Abstract
This study was conducted to determine the immunostimulatory effect of l-proline on inactivated vaccine immunized mice. Ninety-five female KM mice were randomly divided into five groups: (1) mice received dietary supplementation with 0.4 % l-proline and immunized with inactivated vaccine (V–P group); (2) mice received dietary supplementation with 0.3 % l-alanine (isonitrogenous control) and immunized with inactivated vaccine (V–A group, negative control); (3) mice were immunized with inactivated vaccine with oil adjuvant (V–O group, positive control); (4) mice were immunized with inactivated vaccine with aluminum hydroxide adjuvant (V–H group, positive control); (5) mice immunized with phosphate-buffered saline (control group). All mice were dead in the control group between 36 and 48 h post infection. Mice in the V–P group showed 100 % protection after challenge with P. multocida serotype A (CQ2) at dose of 4.4 × 105 CFU (2LD50). Meanwhile, serum antibody titers in the V–P group were higher than those in the V–A group before infection and those in the V–A and V–O groups at 36 h post infection. Moreover, serum IL-1β levels in the V–P group were lower than those in V–O group. Furthermore, serum GSH-PX levels in the V–P group were higher than those in the V–A and V–O groups. Collectively, dietary proline supplementation confers beneficial immunostimulatory effects in inactivated P. multocida vaccine immunized mice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Any substance that helps the vaccine to stimulate the immune system’s response to a target antigen, but does not in itself confer immunity, is referred to as an immunologic adjuvant. Formulation of an effective vaccine, especially for purified, synthetic and subunit vaccine, generally requires an appropriate adjuvant which should be stable, biodegradable, cheap to produce, immunologically inert, have a long shelf life, and promote both the humoral and cellular immune response to an antigen (Sun et al. 2008). However, minimizing toxicity remains as one of the major challenges in adjuvant research and use. For example, aluminium hydroxide, which is common adjuvant sold in the word and has been used in vaccine for over 70 years, is poor adjuvant in inducing cellular immune response and exerts many side effects, such as IgE production, allergenicity and neurotoxicity (Clapp et al. 2011; Goto et al. 1993). Thus, there is a need for identification of new adjuvants that are both safe and efficacious for the use to produce a variety of vaccines.
Traditionally, proline is categorized as a nutritionally nonessential amino acids in mammals (Wu 2009). However, similar to arginine, glutamine and alpha-ketoglutarate, growing evidence shows that proline is a nutritionally essential amino acid for poultry, young mammals, fish and wounded subjects (Li et al. 2007, 2009; Liu et al. 2012; Tan et al. 2009, 2011; Ren et al. 2013; Wu et al. 2011; Yao et al. 2008, 2012). Proline is also a key regulator of multiple biochemical and physiological processes in cells. For example, proline is a signaling molecule, a sensor of cellular energy status, and a source of pyrroline-5-carboxylate and superoxide anion (a free radical) participating in redox reactions in humans and animals (Phang et al. 2008, 2010). In addition, proline plays an important role in conceptus metabolism, growth and development, as well as a potential treatment for intrauterine growth restriction, which is a significant problem in both human medicine and animal production (Rezaei et al. 2011; Wu et al. 2008). Furthermore, proline plays a role in protecting lymphocytes from apoptosis, stimulating cell growth and promoting antibody production (Duval et al. 1991) and a proline-rich polypeptide complex may affect not only adaptive immunity, but also innate immunity, thereby regulating secretions of inflammatory mediators of Zablocka et al. (2010). Collectively, proline exerts an important role in physiological and immunological function. However, not much is known about immunostimulatory effects of proline on vaccine-immunized mammals. Thus, this study was conducted to evaluate immunostimulatory effects of dietary l-proline supplementation in mice immunized with the inactivated Pasteurella multocida vaccine.
