Abstract
Postpartum psychosis is a serious disorder that can cause negative consequences for the mother, infant, and entire family. While reports of this condition date back for centuries, little is known about what interventions are most effective for this population. The purpose of this systematic review was to examine the research evidence on interventions for the prevention and treatment of postpartum psychosis. Studies were searched using CINAHL, EMBASE, MEDLINE, PsycINFO, and PubMed databases. All primary research studies published in English since 1970 that explored interventions for the prevention or treatment of postpartum psychosis were included. The search resulted in 26 studies on interventions for postpartum psychosis, with 10 focusing on prevention and 17 focusing on treatment. Studies on the prevention of postpartum psychosis have examined the effects of mood stabilizers, antipsychotics, and hormone therapy, while those examining treatment have included electroconvulsive therapy, mood stabilizers, antipsychotics, hormones, and the beta blocker propranolol. Only preliminary evidence suggests which interventions may be effective strategies to prevent (e.g., lithium) and treat (e.g., electroconvulsive therapy) postpartum psychosis. Due to methodological limitations in the studies reviewed, extensive evidence-based recommendations for the prevention and treatment of postpartum psychosis cannot be made. The known risk factors and negative consequences of postpartum psychosis point to the importance of preventative and acute treatment measures. Well-designed prospective studies are needed to determine the efficacy of prevention and treatment interventions for women who experience postpartum psychosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Postpartum psychosis (PP) is a psychiatric emergency that requires immediate medical attention. Despite the experience of PP dating back centuries (Brockington 1996), little is known about what interventions are most effective for this vulnerable population. Research on interventions for women who experience PP has been limited due to barriers such as the relative rarity of this disorder and exclusion from drug trials for safety concerns. Affecting one to two per 1,000 deliveries (Kendell et al. 1987), PP can cause several negative consequences, including impaired mother–infant bonding (Hipwell et al. 2000), infant abuse and neglect (Chandra et al. 2006), the risk of recurrent psychiatric illness (Robertson et al. 2005; Terp et al. 1999), suicide (Appleby et al. 1998), and infanticide (Spinelli 2004). PP is at times confused or used interchangeably with postpartum depression. Postpartum depression refers to a non-psychotic depressive episode that often requires treatment and affects approximately 13% of mothers within 12 weeks of giving birth (O’Hara and Swain 1996). PP is more severe than postpartum depression, often requiring hospitalization, and is characterized by delusions, hallucinations, bizarre behavior, depression, mania, and mood lability that usually presents within the first 2 weeks postpartum (Heron et al. 2008; Sit et al. 2006).
Another barrier to conducting research related to PP is that the nosology of this disorder remains disputed. The evidence suggests, however, that PP is usually mania or affective psychosis linked temporally with childbirth (Brockington 1996). The Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV; American Psychiatric Association 2000), and the International Classification of Diseases, Tenth Revision (ICD-10; World Health Organization 1992) do not recognize PP as a distinct condition; rather, PP is diagnosed within various non-postpartum psychiatric disorders if the onset occurs within 4 and 6 weeks postpartum, respectively. The differential diagnosis for PP episodes can include major depression with psychotic features, bipolar I, bipolar II, schizoaffective, unspecified functional psychosis, and brief psychotic disorder. Studies suggest that PP is often a manifestation of a bipolar disorder condition that is triggered by childbirth (Brockington et al. 1981; Jones and Craddock 2001; Kendell et al. 1987). Approximately 25–50% of women who give birth and have a history of bipolar disorder experience an episode of PP (Brockington 1996; Jones and Craddock 2001), while rates as high as 74% have been reported with women who also have a family history of PP (Jones and Craddock 2001). Recent epidemiological research suggests that in primiparous women, the relative risk for hospital admission with bipolar disorder during the first month postpartum is 23 (Munk-Olsen et al. 2006). This finding is four times higher than the relative risk for hospital admission with schizophrenia. Further support for the increased risk of bipolar disorder recurrence postnatally comes from research demonstrating high relapse rates among women who discontinue their lithium (Viguera et al. 2000). In a retrospective study, Viguera et al. (2000) found that 70% of women who stopped their lithium treatment during pregnancy experienced a recurrence of their bipolar disorder in the postpartum period, compared to 24% of non-pregnant matched women. A personal history of PP also predisposes approximately 57% of women to experience another episode after a subsequent pregnancy (Robertson et al. 2005).
