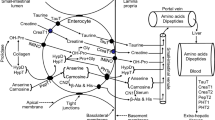
Abstract
Numerous data from both epidemiological and experimental origins indicate that some alimentary proteins and amino acids in supplements can modify the blood LDL cholesterol, HDL cholesterol and total cholesterol. After an initial approval of the health claim for soy protein consumption for the prevention of coronary heart disease, more recently it has been concluded from an overall analysis of literature that isolated soy protein with isoflavones only slightly decrease LDL and total cholesterol. Other plant extracts and also some proteins from animal origin have been reported to exert a lowering effect on blood cholesterol when compared with a reference protein (often casein). The underlying mechanisms are still little understood. Individual amino acids and mixture of amino acids have also been tested (mostly in animal studies) for their effects on cholesterol parameters and on cholesterol metabolism. Methionine, lysine, cystine, leucine, aspartate and glutamate have been tested individually and in combination in different models of either normo or hypercholesterolemic animals and found to be able to modify blood cholesterol and/or LDL cholesterol and/or HDL cholesterol. It is however not known if these results are relevant to human nutrition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the course of studies related to metabolic interactions between nutrients, there have been several studies beginning in the late 1960s, which have examined the influence of dietary proteins from various origins and also of amino acids tested individually or as mixtures on the blood concentrations of total cholesterol, low-density lipoproteins and high-density lipoproteins. These studies have been conducted both in human volunteers and in different experimental animal models in order to gain knowledge on the type of dietary nitrogenous compounds, which may contribute to lower the risk of coronary heart diseases. Briefly, elevated plasma levels of low-density lipoproteins (LDL) and triglycerides present a risk for cardiovascular disease. In contrast, high-density lipoproteins (HDL) are believed to be beneficial. A low ratio of LDL to HDL and low plasma triglyceride levels decrease the risk of cardiovascular disease (Friedman and Brandon 2001). Overall, the studies related to protein and amino acid ingestion and cholesterolemia have led mostly to descriptive results with little mechanistic data. In this review paper, we will examine firstly the effects of various alimentary proteins on cholesterol-related parameters and secondly the effects of several amino acids on such parameters.
Alimentary proteins and cholesterol
There have been several reports on the effects of dietary plant protein extracts on the cholesterolemia in both human and animal studies. Most studies were performed using soy protein extracts. This is probably because according to epidemiological investigations (Anderson et al. 1995), consumption of soy foods has been proposed as being able to contribute in lowering the risk of coronary heart disease. Animal (Lin et al. 2004; Moriyama et al. 2004) and human (Anthony et al. 1998) studies have reported that consumption of soy protein and/or isoflavones has beneficial impacts on the risk factors associated with cardiovascular disease, including the lowering of total and LDL cholesterol levels and increasing the HDL cholesterol and/or the HDL/LDL cholesterol ratio. In 1999, the food-labeling health claim for soy proteins in the prevention of coronary heart disease was approved by the US Food and Drug Administration (1999) (FDA). Similar health claims for soy protein have also been approved in various countries including the UK, Brazil, Japan, South Africa, Philippines, Malaysia, Korea, etc. (Xiao 2008). However, the studies on the effects of soy protein extracts have led to rather heterogeneous results, as will be described later in this review. The Nutrition Committee of the American Heart Association has assessed 22 randomized trials and concluded that, overall, isolated soy protein with isoflavones slightly decreased LDL cholesterol, but has no significant effect on HDL cholesterol (Sacks et al. 2006). The decrease in blood cholesterol concentration related to soy-based plant protein represents approximately between 6 and 12% of total cholesterol (Friedman and Brandon 2001). Although the individual factors that could explain the discrepancies in the reported results are not clear, it is possible that the source of soybeans and processing of the protein extracts are important parameters because of their consequences on the concentrations of bioactive peptides and other minor components in the soy extracts, which may likely play a role in the effect of soy-based protein extracts.
An early human study on the cholesterol-lowering effect was reported in 1967 (Hodges et al. 1967). This study reported that replacement of mixed proteins by isolated soy protein extracts at a dose of 100 g/day reduced cholesterol levels in hypercholesterolemic men. On the basis of a food-frequency questionaire given to 4,800 Japanese men and women, Nagata et al. (1998) noted decreasing total cholesterol plasma concentrations with increasing consumption of soy products. In another epidemiological study, it was found that consumption of soy-based diet induced a decrease in the plasma level of LDL cholesterol of both normocholesterolemic and hypercholesterolemic men (Wong et al. 1998). The extent of LDL cholesterol-lowering by soy-based diets was dependent on the concentrations of naturally occuring estrogenic isoflavones (Crouse et al. 1999). Potter et al. (1998) and Washburn et al. (1999) investigated the role of soy isoflavones on the risk of cardiovascular disease and menopausal symptoms in women consuming soy-based diets. They reported improvement in lipid and lipoprotein levels. A double-blind study suggests that even partial replacement of animal protein with soy protein was effective in lowering plasma cholesterol in 21 severely hypercholesterolemic patients (Sirtori et al. 1999). Two studies have shown that in hypercholesterolemic subjects following a diet with a poor content in fat and a relatively high content in fibers, dietary plant protein extracts obtained mainly from soya exerted no significant beneficial effect in term of hypercholesterolemia (Cuevas et al. 2003; West et al. 2005). Inversely, other studies clearly showed a significant reduction of LDL cholesterol in hypercholesterolemic subjects, who consummed a diet with low lipid content and receiving dietary plant protein extracts that originated mainly from soya (Tonstad et al. 2002; Teixera et al. 2000). The dose effect of these proteic preparations was detectable only after 3 weeks of consumption and was no more detectable after 6 weeks (Teixera et al. 2000) or after 8–16 weeks (Tonstad et al. 2002). The efficiency of the protein extracts on hypercholesterolemia was similar using 30 or 50 g proteins (Tonstad et al. 2002; Teixera et al. 2000). In their publication, Teixera et al. (2000) concluded that a 20 g dose is able to reduce the non-HDL cholesterol by 2.6% after 6 weeks of treatment.