Materials and methods
Preparation of the bacterium and inactivated vaccine
The P. multocida serotype A (CQ2) strain used in the present study was isolated from the lung tissue of clinically infected cattle, which was dead with pneumonia. The bacterium was regarded as P. multocida serotype with 16 s rRNA sequence and multiplex capsular PCR typing system (Townsend et al. 2001). The bacteria isolate was cultured in the Martin’s broth agar medium containing 5 % horse serum. The pathogenicity of the isolate was tested in healthy mice by intraperitoneal inoculation the 2 days culture, and all the mice were succumbed to infection between 36 and 48 h post-infection. Bacteria re-isolated from these infected mice were used to prepare the inactivated vaccine. The bacterial inoculum was identified to be P. multocida serotype A using the PCR method and biochemical characteristics. Before animal experiments, the P. multocida serotype A-inactivated whole cell vaccine was prepared. In brief, bacterial colonies isolated from the liver of mice were cultured in the Martin’s agar at 37 °C overnight, and then an isolate colony from the overnight static culture was transferred to 100 ml of the Martin’s broth to incubate at 37 °C for 12 h with 85 rpm. Then, P. multocida serotype A strains were inactivated by addition of 0.4 % formalin to the culture mix (109 CFU/ml) and incubation at 37 °C for 24 h with continuous agitation.
Experimental design
Ninety-five female KM mice (body weight 18–22 g) were obtained from Laboratory Animal Center of Third Military Medical University, Chongqing, China. The mice were housed in a pathogen-free mouse colony (temperature, 20–30 °C; relative humidity, 45–60 %; lighting cycle, 12 h/day) and had free access to food and drinking water. Animals were randomly divided into five groups (n = 19 per group): (1) mice received dietary supplementation with 0.4 % l-proline (Ajinomoto Inc., Tokyo, Japan) (basal diet + 0.4 % l-proline) from day 0 and immunized with inactivated vaccines at dose of 109 CFU at day 15 and 20 (V–P group); (2) mice received dietary supplementation with 0.3 % l-alanine (isonitrogenous control) from day 0 and immunized with inactivated vaccines at dose of 109 CFU at days 15 and 20 (V–A group, negative control); (3) mice were immunized with inactivated vaccines with oil adjuvant at dose of 109 CFU at days 15 and 20 (V–O group, positive control); (4) mice were immunized with inactivated vaccines with aluminum hydroxide adjuvant at dose of 109 CFU at days 15 and 20 (V–H group, positive control); (5) mice immunized with the same volume of phosphate-buffered saline at days 15 and 20 (control group). As previous study, the amino acid content in the basal diet was measured using Automatic Amino Acid Analyzer (Yin et al. 2011). This basal diet contained 17 % protein. At day 34, all of the mice were challenged by an intraperitioneal injection of P. multocida serotype A (CQ2) at the dose of 4.4 × 105 CFU (2LD50). Ten mice in each group were used to calculate survival rate, whereas the others were euthanized to collect serum at 36 h post-infection for determination of cytokine levels, glutathione peroxidase (GSH-PX) activity, and antibody titers. Serum antibody titers in all groups were also analyzed at day 34 before challenge. This study was performed according to the guidelines of the Laboratory Animal Ethical Commission of the Chinese Academy of Sciences.
Analysis of serum cytokine levels
Interleukin-1β (IL-1β), interleukin-6 (IL-6) and interleukin-8 (IL-8) in serum were measured using ELISA kits in accordance with the manufacturer’s instructions (CUSABIO BIOTECH CO., Ltd., China). An aliquot (100 μl) of the sample or standard was added to duplicate wells of the microtiter plate, which had been pre-coated with antibody (Yin et al. 2008). The buffer was used as a negative control. The plate was incubated for 2 h at 37 °C. A 100 μl of biotin antibody was added to each well after the removal of the liquid and incubated for 1 h at 37 °C. The wells were washed three times with 200 μl of the washing buffer. A 100 μl quantity of HRP–avidin was then added to each well for 1 h at 37 °C. After the final wash, an aliquot (90 μl) of the TMB substrate was added and incubated for 30 min in the dark at 37 °C. The reaction was stopped with 50 μl of the terminating solution and absorbance measured at 450 nm.
Serum GSH-PX activity
Serum GSH-PX activity was measured using spectrophotometric kits in accordance with the manufacturer’s instructions (Nanjing Jiancheng Biotechnology Institute, China).
