Abstract
Na+/H+ exchanger (NHE)-3 is important for intestinal absorption of nutrients and minerals, including calcium. The previous investigations have shown that the intestinal calcium absorption is also dependent on luminal nutrients, but whether aliphatic amino acids and glucose, which are abundant in the luminal fluid during a meal, similarly enhance calcium transport remains elusive. Herein, we used the in vitro Ussing chamber technique to determine epithelial electrical parameters, i.e., potential difference (PD), short-circuit current (Isc), and transepithelial resistance, as well as 45Ca flux in the rat duodenum directly exposed on the mucosal side to glucose or various amino acids. We found that mucosal glucose exposure led to the enhanced calcium transport, PD, and Isc, all of which were insensitive to NHE3 inhibitor (100 nM tenapanor). In the absence of mucosal glucose, several amino acids (12 mM in the mucosal side), i.e., alanine, isoleucine, leucine, proline, and hydroxyproline, markedly increased the duodenal calcium transport. An inhibitor for NHE3 exposure on the mucosal side completely abolished proline- and leucine-enhanced calcium transport, but not transepithelial transport of both amino acids themselves. In conclusion, glucose and certain amino acids in the mucosal side were potent stimulators of the duodenal calcium absorption, but only amino-acid-enhanced calcium transport was NHE3-dependent.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The activity of Na+/H+ exchanger (NHE), especially NHE3 in the apical membrane [also known as solute carrier family 9 member 3 (Slc9a3)], is crucial for intestinal absorption of various nutrients and minerals, such as tripeptides, fatty acids, sodium, and calcium, the latter of which also requires certain nutrients, particularly glucose and amino acids, to enhance its absorption (Comar et al. 1956; Mineo et al. 2002; Lu et al. 2016). Moreover, the function of NHE3 is also essential for proper intracellular acid–base regulation (Donowitz et al. 2013). A recent investigation in NHE3 knockout mice showed that the intestinal calcium absorption was markedly diminished in the absence of apical NHE3 expression (Pan et al. 2012). Since the transepithelial calcium transport is upregulated by a number of luminal nutrients, including sugars and amino acids (Comar et al. 1956; Mineo et al. 2002), we hypothesize that NHE3 may contribute to the intestinal calcium transport induced by those liminal factors. Indeed, NHE3 inhibitors (e.g., tenapanor) have been recognized as drugs capable of restricting intestinal sodium absorption and salt overload for patients with renal diseases and hypertension (Gerritsen et al. 2015), but whether they can reduce the luminal nutrient-induced calcium absorption is not known.
Under normal conditions, calcium ions move across the epithelial sheet via two pathways, i.e., transcellular and paracellular pathways. The transcellular calcium transport is generally a three-step cellular energy-dependent mechanism, consisting of apical calcium entry through calcium channels [e.g., transient receptor potential vanilloid family calcium channel (TRPV)-6 and voltage-dependent calcium channels (Cav)], calbindin-D9k-mediated cytoplasmic calcium transfer, and basolateral calcium extrusion via the plasma membrane Ca2+-ATPase (PMCA) and Na+/Ca2+ exchanger (NCX)-1 (Hoenderop et al. 2005; Kellett 2011). Luminal nutrients, such as glucose and aliphatic amino acids, may contribute considerably to both pathways, because their apical transporters, particularly sodium-dependent glucose transporter (SGLT)-1, and systems B and B0,+ amino-acid transporters, allow sodium influx into the enterocytes (Kellett 2011; Pramod et al. 2013). It has been suggested that the electrogenic sodium uptake depolarizes the apical membrane and, in turn, activates the voltage-dependent calcium channels (e.g., Cav1.3) for cellular calcium entry (Kellett 2011). In addition, in the absence of transepithelial calcium gradient—i.e., luminal and extracellular fluid compartments containing the same calcium concentration—calcium can traverse the paracellular space by solvent drag-induced mechanism (Charoenphandhu and Krishnamra 2007; Kellett 2011). Specifically, the absorbed sodium is pumped by Na+/K+-ATPase lining the lateral membrane into the paracellular space, thus creating osmotic gradient for the lumen-to-plasma water flow, which allows movement of calcium along with the stream of water (for review, please see Larsen et al. 2000; Charoenphandhu and Krishnamra 2007; Kellett 2011). In the rat duodenum, nearly 85% of the glucose-induced calcium transport has been known to occur via the solvent drag mechanism (Tanrattana et al. 2004), but the proportion due to amino-acid transport remains elusive.
Besides sodium-nutrient coupling, the apical NHE3 also mediates sodium uptake into the enterocytes and H+ efflux, thereby preventing intracellular acidification (Thwaites et al. 1999). Relatively low intracellular H+ levels are essential for proper function of cytoplasmic calcium-binding protein, calbindin-D9k with the optimal pH for its calcium-binding affinity between pH 7 and 9 (Kesvatera et al. 2001). Long-lasting absence of NHE3 in NHE3 knockout mice showed impairment of intestinal water, sodium, and calcium absorption, the latter of which was caused by decreases in calcium fluxes through the transcellular and paracellular routes, partially due to downregulated expression of calcium-transporting genes, i.e., calbindin-D9k, TRPV6, claudin-2, and claudin-15 (Pan et al. 2012; Rievaj et al. 2013). However, the effects of acute NHE3 inhibition as well as its mechanism on calcium absorption are still not known. Since NHE3 primarily disturbs intracellular pH balance, acute inhibition of NHE3 is postulated to predominantly compromise the calbindin-D9k-mediated transcellular calcium transport, but not the solvent drag-induced calcium transport. In other words, NHE3 inhibitor is unlikely to affect the glucose-induced calcium transport process, in which most calcium ions move across the paracellular space but not the cytoplasm (Wongdee and Charoenphandhu 2015).
