Abstract
Reactions of (N-isocyanimino)triphenylphosphorane with acenaphthoquinone in the presence of aromatic carboxylic acids proceed smoothly at room temperature and under neutral conditions to afford sterically congested 1,3,4-oxadiazole derivatives in high yields. The reaction proceeds smoothly and cleanly under mild conditions and no side reactions are observed.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multicomponent reactions are capable of achieving high levels of brevity and diversity, as they allow more than two simple and flexible building blocks to be combined in practical, time-saving one-pot operations. Owing to their valued features such as atom economy, inherent simple experimental procedures, and their one-pot character, they are perfectly suited for automated synthesis. Thus, MCRs have attracted considerable interest owing to their exceptional synthetic efficiency [1–4]. Typically, the purification of products resulting from MCRs is also simple because all the organic reagents employed are consumed and incorporated into the target compound [5, 6]. MCRs leading to interesting heterocyclic scaffolds are particularly useful for the construction of diverse chemical libraries of drug-like molecules. The isocyanide-based MCRs are of special importance in this context [7–9].
The intramolecular version of the aza-Wittig-type reaction has attracted considerable attention recently because of its high potential for the synthesis of a wide variety of nitrogen heterocycles, which can be attributed, in good measure, to the rapid progress in the preparation of functionalized iminophosphoranes. Several interesting heterocyclization reactions involving iminophosphoranes have been reported [10–16]. These compounds can easily be converted through aza-Wittig reaction with isocyanates, carbon dioxide, or carbon disulfide into functionalized hetero-cumulenes which exhibit a rich chemistry of unusual synthetic promise [10–16]. The nucleophilicity at the nitrogen is a factor of essential mechanistic importance in the use of these iminophosphoranes as aza-Wittig reagents. Iminophosphoranes are important reagents in synthetic organic chemistry, especially in the synthesis of naturally occurring products, compounds with biological and pharmacological activity [10–16]. (N-Isocyanimino)triphenylphosphorane (3) is expected to have synthetic potential because it provides a reaction system in which the iminophosphorane group can react with a reagent having a carbonyl functionality [17, 18].
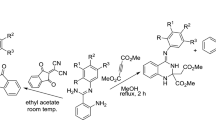
In recent years, several synthetic methods have been reported for the preparation of (N-isocyanimino)triphenylphosphorane (CNNPPh3, 3) (Scheme 1) [17, 18]. There are several reports on the use of (N-isocyanimino)triphenylphosphorane in the synthesis of metal complexes [17, 18]. However, application of 3 in the synthesis of organic compounds is rare [19–29]. As part of our ongoing program to develop efficient and robust methods for the preparation of heterocyclic compounds [19–33], we sought to develop a convenient preparation of sterically congested 1,3,4-oxadiazole derivatives 4a–4p. Herein, we report a hitherto unknown, one-pot three-component reaction, which, starting from readily available acenaphthoquinone (2), affords disubstituted 1,3,4-oxadiazole derivatives 4a–4p (Scheme 1).
1,3,4-Oxadiazoles have attracted interest in medicinal chemistry as surrogates of carboxylic acids, esters, and carboxamides. 1,3,4-Oxadiazoles are an important class of heterocyclic compounds that have a wide range of pharmaceutical and biological activities including antimicrobial, antifungal, anti-inflammatory, and antihypertensive [34–38]. Several methods have been reported in the literature for the synthesis of 1,3,4-oxadiazoles. These protocols are multi-step in nature [39–45]. The most general method involves the cyclization of diacylhydrazides with a variety of reagents, such as SOCl2, POCl3, or H2SO4, usually under harsh reaction conditions. Few reliable and operationally simple examples have been reported for the one-pot synthesis of 1,3,4-oxadiazoles, especially from readily available carboxylic acids and acid hydrazides [46–50].
Results and discussion
The carboxylic acid derivatives 1 with acenaphthoquinone (2) and (N-isocyanimino)triphenylphosphorane (3) in CH3CN react together in a 1:1:1 ratio at room temperature to produce sterically congested 1,3,4-oxadiazole derivatives 4 and triphenylphosphine oxide (5) (Scheme 1, Table 1). The reaction proceeds smoothly and cleanly under mild conditions and no side reactions are observed.