Studies on animal models
In the work reported by Ni et al. (1998), mice were fed a purified diet containing 1 g/100 g cholesterol and cholate (0.25 g/100 g) for 6 weeks together with soy protein isolate or casein as the protein source. In this study, although serum total cholesterol was similar in both groups, the lesion area in the thoracic area of the soy protein group was lower than that of the casein group. In a study published in 2002 (Kern et al. 2002), the effects of soy protein isolate versus casein on blood lipids and adiposity were investigated in rats fed with diets supplemented with l-methionine. As a result, total blood cholesterol concentration was higher for the casein-fed rats than for the soy protein isolate-fed rats suggesting that methionine supplementation did not abolish the hypocholesterolemic effect of soy. In the paper by Horigome and Cho (1992), a diet containing 22.5–23.5 g/100 g of soybean protein was hypocholesterolemic in rats when compared with a diet containing 20.0 g casein/100 g diet. A comparison of serum amino acids in soybean protein-fed versus casein-fed rats showed that whereas concentrations of many amino acids were lower in the soybean protein-fed rats compared with the casein-fed groups, glycine was the only amino acid having a higher concentration. However, this study did not demonstrate a causal link between hypocholesterolemia and modified glycine concentration.
In the study by Potter et al. (1996), the objective was to determine if serum cholesterol concentration differed in the hamster model feeding soy protein from different sources in comparison with caseins. The authors found that serum total cholesterol concentrations were lower in animals fed on isolated soy protein when compared with animals fed on soy protein concentrate. Both of the diets containing soy proteins were able to induce a decrease in blood cholesterol concentration when compared with caseins.
Taken as a whole, it appears that the effects on cholesterolemia attributed to soy-based plant protein extract are most likely complex and may be relevant to a synergistic effect of various soy compounds (Anderson et al. 1995; Francis et al. 2002) including nonprotein compounds such as saponins, fiber, phytic acid, minerals and, last but not least, isoflavones (Potter 1995). But in the case of soy-based plant protein extracts depleted in isoflavones, several authors suggest that the relatively low content of methionine in soy protein would play a role in the hypocholesterolemic effect measured (Morita et al. 1997; Kern et al. 2002).
From a general point of view, the mechanisms underlying the effects of soy-based plant protein extracts may be a decrease in intestinal cholesterol absorption and/or biliary salts, an increase of cholesterol clearance by hepatic LDL receptors and/or changes in hepatic cholesterol metabolism and/or lastly endocrine modifications (Potter 1995; Potter et al. 1998 Belleville 2002, Adams et al. 2002; Baum et al. 1998).
In addition, the situation is further complicated by the fact that interaction between the protein fraction and other compounds of diet may modify the effects of the protein fraction on cholesterol metabolism. For instance, in the work presented by Nagaoka et al. (1999), the authors have studied in rats the effects of casein, soy protein, soy protein with bound phospholipids (SP), soy protein peptic hydrolysate (SPH) or soy protein peptic hydrolysate with bound phospholipids (SPHP) on the plasma and liver cholesterol. Serum total cholesterol was lower in rats fed with SPHP than in those fed with casein. The concentration of cholesterol in liver was lower in the SPHP-fed group compared with all other groups.
Several works strongly suggest that the beta-conglycinin fraction in soya proteins would be implicated in the atheromatous process in mice. However, and in contrast with previous results, this effect would not be explained by an action upon the LDL receptors and also not by an effect through the plama lipoproteins (Adams et al. 2002; Adams et al. 2004).