This study, therefore, aimed to investigate the transepithelial calcium transport in the rat duodenum directly exposed to glucose and various aliphatic amino acids on the mucosal side. Tenapanor, an NHE3 inhibitor, was applied to demonstrate the NHE3-dependent transepithelial calcium transport mechanism. The duodenum was used in this study, since it could transport calcium via both transcellular and paracellular pathways (Tanrattana et al. 2004), and expressed transporters for amino acids (e.g., systems B) and oligopeptides (e.g., H+-coupled peptide transporter-1 (PEPT1)–NHE3 system) (Freeman et al. 1995; Romeo et al. 2006).
Materials and methods
Animals
Female Sprague–Dawley rats (7 weeks old, weighing 180–200 g) were obtained from the National Laboratory Animal Center, Mahidol University. The animals were acclimatized in polystyrene cages (2 animals per cage) under 12:12-h light–dark cycle with average illuminance of 200 lx in the laboratory animal husbandry unit for 7 days before the beginning of the experiment. Room temperature was maintained in the range of 20–25 °C with 50–60% humidity. They were regularly fed standard chow containing 1% wt/wt calcium, 0.9% phosphorus, and 4000 IU/kg vitamin D (CP, Bangkok, Thailand) and reverse osmosis water ad libitum. This study has been approved by the Animal Care and Use Committee of the Faculty of Science, Mahidol University, Thailand.
Chemicals
Radioactive materials, i.e., 45Ca (catalog no. NEZ013), (3H)leucine (catalog no. 460250), and (14C)proline (catalog no. NEC851050), were purchased from PerkinElmer (Boston, MA, USA). The selective NHE3 inhibitor, i.e., tenapanor [N,N′-(10,17,-dioxo-3,6,21,24-tetraoxa-9,11,16,18-tetraazahexacosane-1,26-diyl)bis([(4S)-6,8-dichloro-2-methyl-1,2,3,4-tetrahydroisoquinolin-4-yl]benzenesulfonamide); catalog no. A14011], was purchased from AdooQ Bioscience (CA, USA). The l-amino acids, i.e., glycine, lysine, valine, methionine, isoleucine, alanine, proline, hydroxyproline, and leucine, were purchased from Himedia (Mumbai, India). The l-leucine tripeptide (l-Leu-Leu–Leu; catalog no. 067594) was purchased from the First Base Laboratories (Selangor, Malaysia). TRPV6 blocker (100 µM ruthenium red; catalog no. ab120264) and cyclooxygenase (COX) inhibitor (50 nM indomethacin; catalog no. ab120719) were purchased from Abcam (Cambridge, MA, USA). Inhibitors for PMCA [200 and 400 µM trifluoperazine (TFP); catalog No. T8516] and NCX1 (high-dose KB-R7943 of 150 µM; catalog No. K4144) were purchased from Sigma (St. Louis, MO, USA).
Experimental design
The experiment was divided into two parts, i.e., the mucosal glucose-rich and amino-acid-rich experiments. In the glucose-rich experiment, both apical and basolateral sides of the duodenal epithelium were exposed to bathing solution with equal glucose concentration of 12 mM, whereas the mucosal glucose-free condition was used as a control. In amino-acid-rich experiment, the duodenal epithelium was exposed to 12 mM of various amino acids on the apical side. Glucose was presented only in the basolateral compartment to nourish the tissue. Osmolality of 290 mmol/kg H2O was adjusted with mannitol. The studied amino acids were aliphatic amino acids (i.e., alanine, glycine, isoleucine, leucine, proline, and valine), a sulfur-containing amino acid (methionine), and a basic amino acid (lysine). The measured parameters were transepithelial calcium flux and epithelial electrical parameters, which included potential difference (PD), short-circuit current (Isc), and transepithelial resistance (TER). To demonstrate whether the enhanced transepithelial calcium flux was dependent on NHE3, tenapanor with optimal concentration of 100 nM (Gerritsen et al. 2015) was present into the apical or basolateral compartments.
Intestinal tissue preparation
After the rat was anesthetized by intraperitoneal injection of 50 mg/kg sodium pentobarbitone (Nembutal, Abbott Laboratory, North Chicago, IL, USA), a median laparotomy was made, and the duodenum segment (~5 cm distal to the pylorus) was removed and placed in an ice-cold physiological bathing solution, which contained 118 mM NaCl, 4.7 mM KCl, 1.1 mM MgSO4, 1.25 mM CaCl2, 23 mM NaHCO3, 1 mM 2 d-glucose, and 2 mM mannitol (Sigma), and gassed with 5% CO2 in 95% O2 (carbogen). The duodenal segment was then cut longitudinally along the radix mesenterii in ice-cold physiological bathing solution to expose the mucosa, and rinsed to remove the luminal contents. Subsequently, it was mounted in a modified Ussing chamber with an exposed surface area of 0.69 cm2. The tissue was firmly adhered to the chamber with adhesive silicone grease (Unilab, New South Wales, Australia), and both sides were immersed in 3.5 mL bathing solution maintained at 37 °C. Then, the tissue was equilibrated for 15–20 min before performing a 50-min experiment.