The suggested mechanism for the formation of products 4a–4p is illustrated in Scheme 2. On the basis of the chemistry of isocyanides, it is reasonable to assume that the first step may involve nucleophilic addition of (N-isocyanimino)triphenylphosphorane (3) to acenaphthoquinone (2), which is facilitated by its protonation with the acid 1, leading to nitrilium intermediate 6. This intermediate may be attacked by the conjugate base of acid 1 to form 1:1:1 adduct 7. This adduct may undergo intramolecular aza-Wittig reaction of the iminophosphorane moiety with the ester carbonyl to afford the isolated sterically congested 1,3,4-oxadiazole derivatives 4 by elimination of triphenylphosphine oxide (5) from intermediate 8.
In summary, we believe that the reported method offers a mild, simple, and efficient route for the preparation of 1,3,4-oxadiazole derivatives 4. Its ease of work-up, high yields, and fairly mild reaction conditions make it a useful addition to modern synthetic methodologies. Other aspects of this process are under investigation.
Experimental
(N-Isocyanimino)triphenylphosphorane (3) was prepared based on reported procedures [17, 18]. Other starting materials and solvents were obtained from Merck (Germany) and Fluka (Switzerland) and were used without further purification. The methods used to monitor the reactions are TLC and NMR. Melting points were measured on an Electrothermal 9100 apparatus. IR spectra were measured on a Jasco 6300 FTIR spectrometer. 1H and 13C NMR spectra (CDCl3) were recorded on a Bruker DRX-250 Avance spectrometer at 250.0 and 62.5 MHz, respectively. Elemental analyses were performed using a Heraeus CHN-O-Rapid analyzer. Mass spectra were recorded on a Finnigan MAT-8430 mass spectrometer operating at an ionization potential of 70 eV. Preparative layer chromatography (PLC) plates were prepared from Merck silica gel (F254) powder.
General procedure for the preparation of compounds 4a–4p
A mixture of (N-isocyanimino)triphenylphosphorane (1.0 mmol), acenaphthoquinone (1.0 mmol), and aromatic carboxylic acid (1.0 mmol) in 5 cm3 CH3CN was stirred at room temperature for 24 h. The solvent was removed under reduced pressure, and the viscous residue was purified by PLC [silica gel (F254) powder; petroleum ether/ethyl acetate 4:1].
2-Hydroxy-2-(5-phenyl-1,3,4-oxadiazol-2-yl)-1(2H)-acenaphthylenone (4a, C20H12N2O3)
Yellow powder; yield 80%; R f = 0.36 (petroleum ether/ethyl acetate 4:1); m.p.: 162–164 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.59 (br s, OH), 7.40–8.30 (m, 11CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 87.74 (C–OH), 122.56, 123.88, 126.97, 127.13, 128.79, 128.96, 129.07, 132.02, 132.85 (11CH of arom), 123.22, 127.80, 129.60, 135.12, 142.96 (5C of arom), 161.05, 166.13 (2C=N of oxadiazole), 198.51 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,366, 3,073, 1,718, 1,603, 1,448, 1,012 cm−1; MS (EI, 20 eV): m/z (%) = 328 (M+), 198 (16), 182 (34), 154 (83), 126 (100), 98 (28), 85 (28), 76 (39), 62 (38), 43 (45).
2-[5-(4-Bromophenyl)-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4b, C20H11BrN2O3)
Yellow powder; yield 78%; R f = 0.33 (petroleum ether/ethyl acetate 4:1); m.p.: 151–153 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.57 (br s, OH), 7.57 (d, 3 J HH = 8.3 Hz, 2CHarom), 8.03 (d, 3 J HH = 8.3 Hz, 2CHarom), 7.69–7.86, 8.09–8.30 (m, 6CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 86.50 (C–OH), 122.59, 123.96, 127.03, 128.51, 128.83, 129.08, 132.33, 132.94 (10CH of arom), 122.12, 126.80, 129.50, 130.98, 134.22, 140.25 (6C of arom), 164.02, 165.03 (2C=N of oxadiazole), 202.02 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,204, 3,077, 1,733, 1,600, 1,481, 1,007 cm−1; MS (EI, 20 eV): m/z (%) = 407 (M+), 198 (8), 182 (37), 154 (78), 126 (100), 98 (22), 85 (22), 75 (55), 62 (45), 50 (18).
