Abstract
We introduce a hypothetical model that explains how surface microtubules in euglenids are generated, integrated and inherited with the flagellar apparatus from generation to generation. The Euglenida is a very diverse group of single-celled eukaryotes unified by a complex cell surface called the “pellicle”, consisting of proteinaceous strips that run along the longitudinal axis of the cell and articulate with one another along their lateral margins. The strips are positioned beneath the plasma membrane and are reinforced with subtending microtubules. Euglenids reproduce asexually, and the two daughter cells inherit pellicle strips and associate microtubules from the parent cell in a semi-conservative pattern. In preparation for cell division, nascent pellicle strips develop from the anterior end of the cell and elongate toward the posterior end between two parent (mature) strips, so that the total number of pellicle strips and underlying microtubules is doubled in the predivisional cell. Each daughter cell inherits an alternating pattern of strips consisting of half of the nascent strips and half of the parent (mature) strips. This observation combined with the fact that the microtubules underlying the strips are linked to the flagellar apparatus created a cytoskeletal riddle: how do microtubules associated with an alternating pattern of nascent strips and mature strips maintain their physical relationship to the flagellar apparatus when the parent cell divides? The model of microtubular inheritance articulated here incorporates known patterns of cytoskeletal semi-conservatism and two new inferences: (1) a multigenerational “pellicle microtubule organizing center” (pMTOC) extends from the dorsal root of the flagellar apparatus, encircles the flagellar pocket, and underpins the microtubules of the pellicle; and (2) prior to cytokinesis, nascent pellicle microtubules fall within one of two “left/right” constellations that are linked to one of the two new dorsal basal bodies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diversity in the organization of the microtubular cytoskeleton reflects fundamental differences between the major lineages, or “supergroups”, of eukaryotes. The microtubules in the vast majority of microbial eukaryotes originate from one or more microtubular roots that stem from the basal bodies of the flagellar apparatus (Moestrup 1982, 2000; Sleigh 1988; Beech et al. 1991; Brugerolle 1991; Triemer and Farmer 1991; Roberts and Roberts 1991; Andersen 1991). These microtubules can radiate in complex spatial patterns in order to support the cell surface, a feeding apparatus (if present), systems for locomotion, and a mitotic spindle. The inheritance of microtubules from a parent cell to its daughter cells often requires sophisticated cellular coordination and transformation, especially in groups of eukaryotes with complex cell surface features.
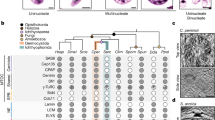
Euglenids, for instance, represent a diverse group of microbial eukaryotes renowned for their complex cytoskeleton, called the “pellicle”, consisting of a superficial framework of microtubules positioned beneath a system of proteinaceous strips oriented along the longitudinal axis of the cell (Fig. 1) (Leander et al. 2007; Sommer 1965). The pellicle strips are positioned beneath the plasma membrane, are S-shaped in transverse section, and articulate along their lateral margins so that they completely envelop the cell (Fig. 1a–b). Some euglenids (e.g., Euglena and Peranema) have deformable cells and exhibit a mode of motility called “euglenoid movement” consisting of twisting and wiggling; this movement is facilitated by the ability of helically arranged strips to slide past each along their lateral margins (Suzaki and Williamson 1985). Other euglenids (e.g., Petalomonas, and Phacus) have stiff cells consisting of a system of longitudinal strips. The number of pellicle strips is (generally) consistent within species and varies considerably between species and more inclusive taxonomic groups (the number of strips in described species ranges from 4 to 120). The microtubules positioned beneath the pellicle strips radiate from the flagellar apparatus, which is located at the base of the flagellar pocket (Fig. 1c). The microtubules extend anteriorly along the inner wall of the flagellar pocket before turning posteriorly at the opening of the flagellar pocket to underlie the superficially arranged pellicle strips (Fig. 1c).