Measurement of transepithelial calcium flux
Transepithelial calcium transport across the duodenal tissue was determined by the method of Charoenphandhu et al. (2009). In brief, the duodenal tissue was equilibrated in Ussing chamber for 10 min, and then the bathing solution was replaced on both sides with fresh solution. The apical hemichamber was filled with either (1) physiological bathing solution (glucose-replete) or (2) glucose-depleted bathing solution with 12 mM of each amino acid. 45Ca (0.45 µCi/ml; final specific activity of 90 mCi/mol) was present in the apical solution. Two samples from the hot side (H; apical chamber; mucosal side) were collected at 10 and 20 min, while five samples were collected from the cold side (C; basolateral chamber; serosal side) every 10 min until the end of the experiment. Unidirectional calcium flux (J H→C; nmol h−1 cm−2) from the hot side to the cold side was calculated by the following equations:
where R H→C is the rate of 45Ca appearance in the cold side (cpm h−1); S H, the specific activity in the hot side (cpm nmol−1); A, the epithelial surface area (cm2); C H, the mean radioactivity in the hot side (cpm); and C To, the total calcium content in the hot side (nmol). 45Ca was analyzed by liquid scintillation spectrophotometry (model Tri-Carb 3100; Packard). Total calcium concentration in bathing solution was analyzed by atomic absorption spectrophotometry (model SpectrAA-300; Varian Techtron). Under the present condition (without transepithelial calcium gradient), calcium ions were transported across the duodenal epithelium via either the transcellular or solvent drag-induced pathways (Wongdee and Charoenphandhu 2015).
Measurement of amino-acid flux
In the study of amino-acid fluxes (l-leucine and l-proline), after 10-min equilibration of the duodenal tissue, both mucosal and serosal bathing solutions were replaced with fresh bathing solution. The mucosal bathing solution was filled with amino-acid-rich (12 mM) or glucose-depleted bathing solution containing a radioactive tracer [56.28 mCi/mol (3H)leucine or 74.5 mCi/mol (14C)proline]. Two samples from the mucosal (hot) side were collected at 10 and 20 min, while five samples were collected from the serosal (cold) side every 10 min until the end of the experiment to calculate unidirectional flux of amino acid from the hot side to the cold side (Eqs. 1 and 2).
Determination of epithelial electrical parameters
PD and Isc were recorded by two pairs of electrodes made of Ag/AgCl half-cells connected to the chamber through 4 M KCl per 3 g% agar bridge. PD-sensing electrodes were placed near the tissue surface, and were connected to a pre-amplifier (model EVC-4000, World Precision Instruments, Sarasota, FL, USA) and a PowerLab 4/30 (AD Instruments, Colorado Springs, CO, USA). A current-applying electrode was placed at the end of each hemichamber. The Isc was recorded by PowerLab 4/30 connected in series to the EVC-4000 current-generating unit. PowerLab was operated by Chart version 5.2.2 for Mac OS X. TER was calculated from Ohm’s equation. Tissue viability was checked by continuous measurement of PD and Isc. Moreover, at the end of the experiment, mucosal exposure to glucose-rich solution could increase PD of the intestinal epithelium.
Statistical analysis
The results are expressed as mean ± SE. Two sets of data were compared by unpaired Student’s t test. Multiple comparisons were performed by one-way analysis of variance (ANOVA) with Dunnett’s post-test. The level of significance was p < 0.05. All data were analyzed by GraphPad Prism 6.0 (GraphPad Software Inc., San Diego, CA, USA).
Results
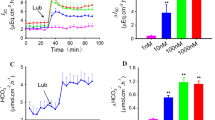
In the present control condition, the apical solution was glucose free, whereas the basolateral solution contained 12 mM glucose. As shown in Fig. 1, the apical glucose markedly increased the transepithelial calcium transport, concurrently with increases in PD and Isc. Apical glucose exposure did not alter TER of the rat duodenum. Exposure to NHE3 inhibitor, tenapanor, on the apical side, had no effect on the glucose-induced transepithelial calcium transport or epithelial electrical property, indicating that this enhanced calcium flux was independent of NHE3.