4-[5-(1,2-Dihydro-1-hydroxy-2-oxo-1-acenaphthylenyl)-1,3,4-oxadiazol-2-yl]benzonitrile (4c, C21H11N3O3)
Yellow powder; yield 73%; R f = 0.30 (petroleum ether/ethyl acetate 4:1); m.p.: 161–163 °C; 1H NMR (250.13 MHz, CDCl3): δ = 3.63 (s, OH), 7.71–8.33 (m, 10CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 77.31 (C–OH), 122.07, 127.13, 127.60, 128.45, 129.09, 132.62, 132.75 (10CH of arom), 109.98, 115.51, 124.00, 128.90, 133.02, 135.05, 143.03 (7C of arom), 161.03, 165.13 (2C=N of oxadiazole), 192.21 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,439, 3,209, 1,729, 1,604, 1,495, 1,014 cm−1; MS (EI, 20 eV): m/z (%) = 353 (M+), 197 (17), 182 (32), 154 (73), 130 (100), 126 (85), 101 (90), 75 (86), 62 (47), 50 (48), 42 (30).
2-Hydroxy-2-[5-(3-methylphenyl)-1,3,4-oxadiazol-2-yl]-1(2H)-acenaphthylenone (4d, C21H14N2O3)
Yellow powder; yield 83%; R f = 0.36 (petroleum ether/ethyl acetate 4:1); m.p.: 169–171 °C; 1H NMR (250.13 MHz, CDCl3): δ = 2.37 (s, CH3), 4.50 (br s, OH), 7.26–8.30 (m, 10CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 21.21 (CH3), 76.57 (C–OH), 122.06, 122.55, 124.29, 126.84, 127.58, 128.44, 128.78, 129.07, 132.60, 132.86 (10CH of arom),123.82, 128.81, 129.10, 130.91, 134.80, 143.35 (6C of arom), 164.18, 166.75 (2C=N of oxadiazole), 198.87 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,399, 3,073, 1,723, 1,603, 1,490, 1,073 cm−1; MS (EI, 20 eV): m/z (%) = 342 (M+), 198 (6), 182 (24), 154 (54), 126 (60), 105 (100), 91 (18), 76 (54), 62 (23), 50 (24).
2-[5-(4-tert-Butylphenyl)-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4e, C24H20N2O3)
Yellow powder; yield 85%; R f = 0.33 (petroleum ether/ethyl acetate 4:1); m.p.: 176–178 °C; 1H NMR (250.13 MHz, CDCl3): δ = 1.30 (s, 3CH3), 5.30 (br s, OH), 7.41 (d, 3 J HH = 8.3 Hz, 2CHarom), 7.98 (d, 3 J HH = 8.3 Hz, 2CHarom), 7.71–7.86, 8.05–8.20 (m, 6CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 31.03 (3CH3), 35.03 (C), 76.60 (C–OH), 122.53, 123.74, 125.89, 126.79, 126.98, 128.70, 129.03, 132.73 (10CH of arom), 122.04, 128.43, 129.75, 130.88, 135.51, 142.33 (6C of arom), 164.28, 166.00 (2C=N of oxadiazole), 198.17 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,209, 2,965, 1,738, 1,614, 1,495, 1,010 cm−1; MS (EI, 20 eV): m/z (%) = 384 (M+), 182 (11), 163 (25), 148 (14), 120 (38), 103 (17), 91 (73), 76 (35), 57 (18), 43 (100).