In addition, it is worth noting that other plant protein extracts than soy-based protein extract and also protein from animal origin have been reported to exert a lowering effect on blood cholesterol. This includes meat proteins, fish proteins and whey proteins (Nagaoka et al. 1991, 1992; Zhang and Beynen 1993; Anderson et al. 1999; McCarty 1999; Washburn et al. 1999; Tomatoke et al. 2000; Wang et al. 2004; Wergedahl et al. 2004; Zhan and Ho 2005; Choi et al. 2005; Mayilvaganan et al. 2004; Debry 2004). In a recent study by Shukla et al. (2006), rats were fed with diets containing fish protein or casein as control and cholesterol in the HDL fraction was determined. This parameter was decreased after fish protein consumption when compared with casein consumption. In contrast, in postmenopausal women, lean white fish compared with other animal protein products induced higher concentration of plasma cholesterol and HDL cholesterol (Jacques et al. 1995). In the study by Zhang and Beynen (1993), the effect of dietary whey protein versus casein on plasma and liver cholesterol concentrations was determined in rats. At a low dietary protein level, whey protein versus caseins did not affect plasma total cholesterol, but lowered the concentration of liver cholesterol. At the high dietary protein level, whey protein lowered plasma and liver cholesterol. This indicates that the effects of soy protein extract versus a reference protein (often casein) on cholesterol blood concentration depend on the amount of proteins in the diet. Therefore, once again, if it appears that the nature of proteins can exert an effect on the blood cholesterol level (at least modestly), the underlying mechanisms remain unclear.
Amino acids and cholesterol
Individual amino acids and mixture of amino acids have been tested for their effects on cholesterol concentrations in blood and tissues as well as on cholesterol metabolism, mostly in animal studies.
Methionine and lysine
From early studies performed using the rabbit model, it was found that high levels of casein or casein amino acids increased serum total and LDL cholesterol. However, this was not observed for soy protein and soy amino acids (Huff et al. 1977; Kurowska et al. 1989). The hypercholesterolemic potential of casein was presumed to be due to its high content in the essential amino acids, rather than nonessential amino acids (Kurowska and Carroll 1990). A working hypothesis was built presuming that alterations in plasma cholesterol concentration could be mediated by differences in the amino acid patterns of the dietary proteins. Indeed, methionine is one of the amino acids that is believed to contribute to hypercholesterolemia in animal models due to the fact that this latter amino acids is more abundant in casein than in soya protein.
Further experiments reported that lysine in combination with methionine, but not lysine or methionine used separately, were the most hypercholesterolemic (Kurowska and Carroll 1994; Giroux et al. 1999). In accordance with these results, once again relatively high levels of lysine and methionine were found in dietary casein and other animal proteins, which are found to be hypercholesterolemic in animal models (Carroll 1981). In a study, Kurowska and Carroll (1994) using the rabbit model reported that a high level of dietary lysine, leucine and methionine produced substancial hypercholesterolemia. In the same study, a combination of lysine and methionine produced a greater effect than either a mixture of lysine and leucine or a mixture of leucine and methionine. The mechanisms by which lysine and methionine modulate cholesterolemia in the rabbit model was studied with special emphasis on the liver metabolism. The results suggested that these amino acids can act directly in the liver by regulating both synthesis and catabolism of LDL protein (Kurowska and Carroll 1992, 1996). Using another animal model i.e., the gerbil model, Hidiroglou et al. (2004) reported that a combination of dietary lipids (saturated and unsaturated fatty acids) as well as methionine supplementation increased both serum total cholesterol and LDL cholesterol. In their study, Hirche et al. (2006) have examined the effect of dietary methionine on plasma and liver cholesterol concentrations using the rat model. They found that rats fed with 3.5 g/kg (or more) methionine had higher cholesterol concentrations in plasma and in liver than rats fed with lower (i.e., 2.6 g/kg) methionine. Interestingly, isolated hepatocytes incubated in media supplemented with 100 or 200 μmol/L methionine had a higher cholesterol synthesis than hepatocytes incubated in a medium supplemented with 50 μmol/L methionine. However, the LDL uptake in hepatocytes was independent of the methionine concentration in the medium. This study is a good indication that in the rat model, dietary methionine induces hypercholesterolemia, in part at least via an enhanced hepatic cholesterol synthesis. In the mouse model, Velez-Carrasco et al. (2008) reported that when comparing two solid food diets; one containing 2% added methionine and the other containing 2% added glycine, the methionine diet provoked a decrease in HDL-cholesterol level. Furthermore, these authors determined that using an HDL turnover study, the HDL-cholesterol production rate was reduced in mice fed on a methionine diet.
Leucine
A study by Zhang et al. (2007) reported that in the mouse model, a leucine supplementation in a high fat diet provided ad libitum decreases markedly in the plasma levels of total and LDL cholesterol when compared with the control experiments, i.e., mice fed on the same diet without leucine. Although the mechanisms involved in such an effect remain unclear, the study determined that the effect of leucine on plasma cholesterol coincided with a reduction in weight gain (without changes in food intake), a decrease in adiposity, and an increase in energy expenditure together with an increase in the expression of uncoupling protein 3 (UCP3). UCP3 mediates mitochondrial uncoupling of oxidative phosphorylation in thermogenic tissues, therefore likely accounting for increased energy expenditure with reduced fuel efficiency in skeletal muscle and adipose tissues.