a Transepithelial calcium transport and b–d epithelial electrical parameters (PD, Isc, and TER) in the rat duodenums with or without 12 mM mucosal glucose. In some experiments, 100 nM tenapanor (NHE3 inhibitor) was present in the mucosal solution throughout the entire experimental period. PD values were the magnitudes of potential difference (the apical side being negative with respect to the basolateral side), and glucose made the apical side more negative. ***p < 0.001 compared with the corresponding control group (open bar). Numbers in parentheses are numbers of animals
In the presence of various amino acids without glucose in the apical solution, transepithelial calcium transport was markedly enhanced, especially after exposure to leucine, proline, hydroxyproline, isoleucine, alanine, and lysine (Fig. 2a). Glycine, valine, and methionine tended to increase the duodenal calcium transport, but such changes were not statistically significant (Fig. 2a). Since the duodenum was well responsive to leucine and proline, these amino acids were used in the subsequent calcium flux studies. Interestingly, a combined glucose and leucine treatment synergistically enhanced calcium flux as compared with glucose treatment alone (p < 0.001) and control condition (p < 0.001; Fig. 2a, inset). Nevertheless, unlike glucose-induced calcium transport, NHE3 inhibitor tenapanor completely abolished the leucine-, proline-, and hydroxyproline-enhanced calcium transport in the rat duodenum (Fig. 2a). Analyses of PD and Isc suggested that several amino acids, i.e., methionine, glycine, alanine, isoleucine, proline, and leucine, induced electrogenic ion transport, because both electrical parameters were significantly elevated above the control levels (Fig. 2b, c). The proline- and leucine-induced PD and Isc changes were diminished by tenapanor. All amino acids lowered TER of the duodenal epithelium, and tenapanor did not alter TER in hydroxyproline-, proline-, or leucine-exposed groups (Fig. 2d).
a Transepithelial calcium transport and b–d epithelial electrical parameters (PD, Isc, and TER) in the rat duodenums exposed on the mucosal side to various amino acids (12 mM each) with or without 100 nM tenapanor. The control group was glucose-free on the mucosal side and glucose-replete on the serosal side (n = 5–10 rats/group). The studied amino acids were methionine (Met), valine (Val), glycine (Gly), lysine (Lys), alanine (Ala), isoleucine (Ile), hydroxyproline (Hyp), proline (Pro), and leucine (Leu). PD values were the magnitudes of potential difference (the apical side being negative with respect to the basolateral side), and amino acids made the apical side more negative. *p < 0.05 compared with the control group (open bar), # p < 0.05 compared with hydroxyproline-exposed group, † p < 0.05 compared with proline-exposed group, ‡ p < 0.05 compared with leucine-exposed group. Inset Transepithelial calcium transport in the rat duodenum exposed to 12 mM glucose or combined 4 mM glucose (Glu) and 4 mM Leu. The control group in the inset was the same as in a. ***p < 0.001 compared with the control group (open bar), ### p < 0.001 compared with the glucose-exposed group
Since NHE3 is believed to mainly localize in the apical membrane but not basolateral membrane (Kulaksiz et al. 2001), we hypothesized that only apical exposure to NHE3 inhibitor could diminish the amino-acid-induced calcium transport. As shown in Fig. 3, the effects of tenapanor on calcium transport and epithelial electrical properties were not observed when tenapanor was present in the basolateral side. On the other hand, the apical application of tenapanor decreased the proline- and leucine-enhanced transepithelial calcium transport, PD, and Isc. The duodenal TER was not altered by apical or basolateral exposure to tenapanor (Fig. 3). Furthermore, tenapanor had no effect on the transepithelial fluxes of proline and leucine in the rat duodenum, as determined by radioactive tracers, (3H)leucine or (14C)proline (Fig. 4).
a Transepithelial calcium transport and b–d epithelial electrical parameters (PD, Isc, and TER) in the rat duodenums exposed on the mucosal side to 12 mM proline or leucine. Tenapanor (100 nM) or its vehicle [0.1% vol/vol dimethyl sulfoxide (DMSO); 3.5 µL) was present in the apical or basolateral solution. PD values were the magnitudes of potential difference (the apical side being negative with respect to the basolateral side), and amino acids made the apical side more negative. ***p < 0.001 compared with the corresponding control group (open bar). Numbers in parentheses are numbers of animals
Transepithelial proline (a) and lysine fluxes (b) in the mucosa-to-serosa direction across the duodenal epithelia. The mucosal solution contained 12 mM proline or leucine. Tenapanor (100 nM) was present in the mucosal solution throughout the entire experimental period. Numbers in parentheses are numbers of animals
In general, calcium moves across the apical membrane via TRPV6 and/or Cav (Wongdee and Charoenphandhu 2015). Since the amino-acid-induced electrogenic ion transport may depolarize the apical membrane and activate Cav, duodenal tissue was exposed on the apical side to nifedipine—a Cav blocker—to determine whether Cav contributed to the leucine-induced calcium transport. As shown in Fig. 5a, nifedipine did not affect the magnitude of leucine-induced calcium flux. This inhibitor also had no effect on the electrical parameters of leucine-treated duodenal epithelia (data not shown). In contrast, other transcellular calcium transport inhibitors, i.e., ruthenium red (TRPV6 blocker), TFP (PMCA inhibitor), and KB-R7943 (NCX1 inhibitor), markedly suppressed the leucine-induced calcium flux (Fig. 5b). Moreover, a COX inhibitor, indomethacin, had no effect on the leucine-induced calcium transport, suggesting the absence of prostaglandin involvement under this condition.