The general organization of a euglenid cell. a Scanning electron micrograph (SEM) of a Euglena species showing the pellicle strips, some of which terminate before reaching the posterior end of the cell. b Transverse transmission electron micrograph (TEM) of the pellicle of Colacium mucronatum showing microtubules (arrowheads) beneath the (S-shaped) proteinaceous strips. c Labeled illustration showing the general ultrastructural features of euglenids. d–e. Identical SEMs of the posterior end of Euglena gracilis showing the multigenerational organization of the pellicle. The strip terminations are linked with lines to show three whorls of exponential reduction, and the different colors superimposed on the SEM correspond to different ages of the strips (e.g., yellow strips were produced from the most recent cell division event). d and e were modified with permission from Esson and Leander (2006)
The microtubular organizing center (MTOC) of euglenid cells consists of a tripartite system of microtubular roots associated with the flagellar apparatus: a dorsal root (DR), an intermediate root (IR) and a ventral root (VR). Two basal bodies—a dorsal basal body and a ventral basal body—provide the nucleation sites for the three microtubular roots. The DR emerges from dorsal basal body and both the IR and VR emerge from the ventral basal body. The microtubules associated with the pellicle strips originate from the dorsal root. The ventral root anchors the microtubules that are associated with the euglenid feeding apparatus, and the role of the intermediate root is unclear. Also unclear is how the microtubules that support the pellicle strips maintain their physical relationship with the flagellar apparatus when cells divide during asexual reproduction. This process is difficult to understand because the euglenid cytoskeleton consists of multigenerational components that are interspersed with one another and inherited in a semi-conservative pattern (Brugerolle 1992; Esson and Leander 2006, 2008, 2010; Farmer and Triemer 1988; Leander et al. 2007; Moestrup 2000; Mignot et al. 1987) (Fig. 1d–e).
The semi-conservative pattern of pellicle strip development
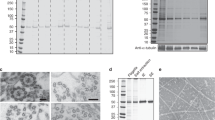
The duplication, segregation, and inheritance of pellicle strips from one generation to the next have been described in detail elsewhere (Esson and Leander 2006; Leander et al. 2007; Mignot et al. 1987). In preparation for cell division, nascent strips develop between two parent (mature) strips near the anterior end of the cell and migrate toward the posterior end, producing twice the number of strips that was present in the interphase cell (Figs. 1d–e and 2a–b). Each daughter cell inherits the same number of strips as the parent, but the nascent and parent strips are interspersed in an alternating pattern over the cell surface. Therefore, each daughter cell inherits half of the nascent strips interspersed between half of the parent (mature) strips.
Electron micrographs of Lepocinclis (Cyclidiopsis) acus showing the doubling of strips and microtubules in preparation for cytokinesis. a SEM showing nascent pellicle strips (arrows) developing between parental strips. b Transverse TEM showing nascent pellicle strips (arrows) emerging between parental strips. c TEM through the canal area showing nascent microtubules (arrows) developing between parental pairs of microtubules. d SEM of a cell undergoing cytokinesis. The canal is partitioned with a wall (arrow) into a left half and a right half. All images were modified with permission from Mignot et al. (1987)
Any given euglenid cell consists of pellicle strips representing several previous generations, and up to four generations may be required for pellicle strips to become fully elongated (mature) (Fig. 1d–e). For instance, Euglena gracilis possesses 40 pellicle strips in the interphase cell (Leander and Farmer 2000; Esson and Leander 2006). The youngest strips, which consist of half the total number of strips present (20), are the shortest and terminate before reaching the posterior end of the cell; the terminating points of these strips form a circular pattern, or “whorl”, when viewed from the posterior end (Fig. 1d). Strips that represent older generations are longer in length and form subsequent whorls at the posterior end of the cell. Each whorl consists of half the number of strips in the previous whorl (e.g., in a cell with 40 strips and three whorls: 20 terminating strips form the first whorl, 10 terminating strips form the next posterior whorl, and 5 terminating strips form the last posterior whorl). A euglenid cell with three whorls of terminating strips reflects at least four generations of strip duplication: each of the three whorls represents a different generation and the strips that reach the posterior tip of the cell represents the fourth (and older) generations (Esson and Leander 2006; Leander et al. 2007).
The microtubules beneath the pellicle duplicate in parallel to the pattern of strip inheritance described above (Mignot et al. 1987). Pairs of new microtubules develop in between existing pairs of microtubules and support the inner wall of the flagellar pocket (Fig. 2c). The new microtubules turn toward the posterior end of the cell at the opening of the flagellar pocket and migrate beneath the nascent pellicle strips. Therefore, prior to cell division, the total number of paired microtubules doubles in association with the doubling of pellicle strips (Fig. 2b–c). Each daughter cell inherits the same number of microtubules as the parent, but the new and parent microtubules are interspersed in an alternating pattern over the cell surface. Therefore, each daughter cell inherits half of the new microtubules interspersed between half of the parent microtubules (Fig. 2d). How these interspersed microtubules segregate during cell division is enigmatic, because both the old and the new microtubules that underlie the pellicle strips are linked to the dorsal root of the flagellar apparatus. The microtubules beneath the pellicle strips do not retract; once established they remain stable throughout the strip developmental process and during cell division (Mignot et al. 1987).
