Summary.
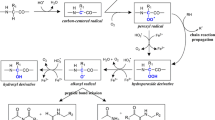
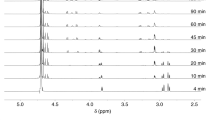
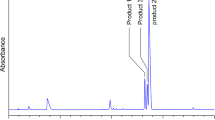
Histidine and lysine are two representative targets of oxidative modifications. Histidine is extremely sensitive to a metal-catalyzed oxidation, generating 2-oxo-histidine and its ring-ruptured products, whereas the oxidation of lysine generates carbonyl products, such as aminoadipic semialdehyde. On the other hand, both histidine and lysine are nucleophilic amino acids and therefore vulnerable to modification by lipid peroxidation-derived electrophiles, such as 2-alkenals, 4-hydroxy-2-alkenals, and ketoaldehydes, derived from lipid peroxidation. Histidine shows specific reactivity toward 2-alkenals and 4-hydroxy-2-alkenals, whereas lysine is a ubiquitous target of aldehydes, generating various types of adducts. Covalent binding of reactive aldehydes to histidine and lysine is associated with the appearance of carbonyl reactivity and antigenecity of proteins.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Uchida, K. Histidine and lysine as targets of oxidative modification. Amino Acids 25, 249–257 (2003). https://doi.org/10.1007/s00726-003-0015-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-003-0015-y