a Effect of Cav inhibitor, nifedipine, on the leucine-induced duodenal calcium transport. The mucosal side of the duodenal tissue was exposed to 12 mM glucose or 12 mM leucine. Nifedipine (10 µM) was present in the mucosal solution throughout the entire experimental period. **p < 0.01 compared with the glucose- and leucine-free group (open bar). NS not significant. b Transepithelial calcium transport in the rat duodenums exposed on the apical side to TRPV6 blocker (100 µM ruthenium red; Ruth) or the basolateral side to PMCA inhibitor (200 and 400 µM trifluoperazine; TFP), NCX1 inhibitor (150 µM KB-R7943), or COX inhibitor (50 nM indomethacin). * p < 0.05, ***p < 0.001 compared with 12 mM Leu-treated group (glucose-free; open bar). Numbers in parentheses are numbers of animals
Finally, contribution of NHE3 was confirmed using a known property of the PEPT1–NHE3 system, in which stimulation of PEPT1 activity could, in turn, enhance NHE3 activity and vice versa (Thwaites et al. 2002; Chen et al. 2010). The results showed that leucine tripeptide—a substrate of PEPT1 but not amino-acid transporter—also enhanced the duodenal calcium transport with similar potency to leucine (Fig. 6). Tenapanor markedly inhibited the stimulatory effect of leucine tripeptide on the duodenal calcium transport (Fig. 6).
Transepithelial calcium transport in the rat duodenum exposed on the mucosal side to 12 mM glucose, 12 mM leucine, or 4 mM l-leucine tripeptide (l-Leu-Leu–Leu). In some experiments, 100 nM tenapanor was also present in the mucosal solution. ***p < 0.001 compared with the control group (open bar). Numbers in parentheses are numbers of animals
Discussion
It has long been known that calcium absorption in the small intestine is dependent on luminal sodium (Kellett 2011; Wongdee and Charoenphandhu 2015). There are a number of explanations to account for the contribution of luminal sodium in the enhancement of calcium transport. First, after entering the enterocytes, cytoplasmic sodium is pumped into the paracellular space to create hyperosmotic environment for the solvent drag-induced paracellular calcium transport (Kellett 2011; Wongdee and Charoenphandhu 2015). Second, the apical calcium influx can depolarize the apical membrane, which, in turn, opens the nifedipine-sensitive Cav for calcium entry (Kellett 2011; Wongdee and Charoenphandhu 2015). The second mechanism is still debatable, since some investigators suggested that its physiological significance might be trivial under normal conditions (Reyes-Fernandez and Fleets 2015). Nevertheless, under conditions in which high-calcium demand drives the enterocytes to recruit every mechanism to obtain adequate calcium supply (e.g., during lactating period), the sodium-induced calcium influx through Cav becomes of greater importance (Nakkrasae et al. 2010; Dorkkam et al. 2013). In this study, we further elaborated the contribution of NHE3—one of the prominent sodium transporters in the small intestine (Donowitz et al. 2013)—in the regulation of intestinal calcium absorption. Specifically, the enhancement of the duodenal calcium transport by amino acids, but not glucose, appeared to be dependent on NHE3. This finding thus extends the current knowledge of how luminal sodium modulates calcium absorption.
Glycine, alanine, and glucose have been reported to enhance calcium absorption (Comar et al. 1956; Mineo et al. 2002). Herein, we further demonstrated that most but not all aliphatic amino acids, i.e., alanine, leucine, isoleucine, and proline, as well as hydroxyproline and lysine were capable of stimulating the duodenal calcium transport in the absence of mucosal glucose. Their apical transporters could be systems B (Slc6a19), B0,+ (Slc6a14), and/or imino transporters (Slc6a20), the functions of which are also dependent on mucosal sodium (Pramod et al. 2013). Previously, both amino acids and glucose were hypothesized to increase calcium absorption through the similar sodium-dependent mechanisms (Turner et al. 1997; Mackenzie and Erickson 2004). However, we found here that their underlying mechanism was totally different. In the presence of glucose, the PD and Isc became greater than those in glucose-free condition, suggesting that apical glucose markedly enhanced the electrogenic ion transport. The ion species that affect both electrical parameters has long been known to be sodium, particularly through SGLT1 (Larsen et al. 2002), leading to a stimulation of the solvent drag-induced calcium transport (Tanrattana et al. 2004; Kellett 2011). Although glucose could induce calcium movement across the transcellular pathway (e.g., through Cav), Tanrattana et al. (2004) reported that the predominant proportion (>80%) was the paracellular calcium transport. Thus, the glucose-enhanced calcium transport in this study probably occurred via the solvent drag-induced pathway rather than the transcellular pathway.
