Summary.
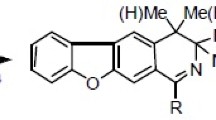
Isoquinolin-1-ylmethylfuranones could be readily prepared from certain Reissert-compounds and isopilopyl bromide by standard procedures. Their reduction to the desired not isolated 1,2,3,4-tetrahydroisoquinoline intermediates was achieved by chemical and/or catalytical hydrogenation, followed by intramolecular amidation conveniently giving the title compounds in a one-pot synthesis. The structures and the stereochemistry of the target compounds were assigned by 13C NMR spectroscopy and X-ray diffraction analysis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A Itoh Y Ikuta T Tanahashi N Nagakura (2000) J Nat Prod 63 725
T Fujii M Ohba A Yonezawa J Sakaguchi (1987) Chem Pharm Bull 35 3470 Occurrence Handle1:CAS:528:DyaL1cXlsV2rsbk%3D
H Takayama M Arai M Kitajima N Aimi (2002) Chem Pharm Bull 50 1141 Occurrence Handle1:CAS:528:DC%2BD38Xmt1Sjtr0%3D Occurrence Handle10.1248/cpb.50.1141
Fujii T, Ohba M, Suzuki H (1985) Chem Pharm Bull 33: 1023; see also: Fujii T, Ohba M, Yoshifuji S (1988) Heterocycles 27: 1009
M Shamma (1972) The Isoquinoline Alkaloids – Chemistry and Pharmacology Academic Press Verlag Chemie GmbH 452
Tridgett R, Clarke I, Turtle R, Johnston G (2005) Brit UK Pat Appl 59; CA (2005) 143: 211845
JC Menéndez MP Díaz C Bellver MM Söllhuber (1992) Eur J Med Chem 27 61 Occurrence Handle10.1016/0223-5234(92)90062-6
Bentley KW (1998) The Isoquinoline Alkaloids. In: Ravindranath B (ed) Chemistry and Biochemistry of Organic Natural Products Vol 1. Harwood Academic Publishers
T Fujii M Ohba K Yoneyama H Kizu S Yoshifuji (1986) Chem Pharm Bull 34 669 Occurrence Handle1:CAS:528:DyaL2sXksVSit7g%3D
T Fujii M Ohba S Akiyama (1985) Chem Pharm Bull 33 5316 Occurrence Handle1:CAS:528:DyaL2sXhvFKlt7w%3D
E Van der Eycken G Deroover SM Toppet GJ Hoornaert (1999) Tetrahedron Letters 40 9147 Occurrence Handle1:CAS:528:DyaK1MXnvF2juro%3D Occurrence Handle10.1016/S0040-4039(99)01943-7
YS Lee SS Kang JH Choi H Park (1997) Tetrahedron 53 3045 Occurrence Handle1:CAS:528:DyaK2sXhs1Ggtbg%3D Occurrence Handle10.1016/S0040-4020(97)00070-7
LF Tietze N Rackelmann I Müller (2004) Chem Eur J 10 2722 Occurrence Handle1:CAS:528:DC%2BD2cXkvFGhtb4%3D Occurrence Handle10.1002/chem.200306039
J Kóbor P Sohár F Fülöp (1994) Tetrahedron 50 4873 Occurrence Handle10.1016/S0040-4020(01)85022-5
O Bassas N Llor MMM Santos R Griera E Molins M Amat J Bosch (2005) Org Lett 7 2817 Occurrence Handle1:CAS:528:DC%2BD2MXkvVGktrY%3D Occurrence Handle10.1021/ol0505609
last communication: Reimann E, Grasberger F (2005) Monatsh Chem 136: 193
E Reimann M Renz H Unger (2002) Monatsh Chem 133 1285 Occurrence Handle1:CAS:528:DC%2BD38Xns1Cmsr0%3D
T Bando S Tanaka K Fugami Y Zen-ichi Y Tamaru (1992) Bull Chem Soc Jp 65 97 Occurrence Handle1:CAS:528:DyaK38Xhsleqs7s%3D Occurrence Handle10.1246/bcsj.65.97
E Reimann A Höglmüller (1985) Arch Pharm 318 559 Occurrence Handle1:CAS:528:DyaL2MXlt1egsb8%3D Occurrence Handle10.1002/ardp.19853180615
A Padwa AG Waterson (2000) Tetrahedron 56 10159 Occurrence Handle1:CAS:528:DC%2BD3MXmtVGjsA%3D%3D Occurrence Handle10.1016/S0040-4020(00)00861-9
E Reimann F Grasberger K Polborn (2003) Monatsh Chem 134 991 Occurrence Handle1:CAS:528:DC%2BD3sXksV2iu7Y%3D Occurrence Handle10.1007/s00706-003-0013-5
NA Preobrazhenskii AM Polyakova WA Preobrazhenskii (1934) Ber Dtsch Chem Ges 67 710
Sheldrick GM (1990/1993), Programs for the Solution and Refinement of Crystal Structures, Göttingen
Author information
Authors and Affiliations
Corresponding author
Additional information
The second author is part of PhD thesis, LMU München, Germany
Rights and permissions
About this article
Cite this article
Reimann, E., Renz, M. & Polborn, K. Synthesis of cis-2,3-Disubstituted Benzo[a]quinolizidin-4-ones via Reissert-Compounds. Monatsh. Chem. 137, 1411–1419 (2006). https://doi.org/10.1007/s00706-006-0540-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-006-0540-y