Abstract
Ruthenium–arene complexes bearing N-heterocyclic carbene (NHC) ligands with the generic formula [RuCl2(p-cymene)(NHC)] are efficient catalyst precursors for the cyclopropanation of activated olefins with ethyl diazoacetate, and the cis/trans diastereoselectivity of the reaction markedly depends on the steric bulk of the NHC. The procedure was successfully applied to styrene, α-methylstyrene, and various other styrenic derivatives bearing electron-withdrawing or donating substituents on their aromatic rings. The reaction of unactivated internal or terminal alkenes was more sluggish, and the use of norbornene as a substrate afforded only olefin metathesis. Further investigation of the ring-opening metathesis polymerization of this strained cycloolefin in the presence of trimethylsilyldiazomethane led to high molecular weight polynorbornene whose microstructure was not significantly affected by the choice of the NHC ancillary ligand.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the first imidazol-2-ylidene derivative was isolated and characterized by Arduengo in 1991 [1, 2], N-heterocyclic carbenes (NHCs) have become ubiquitous ligands in organometallic chemistry and in homogeneous catalysis [3–6]. Over the past 25 years, these stable divalent carbon species have already afforded countless transition metal catalysts that have found important practical applications in olefin chemistry for hydrogenation, hydrosilylation, hydroformylation, hydroamination, or Heck reactions, to name just a few [7–9]. In particular, thanks to the pioneering work of Herrmann [10], Nolan [11], and Grubbs [12], the replacement of a phosphine with an NHC ligand onto ruthenium–alkylidene scaffolds led to a rich portfolio of well-defined catalysts for olefin metathesis [13–16].
In parallel with the development of Grubbs-type 16-electron ruthenium–alkylidene complexes, we found that coordinatively saturated ruthenium–arene species with the generic formula [RuCl2(η 6-arene)(NHC)] were versatile and efficient catalyst precursors for the ring-opening metathesis polymerization (ROMP) of cyclic olefins when activated by visible light [17]. A 1:2:4 molar association of the [RuCl2(p-cymene)]2 dimer (1), an imidazol(in)ium salt, and a base also provided efficient catalytic systems for various types of metathesis reactions [18, 19]. Such a combination required only stable and commercially available reagents to generate active species in situ, thereby affording very simple and straightforward experimental procedures [20].
Early work from our Laboratory had shown that some ruthenium-based catalysts could be engineered and fine-tuned to mediate either olefin metathesis or cyclopropanation reactions [21]. Indeed, it was observed that highly active ruthenium promoters for metathesis were inactive for cyclopropanation and vice versa. With this in mind, we anticipated that the least active or inactive [RuCl2(η 6-arene)(NHC)] initiators for olefin metathesis should be efficient catalyst precursors for olefin cyclopropanation. We therefore launched a detailed study to probe this hypothesis. Herein, we report on the dual catalytic activity of preformed [RuCl2(p-cymene)(NHC)] complexes toward the cyclopropanation and the metathesis of various olefinic substrates.
Results and discussion
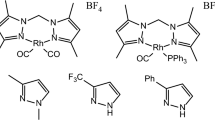
To begin our study, we investigated the catalytic activity of eight representative [RuCl2(p-cymene)(NHC)] complexes bearing imidazol-2-ylidene (2–8) or imidazolin-2-ylidene (9) ligands (Fig. 1) in the cyclopropanation of styrene. This terminal activated olefin is the most common substrate employed to assess new catalytic systems because it is much more reactive than other alkenes with electron-withdrawing substituents or with a high degree of alkyl substitution [22]. Thus, ethyl diazoacetate was slowly added to a solution containing styrene, and a ruthenium complex warmed at 60 °C under an inert atmosphere. The conversion of diazoester was monitored with a gas burette to measure the volume of nitrogen released. After 24 h, the reaction mixture was analyzed by gas chromatography (GC). Products formed included the desired ethyl 2-phenylcyclopropanecarboxylate (10) as a mixture of cis and trans diastereomers (Scheme 1). In most cases, these adducts were accompanied by significant amounts of homologation products, namely ethyl 4-phenylbut-3-enoate (11) and ethyl 3-phenylbut-2-enoate (12). Formally, these compounds result from a carbene insertion into a vinylic C-H bond of styrene, but the exact mechanism of this reaction, for which the intervention of ruthenacyclobutane intermediates was postulated, remains uncertain [23]. We also searched systematically for the peaks of cis- and trans-stilbene (13) in the chromatograms, as they were indicative of styrene metathesis. Other possible side-products, which were identified by GC–MS analyses in previous studies, include ethyl acrylate, ethyl cinnamate, phenylcyclopropane, and 1,2-diphenylcyclopropane [24]. Since they were formed only in trace amounts in the present work, their contribution was neglected. Last but not least, the formal carbene dimers, diethyl maleate, and diethyl fumarate (cis- and trans-14, respectively) accounted for the mass balance.