The absorption of aliphatic amino acids (e.g., leucine, isoleucine, and proline) is usually accompanied by apical sodium entry (Mackenzie and Erickson 2004; Bröer 2008), consistent with changes in PD and Isc. Although the process could lead to depolarization of the apical membrane leading to opening of Cav channels (Morgan et al. 2007; Kellett 2011), the absence of nifedipine effect suggested that Cav did not contribute to the apical calcium entry (Kellett 2011). Hence, it was possible that the conventional TRPV6 channel was the main channel for calcium entry during the mucosal amino-acid exposure (Charoenphandhu and Krishnamra 2007; Kellett 2011), as suggested by the ruthenium red experiment. Unlike the effect of apical glucose, the enhanced PD and Isc, and presumably the resultant solvent drag were not associated with the amino-acid-enhanced calcium transport. Six amino acids, i.e., methionine, glycine, alanine, isoleucine, proline, and leucine, significantly increased PD and Isc, but methionine and glycine did not enhance calcium transport. On the other hand, lysine and hydroxyproline were found to upregulate calcium transport without increasing PD and Isc. Although the paracellular permeability might be increased as indicated by lower TER in the presence of amino acids in apical solution as compared to that in glucose-free (control) condition, increases in the calcium fluxes in the methionine, valine, and glycine groups did not reach a statistically significant level. Meanwhile, since the duodenal epithelia exposed to various amino acids had similar TER, the mucosal amino acids that could enhance calcium transport (e.g., proline and leucine) did not widen the tight junction to increase paracellular calcium movement. Therefore, a lower TER in amino-acid-exposed epithelium did not make the solvent drag-induced calcium transport more favorable. Regarding the absence of effect of some branched-chain amino acids (e.g., valine), although leucine, valine, and isoleucine can use the same apical amino-acid transporter, their affinities to the transporter and transport rates may be different, leading to different potencies to enhance calcium transport. Future experiments are required to elucidate the association between the kinetics of amino-acid uptake and calcium transport across the small intestine.
Interestingly, the NHE3 inhibitor, tenapanor, did not affect the glucose-enhanced calcium transport. However, it completely inhibited the aliphatic amino-acid-induced calcium transport, which strongly indicated the involvement of NHE3 in the amino-acid case. According to the aforementioned hypothesis, the amino-acid-enhanced calcium transport was likely to occur through the NHE3-dependent transcellular pathway rather than the solvent drag-induced paracellular pathway. The exact molecular mechanism, by which tenapanor compromised transcellular calcium transport, remains elusive. The finding that apical tenapanor exposure but not basolateral exposure was able to inhibit proline- and leucine-enhanced calcium transport as well as reduce PD and Isc was consistent with the abundant NHE3 localization in the apical membrane (Hoogerwerf et al. 1996). Since tenapanor did not reduce the transepithelial transport of amino acids proline and leucine themselves, its actions could not possibly be mediated through the inhibition of the amino-acid transporters. In other words, inhibition of the apical NHE3 must have disrupted at least one of the three steps of the transcellular calcium transport pathway. Indeed, sodium influx via NHE3 was crucial for an elimination of excessive cytoplasmic H+ (Thwaites et al. 1999). We previously reported that acute exposure to acidic pH markedly reduced the duodenal calcium transport (Charoenphandhu et al. 2006). Several proteins in the calcium transport machinery, particularly calbindin-D9k, could lose their functions in relatively acidic pH due to protonation of calcium-binding sites (Kesvatera et al. 2001). Therefore, the inhibition of NHE3 and cytoplasmic acidification might partly contribute to a reduction in transcellular calcium absorption. However, since NHE3 has also been reported to modulate other ion transporters or channels through direct protein–protein interaction (Li and Naren 2010), further investigation is required to confirm the possibility that NHE3 directly interacts with and stimulates the apical calcium channels.
To provide evidence that NHE3 really contributed to calcium transport, we employed another transporting system, i.e., PEPT1-mediated peptide transport system, which is known to be coupled with NHE3 (Freeman et al. 1995). This system requires a cluster of H+-coupled PEPT1 and NHE3 arranged in the same vicinity within a microdomain or lipid raft (Watanabe et al. 2005). Under normal conditions (in situ small intestine), NHE3 provides H+ gradient for this peptide symporter (Thwaites et al. 2002). Conversely, high mucosal tri- or dipeptide concentrations and the resultant high PEPT1 activity can induce cytoplasmic acidification, which eventually leads to a stimulation of NHE3 (Watanabe et al. 2005). Hence, the presence of mucosal l-leucine tripeptide would stimulate NHE3—a condition opposite to tenapanor treatment—which, in turn, enhanced calcium transport. Although additional experiments are required to determine the underlying molecular mechanisms of amino-acid- and tripeptide-induced calcium transport, NHE3 is potentially one of the key players in the process.
In conclusion, we demonstrated the amino-acid-enhanced calcium transport across the duodenal epithelia. Although most of studied amino acids were in the aliphatic group, some members in this group were unable to significantly stimulate calcium transport. The efficiency of alanine, leucine, isoleucine, proline, hydroxyproline, and lysine could not be explained simply by changes in PD, Isc or TER. Since the NHE3 inhibitor, tenapanor, completely abolished proline- and leucine-induced calcium transport, the amino-acid-induced calcium transport could be considered as the NHE3-dependent mechanism. Meanwhile, tenapanor was also expected to markedly diminish sodium entry through NHE3, thereby reducing the basolateral sodium efflux through the electrogenic Na+/K+-ATPase; therefore, both calcium transport and PD were eventually reduced by tenapanor. However, amino acids were hypothesized to directly stimulate the transcellular calcium transport independent of PD. Our finding thus corroborates that luminal nutrients, i.e., glucose and certain amino acids, are potent stimulators of calcium absorption, and the apical NHE3 plays an important role in the regulation of the duodenal calcium transport. Limitations of the study included the use of ex vivo intestinal tissues instead of performing experiments in the in situ small intestinal segments of living animals, and the absence of data from male rats. Nevertheless, since both calcium imbalance and osteoporosis often occur in postmenopausal women, the present data have provided foundation for development of calcium supplement product for osteoporotic women.
