensitivity and specificity of the EPDS noted that variation in these statistics may be due to differences in study methodology, language, and diagnostic criteria used (Gibson et al. 2009) and suggested that differences in study settings and thus the populations might account for variations in sensitivity and specificity.
Despite the EPDS being developed as a screening instrument for probable depression, following our data interpreting high scores as indicative of only depression may lead to misdiagnosis and inadequate treatment for women who are indeed experiencing other disorders. Our data support other studies that have referred to the EPDS as detecting general distress in the perinatal period rather than indicating only the presence of probable clinical depression (Brouwers et al. 2001).
Considering the educational level, 47.3 % of the participants had a higher level of education holding a college or university degree, what is usually associated with middle- or upper-class status and not representative for the population of Germany or Europe. Comparable to our results, Grote et al. showed no statistically significance between depressive symptoms and adverse pregnancy outcome in women of middle- or upper-class status (Grote et al. 2010). We know that highly educated women have more antenatal visits than women with lower education (Beeckman et al. 2010) and vice versa women with a history of depression who have fewer prenatal care visits are at an increased risk of LBW, SGA, and preterm birth (Chen and Lin 2011). Thus the higher level of education studying our study population could have possibly counteracted the effect of depression on pregnancy. This factor has to be considered when generalizing our findings, as the risk for preterm birth differs based on educational background.
Limitations
While the prospective design avail, a limitation derives from the fact that we were neither able to comment on our recruitment rate nor to assess previous pre-term labor or patient psychiatric history completely. All potentially eligible patients of our obstetric outpatient department were informed about the study and to those who agreed to participate after contact to our trained interviewers received the first set of questionnaires. One further limitation derives from the fact that elevated depressive symptoms were only assessed in the late second or third trimester of pregnancy. Matthey et al. state in a recent study that a high percentage of women scoring at or above the cutoff score on the EPDS at one point will score below the cutoff score just a few weeks later indicating that every women experiences distress at some point in pregnancy but only part of these women will suffer from enduring stress (Matthey and Ross-Hamid 2012). In consequence, women should be tested repeatedly. Given the missing recruitment rate, the relatively small sample size and the educational status, caution should prevail in the interpretation of our data. Finally, the cutoff score of 12 on the EPDS in this study showed a generally low specificity. Of 36 patients who scored above the cutoff, only 4 could be diagnosed with a depressive disorder and 10 with an Anxiety Disorder according to DSM-IV criteria. Neither of the self-rating scales used in this study could identify most of the women at risk for a depressive disorder or an anxiety disorder correctly. It might be helpful to decrease the detection rate by a change to EPDS cutoff of >9, with the risk of having false positives as already stated by Magnusson et al. (2011).
Conclusions
This study shows that subclinical-elevated depressive symptoms or distress during pregnancy measured with self-rating scales are relatively common in an otherwise healthy sample but provides no evidence that these symptoms are associated with low birth weight or preterm birth. By having diagnosed more cases of anxiety disorders than cases with unipolar depression, our data support the EPDS as a screening tool for general distress in the perinatal period rather than indicating only the presence of probable clinical depression. Therefore these subclinical symptoms should be nevertheless regarded as an indicator for patients at risk beyond unipolar depression that obstetricians need to be aware of during prenatal care.