Some attention was given to the effects of isolated amino acid supplementation on cholesterolemia by (Ohtani et al. (2001). In this latter study, an amino acid mixture containing l-leucine, l-isoleucine, l-valine, l-arginine and l-glutamine was given to 23 rugby players twice a day for 90 days (3.6 g of total amino acids per dose). After this supplementation period, an increase in total cholesterol and LDL in blood was detected. However, as noted by the authors, the blood cholesterol concentrations were maintained in the normal range, i.e., less than 200 mg/dl (Ohtani et al. 2001). Importantly, Ohtani et al. reported that 8 among the 23 volunteers noted an increase in appetite. This points to possible changes in the food intake for some individuals in the course of experiments, changes which may explain (in part at least) some of the reported data. Unfortunately, food intakes were not reported in this publication. Also, the amino acid supplementation gave rise to an increase in hemoglobin and iron blood concentration. This was interpreted by the authors as an enhancement of hematopoiesis capacity due to the amino acid mixture supplementation (Ohtani et al. 2001). However, in our opinion, an increased hematopoiesis would have presumably led to an increased iron requirement. Finally, it is tempting to speculate that the amino acid supplementation performed by Ohtani et al. may have modified the food intake in some individuals and, as a consequence, may have participated in the changes observed in several biochemical and hematological parameters including total cholesterol and LDL cholesterol.
Recently, Zanchi et al. (2008) have published a review on the antiproteolytic effects of leucine emphasizing the inhibitory effect of leucine supplementation on skeletal muscle proteolysis. Several data presented suggest that the concentration of leucine, which is able to diminish protein degradation may be higher than the one required in order to maximally stimulates protein synthesis. This proposition is likely mostly relevant in atrophic conditions. Leucine catabolism is regulated by either of the first degradative steps: transamination to the keto acid alpha-ketoisocaproate (KIC) or subsequent decarboxylation. In an elegant and classical study, Matthews et al. (1981) using in volunteers l-leucine with 15N and 13C double labeling, demonstrated that leucine transamination was found to operate several times faster than the keto acid decarboxylation. Furthermore, it was determined in this latter study that whatever the dietary conditions, i.e., postabsorptive state and fed conditions; the keto acid decarboxylation was of similar magnitude (Matthews et al. 1981). Overall, these findings indicate that leucine metabolism is more related to the transamination pathway than to the decarboxylation pathway. In the course of leucine catabolism, beta-hydroxy-beta-methylbutyrate (HMB) can be formed from leucine. HMB is produced from KIC by the cytosolic enzyme KIC-dioxygenase (Sabourin and Bieber 1983). Numerous studies have reported that HMB is a precursor for cholesterol biosynthesis (Bloch 1944; Rabinowitz 1955; Zabin and Bloch 1951). HMB in the liver and muscle cytosol is converted to beta-hydroxy-beta-methylglutarate CoA (HMGCoA), which can then be utilized for cholesterol biosynthesis (Rudney 1957). In stressed or damaged muscles, it has been proposed that an insufficient synthesis of HMGCoA may limit adequate cholesterol synthesis, thus impairing the proper functioning of cell membranes (Nissen et al. 2000). In the work of Nissen et al. (2000), human volunteers received HMB supplementation (3 g/day) for 3–8 weeks. The authors found that when compared with the control group, HMB supplementation resulted in a 6% decrease in total cholesterol, 7% decrease of LDL cholesterol and a decrease in systolic blood pressure. It thus appears that the role of leucine on cholesterol metabolism is associated with different effects on cell and blood cholesterol. These effects of HMB on surrogate markers of cardiovascular functions lead the authors to propose a possible effect of HMB in decreasing the risk of heart attack and stroke (Nissen et al. 2000).
Cystine
Several works reported on the effect of cystine on cholesterol metabolism. In the study by Aoyama et al. (1999), it was determined that hypercholesterolemia caused in rats by feeding a high-cystine diet was probably due to the stimulation of hepatic cholesterol synthesis. Similar findings were reported in the study by Sérougne et al. (1995), which described hypercholesterolemia in rats induced by dietary cholesterol or by increasing cholesterogenesis in cystine-fed rats. This hypercholesterolemia was characterized by different plasma lipoprotein and apolipoprotein concentration and was associated with different apolipoprotein gene expression in the liver. In a study by Sérougne et al. (1987), rats were fed a cystine-enriched diet and it was determined that this diet increased hepatic cholesterogenesis.
Taurine
In a study by Yokogushi and Oda (2002), it was determined that dietary taurine enhances cholesterol degradation and reduces serum and liver concentration in rats fed on a high-cholesterol diet. The results presented are compatible with the hypothesis that taurine exerts a decreasing effect on cholesterolemia, partly via excretion of bile acid. Similar results were found by Choi et al. (2006) also in the rat model. In this latter study, rats in the control group received a 1.5% cholesterol diet and those in the experimental group received a diet containing 1.5% cholesterol and 1.5% taurine-supplemented diet. In the taurine group, it was found that total cholesterol as well as LDL-cholesterol were reduced and HDL-cholesterol was increased when compared with the data obtained in the control group. Also, the rats receiving taurine showed decreased hepatic content in cholesterol. Overall, these data suggest a role for taurine in rats receiving a high-cholesterol diet. A third study performed on rats (Yamori et al. 2004) reported an effect of dietary taurine in hypercholesterolemic animals. Indeed, taurine was able to lower blood cholesterol concentration in these experiments.