Considering it is well documented that a history of bipolar disorder or PP are significant risk factors for a relapse postnatally, preventative strategies are warranted. Given the seriousness of PP, gaining a better understanding of effective treatment interventions is also critical in order to address the immediate effects and decrease the long-term health consequences.
Aim
While there are general guidelines available on the treatment of bipolar disorder or affective psychosis (e.g., American Psychiatric Association http://www.psychiatryonline.com/pracGuide/pracGuideTopic_8.aspx), specific guidelines on the prevention and treatment of PP for perinatal health professionals are primarily limited to the UK (e.g., NICE, SIGN, and CEMACH). In light of the seriousness of PP, a greater understanding of effective interventions is essential to improve outcomes for this vulnerable population. The purpose of this systematic review was to examine the evidence on interventions for the prevention and treatment of PP. It is beyond the scope of this review to evaluate the evidence of reproductive safety for medications used in pregnancy or breastfeeding; a review examining this specific area has already been previously published (Stowe 2007).
Methods
Search methods
Databases systematically searched for this review included CINAHL (1982–2009), EMBASE (1980–2009), MEDLINE (1950–2009), PsycINFO (1806–2009), and PubMed (1966–2009). Due to the dearth of research on PP, and to ensure that no studies were missed, all studies including the key search terms postpartum psychosis, puerperal psychosis, or postnatal psychosis were reviewed for inclusion. Additional literature was located through reviewing the citations of the retrieved studies and by contacting subject experts.
Inclusion and exclusion criteria
All primary research studies published in English and since 1970 that explored the effectiveness of interventions for the prevention or treatment of PP were included in this review. For the prevention studies, the intervention must have begun prior to the presentation of manic or affective psychotic symptoms either during pregnancy or immediately postpartum. For the treatment studies, the onset of mania or affective psychosis had to have occurred within the first year postpartum. As previously stated, although symptoms generally develop within 2 weeks of delivery, research studies can identify subjects with PP using two different strategies. Some authors use specific diagnostic criteria ascertained through medical records while others use temporal criteria that links symptoms to childbirth. As such, we used the criterion of onset of mania or affective psychosis within 1 year of parturition to ensure that key studies were not omitted. Studies based solely on baby blues or postpartum depression were excluded; however, studies that included mixed samples including both women who experienced postpartum depression and PP were included to ensure that research on prevention and treatment strategies for PP was thoroughly reviewed.
Quality appraisal, data abstraction, and synthesis
Due to the limited number of studies on interventions for PP, all research designs were included. Studies, case series, or case reports of very poor methodological quality were excluded when we could not establish the prevention or treatment intervention used. Data were extracted using an abstraction form and details related to disorder definition (i.e., diagnostic/screening criteria used), population sampled (i.e., participant characteristics, sample size, recruitment process, inclusion/exclusion criteria), study design (i.e., method and timing of assessment, outcome measures, length of follow-up), intervention, and results were collected. Interventions were categorized as prevention or treatment, and were subcategorized according to the specific interventional approach used.
Results
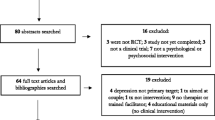
The first author reviewed the titles of approximately 800 papers. After scanning the titles for inclusion, approximately 270 abstracts were specifically examined according to the inclusion criteria. The search resulted in 26 intervention studies (one study included both prevention and treatment interventions). Table 1 summarizes the results of 10 studies that explored the prevention of PP. Table 2 summarizes the results of 17 studies that explored the treatment of PP. All studies that met the inclusion criteria for this review were those that examined biological interventions such as pharmacotherapy and electroconvulsive therapy.
Prevention of PP
Research on the prevention of PP has focused on the effects of mood stabilizers, antipsychotics, and hormone therapy. The sample size for the 10 prevention studies ranged from 1 to 29, while over half (60%) of the studies had between 17 and 29 participants. Half (50%) of the studies were small clinical trials, while the remainder were primarily case studies or retrospective reports.
Mood stabilizers
Six studies evaluated the effects of mood stabilizers for the prevention of PP. In an open clinical trial conducted in Canada, the Netherlands, and the UK, 21 women who met the Research Diagnostic Criteria (RDC) for history of PP were administered lithium at 34 weeks gestation or within 24 h of delivery (Stewart et al. 1991). At 6 months postpartum, only two (10%) women had experienced an episode of PP. In an earlier case study by Stewart (1988) in Canada, four women who had a history of PP were administered prophylactic lithium immediately following delivery and there were no reported relapses of PP.