The semi-conservative pattern of flagellar apparatus development
The ventral and dorsal basal bodies and the associated roots of the flagellar apparatus are also inherited in a semi-conservative pattern and undergo a process called “flagellar transformation” (Brugerolle 1992; Farmer and Triemer 1988; Moestrup 2000). Before cell division, two new dorsal basal bodies develop adjacent to the parental (dorsal and ventral) basal bodies (Fig. 3a and e). The parental dorsal basal body transforms into a fully mature ventral basal body, creating two ventral basal bodies in the predivisional cell. Each of the two daughter cells receives one of the (parental) ventral basal bodies and one of the new dorsal basal bodies (Fig. 3b and e). During flagellar transformation, the dorsal root on the parental dorsal basal body transforms into an intermediate root on the new ventral basal body and a new ventral root develops de novo (Fig. 3c and f). Once established, the ventral basal body and associated ventral root and intermediate root remain intact throughout all future cell divisions. Because the pellicle microtubules are linked to the dorsal root in the interphase cell, the association of these microtubules with the old dorsal root when it transforms into a new intermediate root must be reorganized with microtubules associated with the newly developed dorsal root. This synthesis of previous studies made it extremely difficult to comprehend how the development and inheritance of the microtubules associated with the flagellar apparatus were coordinated with the development and inheritance of the microtubules associated with the pellicle strips. Our goal here was to establish a testable model that explains the process of microtubular reorganization during the interconnected processes of strip duplication and flagellar transformation.
Flagellar transformation in euglenids. a–c Transmission electron micrographs (TEM) of Entosiphon sulcatum. d–f. Corresponding schematic drawings of euglenid flagellar transformation. The parental components of the flagellar apparatus are show in green and the de novo components are shown in red d. The interphase cell contains two basal bodies (DB and VB) and three microtubular roots (DR, IR and VR). a, e Just before cytokinesis, two new basal bodies (arrowheads) form de novo near the parental basal bodies. b New dorsal microtubular roots form on the new dorsal basal bodies; the parent dorsal basal body transforms into a new ventral basal body and develops a ventral root de novo. c, f Separation of two pairs of basal bodies, each consisting of one new dorsal basal body and one ventral basal body. a, b and c were modified with permission from Brugerolle (1992)
Reconciling the inheritance patterns of pellicle and flagellar microtubules
Figures 4, 5 and 6 illustrate our model for the organization and inheritance of the euglenid cytoskeleton. For illustrative purposes, we consider a euglenid cell with 16 pellicle strips, 16 pairs of subtending microtubules, and two generations (i.e., posterior whorls) of strip reduction (Fig. 4a.i–ii). Our model of microtubular integration and inheritance is based on two key inferences.