Aspartate and glutamate
In the work by Yanni et al. (2005), the authors examined the effect of l-aspartate and l-glutamate on serum lipoprotein cholesterol in rabbits fed with high-cholesterol diet using the rabbit model. The animals received a cholesterol/corn oil diet in the control group and the same diet supplemented with aspartate and glutamate for 4 weeks. The animals receiving the amino acid supplement were characterized by an increase in serum HDL-cholesterol. However, no differences were found between the two groups of animals for the serum total cholesterol, LDL-cholesterol and triglycerides.
Conclusion
-
1.
Based on epidemiological studies and some experimental data, in 1999, the US FDA has approved the food-labeling health claim of soy protein in the prevention of coronary heart disease.
-
2.
However later on, the Nutrition Committee of the American Heart Association released a review of 22 randomized trials and concluded that, overall, isolated soy protein with isoflavones slightly decrease LDL cholesterol and total cholesterol. The decrease in blood cholesterol concentration following soy-based protein extracts does not represent more than 6–12% of total cholesterol.
-
3.
It is likely that the source of soybeans and processing of the protein extracts are important for explaining the heterogeneity of the data obtained, since the concentrations of bioactive peptides and other minor components in the soy extracts are different according to the process used.
-
4.
Taken as a whole, it appears that the effect of soy extracts on cholesterolemia is complex and relevant to a synergistic effect of various soy compounds including amino acid composition and nonprotein compounds like isoflavones, fiber, minerals, etc.
-
5.
Other plant extracts than soy-based protein extracts and also protein from animal origin have been reported to exert a lowering effect on blood cholesterol when compared with a reference protein (often casein). The underlying mechanisms are little understood at the present time.
-
6.
Individual amino acid and a mixture of amino acids have been tested (mostly in animal studies) on cholesterol blood and liver concentrations and metabolism. Methionine and lysine have been used in the rabbit model and found to provoke hypercholesterolemia. Although this effect was also found in other experimental animals (rat, gerbil), the relevance of these results to human nutrition remains putative.
-
7.
In the mouse model, leucine supplementation is able to markedly reduce the plasma level of total and LDL cholesterol, but the underlying mechanisms remain to be determined in a causative perspective.
-
8.
In experimental animal models, high dietary supplementation with cystine is associated with hypercholesterolemia. In contrast, taurine supplementation is able to decrease blood cholesterol concentration in hypercholesterolemic animals
-
9.
Aspartate and glutamate supplementation is able to increase HDL-cholesterol in the rabbit model.
References
Adams MR, Golden DL, Anthony MS, Register TC, Williams JK (2002) The inhibitory effect of soy protein on atherosclerosis in mice does not require the presence of LDL receptor or alteration of plasma lipoproteins. J Nutr 132:43–49
Adams MR, Golden DL, Franke AA, Potter SM, Smith HS, Anthony MS (2004) Dietary soy beta-conglycinin (7S globulin) inhibits atherosclerosis in mice. J Nutr 134:511–516
Anderson JW, Johnstone BM, Cook-Newell ME (1995) Meta-analysis of the effects of soy protein on serum lipids. N Engl J Med 333:276–282. doi:10.1056/NEJM199508033330502
Anderson JW, Smith B, Washnock CS (1999) Cardiovascular and renal benefits of dry bean and soybean intake. Am J Clin Nutr 70:464S–474S
Anthony MS, Clarkson TB, Williams JK (1998) Effects of soy isoflavones on atherosclerosis:potential mechanisms. Am J Clin Nutr 68:1390S–1393S
Aoyama Y, Amano N, Yoshida A (1999) Cholesterol synthesis and degradation in normal rats fed a cholesterol-free diet with excess cystine. Lipids 34:583–589. doi:10.1007/s11745-999-0402-8
Baum JA, Teng H, Erdman JW, Weigel RM, Klein BP, Persky VW, Freels S, Surya P, Bakhit RM, Ramos E, Shay NF, Potter SM (1998) Long-term intake of soy protein improves blood lipid profiles and increases mononuclear cell low-density lipoprotein receptor messenger RNA in hypercholesterolemic, postmenopausal women. Am J Clin Nutr 68:545–551
Belleville J (2002) Hypocholesterolemic effect of soy protein. Nutrition 18:684–686. doi:10.1016/S0899-9007(02)00799-2