UK researchers evaluated the effectiveness of lithium prophylaxis in a retrospective study including 17 women who met the RDC criteria for bipolar disorder or history of puerperal affective psychosis (Austin 1992). Within 3 months postpartum, only two (22%) of the nine women who received prophylactic lithium relapsed in comparison to six (75%) of the eight women who did not receive prophylactic lithium. It should be noted that three of the women in the treatment group were also included in the study of Stewart et al. (1991). In another retrospective study including US women who met the DSM-III-R criteria for bipolar disorder, 14 of the 27 women received various combinations of prophylactic mood stabilizers (Cohen et al. 1995). All but one woman received lithium alone or in combination with either carbamazepine or an antidepressant. Only one (7%) of the 14 in the treatment group relapsed, in comparison to eight (62%) of the 13 in the non-treatment group at 3 months postpartum. The relative risk of relapse was 8.6 times greater for women who did not receive prophylactic mood stabilizers.
Support for the prophylactic effect of lithium also comes from a prospective study of 11 women from the Netherlands who met DSM-III criteria for bipolar disorder (van Gent and Verhoeven 1992). Five of the women were studied during two pregnancies. Of the 16 pregnancies, eight women received prophylactic lithium, two received prophylactic carbamazepine, one received prophylactic haloperidol, while five received no medications. Within 3 months postpartum, three (27%) of the 11 women who were treated with either lithium, carbamazepine, or haloperidol experienced a manic or psychotic relapse compared to three (60%) of five women who refused treatment.
The effects of the mood stabilizer divalproex in preventing postpartum relapse were evaluated in an open clinical trial including 26 US women with a DSM-IV diagnosis of bipolar disorder (Wisner et al. 2004). Fifteen of the women received divalproex immediately postpartum, while 11 of the women received only symptom monitoring. Participants were assessed weekly for 20 weeks using the Structured Clinical Interview for DSM-IV, the Hamilton Rating Scale for Depression, the Bech–Rafaelson Mania Scale, the Mania Rating Scale, the Global Assessment of Function, and the Asberg Side Effects Scale. Within 20 weeks postpartum, 10 (67%) of the 15 women who received divalproex relapsed, compared to eight (73%) of the 11 unmedicated women. There were no significant differences between the two groups. While divalproex was not effective, none of the women who relapsed experienced a severe episode that required admission, which may be associated with the close clinical supervision of the participants.
Overall, prophylactic lithium was supported in five studies (Austin 1992; Cohen et al. 1995; Stewart 1988; Stewart et al. 1991; van Gent and Verhoeven 1992). The prophylactic effects of carbamazepine were demonstrated in two studies (Cohen et al. 1995, van Gent and Verhoeven 1992), while prophylactic divalproex was not supported in one study (Wisner et al. 2004).
Antipsychotics
Only one study to date has explored the effects of antipsychotics for the prevention of PP. An open trial exploring the effects of prophylactic olanzapine was conducted in Canada including 25 women with a history of bipolar disorder, of which 11 received olanzapine alone or in combination with an antidepressant or mood stabilizer, while 14 received either antidepressants, mood stabilizers, or no medication (Sharma et al. 2006). The treatment effects were assessed using the Schedule for Affective Disorders and Schizophrenia—Lifetime Version, the Hamilton Rating Scale for Depression—31 items, the Young Mania Rating Scale, and the Personal Mood Diary. At 4 weeks postpartum, only two (18%) of the 11 women who received olanzapine experienced an affective relapse according to the DSM-IV criteria, in comparison to eight (57%) of the 14 who did not receive olanzapine.
Hormone therapy
Three studies reported the effects of hormone therapy for the prevention of psychosis postpartum. To determine the effects of prophylactic oral estrogen, an open clinical trial was conducted with 11 US women who had a history of either PP or postpartum depression (Sichel et al. 1995). None of the 10 women who complied with the estrogen treatment relapsed. The preventive effects of estrogen were less promising in an open clinical trial with 29 pregnant women from the UK who met the RDC for hypomania, mania, or schizoaffective disorder (Kumar et al. 2003). The Schedule for Affective Disorders and Schizophrenia–Lifetime Version was used to assess the treatment effects of prophylactic transdermal estrogen that was administered 48 h after birth in doses of 200, 400, or 800 μg/day. Estrogen was not effective in reducing the rate of relapse; however, of the 12 out of 29 who relapsed, those who received a dose of 800 μg/day required less subsequent psychotropic medication and recovered sooner. In a case study with a woman from the UK, prophylactic progesterone administered during pregnancy and postpartum was not effective in preventing a relapse of PP (Murray 1990). In summary, there are mixed findings for the preventative effects of estrogen and there is no evidence to support the prophylactic use of progesterone.