2-[5-(3-Chlorophenyl)-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4f, C20H11ClN2O3)
Yellow powder; yield 76%; R f = 0.30 (petroleum ether/ethyl acetate 4:1); m.p.: 174–176 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.06 (br s, OH), 7.33–8.29 (m, 10CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 77.25 (C–OH), 122.06, 123.98, 125.24, 127.02, 128.44, 129.09, 130.34, 132.09, 132.62, 132.94 (10CH of arom), 122.59, 128.83, 129.50, 133.82, 135.12, 148.62 (6C of arom), 158.50, 164.82 (2C=N of oxadiazole), 198.27 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,379, 3,080, 1,718, 1,602, 1,551, 1,068 cm−1; MS (EI, 20 eV): m/z (%) = 362 (M+), 197 (18), 182 (44), 154 (95), 126 (100), 98 (23), 85 (19), 75 (47), 62 (34), 50 (17).
2-Hydroxy-2-[5-(3-phenoxyphenyl)-1,3,4-oxadiazol-2-yl]-1(2H)-acenaphthylenone (4g, C26H16N2O4)
Yellow powder; yield 72%; R f = 0.26 (petroleum ether/ethyl acetate 4:1); m.p.: 153–155 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.81 (br s, OH), 6.96–8.29 (m, 15CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 85.02 (C–OH), 117.19, 119.14, 121.80, 122.05, 123.88, 123.97, 126.95, 128.77, 129.04, 129.95, 130.45, 130.92, 132.84 (15CH of arom), 122.52, 128.44, 129.62, 135.22, 145.30, 156.20, 157.85 (7C of arom), 165.45, 164.71 (2C=N of oxadiazole), 198.17 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,307, 3,082, 1,743, 1,563, 1,485, 1,225 cm−1; MS (EI, 20 eV): m/z (%) = 420 (M+), 214 (27), 197 (29), 182 (80), 154 (80), 126 (100), 98 (21), 85 (15), 76 (29), 62 (29), 50 (22).
2-[5-[4-(Bromomethyl)phenyl]-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4h, C21H13BrN2O3)
Yellow powder; yield 82%; R f = 0.33 (petroleum ether/ethyl acetate 4:1); m.p.: 163–165 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.46 (s, CH2), 4.98 (br s, OH), 7.43 (d, 3 J HH = 8.3 Hz, 2CHarom), 7.90 (d, 3 J HH = 8.3 Hz, 2CHarom), 7.70–7.86, 7.99–8.29 (m, 6CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 32.09 (CH2), 76.77 (C–OH), 122.56, 123.89, 126.95, 127.55, 129.07, 129.61, 132.62, 132.87 (10CH of arom), 123.10, 128.44, 128.79, 130.92, 135.24, 141.80 (6C of arom), 165.12, 166.22 (2C=N of oxadiazole), 198.15 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,343, 3,072, 1,717, 1,603, 1,486, 1,012 cm−1.
2-Hydroxy-2-[5-(1-naphthalenyl)-1,3,4-oxadiazol-2-yl]-1(2H)-acenaphthylenone (4i, C24H14N2O3)
Yellow powder; yield 75%; R f = 0.36 (petroleum ether/ethyl acetate 4:1); m.p.: 154–156 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.84 (br s, OH), 7.42–9.07 (m, 13CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 76.39 (C–OH), 122.60, 124.67, 125.95, 126.71, 126.96, 128.22, 128.59, 128.75, 128.79, 129.08, 132.88 (13CH of arom), 123.91, 128.35, 129.90, 131.92, 133.80, 135.22, 142.26 (7C of arom), 162.32, 166.25 (2C=N of oxadiazole), 198.17 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,433, 3,186, 1,741, 1,604, 1,538, 1,089 cm−1.