Analysis of antibodies
Enzyme-linked immunosorbent assays were used for the detection of antibodies. Before detection, antigens were prepared. In brief, 10 ml bacteria culture was crushed by ultrasonic wave, and then diluted with bicarbonate buffer (pH 7.4). A 96-well plate was coated by antigens (100 μl of the prepared antigens) and incubated at 4 °C for 16 h for adsorption. Primary antisera were diluted 1:40 in PBS containing 0.5 % Tween-20 and 1 % bovine serum albumin (BSA). Following five washings, horseradish peroxidase-conjugated goat anti-mouse IgG (Jackson ImmunoResearch, USA) diluted 1:10000 in PBS-T containing 1 % BSA was added to each well and the plates were incubated at 37 °C for 3 h. After the final wash, an aliquot (100 μl) of the TMB substrate was added and incubated for 15 min in the dark at 37 °C. The reaction was stopped with 50 μl of the terminating solution and absorbance measured at 450 nm.
Statistical analysis
All statistical analyses were performed using SPSS 17.0 software. Data are expressed as mean values ± standard error of the mean. Multiple comparisons were performed using the Student–Newman–Keuls method (Ren et al. 2013). Differences were considered significant at P < 0.05 (Wei et al. 2012).
Results
Cytokine profile and GSH-PX activity
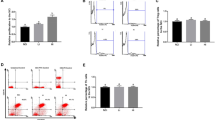
Mouse serum IL-1 β, IL-6 and IL-8 levels were measured at 36 h after infection. As indicated in Table 1, immunization with inactivated vaccines with oil adjuvant increased (P < 0.01) serum IL-1β levels, in comparison to dietary proline or alanine supplementation or the control group. Serum IL-1β levels in the V–H group were (P < 0.01) than those in the control group; while no difference was found among the other groups. Unlike IL-1β, serum IL-6 and IL-8 levels in the control group were higher (P < 0.01) than those in other groups. Serum IL-8 levels in the V–A group were higher (P < 0.05) than those in the V–P, V–O and V–H groups; no difference was detected among these groups (Table 1). Furthermore, serum GSH-PX levels in the V–P, V–H and control groups were higher (P < 0.01) than those in the V–A and V–O groups; while no difference was found among other groups (Fig. 1).
Serum GSH-PX levels in different groups after challenge with P. multocida serotype A (CQ2) at dose of 4.4 × 105 CFU (U/ml). Five groups were: (1) mice received dietary supplementation with 0.4 % l-proline and immunized with inactivated vaccine (V–P group); (2) mice received dietary supplementation with 0.3 % l-alanine (isonitrogenous control) and immunized with inactivated vaccine (V–A group, negative control); (3) mice were immunized with inactivated vaccine with oil adjuvant (V–O group, positive control); (4) mice were immunized with inactivated vaccine with aluminum hydroxide adjuvant (V–H group, positive control); (5) mice immunized with phosphate-buffered saline (control group). GSH-PX: glutathione peroxidase. Data are mean ± SEM, n = 6
Serum antibody titers
Serum antibody titers against P. multocida serotype A were measured before and after challenged with P. multocida serotype A. As indicated in Table 2, serum antibody titers in the V–P, V–A,V–O and V–H groups were much higher (P < 0.01) than those in the control group. Immunization with inactivated vaccines and dietary proline supplementation increased (P < 0.05) serum antibody titers, when compared with the immunization with inactivated vaccines and dietary alanine supplementation before infection; furthermore, serum antibody titers in the V–P and V–H groups were higher (P < 0.05) than those in the V–A and V–O groups at 36 h post infection; while no difference was detected among the other groups.
Survival rates
The survival rates of immunized mice were calculated every day after infection with P. multocida serotype A. All mice were dead in the control group between 36 and 48 h post infection, whereas two mice died in each of the V–A and V–O adjuvant groups and no death was observed in the V–P or V–H group (Fig. 2).