Treatment of PP
Research on the treatment of PP has examined the effects of electroconvulsive therapy (ECT), antipsychotics, mood stabilizers, hormones, and the beta-blocker propranolol. The sample size for the 17 treatment studies and case reports ranged from 1 to 114, with 15 (88%) studies including 10 or fewer participants; over half (53%) of the studies had only one participant. Not surprising, the majority (76%) of treatment studies were case reports.
Electroconvulsive therapy
Three studies have explored the effects of ECT in the treatment of PP. In a retrospective study with 114 women, UK researchers compared the clinical response to ECT in women with postpartum and non-postpartum psychosis (Reed et al. 1999). According to blind ratings, the postpartum group was found to have greater clinical improvement following ECT compared to the non-postpartum group. In a case study of a woman from the UK, treatment with chlorpromazine alone resulted in little clinical improvement, while the addition of ECT resulted in positive outcomes (Stanworth 1982). Another case study described five US women who had severe and treatment refractory PP or depression with onset between 3 weeks and 11 months postpartum (Forray and Ostroff 2007). All women were treated with ECT and improved within three to six sessions. Overall, these studies provide beginning support for the beneficial effects of ECT in the treatment of PP.
Mood stabilizers
Three studies have examined the effects of lithium in the treatment of PP. A case study of an Israeli woman revealed a positive response to lithium within 10 days of initiating treatment and patient recovery within a month (Lichtenberg et al. 1988). The woman also had a history of GM2 Gangliosidosis, wherein approximately one third who have this disorder also experience psychosis. A prospective study of 19 women from the Netherlands compared the treatment effects of a group who received lithium combined with perfenazine to a group who received one or more antipsychotic treatments (Silbermann et al. 1975). The lithium treatment group was symptom free within 7–15 days, was less likely to relapse, and had significantly faster recovery times, with a mean of 12 weeks compared to 20 weeks. In a case study, two US women with a history of bipolar disorder experienced PP within 2 weeks postpartum (Targum et al. 1979). One woman was treated with lithium and thioridazine and the other woman with lithium and chlorpromazine. Both women recovered within 17 days. In summary, there have been very few studies examining the effect of lithium. This treatment option was found to be effective in one case study where it was used as monotherapy (Lichtenberg et al. 1988) and in two studies where it was used as adjunct therapy (Silbermann et al. 1975; Targum et al. 1979).
Antipsychotics
The effects of antipsychotics in the treatment of PP have been examined in four studies. One study described a UK woman who experienced PP after two separate pregnancies and reported that treatment with chlorpromazine was successful during both episodes (Murray 1990). In another case study with a UK woman experiencing PP, chlorpromazine combined with lorazepam was ineffective (Marshall and Nursing Care Study 1981). However, when chlorpromazine was increased and combined with imipramine, the woman’s psychotic symptoms decreased within 1 week and she remained symptom free at 12 weeks.
In a case study of a woman from Germany with PP, treatment with clozapine was effective within a week (Kornhuber and Weller 1991). In another case study, a Spanish woman was refractory to treatment with various drugs, including antidepressants, anxiolytics, and the antipsychotics haloperidol and methotrimeprazine (Iruela et al. 1992). Another antipsychotic, pimozide, was administered and was effective within 4 days. Overall, very few studies have explored the effects of antipsychotics in the treatment of PP. There are reports of successful treatment with chlorpromazine, clozapine, and pimozide.
Hormone therapy
Five studies have examined the effects of hormonal treatment among women diagnosed with PP. Three studies conducted in Finland examined the treatment effects of sublingual estrogen in women who had estrogen deficiency and PP (Ahokas and Aito 1999; Ahokas et al. 2000a; Ahokas et al. 2000b). A total of 14 women were described in the three reports. Using the Brief Psychiatric Rating Scale (BPRS), psychiatric symptoms were reported to have decreased within 2 weeks of estrogen treatment. Four women discontinued their estrogen treatment and experienced a relapse within 2 weeks. The case study of a woman from the UK reported a positive outcome for the effects of progesterone in the treatment of PP after the woman did not respond to an unspecified tranquilizer (Atkinson and Atkinson 1983). In a recent case study, a Taiwanese woman with a history of mania was treated for PP with hormone replacement therapy, which consists of estrogen and progesterone (Huang et al. 2008). This treatment was reported to be effective within 2 weeks after the woman was refractory to antipsychotics and mood stabilizers. With only a few individual case reports, it is not possible at present to conclude that any evidence exists for the hormonal treatment of PP.