References
Bröer S (2008) Amino acid transport across mammalian intestinal and renal epithelia. Physiol Rev 88:249–286. doi:10.1152/physrev.00018.2006
Charoenphandhu N, Krishnamra N (2007) Prolactin is an important regulator of intestinal calcium transport. Can J Physiol Pharmacol 85:569–581. doi:10.1139/y07-041
Charoenphandhu N, Tudpor K, Pulsook N, Krishnamra N (2006) Chronic metabolic acidosis stimulated transcellular and solvent drag-induced calcium transport in the duodenum of female rats. Am J Physiol Gastrointest Liver Physiol 291:G446–G455. doi:10.1152/ajpgi.00108.2006
Charoenphandhu N, Nakkrasae LI, Kraidith K, Teerapornpuntakit J, Thongchote K, Thongon N, Krishnamra N (2009) Two-step stimulation of intestinal Ca2+ absorption during lactation by long-term prolactin exposure and suckling-induced prolactin surge. Am J Physiol Endocrinol Metab 297:E609–E619. doi:10.1152/ajpendo.00347.2009
Chen MM, Singh A, Xiao F, Dringenberg U, Wang J, Engelhardt R, Yeruva S, Rubio-Aliaga I, Nässl AM, Kottra G, Daniel H, Seidler U (2010) Gene ablation for PEPT1 in mice abolishes the effects of dipeptides on small intestinal fluid absorption, short-circuit current, and intracellular pH. Am J Physiol Gastrointest Liver Physiol 299:G265–G274. doi:10.1152/ajpgi.00055.2010
Comar CL, Nold MM, Wasserman RH (1956) The influence of amino acids and other organic compounds on the gastrointestinal absorption of calcium45 and strontium89 in the rat. J Nutr 59:371–383
Donowitz M, Ming Tse C, Fuster D (2013) SLC9/NHE gene family, a plasma membrane and organellar family of Na+/H+ exchangers. Mol Aspects Med 34:236–251. doi:10.1016/j.mam.2012.05.001
Dorkkam N, Wongdee K, Suntornsaratoon P, Krishnamra N, Charoenphandhu N (2013) Prolactin stimulates the L-type calcium channel-mediated transepithelial calcium transport in the duodenum of male rats. Biochem Biophys Res Commun 430:711–716. doi:10.1016/j.bbrc.2012.11.085
Freeman TC, Bentsen BS, Thwaites DT, Simmons NL (1995) H+/di-tripeptide transporter (PepT1) expression in the rabbit intestine. Pflugers Arch 430:394–400
Gerritsen KG, Boer WH, Joles JA (2015) The importance of intake: a gut feeling. Ann Transl Med 3:49. doi:10.3978/j.issn.2305-5839.2015.03.21
Hoenderop JG, Nilius B, Bindels RJ (2005) Calcium absorption across epithelia. Physiol Rev 85:373–422. doi:10.1152/physrev.00003.2004
Hoogerwerf WA, Tsao SC, Devuyst O, Levine SA, Yun CH, Yip JW, Cohen ME, Wilson PD, Lazenby AJ, Tse CM, Donowitz M (1996) NHE2 and NHE3 are human and rabbit intestinal brush-border proteins. Am J Physiol 270:G29–G41
Kellett GL (2011) Alternative perspective on intestinal calcium absorption: proposed complementary actions of Cav1.3 and TRPV6. Nutr Rev 69:347–370. doi:10.1111/j.1753-4887.2011.00395.x
Kesvatera T, Jonsson B, Telling A, Tougu V, Vija H, Thulin E, Linse S (2001) Calbindin D9k: a protein optimized for calcium binding at neutral pH. Biochemistry 40:15334–15340. doi:10.1021/bi0114022
Kulaksiz H, Bektas H, Cetin Y (2001) Expression and cell-specific and membrane-specific localization of NHE-3 in the human and guinea pig upper gastrointestinal tract. Cell Tissue Res 303:337–343. doi:10.1007/s004410000329
Larsen EH, Nedergaard S, Ussing HH (2000) Role of lateral intercellular space and sodium recirculation for isotonic transport in leaky epithelia. Rev Physiol Biochem Pharmacol 141:153–212
Larsen EH, Sorensen JB, Sorensen JN (2002) Analysis of the sodium recirculation theory of solute-coupled water transport in small intestine. J Physiol 542:33–50. doi:10.1113/jphysiol.2001.013248
Li C, Naren AP (2010) CFTR chloride channel in the apical compartments: spatiotemporal coupling to its interacting partners. Integr Biol (Camb) 2:161–177. doi:10.1039/b924455g
Lu Z, Yao L, Jiang Z, Aschenbach JR, Martens H, Shen Z (2016) Acidic pH and short-chain fatty acids activate Na+ transport but differentially modulate expression of Na+/H+ exchanger isoforms 1, 2, and 3 in omasal epithelium. J Dairy Sci 99:733–745. doi:10.3168/jds.2015-9605
Mackenzie B, Erickson JD (2004) Sodium-coupled neutral amino acid (System N/A) transporters of the SLC38 gene family. Pflugers Arch 447:784–795. doi:10.1007/s00424-003-1117-9
Mineo H, Hara H, Shigematsu N, Okuhara Y, Tomita F (2002) Melibiose, difructose anhydride III and difructose anhydride IV enhance net calcium absorption in rat small and large intestinal epithelium by increasing the passage of tight junctions in vitro. J Nutr 132:3394–3399