The mortality in each group after challenge with P. multocida serotype A (CQ2) at dose of 4.4 × 105 CFU. Mice were immunized with inactivated vaccines and dietary 0.4 % proline supplementation as V–P group; mice were immunized with inactivated vaccines and dietary 0.3 % alanine supplementation as V–A group; mice were immunized with inactivated vaccines and oil adjuvant as V–O group; mice were immunized with inactivated vaccines and aluminum hydroxide adjuvant as V–H group; mice immunized with PBS as control group (n = 10/group)
Discussion
There is growing interest in the dietary requirements of traditionally classified “nutritionally nonessential amino acids” by animals (Hou et al. 2012; Li et al. 2011; Tan et al. 2012a, b; Wu 2010a, b, 2013). This study was conducted to evaluate the immunostimulatory effects of dietary l-proline supplementation on inactivated P. multocida vaccine-immunized mice. Based on the challenge experiment, no death was observed in the V–P and V–H groups, and only 10.5 % of mice died in the V–A or the V–O group, in striking contract to 100 % mortality in the control group. These compelling results indicate that proline, similar to the Al (OH)3 adjuvant, exerts a significant immunostimulatory effect on inactivated P. multocida vaccine-immunized mice. To illustrate this promising result, we analyzed serum antibody titers before and post the challenge to study the stimulatory effects of proline in the systemic humoral immune response. A significant increase was observed in all the mice immunized with the inactivated P. multocida vaccine, in comparison with the mice immunized with PBS. Notably, the most promising results are that antibody titers in the V–P group were much higher than those in the V–A group on day 34 and that antibody titers in the V–P group were also higher than those in the V–O and V–A groups at 36 h post challenge. Similarly, serum antibody titers in the V–H group were higher than those in the V–O and V–A groups at 36 h post challenge. These results indicate that dietary supplementation with 0.4 % proline increases serum antibody titers in the inactivated P. multocida vaccine-immunized mice. The findings also help explain the observation that the mortality rate in the control group was 100 %, while no mortality was observed in the V–P or V–H group. At present, the molecular mechanisms responsible for the effects of proline on enhancing antibody production are unknown.
The production of cytokines is closely related to immune responses and mortality in animals (Praveena et al. 2010; Ren et al. 2012). A novel and important finding of this study is that dietary supplementation with proline affected concentrations of inflammatory cytokines in serum. Particularly, IL-1β is one of the pivotal early response pro-inflammatory cytokines that, through up- or down regulation of other cytokine, enables organisms to respond to infectious non-self challenges and induces a cascade of effects leading to inflammation (Dinarello 1997). IL-6 plays a very complex role in biological events, including immune responses, hematopoiesis, and regulation of the endocrine and nervous systems (Biffl et al. 1996; Naugler and Karin 2008). IL-8 is often associated with inflammation by acting preferentially on neutrophils (Baggiolini et al. 1995). Similarly, we found that serum IL-6 and IL-8 levels in the control group were much higher than those in other groups and that serum IL-8 levels in the V–A group is higher than those in the V–P, V–O and V–H groups. These compelling results are in consistent with the previous studies that indicated that serum concentrations of pro-inflammatory cytokines increased in mice after a challenge with P. multocida serotype A, while high antibody titers in other groups protected mice from high levels of pro-inflammatory cytokines (Praveena et al. 2010; Ren et al. 2012). Moreover, serum IL-1β levels in the V–P and V–H groups were neither the lowest nor the highest. This intermediate levels of IL-1β may contribute to the high protection against mortality in the V–P and V–H groups because an optimal concentration of IL-1β functions as a mediator of the host inflammatory response to infection and other inflammatory stimuli, whereas larger quantities of IL-1β induce many negative effects, including inducing synthesis of acute-phase plasma proteins, initiating metabolic wasting (Dinarello 1997). Although serum IL-1β levels in the V–A group was similar to those in the V–P group, only 10.5 % of mice in the V–A group died, indicating that factors, including low antibody titers, may also affect the protection.
As an indicator of anti-oxidative ability, serum GSH-PX levels in each group were determined to explain the clinical results. GSH-PX is an important peroxidase widely spread in the body (Wang et al. 2009). It can remove harmful peroxide metabolites from cells and block the lipid peroxidation chain reaction, thereby protecting the integrity of cell membrane structure and function (Wu et al. 2004). Consistent with the clinical results, serum GSH-PX levels in the V–P and V–H groups were substantially higher than those in the V–A and V–O groups. As alluded to above concerning IL-1β, a mere increase in serum GSH-PX levels in the control group could not ensure high protection. A major reason for high GSH-PX levels in the proline group may be that proline can scavenge free radicals, and thus has vital antioxidant properties (Kaul et al. 2008). In fact, there are reports that proline concentration increases markedly in response to cellular oxidative stress (Verbruggen and Hermans 2008).