Propranolol
This drug is a beta-adrenergic blocker that is primarily used to treat hypertension (Prichard and Gillam 1996). Propranolol was first used to treat PP in 1973 (Steiner et al. 1973), although the mechanism in which propranolol works on treating psychiatric conditions is not well understood. To compare the effects of propranolol and chlorpromazine, an open clinical trial was conducted with 10 Israeli women experiencing PP (Steiner et al. 1973). Both treatments were effective in reducing the severity of psychotic symptoms as measured by the BPRS and the Cognitive Global Impression (CGI) instruments; however, women treated with propranolol were discharged sooner (61 days compared to 104 days), symptoms improved more rapidly (3 days compared to 55 days), and demonstrated greater mean improvement on the BPRS and the CGI. The use of propranolol was also supported in a case study of a Canadian woman who was unresponsive to antipsychotics, antidepressants, lithium, and ECT (O’Reagan 1981). Propranolol was said to have “cured” the patient’s psychosis. Overall, only two studies to date have supported the effects of propranolol in the treatment of PP (O’Reagan 1981; Steiner et al. 1973).
Discussion
The aim for this systematic review was to examine the effectiveness of interventions for the prevention and treatment of PP. A total of 26 studies were reviewed, with 10 examining preventative interventions and 17 examining treatment interventions (one study included both prevention and treatment interventions). Studies were conducted in nine countries, with the majority conducted in the UK (33%), followed by the US (19%). The years of publication for the included studies ranged from 1973 to 2008, while approximately two thirds (65%) of the studies were published more than 10 years ago. The methodological quality of the studies differed significantly, resulting in a limited evidence base to guide current practice and decision making. In particular, the study sample sizes were generally small, reports were often retrospective, and the designs were primarily case reports. While the evidence provided from this research is limited, it does highlight promising areas for additional research and may provide clinicians with some suggestions to guide management. In addition, for some treatment approaches with a considerable evidence base in non-postpartum samples—mood stabilizers and antipsychotics for example—further evidence in postpartum cases may be reassuring. For novel treatment options such as hormonal manipulation, however, the lack of data is far more concerning reflecting the need for additional research before conclusions about efficacy can be drawn. Overall, further research is warranted using rigorous methodology to evaluate current preventative and treatment options.
The studies included in this review used a variety of diagnostic criteria to determine participant study inclusion and intervention efficacy. The standardized diagnostic criteria used for inclusion in the preventative studies included the DSM III (van Gent and Verhoeven 1992), DSM III-R (Cohen et al. 1995; Sichel et al. 1995), DSM-IV (Sharma et al. 2006; Wisner et al. 2004), and the RDC (Austin 1992; Kumar et al. 2003; Stewart et al. 1991). Five prevention studies used standardized questionnaires to determine the relapse of postpartum psychosis, including the Schedule for Affective Disorders and Schizophrenia—Lifetime Version (Kumar et al. 2003; Sharma et al. 2006), the Mania Rating Scale (Wisner et al. 2004), the Young Mania Rating Scale (Sharma et al. 2006), the Bech–Rafaelson Mania Scale (Wisner et al. 2004), the Hamilton Rating Scale for Depression (Sharma et al. 2006; Wisner et al. 2004), the Global Assessment of Function (Wisner et al. 2004), the Asberg Side Effects Scale (Wisner et al. 2004), the Personal Mood Diary (Sharma et al. 2006), the DSM III-R checklist (Sichel et al. 1995), the Structured Clinical Interview for DSM-IV (Wisner et al. 2004), and a “standard data form” (Stewart et al. 1991). Only one treatment study (Ahokas et al. 2000a) reported using standardized diagnostic criteria to diagnose postpartum psychosis, namely the ICD-10. Questionnaires were used to assess the treatment effects in seven studies; specifically the BPRS (Ahokas and Aito 1999; Ahokas et al. 2000b; Steiner et al. 1973), the CGI (Steiner et al. 1973), and various unstandardized scales (Forray and Ostroff 2007; Reed et al. 1999; Silbermann et al. 1975). Overall, eight of the prevention studies and only seven of the treatment studies used some form of a standard diagnostic assessment. The diagnostic measures were diverse, making comparisons between studies difficult to interpret. Moreover, the studies included in this review represent a variety of prevention and treatment approaches in various combinations, further precluding any clear comparisons between studies. For these reasons, results from this review should be interpreted with caution.
