Summary.
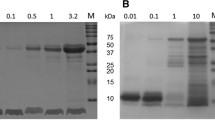
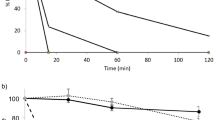
It was the aim of this study to evaluate the stability of two novel cystine-knot microproteins (CKM) SE-ET-TP-020 and SE-MC-TR-020 with potential clinical relevance towards luminally secreted proteases of the gastrointestinal tract in order to gain information about their potential for oral administration. Therefore, the stability of the two CKM and the model-drug insulin towards collected porcine gastric and small intestinal juice as well as towards isolated proteolytic enzymes was evaluated under physiological conditions. No intact SE-ET-EP-020 was detected after few seconds of incubation with porcine small intestinal juice. SE-ET-TP-020 was also degraded in porcine gastric juice. Furthermore, SE-ET-TP-020 was extensively degraded by isolated chymotrypsin, trypsin and pepsin. Moreover, it was degraded by elastase. SE-MC-TR-020 was degraded entirely within approximately 2 h when incubated in porcine small intestinal juice, whereas no degradation was observed within a 3 h incubation period with porcine gastric juice. In presence of the isolated proteolytic enzymes, SE-MC-TR-020 was only slightly degraded by trypsin and pepsin, whereas elastase caused no degradation to SE-MC-TR-020 at all. Chymotrypsin was the protease that caused most degradation to SE-MC-TR-020. The model drug insulin was degraded extensively by chymotrypsin, elastase, pepsin and trypsin as well as by porcine gastric and porcine small intestinal juice. In conclusion, a precise characterisation of SE-ET-TP-020 and SE-MC-TR-020 degrading luminally secreted GI enzymes has been made, which is an important and substantial prerequisite for the further optimisation of these CKM.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
O Avrutina HU Schmoldt D Gabrijelcic-Geiger D Le Nguyen CP Sommerhoff U Diederichsen H Kolmar (2005) ArticleTitleTrypsin inhibition by macrocyclic and open-chain variants of the squash inhibitor MCoTI-II Biol Chem 386 1301–1306 Occurrence Handle16336125 Occurrence Handle10.1515/BC.2005.148 Occurrence Handle1:CAS:528:DC%2BD28XhslOltQ%3D%3D
A Bernkop-Schnürch (1998) ArticleTitleThe use of inhibitory agents to overcome the enzymatic barrier to perorally administered therapeutic peptides and proteins J Control Rel 52 1–16 Occurrence Handle10.1016/S0168-3659(97)00204-6
A Bernkop-Schnürch R Fragner (1996) ArticleTitleInvestigations into the diffusion behaviour of polypeptides in native intestinal mucus with regard to their peroral administration Pharm Sci 2 361–363
A Christmann K Walter A Wentzel R Kratzner H Kolmar (1999) ArticleTitleThe cystine knot of a squash-type protease inhibitor as a structural scaffold for Escherichia coli cell surface display of conformationally constrained peptides Protein Eng 12 797–806 Occurrence Handle10506290 Occurrence Handle10.1093/protein/12.9.797 Occurrence Handle1:CAS:528:DyaK1MXmvVagtrk%3D
ML Colgrave DJ Craik (2004) ArticleTitleThermal, chemical, and enzymatic stability of the cyclotide kalata B1: the importance of the cyclic cystine knot Biochemistry 43 5965–5975 Occurrence Handle15147180 Occurrence Handle10.1021/bi049711q Occurrence Handle1:CAS:528:DC%2BD2cXjsFGgsL4%3D
DJ Craik NL Daly T Bond C Waine (1999) ArticleTitlePlant cyclotides: a unique family of cyclic and knotted proteins that defines the cyclic cystine knot structural motif J Mol Biol 294 1327–1336 Occurrence Handle10600388 Occurrence Handle10.1006/jmbi.1999.3383 Occurrence Handle1:CAS:528:DyaK1MXnvFaqsLs%3D