Illustration of the new model of cytoskeletal development and inheritance in euglenids using an interphase cell with16 pellicle strips and 16 pairs of microtubules. For the sake of clarity, the ventral root has been omitted. The colors of pellicle microtubules, pellicle strips, and components of the flagellar apparatus correspond to different generations. See also Online Supplementary Material Fig. S1. a The initial generation of the model. i. Posterior view of a cell showing pellicle strips with two whorls of pellicle strip termination. ii. Cross section of the flagellar pocket showing 16 pairs of microtubules supporting the wall. iii. The flagellar apparatus. The microtubules supporting the wall of the flagellar pocket and the pellicle strips originate from a “pellicle microtubule organizing center” (pMTOC) associated with the dorsal root. iv. The total number of microtubule pairs that originated from the dorsal root (orange).b Developmental stage of the cell showing the doubling of the flagellar apparatus, micotubular pairs and pellicle strips prior to cytokinesis. i. Posterior view of a cell showing pellicle strips with two whorls of pellicle strip termination. The eight blue and eight red nascent strips emerge between the 16 parental orange strips. ii. Cross section of the flagellar pocket showing eight blue and eight red pairs of nascent microtubules between the 16 parental pairs of microtubules (orange). The arrows mark the cleavage furrow, which is determined by the “right/left” clusters of nascent strip units (shown in blue and red). iii. The flagellar apparatus. Two new dorsal basal bodies (blue and red) form de novo near the parental basal bodies (white and orange). The parental dorsal basal body (orange) transforms into a new ventral basal body. All of the orange pairs of microtubules that support the flagellar pocket and pellicle remain associated with the parental (orange) pMTOC on the new (orange) intermediate root (previous parental dorsal root). Eight new pairs of microtubules develop from each of the new dorsal roots and associated pMTOCs (shown in blue and red, respectively). The new pairs of microtubles are placed in alternative pattern with the parental pairs of microtubules (orange). iv. The total number of microtubule pairs that originated from each dorsal roots under consideration. c The daughter cell that inherited the blue strip units and half of the orange strip units following cytokinesis. i. Posterior view of a cell showing eight blue nascent strips positioned between eight orange parental strips with two whorls of pellicle strip termination. ii. Cross section of the flagellar pocket showing eight nascent blue strip units in between eight orange strip units. iii. The flagellar apparatus showing that all of the microtubules supporting the flagellar pocket and pellicle strips originate from the pMTOC on the blue dorsal root. The pMTOCs are shown in two colors in order to track the generations. iv. The total number of microtubule pairs that originated from each dorsal roots under consideration. d The daughter cell that inherited the red strip units and half of the orange strip units following cytokinesis. i. Posterior view of a cell showing eight red nascent strips positioned between eight orange parental strips with two whorls of pellicle strip termination. ii. Cross section of the flagellar pocket showing eight red nascent strip units in between eight orange strip units. iii. The flagellar apparatus showing that all of the microtubules supporting the flagellar pocket and pellicle strips originate from the pMTOC on the red dorsal root. The pMTOCs are shown in two colors in order to track the generations. iv. The total number of microtubule pairs that originated from each dorsal roots under consideration
Illustration of the new model of cytoskeletal development and inheritance in subsequent generations using an interphase cell with16 pellicle strips and 16 pairs of microtubules. For the sake of clarity, the ventral root has been omitted. The colors of pellicle microtubules, pellicle strips, and components of the flagellar apparatus correspond to different generations. a One of the daughter cells derived from the initial generation is shown in Fig. 4c. b Developmental stage of the cell showing the doubling of the flagellar apparatus, microtubular pairs and pellicle strips prior to cytokinesis. i. Posterior view of a cell showing a pellicle with two whorls of pellicle strip termination. The eight purple and eight green nascent strips emerge between the 16 parental strips (orange and blue). ii. Cross section of the flagellar pocket showing eight purple and eight green pairs of nascent microtubules between the 16 parental pairs of microtubules (orange and blue). The arrows mark the cleavage furrow, which is determined by the “right/left” clusters of nascent strip units (shown in purple and green). iii. The flagellar apparatus. The parental dorsal basal body (blue) transforms into a new ventral basal body. All of the blue and orange pairs of microtubules that support the flagellar pocket and pellicle remain associated with the parental (blue/orange) pMTOC on the new (blue) intermediate root (previous parental dorsal root). Eight new pairs of microtubules develop from each of the new dorsal roots and associated pMTOCs (shown in purple and green, respectively). The new pairs of microtubles are placed in alternative pattern with the parental pairs of microtubules (blue and orange). iv. The total number of microtubule pairs that