Prevention
Research on the prevention of PP has focused on the effects of mood stabilizers, antipsychotics, and hormone therapy. Lithium was the most commonly studied prevention approach with research suggesting it may decrease the rate of relapse from approximately 50% to less than 22% (Austin 1992; Cohen et al. 1995; Stewart 1988; Stewart et al. 1991). Further support for the use of lithium comes from studies demonstrating high relapse rates among women who discontinue their lithium during pregnancy (Targum et al. 1979; Viguera et al. 2000). There is insufficient evidence to suggest whether lithium is equally effective when administered prophylactically through pregnancy or started immediately in the postpartum period.
Only one study has focused on the effects of antipsychotics in the prevention of PP and positive results have been found in a small trial of olanzapine (Sharma et al. 2006). Additional research is clearly required, but the dearth of evidence for prophylactic antipsychotic medication in the postpartum context must be considered in light of the more robust evidence base for these medications in non-postpartum samples. In the case of hormonal prophylaxis, however, the evidence base is poor and without the studies demonstrating efficacy in mood disorders more generally there is insufficient evidence to support their use at present. What is very clear is that women with a history of PP or bipolar disorder who do not receive prophylactic treatment are at an increased risk for relapse of affective symptoms (Austin 1992; Cohen et al. 1995). Larger naturalistic and possibly randomized controlled trials are required to examine the effects of prophylactic interventions.
Treatment
Research on the treatment of PP has examined the effects of ECT, mood stabilizers, antipsychotics, hormones, and propranolol. The limited evidence supports the use of ECT in the treatment of PP when administered alone (Forray and Ostroff 2007; Reed et al. 1999), as well as when combined with the antipsychotic chlorpromazine (Stanworth 1982). Overall, the use of antipsychotics in the treatment of PP requires further investigation before any conclusions can be drawn about their effectiveness postnatally. Their use at this time, however, must be considered in light of the large body of evidence of efficacy in non-postpartum episodes.
Lithium is commonly used in the treatment of PP; however, the evidence to support this treatment option is scarce. In the few studies conducted to date (Lichtenberg et al. 1988; Silbermann et al. 1975; Targum et al. 1979), lithium treatment for PP is generally supported. The effectiveness of lithium in the treatment of bipolar disorder is well documented and it has been suggested that lithium is equally effective with postpartum women as it is with non-postpartum women (Abou-Saleh and Coppen 1983). Further comparative investigations are needed that examine whether the effects of lithium are equally effective in treating episodes of PP.
Three studies conducted by the same group (Ahokas and Aito 1999; Ahokas et al. 2000a, b) have found beneficial effects of estrogen. The potential beneficial use of progesterone (Atkinson and Atkinson 1983) and hormone replacement therapy (Huang et al. 2008) were described through case studies. There is insufficient research to recommend hormone treatment; however, preliminary results would suggest that further studies are indicated—particularly as the involvement of hormonal factors in etiology come to be better understood.
There is an array of differences reported across studies on the effect of interventions for PP. Health professionals should therefore gain knowledge on what methods have been effective for women in the past and if any adverse effects were experienced (Sharma 2003). Although we have focused in this review on biological approaches, treatment should also go beyond the medical symptoms and include psychosocial and psychological therapy given the complex and multiple needs of women diagnosed with PP.
Safety of medication during pregnancy and breastfeeding
An important consideration is the reproductive safety of medication in pregnancy and breastfeeding. While beyond the scope of this review, there is clearly a need to conduct a risk benefit analysis with each individual woman. Although some medications are associated with particular problems, there are no universal recommendations that can be made about whether to continue or stop and the decision must ultimately lie with the woman and her family. Moreover, women with bipolar disorder need to be informed early in their illness about the risks in childbirth and the reproductive safety of medication and family planning should be discussed when drugs are prescribed.
Limitations
The review only included studies published in English. Although most cases of PP occur within the first 2 weeks after giving birth (Heron et al. 2007), we included onset within 1 year postpartum. In 10 (38%) of the studies included in this review, women had onset times beyond 2 weeks postpartum. This may preclude any clear inferences to be made about the participants who exceeded 2 weeks in onset, as they may be cases that are coincidentally psychotic in the postpartum period, and not truly cases of PP.