Bloch K (1944) Some aspects of the metabolism of leucine and valine. J Biol Chem 115:255–263
Carroll KK (1981) Dietary protein and cardiovascular disease. In: New trends in nutrition, Lipid research and cardiovascular diseases. Alan Liss Inc., New York
Choi YY, Osada K, Ito Y, Nagasawa T, Choi MR, Nishizawa N (2005) Effects of dietary protein of Korean foxtail millet on plasma adiponectin, HDL-cholesterol, and insulin levels in genetically type 2 diabetic mice. Biosci Biotechnol Biochem 69:31–37. doi:10.1271/bbb.69.31
Choi MJ, Kim JH, Chang KJ (2006) The effect of dietary taurine supplementation on plasma and liver lipid concentrations and free amino acid concentrations in rats fed a high-cholesterol diet. Adv Exp Med Biol 583:235–242. doi:10.1007/978-0-387-33504-9_25
Crouse JR, Morgan T, Terry JG, Ellis G, Vitolons M, Burke GL (1999) A randomised trial comparing the effect of casein with that of soy protein containing varying amounts of isoflavones on plasma concentrations of lipids and lipoproteins. Arch Intern Med 159:2070–2076. doi:10.1001/archinte.159.17.2070
Cuevas AM, Irribarra VL, Castillo OA, Yanez MD (2003) Isolated soy protein improves endothelial function in postmenopausal hypercholesterolemic women. Eur J Clin Nutr 57:889–894. doi:10.1038/sj.ejcn.1601622
Debry G (2004) Experimental data on animals: dietary proteins and atherosclerosis. CRC, West Palm Beach
Francis G, Kerem Z, Makkar HP, Becker K (2002) The biological action of saponins in animal systems: a review. Br J Nutr 88:587–605. doi:10.1079/BJN2002725
Friedman M, Brandon DL (2001) Nutritional and health benefits of soy proteins. J Agric Food Chem 49:1069–1086. doi:10.1021/jf0009246
Giroux I, Kurowska EM, Carroll KK (1999) Role of dietary lysine, methionine, and arginine in the regulation of hypercholesterolemia in rabbits. J Nutr Biochem 10:166–171. doi:10.1016/S0955-2863(98)00091-6
Hidiroglou N, Gilani GS, Long L, Zhao X, Madere R, Cockell K, Belonge B, Ratnayake WM, Peace R (2004) The influence of dietary vitamin E, fat, and methionine on blood cholesterol profile, homocysteine levels, and oxidizability of low-density lipoprotein in the gerbil. J Nutr Biochem 15:730–740. doi:10.1016/j.jnutbio.2004.04.009
Hirche F, Schröder A, Knoth GI, Stangl GI, Eder K (2006) Effect of dietary methionine on plasma and liver cholesterol concentrations in rats and expression of hepatic genes involved in cholesterol metabolism. Br J Nutr 95:879–888. doi:10.1079/BJN20061729
Hodges RE, Krehl WA, Stone DB, Lopez A (1967) Dietary carbohydrates and low cholesterol diets: effects on serum lipids on man. Am J Clin Nutr 20:198–208
Horigome T, Cho YS (1992) Dietary casein and soybean protein affect the concentrations of serum cholesterol, triglycerides and free amino acids in rats. J Nutr 122:2273–2282
Huff MW, Hamilton RMG, Carroll KK (1977) Effects of dietary proteins and amino acids on the plasma cholesterol concentrations of rabbits fed cholesterol-free diets. Adv Exp Med Biol 82:275–277
Jacques H, Gascon A, Bergeron N, Lavigne C, Hurley C, Deshaies Y, Moorjani S, Julien P (1995) Role of dietary fish protein in the regulation of plasma lipids. Can J Cardiol G:63G–71G
Kern M, Ellison D, Marroquin Y, Ambrose M, Mosier K (2002) Effects of soy protein supplemented with methionine on blood lipids and adiposity of rats. Nutrition 18:654–656. doi:10.1016/S0899-9007(02)00783-9
Kurowska EM, Carroll KK (1990) Essential amino acids in relation to hypercholesterolemia induced in rabbits by dietary casein. J Nutr 120:831–836
Kurowska EM, Carroll KK (1992) Effect of high levels of selected dietary essential amino acids on hypercholesterolemia and down-regulation of hepatic LDL receptors in rabbits. Biochim Biophys Acta 1126:185–191
Kurowska EM, Carroll KK (1994) Hypercholesterolemic responses in rabbits to selected groups of dietary essential amino acids. J Nutr 124:364–370
Kurowska EM, Carroll KK (1996) LDL versus apolipoprotein B responses to variable proportions of selected amino acids in semipurified diets fed to rabbits and in the media of hepG2 cells. J Nutr Biochem 7:418–424. doi:10.1016/0955-2863(96)00064-2
Kurowska EM, Hrabek-Smith JM, Carroll KK (1989) Compositional changes in serum lipoproteins during development of casein-induced hypercholesterolemia in rabbits. J Nutr 116:1395–1404
Lin Y, Meijer GW, Vermeer MA, Trautwein EA (2004) Soy protein enhances the cholesterol-lowering effect of plant sterol esters in cholesterol-fed hamsters. J Nutr 134:143–148