2-[5-(4-Chlorophenyl)-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4j, C20H11ClN2O3)
Yellow powder; yield 72%; R f = 0.30 (petroleum ether/ethyl acetate 4:1); m.p.: 169–171 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.58 (br s, OH), 7.77 (d, 3 J HH = 8.3 Hz, 2CHarom), 8.24 (d, 3 J HH = 8.3 Hz, 2CHarom), 7.40–7.73, 7.83–8.13 (m, 6CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 83.23 (C–OH), 122.07, 122.58, 128.40, 128.45, 129.09, 129.37, 132.63, 132.94 (10CH of arom), 123.95, 127.02, 128.83, 135.02, 137.52, 138.45 (6C of arom), 164.12, 165.23 (2C=N of oxadiazole), 202.12 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,227, 3,022, 1,732, 1,604, 1,486, 1,078 cm−1.
2-[5-(4-Fluorophenyl)-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4k, C20H11FN2O3)
Yellow powder; yield 75%; R f = 0.33 (petroleum ether/ethyl acetate 4:1); m.p.: 156–158 °C; 1H NMR (250.13 MHz, CDCl3): δ = 4.40 (s, OH), 7.10–8.31 (m, 10CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 82.65 (C–OH), 116.38 (d, 2 J CF = 20.1 Hz, 2CH of arom), 122.06, 122.55, 128.44, 129.09, 132.62 (6CH of arom), 129.53 (d, 3 J CF = 12.5 Hz, 2CH of arom), 123.93, 127.00, 128.94, 135.02, 140.74 (5C of arom), 160.52 (d, 1 J CF = 252.0 Hz, C of arom), 164.48, 166.53 (2C=N of oxadiazole), 189.51 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,375, 3,082, 1,725, 1,606, 1,497, 1,013 cm−1.
2-Hydroxy-2-[5-(4-methylphenyl)-1,3,4-oxadiazol-2-yl]-1(2H)-acenaphthylenone (4l, C21H14N2O3)
Yellow powder; yield 80%; R f = 0.36 (petroleum ether/ethyl acetate 4:1); m.p.: 170–172 °C; 1H NMR (250.13 MHz, CDCl3): δ = 2.37 (s, CH3), 4.30 (br s, OH), 7.20–8.29 (m, 10CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 21.61 (CH3), 76.64 (C–OH), 122.05, 123.81, 127.07, 128.44, 129.05, 129.64, 132.61, 132.80 (10CH of arom), 122.52, 128.60, 128.75, 126.89, 135.22, 142.85 (6C of arom), 162.08, 164.13 (2C=N of oxadiazole), 198.10 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,355, 3,068, 1,725, 1,603, 1,498, 1,012 cm−1.
2-Hydroxy-2-[5-(4-methoxyphenyl)-1,3,4-oxadiazol-2-yl]-1(2H)-acenaphthylenone (4m, C21H14N2O4)
Yellow powder; yield 68%; R f = 0.26 (petroleum ether/ethyl acetate 4:1); m.p.: 195–197 °C; 1H NMR (250.13 MHz, CDCl3): δ = 3.84 (s, CH3), 4.33 (s, OH), 6.85–8.31 (m, 10CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 55.55 (OCH3), 77.55 (C–OH), 114.40, 122.54, 123.92, 126.92, 128.93, 128.95, 132.50, 132.89 (10CH of arom), 122.54, 128.86, 131.04, 135.22, 140.04, 158.82 (6C of arom), 164.08, 166.53 (2C=N of oxadiazole), 194.51 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,244, 3,083, 1,739, 1,614, 1,500, 1,185 cm−1.
2-[5-(3,5-Dimethoxyphenyl)-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4n, C22H16N2O5)
Yellow powder; yield 67%; R f = 0.20 (petroleum ether/ethyl acetate 4:1); m.p.: 168–170 °C; 1H NMR (250.13 MHz, CDCl3): δ = 3.80 (s, 2OCH3), 4.43 (s, OH), 6.56–8.30 (m, 9CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 55.64 (2OCH3), 87.00 (C–OH), 105.00, 107.05, 122.04, 123.94, 128.43, 132.59, 132.84 (9CH of arom), 124.75, 127.00, 131.04, 135.03, 142.10, 162.15 (7C of arom), 164.02, 166.43 (2C=N of oxadiazole), 189.51 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,380, 3,094, 1,725, 1,604, 1,463, 1,162 cm−1.