Implications for future research
There are numerous gaps in the research on strategies to prevent and treat PP. Agreement is needed on the diagnosis and classification of the disorder that is not dealt with in a particularly helpful way in current DSM-IV and ICD-10 criteria. While acquiring large representative samples and conducting randomized controlled trials is problematic, collaborative multicenter naturalistic studies are possible and would provide very helpful data.
Future research could also focus on gaining a better understanding of the causes of PP, as this will act as a guide for research on prevention and treatment. Given the equivocal evidence on hormone therapy in both the prevention and treatment of PP, research on the etiology of PP could further examine the role of hormonal factors to guide management.
Conclusion
Firm recommendations on effective interventions are limited until further research is conducted on all forms of prevention and treatment for PP. PP represents a psychiatric emergency, thus the timely recognition of symptoms and early treatment is critical to the well-being of the affected woman and her family. The well-established risk factors (e.g., bipolar disorder) and potentially severe consequences (e.g., suicide and infanticide) of PP point to the importance of preventive measures. Multicenter prospective studies are desperately needed to provide guidance on prevention and treatment interventions.
References
Abou-Saleh MT, Coppen A (1983) Puerperal affective disorders and response to lithium. Br J Psychiatry 142:539
Ahokas A, Aito M (1999) Role of estradiol in puerperal psychosis. Psychopharmacology 147:108–110
Ahokas A, Aito M, Rimon R (2000a) Positive treatment effect of estradiol in postpartum psychosis: a pilot study. J Clin Psychiatry 61:166–169
Ahokas A, Aito M, Turtiainen S (2000b) Association between oestradiol and puerperal psychosis. Acta Psychiatr Scand 101:167–170
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th edn. American Psychiatric Association, Washington, text revision
Appleby L, Mortensen PB, Faragher EB (1998) Suicide and other causes of mortality after post-partum psychiatric admission. Br J Psychiatry 173:209–211
Atkinson S, Atkinson T (1983) Puerperal psychosis: a personal experience. Health Visit 56:17–19
Austin MV (1992) Puerperal affective psychosis: is there a case for lithium prophylaxis? Br J Psychiatry 161:692–694
Brockington IF (1996) Motherhood and mental health. Oxford University Press, Oxford
Brockington IF, Cernik KF, Schofield EM, Downing AR, Francis AF, Keelan C (1981) Puerperal psychosis: phenomena and diagnosis. Arch Gen Psychiatry 38:829–833
Chandra PS, Bhargavaraman RP, Raghunandan VNGP, Shaligram D (2006) Delusions related to infant and their association with mother–infant interactions in postpartum psychotic disorders. Arch Womens Ment Health 9:285–288
Cohen LS, Sichel DA, Robertson LM, Heckscher E, Rosenbaum JF (1995) Postpartum prophylaxis for women with bipolar disorder. Am J Psychiatry 152:1641–1645
Forray A, Ostroff RB (2007) The use of electroconvulsive therapy in postpartum affective disorders. J ECT 23:188–193
Heron J, Robertson-Blackmore E, McGuinness M, Craddock N, Jones I (2007) No latent period in bipolar affective puerperal psychosis. Arch Womens Ment Health 10:79–81
Heron J, McGuinness M, Robertson Blackmore E, Craddock N, Jones I (2008) Early postpartum symptoms in puerperal psychosis. Br J Obstet Gynaecol 115:348–353
Hipwell AE, Goossens FA, Melhuish EC, Kumar R (2000) Severe maternal psychopathology and infant–mother attachment. Dev Psychopathol 12:157–175
Huang MC, Wang YB, Chan CH (2008) Estrogen-progesterone combination for treatment-refractory post-partum mania. Psychiatry Clin Neurosci 62:126
Iruela LM, Ibañez-Rojo V, Gilaberte I, Oliveros SC (1992) New possible indications of pimozide. J Clin Psychiatry 53:172–173
Jones I, Craddock N (2001) Familiarity of the puerperal trigger in bipolar disorder: results of a family study. Am J Psychiatry 158:913–917
Kendell R, Chalmers J, Platz C (1987) Epidemiology of puerperal psychoses. Br J Psychiatry 150:662–673
Kornhuber J, Weller M (1991) Postpartum psychosis and mastitis: a new indication for clozapine? Am J Psychiatry 148:1751–1752