Collectively, our results clearly demonstrate that dietary proline supplementation provides an immunostimulatory effect in inactivated P. multocida vaccine-immunized mice. The underlying mechanism may involve a crucial role for proline in modulating multiple biochemical, physiological and immunological processes in humans and animals by acting as a signaling molecule, a sensor of cellular energy status, and a source of pyrroline-5-carboxylate and superoxide anion (Phang and Liu 2012; Wang et al. 2012; Wu et al. 2011). In addition to proline itself, other substances produced from proline metabolism, such as arginine, glutamine and H2O2, may also play a vital role in immune function (Dai et al. 2012a, b; Gao et al. 2012; Hou et al. 2011; Li et al. 2007; Xi et al. 2012). Arginine, de novo synthesis from proline, is essential substance for T-lymphocyte responses for low l-arginine levels down-regulated the CD3 ξ chain (Bronte et al. 2003; Rodriguez et al. 2003; Munder et al. 2006; Peranzoni et al. 2007). Glutamine is another metabolite that is crucial for optimal nutrition and immune response in animals (Li et al. 2007), including mice (Ren et al. 2011). Moreover, H2O2, a major product of proline oxidation, is a signaling molecule and a cytotoxic agent toward pathogenic bacteria (Rezaei et al. 2013a, b; Shi et al. 2004). Further studies are warranted to fully understand the molecular mechanisms for the adjuvant effect of proline in mammals.
In conclusion, dietary proline supplementation, like the traditionally used aluminum hydroxide adjuvant, increases serum antibody titers, ameliorates the serum cytokines profile, and elevates serum GSH-PX levels in inactivated P. multocida vaccine-immunized mice. These beneficial results eventually contribute to the full protection after a challenge with P. multocida serotype A. To our knowledge, this is the first study to determine the adjuvant effect of proline in mammals. Our findings have important implications for preventive medicine in humans and animals.
Abbreviations
- BSA:
-
Bovine serum albumin
- GSH-PX:
-
Glutathione peroxidase
- IL:
-
Interleukin
- PBS:
-
Phosphate-buffered saline
- TNF-α:
-
Tumor necrosis factor alpha
References
Baggiolini M, Loetscher P, Moser B (1995) Interleukin-8 and the chemokine family. Int J Immunopharmacol 17:103–108
Biffl WL, Moore EE, Moore FA et al (1996) Interleukin-6 in the injured patient. Marker of injury or mediator of inflammation? Ann Surg 224:647–664
Bronte V, Serafini P, Mazzoni A et al (2003) l-arginine metabolism in myeloid cells controls T-lymphocyte functions. Trends Immunol 24:302–306
Clapp T, Siebert P, Chen D et al (2011) Vaccines with aluminum-containing adjuvants: optimizing vaccine efficacy and thermal stability. J Pharm Sci 100:388–401
Dai ZL, Li XL, Xi PB et al (2012a) Regulatory role for l-arginine in the utilization of amino acids by pig small-intestinal bacteria. Amino Acids 43:233–244
Dai ZL, Li XL, Xi PB et al (2012b) Metabolism of select amino acids in bacteria from the pig small intestine. Amino Acids 42:1597–1608
Dinarello CA (1997) Interleukin-1. Cytokine Growth Factor Rev 8:253–265
Duval D, Demangel C, Munier-Jolain K et al (1991) Factors controlling cell proliferation and antibody production in mouse hybridoma cells: I. Influence of the amino acid supply. Biotechnol Bioeng 38:561–570
Gao KG, Jiang ZY, Lin YC et al (2012) Dietary l-arginine supplementation enhances placental growth and reproductive performance in sows. Amino Acids 42:2207–2214
Goto N, Kato H, Maeyama J et al (1993) Studies on the toxicities of aluminium hydroxide and calcium phosphate as immunological adjuvants for vaccines. Vaccine 11:914–918
Hou YQ, Wang L, Ding BY et al (2011) Alpha-ketoglutarate and intestinal function. Front Biosci 16:1186–1196
Hou Y, Wang QL, Zhang W et al (2012) Protective effects of N-acetylcysteine on intestinal functions of piglets challenged with lipopolysaccharide. Amino Acids 43:1233–1242