originated from each dorsal roots under consideration. c The daughter cell that inherited the purple strip units and half of the orange/blue strip units following cytokinesis. i. Posterior view of a cell showing eight purple nascent strips positioned between eight blue/orange parental strips with two whorls of pellicle strip termination. ii. Cross section of the flagellar pocket showing eight nascent purple strip units in between eight blue/orange strip units. iii. The flagellar apparatus showing that all of the microtubules supporting the flagellar pocket and pellicle strips originate from the pMTOC on the purple dorsal root. The pMTOCs are shown in several colors in order to track the merging of several different generations. iv. The total number of microtubule pairs that originated from each dorsal roots under consideration. d The daughter cell that inherited the green strip units and half of the blue/orange strips units following cytokinesis. i. Posterior view of a cell showing eight green nascent strips positioned between eight blue/orange parental strips with two whorls of pellicle strip termination. ii. Cross section of the flagellar pocket showing eight green nascent strip units in between eight blue/orange strip units. iii. The flagellar apparatus showing that all of the microtubules supporting the flagellar pocket and pellicle strips originate from the pMTOC on the green dorsal root. The pMTOCs are shown in several colors in order to track the merging of several different generations. iv. The total number of microtubule pairs that originated from each dorsal roots (DR) under consideration
Illustration of the overall multigenerational pattern of cytoskeletal development and inheritance in euglenids through several generations, starting with an interphase cell with16 pellicle strips and 16 pairs of microtubules. For the sake of clarity, the ventral root has been omitted. The colors of pellicle microtubules, pellicle strips, and components of the flagellar apparatus (e.g., dorsal roots and pMTOCs) correspond to different generations
Inference 1
The pellicle microtubules do not extend directly from the dorsal root, which stands in contrast to previous interpretations that have been illustrated in the literature (Shin et al. 2001; Solomon et al. 1987; Willey and Wibel 1987; Supplementary Fig. 1). Instead, we propose that the pellicle microtubules extend from an intermediary “pellicle microtubule organizing center” (pMTOC) that branches from the dorsal root of the dorsal basal body (Fig. 4a.iii). The pMTOC encircles the posterior end of the flagellar pocket and supports all of the microtubules that underlie the pellicle strips. The distal end of the pMTOC terminates near the intermediate root, which branches from the ventral basal body (Fig. 4a.iii).
Inference 2
In preparation for cell division, the pellicle microtubules and associated proteinaceous strips are organized within one of two “left/right” constellations (Fig. 4b.i–ii). Each constellation of pellicle microtubules is linked to a different pMTOC that stems from one of the two new dorsal roots (and dorsal basal bodies) (Fig. 4b.iii). Therefore, although all of the nascent pellicle strips and microtubules that emerge between the parent strips look alike, half of them come from one dorsal basal body (i.e., the “left” constellation) and the other half come from the second dorsal basal body (i.e., the “right” constellation) (Fig. 4b.iii–iv). This organization of microtubules provides the fundamental mechanism for the intercalation of nascent strip units (a proteinaceous strip plus associated microtubules) between parent strip units and the subsequent separation of this multigenerational pattern into daughter cells. Below, we break down this process through several generations of cell division in order to emphasize the integration and continuity of euglenid microtubules through time (Figs. 4 and 5 and Supplementary Fig. 1).
First generation
In our chosen example, 16 pairs of pellicle microtubules (orange) originate from a pMTOC, extend anteriorly along the wall of the flagellar pocket, bend posteriorly at the opening of the flagellar pocket, and ultimately reside beneath the 16 pellicle strips (Figs. 1c and 4a). In preparation for cell division, two new dorsal basal bodies and associated dorsal roots develop de novo, and the parental dorsal root is transformed into the intermediate root of the newly transformed ventral basal body; the predivisional cell is now equipped with two dorsal basal bodies and two ventral basal bodies (Fig. 4b). A new pMTOC develops from each of the two new dorsal roots, and the new pMTOCs are superimposed onto the parental pMTOC that is now attached to the intermediate root of the newly transformed ventral basal body (Fig. 4b.iii). However, each of the two new pMTOCs occupy opposite (left–right) halves of the parental pMTOC (Fig. 4b.iii). Eight nascent pairs of microtubules develop from the “right” (red) pMTOC, and eight nascent pairs of microtubules develop from the “left” (blue) pMTOC (Fig. 4b.iii). All 16 nascent pairs of microtubules elongate between the 16 parent (orange) pairs of microtubules, generating an alternating pattern of parent and nascent microtubules in the wall of the flagellar pocket (Fig. 4b.ii) and beneath the pellicle strips (Fig. 5b.i). Therefore, the total number of microtubule pairs and corresponding pellicle strips is doubled to 32: 16 are from the parent, 8 are from the “right” (red) pMTOC, and 8 are from the “left” (blue) pMTOC (Fig. 4b).