2-Hydroxy-2-[5-(2-methylphenyl)-1,3,4-oxadiazol-2-yl]-1(2H)-acenaphthylenone (4o, C21H14N2O3)
Yellow powder; yield 72%; R f = 0.34 (petroleum ether/ethyl acetate 4:1); m.p.: 154–156 °C; 1H NMR (250.13 MHz, CDCl3): δ = 2.57 (s, CH3), 4.09 (br s, OH), 7.06–8.64 (m, 10CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 22.32 (CH3), 77.70 (C–OH), 122.06, 126.06, 126.98, 128.44, 129.09, 129.13, 131.50, 132.61, 132.80 (10CH of arom), 123.82, 128.60, 131.65, 132.00, 138.62, 140.35 (6C of arom), 164.18, 166.73 (2C=N of oxadiazole), 198.03 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,416, 3,072, 1,725, 1,604, 1,491, 1,014 cm−1.
2-[5-(3,4-Dimethylphenyl)-1,3,4-oxadiazol-2-yl]-2-hydroxy-1(2H)-acenaphthylenone (4p, C22H16N2O3)
Yellow powder; yield 70%; R f = 0.32 (petroleum ether/ethyl acetate 4:1); m.p.: 167–169 °C; 1H NMR (250.13 MHz, CDCl3): δ = 2.26, 2.28 (s, 2CH3), 4.42 (br s, OH), 7.07–8.75 (m, 9CHarom) ppm; 13C NMR (62.53 MHz, CDCl3): δ = 19.65, 19.93 (2CH3), 80.00 (C–OH), 122.05, 122.54, 126.89, 128.43, 128.59, 128.85, 130.16, 132.61, 132.75 (9CH of arom), 122.05, 123.92, 127.50, 128.05, 129.02, 135.32, 145.90 (7C of arom), 164.18, 166.33 (2C=N of oxadiazole), 188.07 (C=O) ppm; IR (KBr): \( \bar{\nu } \) = 3,376, 3,083, 1,721, 1,603, 1,494, 1,184 cm−1.
References
Sapi J, Laronze J-Y (2004) Arkivoc (vii):208
Zhu J, Bienayme H (2005) Multicomponent reactions. Wiley-VCH, Weinheim
Hazeri N, Maghsoodlou MT, Habibi-Khorassani SM, Ziyaadini M, Marandi G, Khandan-Barani K, Bijanzadeh HR (2007) Arkivoc (viii):34
Dömling A, Beck B, Herdtweck E, Antuch W, Oefner C, Yehia N, Gracia-Marques A (2007) Arkivoc (vxii):99
Ramon DJ, Yus M (2005) Angew Chem Int Ed 44:1602
Basso A, Banfi L, Riva R, Guanti G (2005) J Org Chem 70:575
Dömling A, Ugi I (2000) Angew Chem Int Ed 39:3168
Dömling A (2006) Chem Rev 106:17
Bayat M, Imanieh H, Zabarjad Shiraz N, Shah Qavidel M (2010) Monatsh Chem 141:333
Molina P, Vilaplana M (1994) Synthesis 1197
Palacios F, Aparicio D, Rubiales G, Alonso C, de los Santos JM (2009) Curr Org Chem 13:808
Palacios F, Aparicio D, Rubiales G, Alonso C, de los Santos JM (2006) Curr Org Chem 10:2371
Hajós G, Nagy I (2008) Curr Org Chem 12:39
Palacios F, Alonso C, Aparicio D, Rubiales G, de los Santos JM (2007) Tetrahedron 63:523
Cossio FP, Alonso C, Lecea B, Ayerbe M, Rubiales G, Palacios F (2006) J Org Chem 71:2839
Palacios F, Herrán E, Alonso C, Rubiales G, Lecea B, Ayerbe M, Cossío FP (2006) J Org Chem 71:6020
Stolzenberg H, Weinberger B, Fehlhammer WP, Pühlhofer FG, Weiss R (2005) Eur J Inorg Chem 21:4263
Chiu TW, Liu YH, Chi KM, Wen YS, Lu KL (2005) Inorg Chem 44:6425
Souldozi A, Ramazani A, Bouslimani N, Welter R (2007) Tetrahedron Lett 48:2617