Morgan EL, Mace OJ, Affleck J, Kellett GL (2007) Apical GLUT2 and Cav1.3: regulation of rat intestinal glucose and calcium absorption. J Physiol 580:593–604. doi:10.1113/jphysiol.2006.124768
Nakkrasae LI, Thongon N, Thongbunchoo J, Krishnamra N, Charoenphandhu N (2010) Transepithelial calcium transport in prolactin-exposed intestine-like Caco-2 monolayer after combinatorial knockdown of TRPV5, TRPV6 and Cav1.3. J Physiol Sci 60:9–17. doi:10.1007/s12576-009-0068-0
Pan W, Borovac J, Spicer Z, Hoenderop JG, Bindels RJ, Shull GE, Doschak MR, Cordat E, Alexander RT (2012) The epithelial sodium/proton exchanger, NHE3, is necessary for renal and intestinal calcium (re)absorption. Am J Physiol Renal Physiol 302:F943–F956. doi:10.1152/ajprenal.00504.2010
Pramod AB, Foster J, Carvelli L, Henry LK (2013) SLC6 transporters: structure, function, regulation, disease association and therapeutics. Mol Aspects Med 34:197–219. doi:10.1016/j.mam.2012.07.002
Reyes-Fernandez PC, Fleet JC (2015) Luminal glucose does not enhance active intestinal calcium absorption in mice: evidence against a role for Cav1.3 as a mediator of calcium uptake during absorption. Nutr Res 35:1009–1015. doi:10.1016/j.nutres.2015.08.004
Rievaj J, Pan W, Cordat E, Alexander RT (2013) The Na+/H+ exchanger isoform 3 is required for active paracellular and transcellular Ca2+ transport across murine cecum. Am J Physiol Gastrointest Liver Physiol 305:G303–G313. doi:10.1152/ajpgi.00490.2012
Romeo E, Dave MH, Bacic D, Ristic Z, Camargo SM, Loffing J, Wagner CA, Verrey F (2006) Luminal kidney and intestine SLC6 amino acid transporters of B0AT-cluster and their tissue distribution in Mus musculus. Am J Physiol Renal Physiol 290:F376–F383. doi:10.1152/ajprenal.00286.2005
Tanrattana C, Charoenphandhu N, Limlomwongse L, Krishnamra N (2004) Prolactin directly stimulated the solvent drag-induced calcium transport in the duodenum of female rats. Biochim Biophys Acta 1665:81–91. doi:10.1016/j.bbamem.2004.06.017
Thwaites DT, Ford D, Glanville M, Simmons NL (1999) H+/solute-induced intracellular acidification leads to selective activation of apical Na+/H+ exchange in human intestinal epithelial cells. J Clin Invest 104:629–635. doi:10.1172/JCI7192
Thwaites DT, Kennedy DJ, Raldua D, Anderson CM, Mendoza ME, Bladen CL, Simmons NL (2002) H+/dipeptide absorption across the human intestinal epithelium is controlled indirectly via a functional Na+/H+ exchanger. Gastroenterology 122:1322–1333. doi:10.1053/gast.2002.32992
Turner JR, Rill BK, Carlson SL, Carnes D, Kerner R, Mrsny RJ, Madara JL (1997) Physiological regulation of epithelial tight junctions is associated with myosin light-chain phosphorylation. Am J Physiol 273:C1378–C1385
Watanabe C, Kato Y, Ito S, Kubo Y, Sai Y, Tsuji A (2005) Na+/H+ exchanger 3 affects transport property of H+/oligopeptide transporter 1. Drug Metab Pharmacokinet 20:443–451. doi:10.2133/dmpk.20.443
Wongdee K, Charoenphandhu N (2015) Vitamin D-enhanced duodenal calcium transport. Vitam Horm 98:407–440. doi:10.1016/bs.vh.2014.12.010
Acknowledgements
This work was supported by grants from the Thailand Research Fund (TRF)–Mahidol University through Distinguished Research Professor Grant (DPG5780004 to N. Krishnamra), the TRF–Mahidol University through the TRF Senior Research Scholar Grant (RTA5780001 to N. Charoenphandhu), the Faculty of Science, Mahidol University (to N. Charoenphandhu and J. Thongbunchoo), the Faculty of Allied Health Sciences, Burapha University and TRF through TRF Research Scholar Award (RSA5780041 to K. Wongdee), the Higher Education Research Promotion and National Research University Project of Thailand, Office of the Higher Education Commission (185100 to K. Wongdee), and TRF International Research Network Program (IRN59W0002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Handling Editor: E. I. Closs.
Rights and permissions
About this article
Cite this article
Thammayon, N., Wongdee, K., Lertsuwan, K. et al. Na+/H+ exchanger 3 inhibitor diminishes the amino-acid-enhanced transepithelial calcium transport across the rat duodenum. Amino Acids 49, 725–734 (2017). https://doi.org/10.1007/s00726-016-2374-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-016-2374-1