A preliminary test was carried out in the presence of the [RuCl2(p-cymene)]2 dimer (1). When ethyl diazoacetate was slowly added to a solution of styrene and of this simple catalyst precursor at 60 °C, ethyl 2-phenylcyclopropanecarboxylate (10) was obtained in a modest 58 % yield, and the cis/trans ratio was 0.56 (Table 1). The introduction of an NHC ancillary ligand onto the metal center had a beneficial influence on the reaction course as the cyclopropanation yield reached 78–85 % with a cis/trans ratio ranging from 0.5 to 0.9. Complex 2 bearing the least sterically demanding NHC ligand under examination led to the fastest conversion of the diazoester into active species (Fig. 2) and afforded the highest cis selectivity. The replacement of its N-methyl substituents with bulky and rigid mesityl groups in complex 8, on the other hand, led to the highest proportion of trans-isomers. All the ruthenium compounds of our screening promoted the insertion of the reactive carbene into the various Csp 2-H bonds of styrene in addition to its C=C double bond, thereby affording products 11 and 12 in 6–16 % yields. GC analysis also evidenced the formation of carbene dimers 14 in 4–14 % yields when the [RuCl2(p-cymene)(NHC)] complexes 2–9 served as catalyst precursors. In the presence of dimer 1, which was less efficient at promoting cyclopropanation, the formation of ethyl maleate and fumarate accounted for 35 % of the diazoester conversion. Conversely, styrene metathesis was essentially repressed under the experimental conditions adopted. Indeed, in most cases, it accounted for less than 1 % of the olefin consumption. Only with [RuCl2(p-cymene)(IMes)] (7) (IMes is 1,3-dimesitylimidazol-2-ylidene) was stilbene (mostly its trans-isomer) formed in small but detectable amount. This observation is in line with previous results from our group, which had highlighted the superior catalytic activity of complex 7 in the visible light-induced ROMP of cyclooctene promoted by [RuCl2(p-cymene)(NHC)] compounds 2–9 [17].
Next, we examined the reaction of ethyl diazoacetate with cyclooctene, a non-activated cycloolefin with respect to cyclopropanation. Only complexes 2–6 bearing NHC ligands with alkyl substituents on their nitrogen atoms served as catalyst precursors in these experiments (cf. Fig. 1). We reasoned that the use of [RuCl2(p-cymene)(NHC)] complexes 7–9 bearing the IMes ligand or its variants would favor olefin metathesis instead of cyclopropanation. The experimental procedure set up previously for styrene was adapted by increasing the alkene concentration to compensate for its low reactivity. Thus, the ruthenium complexes and the diazoester were diluted with neat cyclooctene, and no chlorobenzene was added. The amount of catalyst (0.5 mol % relative to the diazoester), the reaction time (24 h), and the temperature (60 °C) were kept unchanged. Under these conditions, ethyl bicyclo[6.1.0]nonane-9-carboxylate (15) was obtained as a mixture of endo- and exo-diastereomers (Scheme 2). Yields were, however, far from quantitative (Table 2), and the formation of ethyl fumarate and maleate (14) prevailed (up to 60 % based on ethyl diazoacetate). The cyclopropanation diastereoselectivities were also unexceptional with endo/exo ratios in the 0.4–0.5 range. Furthermore, the decomposition rate of the diazo compound was rather slow and remained independent of the substitution pattern on the NHC ligand (Fig. 3). Beside formal carbene dimers (14) and cyclopropanes (15), ethyl 2-(cyclooct-2-enyl)acetate (16) resulting from the insertion of the carbene into an allylic C-H bond of cyclooctene was also formed in low yield (<1 %). Competitive ROMP of the cycloolefin constituted a more important side-reaction and afforded polyoctenamer (17) in small but nonetheless significant yields (3–7 %).