Kumar C, McIvor RJ, Davies T (2003) Estrogen administration does not reduce the rate of recurrence of affective psychosis after childbirth. J Clin Psychiatry 64:112–118
Lichtenberg P, Navon R, Wertman E, Dasberg H, Lerer B (1988) Post-partum psychosis in adult GM2 Gangliosidosis: a case report. Br J Psychiatry 153:387–389
Marshall S, Nursing care study (1981) Puerperal psychosis: with a lot of help from her friends. Nurs Mirror 152:46–47
Munk-Olsen T, Laursen TM, Pedersen CB, Mors O, Mortensen PB (2006) New parents and mental disorders: a population-based register study. JAMA 296:2582–2589
Murray D (1990) Recurrence of puerperal psychosis not prevented by prophylactic progesterone administration. J Nerv Ment Dis 178:537–538
O’Hara MW, Swain AM (1996) Rates and risk of postpartum depression: a meta-analysis. Int Rev Psychiatry 8:37–54
O’Reagan JB (1981) Treatment of acute postpartum psychosis. Can Med Assoc J 125:1083
Prichard BNC, Gillam PMS (1996) Treatment of hypertension with propranolol. Br Med J 1:7–16
Reed P, Sermin N, Appleby L, Faragher B (1999) A comparison of clinical response to electroconvulsive therapy in puerperal and non-puerperal psychoses. J Affect Disord 54:255–260
Robertson E, Jones I, Haque S, Holder R, Craddock N (2005) Risk of puerperal and non-puerperal recurrence of illness following bipolar affective puerperal (post-partum) psychosis. Br J Psychiatry 186:258–259
Sharma V (2003) Pharmacotherapy of postpartum psychosis. Expert Opin Pharmacother 4:1651–1658
Sharma V, Smith A, Mazmanian D (2006) Olanzapine in the prevention of postpartum psychosis and mood episodes in bipolar disorder. Bipolar Disord 8:400–404
Sichel DA, Cohen LS, Robertson LM, Ruttenberg A, Rosenbaum JF (1995) Prophylactic estrogen in recurrent postpartum affective disorder. Biol Psychiatry 38:814–818
Silbermann RM, Beenen F, de Jong H (1975) Clinical treatment of post partum delirium with perfenazine and lithium carbonate. Psychiatr Clin 8:314–326
Sit D, Rothschild AJ, Wisner KL (2006) A review of postpartum psychosis. J Womens Health 15:352–368
Spinelli MG (2004) Maternal infanticide associated with mental illness: prevention and promise of saved lives. Am J Psychiatry 161:1548–1557
Stanworth HM (1982) After-care of puerperal psychosis in the community. Nurs Times 78:922–925
Steiner M, Latz A, Blum I, Atsmon A, Wijsenbeek H (1973) Propranolol versus chlorpromazine in the treatment of psychoses associated with childbearing. Psychiatr Neurol Neurochir 76:421–426
Stewart DE (1988) Prophylactic lithium in postpartum affective psychosis. J Nerv Ment Dis 176:485–489
Stewart DE, Klompenhouwer JL, Kendall RE, Van Hulst AM (1991) Prophylactic lithium in puerperal psychosis: the experience of three centres. Br J Psychiatry 158:393–397
Stowe ZN (2007) The use of mood stabilizers during breastfeeding. J Clin Psychiatry 68(Suppl 9):22–28
Targum S, Davenport Y, Webster M (1979) Postpartum mania in bipolar manic-depressive patients withdrawn from lithium carbonate. J Nerv Ment Disord 167:572–574
Terp IM, Engholm G, Moller H, Mortenson PB (1999) A follow-up study of postpartum psychoses: prognosis and risk factors for readmission. Acta Psychiatr Scand 100:40–46
van Gent EM, Verhoeven WM (1992) Bipolar illness, lithium prophylaxis, and pregnancy. Pharmacopsychiatry 25:187–191
Viguera AC, Nonacs R, Cohen LS, Tondo L, Murray A, Baldessarini RJ (2000) Risk of recurrence of bipolar disorder in pregnant and nonpregnant women after discontinuing lithium maintenance. Am J Psychiatry 157:179–184
Wisner KL, Hanusa BH, Peindl KS, Perel JM (2004) Prevention of postpartum episodes in women with bipolar disorder. Biol Psychiatry 56:592–596
World Health Organization (1992) The ICD-10 Classification of Mental and Behavioral Disorders. World Health Organization, Geneva
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Doucet, S., Jones, I., Letourneau, N. et al. Interventions for the prevention and treatment of postpartum psychosis: a systematic review. Arch Womens Ment Health 14, 89–98 (2011). https://doi.org/10.1007/s00737-010-0199-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00737-010-0199-6