Kaul S, Sharma SS, Mehta IK (2008) Free radical scavenging potential of l-proline: evidence from in vitro assays. Amino Acids 34:315–320
Li P, Yin YL, Li D et al (2007) Amino acids and immune function. Br J Nutr 98:237–252
Li P, Mai KS, Trushenski J et al (2009) New developments in fish amino acid nutrition: towards functional and environmentally oriented aquafeeds. Amino Acids 37:43–53
Li XL, Rezaei R, Li P et al (2011) Composition of amino acids in feed ingredients for animal diets. Amino Acids 40:1159–1168
Liu XD, Wu X, Yin YL et al (2012) Effects of dietary l-arginineor N-carbamylglutamate supplementation during late gestation of sows on the miR-15b/16, miR-221/222, VEGFA and eNOS expression in umbilical vein. Amino Acids 42:2111–2119
Munder M, Schneider H, Luckner C et al (2006) Suppression of T-cell functions by human granulocyte arginase. Blood 108:1627–1634
Naugler WE, Karin M (2008) The wolf in sheep’s clothing: the role of interleukin-6 in immunity, inflammation and cancer. Trends Mol Med 14:109–119
Peranzoni E, Marigo I, Dolcetti L et al (2007) Role of arginine metabolism in immunity and immunopathology. Immunobiology 212:795–812
Phang JM, Liu W (2012) Proline metabolism and cancer. Front Biosci 17:1835–1845
Phang JM, Donald SP, Pandhare J et al (2008) The metabolism of proline, a stress substrate, modulates carcinogenic pathways. Amino Acids 35:681–690
Phang JM, Liu W, Zabirnyk O (2010) Proline metabolism and microenvironmental stress. Annu Rev Nutr 30:441–463
Praveena PE, Periasamy S, Kumar AA et al (2010) Cytokine profiles, apoptosis and pathology of experimental Pasteurella multocida serotype A1 infection in mice. Res Vet Sci 89:332–339
Ren W, Luo W, Wu M et al (2011) Dietary l-glutamine supplementation improves pregnancy outcome in mice infected with type-2 porcine circovirus. Amino Acids. doi:10.1007/s00726-011-1134-5
Ren W, Yin Y, Liu G et al (2012a) Effect of dietary arginine supplementation on reproductive performance of mice with porcine circovirus type 2 infection. Amino Acids 42:2089–2094
Ren W, Zou L, Li N et al (2012b) Dietary arginine supplementation enhances immune responses to inactivated Pasteurella multocida vaccination in mice. Br J Nutr 109:867–872
Ren W, Yu R, Liu G et al (2013a) DNA vaccine encoding the major virulence factors of Shiga toxin type 2e (Stx2e)-expressing Escherichia coli induces protection in mice. Vaccine 31:367–372
Ren W, Wu M, Wei L et al (2013b) Dietary supplementation with proline confers a positive effect in both porcine circovirus-infected pregnant and non-pregnant mice. Br J Nutr. doi:10.1017/S0007114513000652
Rezaei R, Knabe DA, Li XL et al (2011) Enhanced efficiency of milk utilization for growth in surviving low-birth-weight piglets. J Anim Sci Biotech 2:73–83
Rezaei R, Wang WW, Wu ZL et al (2013a) Biochemical and physiological bases for utilization of dietary amino acids by young pigs. J Anim Sci Biotech 4:7
Rezaei R, Knabe DA, Tekwe CD et al (2013b) Dietary supplementation with monosodium glutamate is safe and improves growth performance in postweaning pigs. Amino Acids 44:911–923
Rodriguez PC, Zea AH, DeSalvo J et al (2003) l-arginine consumption by macrophages modulates the expression of CD3 zeta chain in T lymphocytes. J Immunol 171:1232–1239
Shi W, Meininger CJ, Haynes TE et al (2004) Regulation of tetrahydrobiopterin synthesis and bioavailability in endothelial cells. Cell Biochem Biophys 41:415–434
Sun Y, Li M, Liu J (2008) Haemolytic activities and adjuvant effect of Anemone raddeana saponins (ARS) on the immune responses to ovalbumin in mice. Int Immunopharmacol 8:1095–1102
Tan BE, Yin YL, Liu ZQ et al (2009) Dietary l-arginine supplementation increases muscle gain and reduces body fat mass in growing-finishing pigs. Amino Acids 37:169–175
Tan BE, Yin YL, Liu ZQ et al (2011) Dietary l-arginine supplementation differentially regulates expression of fat-metabolic genes in porcine adipose tissue and skeletal muscle. J Nutr Biochem 22:441–445
Tan BE, Li XG, Yin YL et al (2012a) Regulatory roles for l-arginine in reducing white adipose tissue. Front Biosci 17:2237–2246