During cytokinesis, the parental (orange) pMTOC (and associated microtubules and pellicle strips) is pulled in half at the junction where the new “left” (blue) pMTOC ends and the new “right” (red) pMTOC begins (Fig. 4b–d). This organization determines the cleavage furrow and ensures that each daughter cell receives 16 pairs of microtubules and corresponding pellicle strips with the same alternating pattern of old and new strip units that were present in the parent cell (Fig. 4c–d). Each daughter cell also receives one new dorsal basal body (with one of the new pMTOCs attached to the dorsal root) and one of the ventral basal bodies (and associated roots). Therefore, the daughter cell that received the newly transformed ventral basal body, which segregated with the new dorsal basal body on the left, could still have the parent pMTOC attached to the intermediate root (Fig. 4b–c). The daughter cell that received the older ventral basal body would definitely not have a pMTOC attached to the intermediate root because it would have segregated with the new dorsal body on the right (Fig. 4b and d). This configuration could be considered the fully mature state for a ventral basal body in euglenids, which requires a minimum of three generations to achieve.
Subsequent generations
One of the daughter cells from the above scenario, namely the configuration shown in Fig. 4c, will be followed further for additional explanation of the model. The color of the microtubules and the pMTOC in all of the illustrations has been kept constant in order to keep track of different generational origins. For instance, at least two different generations of microtubules, shown in orange and blue, extend from the pMTOC linked to the dorsal root of the parent cell shown in Fig. 5a. Prior to cytokinesis, each of the two new dorsal roots develops a new pMTOC and eight pairs of microtubules, shown in green and purple (Fig. 5b). As described previously, the new pMTOCs are superimposed onto the parental (multigenerational) pMTOC, which is ultimately pulled in half at the junction where the new “right” (green) pMTOC ends and the new “left” (purple) pMTOC begins (Fig. 5b–d). The pMTOCs in each new daughter cell supports four pairs of microtubules derived from the oldest generation (orange), four pairs of microtubules derived from the subsequent generation (blue), and eight pairs of microtubules derived from the newest generation of pMTOCs (either purple or green) (Fig. 5c–d). The configuration of microtubules in these daughter cells (and subsequent generations of daughter cells) demonstrates the complex multigenerational pattern of cytoskeletal components in euglenids (Fig. 6). Theoretically, an extant cell could still possess basal bodies, microtubules and an associated proteinaceous strip that was derived from the very first euglenid cell with an indisputable pellicle (shown in orange in our example; Fig. 6) (Leander et al. 2007).
Summary of our model
The hypothetical model we introduce here, in our opinion, explains how microtubules in euglenid cells are generated, organized, synchronized, and inherited during cell division. Prior to cell division, two new dorsal basal bodies each with a new dorsal root develop de novo. The original ventral basal body and its associated roots remain stable from generation to generation. However, the original dorsal basal body and its associated dorsal root transform into a second ventral basal body and its associated intermediate root, respectively; this new ventral basal body also develops a new ventral root de novo (Farmer and Triemer 1988; Moestrup 2000). The model of microtubular inheritance we propose incorporates the above observations and the following insights: (1) a multigenerational “pellicle microtubule organizing center” (pMTOC) extends from the dorsal root of the flagellar apparatus, encircles the flagellar pocket, and underpins the microtubules of the pellicle; (2) prior to cell division, each of the two new dorsal roots develop separate nascent pMTOCs; (3) each nascent pMTOC is superimposed upon the parent pMTOC; (4) the nascent pMTOCs take over the role of microtubular nucleation once they merge with the parent pMTOC; (5) the two nascent pMTOCs occupy opposite halves of the parent pMTOC, and each supports half of the nascent pellicle strip microtubules; (6) this organization creates two “left/right” clusters of nascent pellicle strip microtubules that elongate between the parent strip microtubules, which maintain their connection to the now fused pMTOCs; (7) the relative “left/right” positions of the two nascent pMTOCs determine the position of the cleavage furrow; and (8) following cell division, each daughter cell inherits a ventral basal body, a nascent dorsal basal body with its associated dorsal root, pMTOC, and nascent pellicle strip microtubules, and half of the mature (parent) strip microtubules.