To further expand the scope of the olefin cyclopropanation with ethyl diazoacetate catalyzed by [RuCl2(p-cymene)(NHC)] complexes, we probed other substrates than just styrene and cyclooctene. For this purpose, we elected catalyst precursor 2, which afforded the fastest conversion of ethyl diazoacetate into dinitrogen (cf. Fig. 2) and led to the highest cis-diastereoselectivity in the cyclopropanation of styrene (cf. Table 1). Among the various styrene derivatives investigated (Table 3), α-methylstyrene afforded the highest yield of the analog of 10 (93 %). It also gave the highest proportion of cis- vs. trans-isomer (49 %). Para-substitution of the aromatic ring of styrene with electron-withdrawing (chloro) or donating groups (methyl, tert-butyl, methoxy) did not affect the cyclopropanation yield, which remained close to 85 % in all cases, but influenced the cis/trans ratio, which fluctuated between 0.5 and 0.6, down from 0.9 with styrene. Two terminal linear alkenes (1-octene and 1-decene) were also cyclopropanated, albeit in modest yields (ca. 40 %). Internal olefins such as cyclohexene and cyclooctene were much less reactive, and cyclopropanation yields culminated at roughly 15–20 %. Except with the latter substrate, which afforded 7 % of polyoctenamer (17), only traces of metathesis products were detected in most of the reaction mixtures. In the case of α-methylstyrene and cyclohexene, they were completely absent. This is most likely due to the steric constraints encountered in the formation of a tetrasubstituted olefin from α-methylstyrene or to the adverse thermodynamics for the ring-opening of cyclohexene [25].
Unlike the other olefinic substrates investigated so far, norbornene was not cyclopropanated by ethyl diazoacetate in the presence of complex 2 (Table 3). Instead, only ROMP occurred, and the reaction mixture gelled after the addition of a few drops of diazoester. After 24 h, the solid mass was dissolved again in chloroform, and polynorbornene was precipitated in methanol. Size-exclusion chromatography indicated the formation of a high molecular weight polymer (M n = 140,000) with a broad dispersity (M w/M n = 4.75). This result prompted us to further investigate the reaction of norbornene employing ruthenium initiators 2–6 under experimental conditions more appropriate for the ROMP of this strained cycloolefin [26, 27]. Thus, trimethylsilyldiazomethane (TMSD) was elected as a carbene precursor, and the reactions were run in chlorobenzene using higher dilution conditions than for cyclopropanation in order to prevent instant jellification (Scheme 3). Using this revised catalytic system, moderate to high yields of polynorbornene (up to 81 %) were obtained after 5 h at 60 °C (Table 4). The highest conversion was reached with complex 5, which afforded a very high molecular weight polymer. Using NMR spectroscopy, we were able to determine the proportion of cis and trans double bonds within the unsaturated chains and to ascertain their block or random distribution by computing the σ cis and r cis r trans parameters, respectively [28, 29]. Whichever ligand was used, polynorbornenes that contained roughly two trans double bonds for one cis were obtained. The cis/trans distributions were not completely random, as indicated by values of the r cis r trans parameter greater than 1. However, the block arrangement of the polymers was rather limited and remained unaffected by the exact nature of the NHC ancillary ligand.