Souldozi A, Ramazani A (2007) Tetrahedron Lett 48:1549
Souldozi A, Ramazani A (2009) Phosphorus Sulfur Silicon Relat Elem 184:3191
Souldozi A, Ramazani A (2009) Phosphorus Sulfur Silicon Relat Elem 184:2344
Souldozi A, Ramazani A (2008) Arkivoc (xvi):235
Ramazani A, Salmanpour S, Souldozi A (2010) Phosphorus Sulfur Silicon Relat Elem 185:97
Souldozi A, Ślepokura K, Lis T, Ramazani A (2007) Z Naturforsch 62b:835
Ramazani A, Morsali A, Ganjeie B, Kazemizadeh AR, Ahmadi E, Kempe R, Hertle I (2005) Z Naturforsch 60b:569
Ramazani A, Rezaei A (2010) Org Lett 12:2852
Ramazani A, Ahmadi Y, Rouhani M, Shajari N, Souldozi A (2010) Heteroat Chem 21:368
Ramazani A, Shajari N, Mahyari A, Ahmadi Y (2010) Mol Divers 14. doi:10.1007/s11030-010-9275-0
Ramazani A, Rezaei A, Mahyari TA, Rouhani M, Khoobi M (2010) Helv Chim Acta 93:2033
Mahyari TA, Shajari N, Kazemizadeh AR, Ślepokura K, Lis T, Ramazani A (2007) Z Naturforsch 62b:829
Adib M, Riazati Kesheh M, Ansari S, Bijanzadeh HR (2009) Synlett 1575
Ramazani A, Mahyari TA, Rouhani M, Rezaei A (2009) Tetrahedron Lett 50:5625
Tully WR, Gardner CR, Gillespie RJ, Westwood R (1991) J Med Chem 34:2060
Chen C, Senanayake CH, Bill TJ, Larsen RD, Verhoeven TR, Reider PJ (1994) J Org Chem 59:3738
Holla BS, Gonsalves R, Shenoy S (2000) Eur J Med Chem 35:267
Crimmin MJ, O’Hanlon PJ, Rogers NH, Walker G (1989) J Chem Soc Perkin Trans 1 2047
Laddi UV, Desai SR, Bennur RS, Bennur SC (2002) Ind J Heterocycl Chem 11:319
Baxendale IR, Ley SV, Martinelli M (2005) Tetrahedron 61:5323
Liras S, Allen MP, Segelstein BE (2000) Synth Commun 30:437
Brown BJ, Clemens IR, Neesom JK (2000) Synlett 131
Coppo FT, Evans KA, Graybill TL, Burton G (2004) Tetrahedron Lett 45:3257
Brain CT, Paul JM, Loong Y, Oakley PJ (1999) Tetrahedron Lett 40:3275
Brain CT, Brunton SA (2001) Synlett 382
El-Sayed WA, El-Essawy FA, Ali OM, Nasr BS, Abdalla MM, Abdel-Rahman AAH (2010) Monatsh Chem 141:1021
Tandon VK, Chhor RB (2001) Synth Commun 31:1727
Mashraqui SH, Ghadigaonkar SG, Kenny RS (2003) Synth Commun 33:2541
Bentiss F, Lagrenee M, Barbry D (2001) Synth Commun 31:935
Jedlovska E, Lesko J (1994) Synth Commun 24:1879
Wang Y, Sauer DR, Djuric SW (2006) Tetrahedron Lett 47:105
Acknowledgments
The authors are thankful to the Zanjan University for partial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramazani, A., Zeinali Nasrabadi, F., Mashhadi Malekzadeh, A. et al. Synthesis of sterically congested 1,3,4-oxadiazole derivatives from aromatic carboxylic acids, acenaphthoquinone, and (N-isocyanimino)triphenylphosphorane. Monatsh Chem 142, 625–630 (2011). https://doi.org/10.1007/s00706-011-0488-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0488-4