Matthews DE, Bier DM, Rennie MJ, Edwards RH, Halliday D, Millward DJ, Clugston GA (1981) Regulation of leucine metabolism in man: a stable isotope study. Science 214:1129–1131. doi:10.1126/science.7302583
Mayilvaganan M, Singh SP, Johari RP (2004) Hypocholesterolemic effect of protein prepared from Phaseolus aconitifolius (Jacq.). Indian J Exp Biol 42:904–908
McCarty (1999) Vegan proteins may reduce risk of cancer, obesity, and cardiovascular disease by promoting increased glucagon activity. Med Hypotheses 53:459–485. doi:10.1054/mehy.1999.0784
Morita T, Oh-Hashi A, Takei K, Ikai M, Kasaoka S, Kiriyama S (1997) Cholesterol-lowering effects of soybean, potato and rice proteins depend on their low methionine contents in rats fed a cholesterol-free purified diet. J Nutr 127:470–477
Moriyama T, Kishimoto K, Nagai K, Urade R, Ogawa T, Utsumi S, Maruyama N, Maebuchi M (2004) Soybean beta-conglycinin diet suppresses serum triglyceride levels in normal and genetically obese mice by induction of beta-oxidation, downregulation of fatty acid synthase, and inhibition of triglyceride absorption. Biosci Biotechnol Biochem 68:352–359. doi:10.1271/bbb.68.352
Nagaoka S, Kanamaru Y, Kuzuya Y (1991) Effects of whey protein and casein on the plasma and liver lipids in rats. Agric Biol Chem 55:813–818
Nagaoka S, Kanamaru Y, Kuzuya Y, Kojima S (1992) Comparative studies on the serum cholesterol lowering action of whey protein and soybean protein in rats. Biosci Biotechnol Biochem 56:1484–1485
Nagaoka S, Miwa K, Eto M, Kuzuya Y, Hori G, Yamamoto K (1999) Soy protein peptic hydrolysate with bound phospholipids decreases micellar solubility and cholesterol absorption in rats and caco-2 cells. J Nutr 129:1725–1730
Nagata C, Takatsuka N, Kurisu Y, Shimizu H (1998) Decreased serum total cholesterol concentration associated with high intake of soy products in Japanese men and women. J Nutr 128:209–213
Ni W, Tsuda Y, Sakono M, Imaizumi K (1998) Dietary soy protein isolate, compared with casein, reduces atherosclerotic lesion area in apolipoprotein E-deficient mice. J Nutr 128:1884–1889
Nissen S, Sharp RL, Panton L, Vukovitch M, Trappe S, Fuller JC (2000) Beta-hydroxy-beta-methylbutyrate (HMB) supplementation in humans is safe and may decrease cardiovascular risk factors. J Nutr 130:1937–1945
Ohtani M, Maruyama K, Sugita M, Kobayashi K (2001) Amino acid supplementation affects hematological and biochemical parameters in elite rugby players. Biosci Biotechnol Biochem 65:1970–1976. doi:10.1271/bbb.65.1970
Potter SM (1995) Overview of proposed mechanisms for the hypercholesterolemic effect of soy. J Nutr 125:606S–611S
Potter SM, Pertile J, Berber-Jimenez MD (1996) Soy protein concentrate and isolated soy protein similarly lower blood serum cholesterol but differently affect thyroid hormones in hamsters. J Nutr 126:2007–2011
Potter SM, Baum JA, Stillman RJ, Shay NF, Erdman JW (1998) Soy protein isiflavones: their effects on blood lipids and bone density in postmenopausal women. Am J Clin Nutr 68:1375S–1379S
Rabinowitz JL (1955) The biosynthesis of radioactive beta-hydroxyisovaleric acid in rat liver. J Am Chem Soc 77:1295–1297. doi:10.1021/ja01610a069
Rudney H (1957) The biosynthesis of beta-hydroxy-beta-methylglutaric acid. J Biol Chem 227:363–377
Sabourin PJ, Bieber LL (1983) Formation of beta-hydroxyisovalerate by an alpha-ketoisocaproate oxygenase in human liver. Metabolism 32:160–164. doi:10.1016/0026-0495(83)90223-8
Sacks FM, Lichtenstein A, Van Horn L, Harris W, Kris-Etherton P, Winston M (2006) Soy protein, isoflavones, and cardiovascular health: an American Heart Association science advisory for professionals from the Nutrition Committee. Circulation 113:1034–1044. doi:10.1161/CIRCULATIONAHA.106.171052
Sérougne C, Férézou J, Rukaj A (1987) A new relatioship between cholesterolemia and cholesterol synthesis determined in rats fed an excess of cystine. Biochim Biophys Acta 921:522–530
Sérougne C, Felgines C, Férézou J, Hajri T, Bertin C, Mazur A (1995) Hypertcholesterolemia induced by cholesterol- or cystine-enriched diet is characterized by different plasma lipoprotein and apolipoprotein concentrations in rats. J Nutr 125:35–41
Shukla A, Bettzieche A, Hirche F, Brandsch C, Stangl GI, Eder K (2006) Dietary fish protein alters blood lipid concentrations and hepatic genes involved in cholesterol homeostasis in the rat model. Br J Nutr 96:674–682