In conclusion, we have shown that ruthenium–arene complexes bearing N-heterocyclic carbene ligands were efficient catalyst precursors for the cyclopropanation of activated olefins with ethyl diazoacetate. Compared to the [RuCl2(p-cymene)]2 dimer (1), the [RuCl2(p-cymene)(NHC)] complexes 2–9 afforded higher yields of ethyl 2-phenylcyclopropanecarboxylate (10) in the cyclopropanation of styrene. The steric bulk of the NHC ligand also markedly influenced the cis/trans selectivity of the reaction. The procedure was successfully extended to α-methylstyrene and to various other styrenic derivatives bearing electron-withdrawing or donating substituents on their aromatic rings. The reaction of unactivated internal or terminal alkenes was more sluggish, and the use of norbornene as a substrate afforded only olefin metathesis. Further investigation of the ROMP of this strained cycloolefin in the presence of trimethylsilyldiazomethane showed that high molecular weight polynorbornene was formed and that the choice of the NHC ancillary ligand had little impact on its microstructure.
Experimental
All the syntheses were carried out under a dry inert atmosphere using standard Schlenk techniques. Solvents and olefins were distilled from appropriate drying agents and deoxygenated prior to use. The [RuCl2(p-cymene)]2 dimer (1) was purchased from Strem. The [RuCl2(p–cymene)(NHC)] complexes 2–9 were prepared according to literature procedures [17, 30–33]. All the other chemicals were purchased from Aldrich and used as received. 1H and 13C NMR spectra were recorded at 298 K on a Bruker DRX 400 spectrometer operating at 400.13 and 100.62 MHz, respectively. Gas chromatography was carried out on a Varian 3900 instrument equipped with a flame ionization detector and a WCOT fused silica column (stationary phase: CP-Sil 5CB, column length: 15 m, inside diameter: 0.25 mm, outside diameter: 0.39 mm, film thickness: 0.25 μm). Size-exclusion chromatography was performed in THF at 45 °C on a SFD S5200 autosampler liquid chromatograph equipped with a SFD 2000 refractive index detector and a battery of 4 PL gel columns fitted in series (particle size: 5 μm; pore sizes: 105, 104, 103, and 102 Å; flow rate: 1 cm3/min). The molecular weights (not corrected) are reported vs. monodisperse polystyrene standards used to calibrate the instrument.
Typical procedure for the reaction of styrene with ethyl diazoacetate
A 10 cm3 two-neck flask equipped with a magnetic stirring bar and capped with a three-way stopcock was charged with a ruthenium complex (0.005 mmol). The reactor was purged of air by applying three vacuum/nitrogen cycles before 1 cm3 dry chlorobenzene and 2 cm3 styrene were added. The solution was stirred for a few minutes in an oil bath thermostated at 60 °C. Ethyl diazoacetate (0.1141 g, 1 mmol) was diluted up to 1 cm3 with styrene in a 1 cm3 syringe. This diazoester solution was slowly added to the reaction mixture with a syringe pump over a 4-h period. Stirring was maintained for an additional 20 h at 60 °C. The rate of nitrogen evolution was monitored by a water column connected to the reaction flask via the three-way stopcock and a metallic cannula. After 24 h, the reaction mixture was analyzed by gas chromatography, and its composition was established by comparison with authentic samples.
Typical procedure for the ROMP of norbornene
A ruthenium complex (0.015 mmol) was placed in a 25 cm3 round-bottom flask equipped with a magnetic stirring bar and capped with a three-way stopcock. The reactor was purged of air by applying three vacuum/argon cycles before 5 cm3 dry chlorobenzene and a solution of 0.5 g norbornene (5.3 mmol) in 5 cm3 chlorobenzene were added with syringes. The reaction mixture was stirred for a few minutes in an oil bath thermostated at 60 °C before trimethylsilyldiazomethane (0.1 M in chlorobenzene, 0.10 mmol) was added via a syringe. It was kept at 60 °C for 5 h, cooled to room temperature, and diluted in chloroform. The resulting viscous solution was slowly poured in 600 cm3 methanol under vigorous stirring. The precipitated polymer was washed with methanol and dried overnight under high vacuum. It was analyzed by NMR spectroscopy and by size-exclusion chromatography.