DJ Craik NL Daly C Waine (2001) ArticleTitleThe cystine knot motif in toxins and implications for drug design Toxicon 39 43–60 Occurrence Handle10936622 Occurrence Handle10.1016/S0041-0101(00)00160-4 Occurrence Handle1:CAS:528:DC%2BD3cXmsFKisrw%3D
DJ Craik M Cemazar NL Daly (2006) ArticleTitleThe cyclotides and related macrocyclic peptides as scaffolds in drug design Curr Opin Drug Discov Devel 9 251–260 Occurrence Handle16566295 Occurrence Handle1:CAS:528:DC%2BD28XislSmtL4%3D
SA Doggrell (2004) ArticleTitleIntrathecal ziconotide for refractory pain Expert Opin Investig Drugs 13 875–877 Occurrence Handle15212625 Occurrence Handle10.1517/13543784.13.7.875 Occurrence Handle1:CAS:528:DC%2BD2cXltVKmu70%3D
L Grain (1973) ArticleTitleIsolation of oxytocic peptides from Oldenlandia affinis by solvent extraction of tetraphenylborate complexes and chromatography on sephadex LH-20 Lloydia 36 207–208 Occurrence Handle4744557 Occurrence Handle1:STN:280:DyaE2c%2FgvFamsw%3D%3D
R Kratzner JE Debreczeni T Pape TR Schneider A Wentzel H Kolmar GM Sheldrick I Uson (2005) ArticleTitleStructure of Ecballium elaterium trypsin inhibitor II (EETI-II): a rigid molecular scaffold Acta Crystallogr D Biol Crystallogr 61 1255–1262 Occurrence Handle16131759 Occurrence Handle10.1107/S0907444905021207 Occurrence Handle1:CAS:528:DC%2BD2MXpsFWgtr0%3D
B Loretz FA Föger M Werle A Bernkop-Schnürch (2006) ArticleTitleOral gene delivery: strategies to improve stability of pDNA towards intestinal digestion J Drug Targ 14 311–319 Occurrence Handle10.1080/10611860600823766 Occurrence Handle1:CAS:528:DC%2BD28Xotlygt70%3D
PK Pallaghy KJ Nielsen DJ Craik RS Norton (1994) ArticleTitleA common structural motif incorporating a cystine knot and a triple-stranded beta-sheet in toxic and inhibitory polypeptides Protein Sci 3 1833–1839 Occurrence Handle7849598 Occurrence Handle1:CAS:528:DyaK2MXit1yjsLs%3D Occurrence Handle10.1002/pro.5560031022
J Swarbrick JC Boylan (2002) Encyclopedia of pharmaceutical technology EditionNumber2 Marcel Dekker, Inc. New York 885
M Werle T Schmitz H Huang A Wentzel H Kolmar A Bernkop-Schnürch (2006a) ArticleTitleThe potential of cystine-knot microproteins as novel pharmacophoric scaffolds in oral peptide drug delivery J Drug Targ 14 137–146 Occurrence Handle10.1080/10611860600648254 Occurrence Handle1:CAS:528:DC%2BD28XmtVamsbg%3D
M Werle A Samhaber A Bernkop-Schnürch (2006b) ArticleTitleDegradation of teriparatide by gastro-intestinal proteolytic enzymes J Drug Targ 14 109–115 Occurrence Handle10.1080/10611860600647934 Occurrence Handle1:CAS:528:DC%2BD28XmtVamsb0%3D
M Werle A Bernkop-Schnürch (2006c) ArticleTitleStrategies to improve plasma half life time of peptide and protein drugs Amino Acids 30 351–367 Occurrence Handle10.1007/s00726-005-0289-3 Occurrence Handle1:CAS:528:DC%2BD28Xls1egsrw%3D
JF Woodley (1994) ArticleTitleEnzymatic barriers for GI peptide and protein delivery Crit Rev Ther Drug Carrier Syst 11 61–95 Occurrence Handle7600588 Occurrence Handle1:CAS:528:DyaK2MXlsVClu7g%3D
Author information
Authors and Affiliations
Additional information
Authors’ address: Martin Werle, Ph.D., Department of Pharmaceutical Technology, Institute of Pharmacy, Innrain 52, 6020 Innsbruck, Austria
Rights and permissions
About this article
Cite this article
Werle, M., Kolmar, H., Albrecht, R. et al. Characterisation of the barrier caused by luminally secreted gastro-intestinal proteolytic enzymes for two novel cystine-knot microproteins. Amino Acids 35, 195–200 (2008). https://doi.org/10.1007/s00726-007-0569-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-007-0569-1