Conclusions
Unlike the model described here for euglenids, many other eukaryotes possess cytoskeletal microtubules that stem directly from a dorsal root, such as stramenopiles (e.g. Ochoromonas, Apoikia) (Andersen 1991; Kim et al. 2010), alveolates (e.g. Amphidinium, Gymnodinium) (Farmer and Robert 1989; Hansen and Moestrup 2005), excavates (e.g. Carpediemonas, Malawimonas) (O’Kelly and Nerad 1999; Simpson and Patterson 1999), and amoebozoans (e.g. Physarum, Covostelium) (Spiegel 1981; Wright et al. 1979). Most of these ultrastructural reconstructions are based on TEM sections through the flagellar apparatus taken from several different angles. Similar TEM data from euglenids do not show a direct connection between the pellicle microtubules and the dorsal root; instead, the proximal ends of the pellicle microtubules terminate before reaching the dorsal root. These data indicate that some other relationship between the dorsal root and the pellicle microtubules exists. The presence of a pMTOC is not only consistent with these observations but, perhaps even more importantly, is necessary to explain how the intercalation of alternating nascent and parent microtubules stay connected to the new dorsal basal bodies during cell division. The physical connection and the left–right clustering of the nascent microtubules provide a mechanism for determining the cleavage furrow during cytokinesis. Moreover, there is solid evidence in other euglenozoans (e.g., trypanosomatids) that a pMTOC-like structure branches from the dorsal basal body and encircles the flagellar pocket. Similar experiments using detergent-based cell lysis protocols and whole mount preparations of the flagellar apparatus (e.g., TEM negative staining) on euglenid cells provide alternative ways to observe pMTOC-like structures in this diverse group of eukaryotes. Nonetheless, our model is fundamentally different from any previous eukaryotic model for the integration of the flagellar apparatus with cell surface microtubules. Although the relatively high level of morphological diversity in euglenids is a direct reflection of evolutionary modifications of the cytoskeleton, the intricate mechanism of microtubular inheritance appears to be highly conserved within the group. How widely this particular mechanism is used in other lineages within the Euglenozoa and perhaps across the entire tree of excavates remains to be determined.
Abbreviations
- DB:
-
Dorsal basal body
- DR:
-
Dorsal root
- IR:
-
Intermediate root
- MTOC:
-
Microtubular organizing center
- pMTOC:
-
Pellicle microtubule organizing center
- VB:
-
Ventral basal body
- VR:
-
Ventral root
References
Andersen RA (1991) The cytoskeleton of chromophyte algae. Protoplasma 164:143–159
Beech PL, Heimann K, Melkonian M (1991) Development of the flagellar apparatus during the cell cycle in unicellular algae. Protoplasma 164:23–37
Brugerolle G (1991) Flagellar and cytoskeletal systems in amitochondrial flagellates: Archamoeba, Metamonada and Parabasala. Protoplasma 164:70–90
Brugerolle G (1992) Flagellar apparatus duplication and partition, flagellar transformation during division in Entosiphon sulcatum. BioSystems 28(1–3):203–209
Esson HJ, Leander BS (2006) A model for the morphogenesis of strip reduction patterns in phototrophic euglenids: evidence for heterochrony in pellicle evolution. Evol Dev 8(4):378–388. doi:10.1111/j.1525-142X.2006.00110.x
Esson HJ, Leander BS (2008) Novel pellicle surface patterns on Euglena obtusa (Euglenophyta) from the marine benthic environment: implications for pellicle development and evolution. J Phycol 44(1):132–141
Esson HJ, Leander BS (2010) Evolution of distorted pellicle patterns in rigid photosynthetic euglenids (Phacus Dujardin). J Eukaryot Microbiol 57(1):19–32. doi:10.1111/j.1550-7408.2009.00447.x
Farmer MA, Robert KR (1989) Comperative analyses of the dinoflagellate flagellar apparatus. III. Freeze substitution of Amphydinium rhyncocephalum. J Phycol 25:280–292
Farmer MA, Triemer RE (1988) Flagellar systems in the euglenoid flagellates. BioSystems 21(3–4):283–291
Hansen G, Moestrup Ø (2005) Flagellar apparatus and nuclear chambers of the green dinoflagellate Gymnodinium chlorophorum. Phycol Res 53(2):169–181
Kim E, Yubuki N, Leander BS, Graham LE (2010) Ultrastructure and 18S rDNA Phylogeny of Apoikia lindahlii comb. nov. (Chrysophyceae) and its epibiontic protists, Filos agilis gen. et sp. nov. (Bicosoecida) and Nanos amicus gen. et sp. nov. (Bicosoecida). Protist 161(2):177–196. doi:10.1016/j.protis.2009.09.003