References
Arduengo AJ III, Harlow RL, Kline M (1991) J Am Chem Soc 113:361
Arduengo AJ III (1999) Acc Chem Res 32:913
Nolan SP (ed) (2006) N-Heterocyclic carbenes in synthesis. Wiley-VCH, Weinheim
Glorius F (ed) (2007) N-Heterocyclic carbenes in transition metal catalysis. Topics in Organometallic Chemistry, vol 21. Springer, Berlin
Díez-González S (ed) (2010) N-Heterocyclic carbenes: from laboratory curiosities to efficient synthetic tools. RSC Catalysis Series, vol 6. Royal Society of Chemistry, Cambridge
Cazin CSJ (ed) (2011) N-Heterocyclic carbenes in transition metal catalysis and organocatalysis. Catalysis by Metal Complexes, vol 32. Springer, Dordrecht
Herrmann WA (2002) Angew Chem Int Ed 41:1290
Kantchev EAB, O’Brien CJ, Organ MG (2007) Angew Chem Int Ed 46:2768
Díez-González S, Marion N, Nolan SP (2009) Chem Rev 109:3612
Weskamp T, Kohl FJ, Hieringer W, Gleich D, Herrmann WA (1999) Angew Chem Int Ed 38:2416
Huang J, Stevens ED, Nolan SP, Petersen JL (1999) J Am Chem Soc 121:2674
Scholl M, Trnka TM, Morgan JP, Grubbs RH (1999) Tetrahedron Lett 40:2247
Deshmukh PH, Blechert S (2007) Dalton Trans 2479
Samojłowicz C, Bieniek M, Grela K (2009) Chem Rev 109:3708
Vougioukalakis GC, Grubbs RH (2010) Chem Rev 110:1746
Hamad FB, Sun T, Xiao S, Verpoort F (2013) Coord Chem Rev 257:2274
Delaude L, Demonceau A, Noels AF (2001) Chem Commun 986
Delaude L, Szypa M, Demonceau A, Noels AF (2002) Adv Synth Catal 344:749
Delaude L, Demonceau A, Noels AF (2006) Curr Org Chem 10:203
Delaude L, Demonceau A (2012) Dalton Trans 41:9257
Noels AF, Demonceau A (1998) J Phys Org Chem 11:602
Maas G (2004) Chem Soc Rev 33:183
Simal F, Demonceau A, Noels AF, Knowles DRT, O’Leary S, Maitlis PM, Gusev O (1998) J Organomet Chem 558:163
Noels AF, Demonceau A, Carlier E, Hubert AJ, Márquez-Silva R-L, Sánchez-Delgado RA (1988) Chem Commun 783
Patton PA, Lillya CP, McCarthy TJ (1986) Macromolecules 19:1266
Stumpf AW, Saive E, Demonceau A, Noels AF (1995) Chem Commun 1127
Demonceau A, Stumpf AW, Saive E, Noels AF (1997) Macromolecules 30:3127
Ivin KJ, Laverty DT, Rooney JJ (1977) Makromol Chem 178:1545
Ivin KJ, Laverty DT, Rooney JJ (1978) Makromol Chem 179:253
Herrmann WA, Elison M, Fischer J, Köcher C, Artus GRJ (1996) Chem Eur J 2:772
Herrmann WA, Köcher C, Goossen LJ, Artus GRJ (1996) Chem Eur J 2:1627
Jafarpour L, Huang J, Stevens ED, Nolan SP (1999) Organometallics 18:3760
Lo C, Cariou R, Fischmeister C, Dixneuf PH (2007) Adv Synth Catal 349:546
Acknowledgments
The financial support of the “Fonds de la Recherche Scientifique-FNRS,” Brussels, is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the lifework of Professor Franz Stelzer and his achievements in olefin metathesis and polymer chemistry.
Rights and permissions
About this article
Cite this article
Méret, M., Maj, A.M., Demonceau, A. et al. Ruthenium–arene catalysts bearing N-heterocyclic carbene ligands for olefin cyclopropanation and metathesis. Monatsh Chem 146, 1099–1105 (2015). https://doi.org/10.1007/s00706-015-1492-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-015-1492-x