References
Alder J, Fink N, Bitzer J, Hösli I, Holzgreve W (2007) Depression and anxiety during pregnancy: a risk factor for obstetric, fetal and neonatal outcome? A critical review of the literature. J Matern Fetal Neonatal Med 20(3):189–209, Review
Andersson L, Sundström-Poromaa I, Wulff M, Aström M, Bixo M (2004) Neonatal outcome following maternal antenatal depression and anxiety: a population-based study. Am J Epidemiol 159(9):872–881
Beeckman K, Louckx F, Putman K (2010) Determinants of the number of antenatal visits in a metropolitan region. BMC Publ Health 10:527
Bennett HA, Einarson A, Taddio A, Koren G, Einarson TR (2004) Prevalence of depression during pregnancy: systematic review. Obstet Gynecol 103(4):698–709, Review. Erratum in: Obstet Gynecol. 2004 Jun;103(6):1344
Bergant AM, Nguyen T, Heim K, Ulmer H, Dapunt O (1998) German language version and validation of the Edinburgh postnatal depression scale. Dtsch Med Wochenschr 123(3):35–40, German
Bonari L, Pinto N, Ahn E, Einarson A, Steiner M, Koren G (2004) Perinatal risks of untreated depression during pregnancy. Can J Psychiatry 49(11):726–735
Bortz J (2005) Statistik: Für Human- und Sozialwissenschaftler, 6th edn. Springer, Berlin
Brouwers EP, van Baar AL, Pop VJ (2001) Does the Edinburgh postnatal depression scale measure anxiety? J Psychosom Res 51(5):659–663
Chen CH, Lin HC (2011) Prenatal care and adverse pregnancy outcomes among women with depression: a nationwide population-based study. Can J Psychiatry 56(5):273–280
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Erlbaum, Hillsdale
Cox JL, Holden JM, Sagovsky R (1987) Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br J Psychiatry 150:782–786
Cox JL, Chapman G, Murray D, Jones P (1996) Validation of the Edinburgh Postnatal Depression Scale (EPDS) in non-postnatal women. J Affect Disord 39(3):185–189
Davis EP, Glynn LM, Schetter CD, Hobel C, Chicz-Demet A, Sandman CA (2007) Prenatal exposure to maternal depression and cortisol influences infant temperament. J Am Acad Child Adolesc Psychiatry 46(6):737–746
Dawson G, Ashman SB, Carver LJ (2000) The role of early experience in shaping behavioral and brain development and its implications for social policy. Dev Psychopathol 12(4):695–712
Diego MA, Field T, Hernandez-Reif M, Schanberg S, Kuhn C, Gonzalez-Quintero VH (2009) Prenatal depression restricts fetal growth. Early Hum Dev 85(1):65–70
Essau CA, Lewinsohn PM, Seeley JR, Sasagawa S (2010) Gender differences in the developmental course of depression. J Affect Disord 127(1–3):185–190, Epub 2010 Jun 22
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Faul F, Erdfelder E, Buchner A, Lang A-G (2009) Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav Res Methods 41:1149–1160
Field T, Diego M, Hernandez-Reif M, Figueiredo B, Deeds O, Ascencio A, Schanberg S, Kuhn C (2010) Comorbid depression and anxiety effects on pregnancy and neonatal outcome. Infant Behav Dev 33(1):23–29
Fishell A (2010) Depression and anxiety in pregnancy. J Popul Ther Clin Pharmacol 17(3):e363–e369
Fransson E, Ortenstrand A, Hjelmstedt A (2011) Antenatal depressive symptoms and preterm birth: a prospective study of a Swedish national sample. Birth 38(1):10–16. doi:10.1111/j.1523-536X.2010.00441.x
Giardinelli L, Innocenti A, Benni L, Stefanini MC, Lino G, Lunardi C, Svelto V, Afshar S, Bovani R, Castellini G, Faravelli C (2012) Depression and anxiety in perinatal period: prevalence and risk factors in an Italian sample. Arch Womens Ment Health 15(1):21–30
Gibson J, McKenzie-McHarg K, Shakespeare J, Price J, Gray R (2009) A systematic review of studies validating the Edinburgh Postnatal Depression Scale in antepartum and postpartum women. Acta Psychiatr Scand 119(5):350–364
Goedhart G, Snijders AC, Hesselink AE, van Poppel MN, Bonsel GJ, Vrijkotte TG (2010) Maternal depressive symptoms in relation to perinatal mortality and morbidity: results from a large multiethnic cohort study. Psychosom Med 72(8):769–776
Goldenberg RL, Culhane JF, Iams JD, Romero R (2008) Epidemiology and causes of preterm birth. Lancet 371(9606):75–84
Grigoriadis S, de Camps Meschino D, Barrons E, Bradley L, Eady A, Fishell A, Mamisachvili L, Cook GS, O’Keefe M, Romans S, Ross LE (2011) Mood and anxiety disorders in a sample of Canadian perinatal women referred for psychiatric care. Arch Womens Ment Health 14(4):325–333