Leander BS, Farmer MA (2000) Comparative morphology of the euglenid oellicle. I. Patterns of strips and pores. J Eukaryot Microbiol 47(5):469–479
Leander BS, Esson HJ, Breglia SA (2007) Macroevolution of complex cytoskeletal systems in euglenids. BioEssays 29(10):987–1000. doi:10.1002/bies.20645
Mignot JP, Brugerolle G, Bricheux G (1987) Intercalary strip development and dividing cell morphogenesis in the euglenid Cyclidiopsis acus. Protoplasma 139(1):51–65
Moestrup Ø (1982) Flagellar structure in algae: a review, with new observations particularly on the Chrysophyceae, Phaeophyceae (Fucophyceae), Euglenophyceae and Reckertia. Phycologia 21(4):427–528
Moestrup Ø (2000) The flagellate cytoskeleton. Introduction of a general terminology for microtubular flagellar roots in protists. In: Leadbeater BSC, Green JC (eds) The flagellate. Unity, diversity and evolution. The systematics association special volume series 59. Taylor & Francis Limited, London, pp 69–94
O’Kelly CJ, Nerad TA (1999) Malawimonas jakobiformis n. gen., n. sp. (Malawimonadidae n. fam.): a Jakoba-like heterotrophic nanoflagellate with discoidal mitochondrial cristae. J Euk Microbiol 46(5):522–531. doi:10.1111/j.1550-7408.1999.tb06070.x
Roberts KR, Roberts JE (1991) The flagellar apparatus and cytoskeleton of the dinoflagellates. A comparative overview. Protoplasma 164:105–122
Shin W, Boo SM, Triemer RE (2001) Ultrastructure of the basal body complex and putative vestigial feeding apparatus in Phacus pleuronectes (Euglenophyceae). J Phycol 37(5):913–921
Simpson AGB, Patterson DJ (1999) The ultrastructure of Carpediemonas membranifera (Eukaryota) with reference to the “excavate hypothesis”. Eur J Protistol 35:353–370
Sleigh MA (1988) Flagellar root maps allow speculative comparisons of root patterns and of their ontogeny. BioSystems 21(3–4):277–282
Solomon JA, Walne PL, Kivic PA (1987) Entosiphon sulcatum (Euglenophyceae): flagellar roots of the basal body complex and reservoir region. J Phycol 23(1):85–98
Sommer JR (1965) The Ultrastructure of the pellicle complex of Euglena gracilis. J Cell Biol 24:253–257
Spiegel FW (1981) Phylogenetic significance of the flagellar apparatus in protostelids (Eumycetozoa). BioSystems 14(3–4):491–499
Suzaki T, Williamson RE (1985) Euglenoid movement in Euglena fusca: evidence for sliding between pellicular strips. Protoplasma 124(1–2):137–146
Triemer RE, Farmer MA (1991) An ultrastructural comparison of the mitotic apparatus, feeding apparatus, flagellar apparatus and cytoskeleton in euglenoids and kinetoplastids. Protoplasma 164:91–104
Willey RL, Wibel RG (1987) Flagellar roots and the reservoir cytoskeleton of Colacium libellae (Euglenophyceae). J Phycol 23(s2):283–288
Wright M, Moisand A, Mir L (1979) The structure of the flagellar apparatus of the swarm cells of Physarum polycephalum. Protoplasma 100(3):231–250. doi:10.1007/bf01279314
Acknowledgements
This work was supported by grants from the Tula Foundation (Centre for Microbial Diversity and Evolution at the University of British Columbia) and the Canadian Institute for Advanced Research, Program in Integrated Microbial Biodiversity.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: David Robinson
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Related to Fig. 4. Illustration of the conventional model of cytoskeletal organization in euglenids using an interphase cell with16 pellicle strips and 16 pairs of microtubules. For the sake of clarity, the ventral root has been omitted. In this model, all of the microtubules that support the wall of the flagellar pocket and the pellicle strips originate directly from the dorsal root. This configuration is physically impossible to maintain during cell division because new microtubule pairs that emerge prior to cytokinesis are associated with new dorsal roots on new dorsal basal bodies and the dorsal root of the parent transforms into an intermediate root on a new ventral basal body. Therefore, the semi-conservative inheritance of new microtubules intercalated between parent microtubules requires a different explanatory model than the one illustrated in previous interpretations. (JPG 1.97 mb)
Rights and permissions
About this article
Cite this article
Yubuki, N., Leander, B.S. Reconciling the bizarre inheritance of microtubules in complex (euglenid) microeukaryotes. Protoplasma 249, 859–869 (2012). https://doi.org/10.1007/s00709-011-0340-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-011-0340-z