Tan BE, Li XG, Wu G et al (2012b) Dynamic changes in blood flow and oxygen consumption in the portal-drained viscera of growing pigs receiving acute administration of l-arginine. Amino Acids 43:2481–2489
Townsend KM, Boyce JD, Chung JY et al (2001) Genetic organization of Pasteurella multocida cap Loci and development of a multiplex capsular PCR typing system. J Clin Microbiol 39:924–929
Verbruggen N, Hermans C (2008) Proline accumulation in plants: a review. Amino Acids 35:753–759
Wang XQ, Ou DY, Yin JD et al (2009) Proteomic analysis reveals altered expression of proteins related to glutathione metabolism and apoptosis in the small intestine of zinc oxide-supplemented piglets. Amino Acids 37:209–218
Wang JJ, Wu ZL, Li DF et al (2012) Nutrition, epigenetics, and metabolic syndrome. Antioxid Redox Signal 17:282–301
Wei JW, Carroll RJ, Harden KK et al (2012) Comparisons of treatment means when factors do not interact in two-factorial studies. Amino Acids 42:2031–2035
Wu G (2009) Amino acids: metabolism, functions, and nutrition. Amino Acids 37:1–17
Wu G (2010a) Recent advances in swine amino acid nutrition. J Anim Sci Biotech 1:49–61
Wu G (2010b) Functional amino acids in growth, reproduction and health. Adv Nutr 1:31–37
Wu G, Fang YZ, Yang S et al (2004) Glutathione metabolism and its implications for health. J Nutr 134:489–492
Wu G, Bazer FW, Datta S et al (2008) Proline metabolism in the conceptus: implications for fetal growth and development. Amino Acids 35:691–702
Wu G, Bazer FW, Burghardt RC et al (2011) Proline and hydroxyproline metabolism: implications for animal and human nutrition. Amino Acids 40:1053–1063
Wu G, Wu ZL, Dai ZL et al (2013) Dietary requirements of “nutritionally nonessential amino acids” by animals and humans. Amino Acids 44:1107–1113
Xi PB, Jiang ZY, Dai ZL et al (2012) Regulation of protein turnover by l-glutamine in porcine intestinal epithelial cells. J Nutr Biochem 23:1012–1017
Yao K, Yin YL, Chu WY et al (2008) Dietary arginine supplementation increases mTOR signaling activity in skeletal muscle of neonatal pigs. J Nutr 138:867–872
Yao K, Yin YL, Li XL et al (2012) Alpha-ketoglutarate inhibits glutamine degradation and enhances protein synthesis in intestinal porcine epithelial cells. Amino Acids 42:2491–2500
Yin YL, Tang ZR, Sun ZH et al (2008) Effect of galacto-mannan-oligosaccharides or chitosan supplementation on cytoimmunity and humoral immunity response in early-weaned piglets. Asian Aust J Anim Sci 21:723–731
Yin FG, Zhang ZZ, Huang J et al (2010) Digestion rate of dietary starch affects systemic circulation of amino acids in weaned pigs. Br J Nutr 103:1404–1412
Zablocka A, Ogorzalek A, Macala J et al (2010) A proline-rich polypeptide complex (PRP) influences inducible nitric oxide synthase in mice at the protein level. Nitric Oxide 23:20–25
Acknowledgments
This research was jointly supported by MATS-Beef Cattle Yak system(CARS-38), National Basic Research Program of China (2012CB124704, 2013CB127301), National Natural Science Foundation of China (31110103909,30901040, 31272463, 30928018, and 31101729), the State Key Laboratory of Food Science and Technology, the Nanchang University project (SKLF-TS-201108, SKLF-KF-201005, SKLF-KF-201216), the CAS/SAFEA International Partnership Program for Creative Research Teams, Chinese Universities Scientific Funds (2012RC024), and the Thousand-People-Talent program at China Agricultural University, Texas AgriLife Research project (H-8200).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
W. Ren and L. Zou contributed equally to this study.
Rights and permissions
About this article
Cite this article
Ren, W., Zou, L., Ruan, Z. et al. Dietary l-proline supplementation confers immunostimulatory effects on inactivated Pasteurella multocida vaccine immunized mice. Amino Acids 45, 555–561 (2013). https://doi.org/10.1007/s00726-013-1490-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-013-1490-4