Grote NK, Bridge JA, Gavin AR, Melville JL, Iyengar S, Katon WJ (2010) A meta-analysis of depression during pregnancy and the risk of preterm birth, low birth weight, and intrauterine growth restriction. Arch Gen Psychiatry 67(10):1012–1024
Kaitz M, Maytal HR, Devor N, Bergman L, Mankuta D (2010) Maternal anxiety, mother–infant interactions, and infants’ response to challenge. Infant Behav Dev 33(2):136–148
Kaplan LA, Evans L, Monk C (2008) Effects of mothers’ prenatal psychiatric status and postnatal caregiving on infant biobehavioral regulation: can prenatal programming be modified? Early Hum Dev 84(4):249–256
Kelly RH, Russo J, Holt VL, Danielsen BH, Zatzick DF, Walker E, Katon W (2002) Psychiatric and substance use disorders as risk factors for low birth weight and preterm delivery. Obstet Gynecol 100(2):297–304
Liu Y, Murphy SK, Murtha AP, Fuemmeler BF, Schildkraut J, Huang Z, Overcash F, Kurtzberg J, Jirtle R, Iversen ES, Forman MR, Hoyo C (2012) Depression in pregnancy, infant birth weight and DNA methylation of imprint regulatory elements. Epigenetics 7(7):735–746
Löwe B, Spitzer RL, Gräfe K, Kroenke K, Quenter A, Zipfel S, Buchholz C, Witte S, Herzog W (2004) Comparative validity of three screening questionnaires for DSM-IV depressive disorders and physicians’ diagnoses. J Affect Disord 78(2):131–140
Magnusson M, Lagerberg D, Sundelin C (2011) How can we identify vulnerable mothers who do not reach the cut off 12 points in EPDS? J Child Health Care 15(1):39–49
Marcus SM (2009) Depression during pregnancy: rates, risks and consequences—Motherisk Update 2008. Can J Clin Pharmacol 16(1):e15–e22
Marcus S, Lopez JF, McDonough S, Mackenzie MJ, Flynn H, Neal CR Jr, Gahagan S, Volling B, Kaciroti N, Vazquez DM (2011) Depressive symptoms during pregnancy: impact on neuroendocrine and neonatal outcomes. Infant Behav Dev 34(1):26–34
Matthey S, Ross-Hamid C (2012) Repeat testing on the Edinburgh Depression Scale and the HADS-A in pregnancy: differentiating between transient and enduring distress. J Affect Disord. doi:10.1016/j.jad.2012.02.037
Matthey S, Barnett B, Kavanagh DJ, Howie P (2001) Validation of the Edinburgh Postnatal Depression Scale for men, and comparison of item endorsement with their partners. J Affect Disord 64(2–3):175–184
Matthey S, Henshaw C, Elliott S, Barnett B (2006) Variability in use of cut-off scores and formats on the Edinburgh Postnatal Depression Scale: implications for clinical and research practice. Arch Womens Ment Health 9(6):309–315
Meades R, Ayers S (2011) Anxiety measures validated in perinatal populations: a systematic review. J Affect Disord 133(1–2):1–15
Olkin E, Pratt JW (1958) Unbiased estimation of certain correlation coefficients. Ann Math Stat 29:201–211
Reck C, Hunt A, Fuchs T, Weiss R, Noon A, Moehler E, Downing G, Tronick EZ, Mundt C (2004) Interactive regulation of affect in postpartum depressed mothers and their infants: an overview. Psychopathology 37(6):272–280
Saigal S, Doyle LW (2008) An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet 371(9608):261–269
Salomon A et al (2001) Is clinical depression distinct from subthreshold depressive symptoms? A review of the continuity issue in depression. ResearchJ Nerv Ment Dis 189:498–506
Spielberger CD, Gorsuch RL, Lushene RE (1970) Manual for the state-trait anxiety inventory (self-evaluation questionnaire). Consulting Psychologists Press, Palo Alto
Spitzer RL, Kroenke K, Williams JB (1999) Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary care evaluation of mental disorders. Patient health questionnaire. JAMA 282(18):1737–1744
Stewart RC (2007) Maternal depression and infant growth: a review of recent evidence. Matern Child Nutr 3(2):94–107, Review
Strohle A, Holsboer F (2003) Stress responsive neurohormones in depression and anxiety. Pharmacopsychiatry 36:207–214
Tegethoff M, Greene N, Olsen J, Meyer AH, Meinlschmidt G (2010) Maternal psychosocial adversity during pregnancy is associated with length of gestation and offspring size at birth: evidence from a population-based cohort study. Psychosom Med 72(4):419–426
Wittchen HU, Wunderlich U, Gruschwitz S, Zaudig M (1997) SCID. Structured clinical interview for DSM-IV, Axis I. Hogrefe, Goettingen
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gawlik, S., Waldeier, L., Müller, M. et al. Subclinical depressive symptoms during pregnancy and birth outcome—a pilot study in a healthy German sample. Arch Womens Ment Health 16, 93–100 (2013). https://doi.org/10.1007/s00737-012-0320-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00737-012-0320-0