Sirtori CR, Pazzucconi F, Colombo L, Battistin P, Bondioli A, Dezscheemaeker K (1999) Double blind study of the addition of high-protein soya milk vs. cow’s milk and resistance to or intolerance of statins. Br J Nutr 82:91–96
Teixera SR, Potter SM, Weigel R, Hannum S, Erdman JW, Hasler CM (2000) Effects of feeding 4 levels of soy protein for 3 and 6 wk on blood lipids and apolipoproteins in moderatly hypercholesterolemic men. Am J Clin Nutr 71:1077–1084
Tomatoke H, Shimaoka I, Kayashita J, Yokoyama F, Nakajoh M, Kato N (2000) A buckwheat protein product suppresses gallstone formation and plasma cholesterol more strongly than soy protein isolate in hamsters. J Nutr 130:1670–1674
Tonstad S, Smerud K, Hoie L (2002) A comparison of the effects of 2 doses of soy protein or casein on serum lipids, serum lipoproteins, and plasma total homocysteine in hypercholesterolemic subjects. Am J Clin Nutr 76:78–84
US Food and Drug Administration (1999) Food labelling health claims: soy protein and coronary heart disease. Food and Drug Administration, HHS. Final rule. Fed Regist 64:57700–57733
Velez-Carrasco W, Merkel M, Twiss CO, Smith JD (2008) Dietary methionine effects on plasma homocysteine and HDL metabolism in mice. J Nutr Biochem 19:362–370. doi:10.1016/j.jnutbio.2007.05.005
Wang Y, Jones PJ, Ausman LM, Lichtenstein AH (2004) Soy protein reduces triglycerides levels and triglycerides fatty acid fractional synthesis rate in hypercholesterolemic subjects. Atherosclerosis 173:269–275. doi:10.1016/j.atherosclerosis.2003.12.015
Washburn S, Burke GL, Morgan T, Anthony M (1999) Effect of soy protein supplementation on serum lipoproteins, blood pressure, and menopausal symptoms in perimenopausal women. Menopause 6:7–13. doi:10.1097/00042192-199906010-00004
Wergedahl H, Liaset B, Gudbrandsen OA, Lied E, Espe M, Muna Z, Mork S, Berge RK (2004) Fish protein hydrolysate reduces plasma total cholesterol, increases the proportion of HDL cholesterol, and lowers acyl-CoA:cholesterol acyltransferase activity in liver of Zucker rats. J Nutr 134:1320–1327
West SG, Hilpert KF, Juturu V, Bordi PL, Lampe JW, Mousa SA, Kris-Etherton PM (2005) Effect of including soy protein in a blood cholesterol-lowering diet on markers of cardiac risk in men and in postmenopausal women with and without hormone replacement therapy. J Womens Health 14:353–362. doi:10.1089/jwh.2005.14.253
Wong WW, Smith EO, Stuff JE, Hachey DL, Heird WC, Pownell HJ (1998) Cholesterol-lowering effect of soy protein in normocholesterolemic and hypercholesterolemic men. Am J Clin Nutr 68:1385S–1389S
Xiao CW (2008) Health effects of soy protein and isoflavones in humans. J Nutr 138:1244S–1249S
Yamori Y, Murakami S, Ikeda K, Nara Y (2004) Fish and lifestyle-related disease prevention: experimental and epidemiological evidence for anti-atherogenic potential of taurine. Clin Exp Pharmacol Physiol 31:S20–S23. doi:10.1111/j.1440-1681.2004.04122.x
Yanni AE, Perrea DN, Yatzidis HA (2005) Effects of antiatherogenic l-aspartate and l-glutamate on serum lipoproteins cholesterol and apolipoproteins A-1 and B in rabbits fed with high-cholesterol diet. Nutr Metab Cardiovasc Dis 15:161–165. doi:10.1016/j.numecd.2004.06.001
Yokogushi H, Oda H (2002) Dietary taurine enhances cholesterol degradation and reduces serum and liver cholesterol concentrations in rats fed a high-cholesterol diet. Amino Acids 23:433–439. doi:10.1007/s00726-002-0211-1
Zabin I, Bloch K (1951) The utilization of butyric acid for the synthesis of cholesterol and fatty acids. J Biol Chem 192:261–266
Zanchi NE, Nicastro H, Lancha AH (2008) Potential antiproteolytic effects of l-leucine: observations of in vitro and in vivo studies. Nutr Metab 5:20–26. doi:10.1186/1743-7075-5-20
Zhan S, Ho SC (2005) Meta-analysis of the effects of soy protein containing isoflavones on the lipid profile. Am J Clin Nutr 81:397–408
Zhang X, Beynen AC (1993) Influence of dietary fish proteins on plasma and liver cholesterol concentrations in rats. Br J Nutr 69:767–777. doi:10.1079/BJN19930077
Zhang Y, Guo K, LeBlanc RE, Loh D, Schwartz GJ, Yu YH (2007) Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes 56:1647–1654. doi:10.2337/db07-0123
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blachier, F., Lancha, A.H., Boutry, C. et al. Alimentary proteins, amino acids and cholesterolemia. Amino Acids 38, 15–22 (2010). https://doi.org/10.1007/s00726-009-0239-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-009-0239-6