Abstract
HIV-1 transmitted drug resistance (TDR) mutations may reduce the efficacy of antiretroviral therapy (ART), but pre-treatment testing to determine the virus genotype can improve the efficacy of ART. Unfortunately, issues related to cost and logistics of pre-treatment testing limit its use in resource-limited settings. We studied 596 ART-naive individuals who were newly diagnosed from 2014 to 2016 in São Paulo, Brazil, to evaluate TDR and virological outcome after 48 weeks of genotype-guided therapy. One or more TDR (based on the WHO surveillance list) was observed in 10.9% (CI 95%, 8.6–13.6) of the sequences, the most common of which was the K103 N mutation, which confers resistance to first-generation drugs of the non-nucleoside reverse transcriptase inhibitor (NNRTI) antiretroviral drug class. Dual-class (1%, 6/596) and triple-class (0.34%, 2/596) resistance were uncommon. After 48 weeks of treatment with ART, infection was suppressed to below 200 copies/mL in most patients (95%), with full suppression (RNA target not detected) in 65%. The following characteristics at patient enrollment were independently associated with a lack of full suppression: CD4 T cell counts below 500 cells/µL, viremia above 100,000 copies/mL, older age, and TDR to NNRTI. The rates of resistance were intermediate, but genotype-guided therapy resulted in high rates of viral suppression. The observed resistance profile should not be an obstacle to the use of the dolutegravir-based regimen now recommended in Brazil, but genotype testing may be warranted before initiating first-generation NNRTI-based regimens.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The benefits of combination antiretroviral therapy (ART) to treat human immunodeficiency virus (HIV)-1 infection are well established, but virus resistance is an important factor in treatment failure [1]. Viral suppression is associated with immunological recovery, favorable clinical outcome [2], and improved life expectancy, which in some recent studies approaches that of the general population [3]. However, incomplete viral suppression favors the emergence of drug resistance, increases the probability of virologic failure, and may compromise subsequent treatment [4]. Some circulating viruses may be resistant and, if transmitted, may impact treatment response. Therefore, the use of a pre-treatment genotype test is recommended in many countries [1]. However, issues related to cost and logistics limit the use of the test in resource-limited settings. Plasma HIV-1 RNA quantitation is the preferred method for measuring the viral load (VL) to monitor the response to ART [5]. Viral suppression should be achieved within three to six months on ART. The limit of detection (LoD) of different commercial and in-house assays varies from 20 to 400 copies per mL (c/mL), and the results are reported either by numeric value or as “below the LoD.” The Abbott rtPCR assay (USA) that is currently used in the Brazilian public system has an LoD of 40 c/mL. The results that are below the LoD and with no viral RNA detected are reported as “target not detected” (TND). The level of viremia that defines satisfactory viral suppression varies with the definition purpose. The Brazilian Ministry of Health and the World Health Organization (WHO) adopt a VL of 1,000 c/mL to define viral suppression in the cascade of care framework [6]. Many clinical trials use < LoD (below limit of detection) to define virological success [7, 8]. Many clinical guidelines define virological failure as a VL above 200 c/mL [9,10,11]. Although the benefit of very stringent suppression levels has not been clearly proven [12], TND, full viral suppression, has been correlated with a lower risk of viral rebound when compared to very low viremia levels, e.g., < 20 c/mL [13] and < 40 c/mL or 40-49 c/mL [14]. Additionally, observational studies in Canada [15] and Africa [16] have shown an association between lowest viremia levels and favorable clinical outcome. These findings support the concept that the lower the viremia, the better. The Brazilian Ministry of Health has provided free access to ART since the nineties and started recommending universal treatment in 2013. An efavirenz-based regimen was recommended as the first-line choice of treatment from 2002 to 2017 when efavirenz was substituted for dolutegravir. Low or intermediate TDR rates have been documented in São Paulo and other areas of Brazil in most of the studies using Sanger population sequencing from plasma HIV RNA. [17,18,19,20,21]. To investigate the evolution of TDR prevalence in the area and the impact of TDR on ART response, we analyzed HIV sequences from 596 ART-naïve patients from São Paulo diagnosed from 2014 to 2016. The state of São Paulo has the largest population of people living with HIV/AIDS in Brazil, with over 200,000 patients currently on ART. In this study, the virological response after 48 weeks on ART was documented and the predictors of failure evaluated for individuals followed at the public HIV outpatient clinics in São Paulo.
Patients and methods
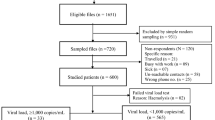
As part of an open access initiative to provide pre-treatment genotype testing in the São Paulo public health system, consecutive blood samples from ART-naïve HIV-positive patients who were newly diagnosed at public outpatient clinics between January 2014 and April 2016 were sent to the Adolfo Lutz Retrovirus Laboratory. The National ART Dispensation database SICLOM was reviewed to eliminate cases of unreported ART at study inclusion and to identify the regimen start date, ART regimen used, and subsequent ART changes up to the virological outcome assessment date. The virological outcome was assessed in the National HIV Laboratory database, SISCEL. The first VL result available after 48 weeks of ART initiation was recorded, and suppression rates were calculated considering four different metrics: 1,000 c/mL; 200 c/mL; < 40 c/mL (below LoD); and RNA target not detected (TND).
Sequences were obtained from plasma viral RNA that was extracted either manually (QIAmp Viral RNA Mini Kit, QIAGEN, Hilden Germany) or automatically (M2000 extractor, Abbott, USA) and retrotranscribed and amplified using an in-house PCR assay [19]. Briefly, amplification of a portion of the HIV polymerase region (codon 1-235 related to HXB2, accession number MG211834-MG212426) was carried out using retrotranscribed RNA from a one-step reaction (RT-PCR), followed by nested PCR using SuperScript III and High-Fidelity Taq polymerase (Life Technologies, USA). A unique fragment of approximately 1,112 bp was sequenced after Big Dye incorporation using eight primers to cover part of the pol region (codon 1 to 235). Sequences were edited manually using Sequencher 4.7 software (Gene Codes, USA), ReCall (beta v3.01), or both.
HIV genotyping was performed according to Stanford HIV Database Genotyping Resistance Interpretation (GRI-HIVdb), and the results were reported to clinical sites to support patient management strategies. For purposes of the study, sequences were reanalyzed by using the Calibrated Population Resistance (CPR) tool (CPR Version 6.0, Stanford Database, SDRM 2009) and referencing a standard mutation list of surveillance drug-resistance mutations adopted by WHO as transmitted resistance (TDR).
HIV subtypes were determined using the NCBI Genotyping and REGA HIV Subtyping tools and confirmed by phylogenetic analysis using BEAST v1.7.4 under the GTR + I + G model. Recombinant genomes were evaluated by jpHMM (jumping profile Hidden Markov Model, http://jphmm.gobics.de/) and SCUEAL Datamonkey, http://www.datamonkey.org/dataupload_scueal.php.
Statistical analysis
The data were analyzed using Epi Info 6 (CDC, USA), STATA (v8 Stata Corp, USA), and Graph Pad Prism (v5, USA), applying a level of statistical significance of p < 0.05 (two-tailed test). Results of continuous variables were expressed as the median and interquartile range (IQR: 25-75). Mid-p 95% was used to calculate the confidence interval. Dichotomous variables were compared using the Pearson chi or Fisher exact test, and continuous variables were compared using the Mann-Whitney or Kruskal-Wallis test. For logistic regression, variables were dichotomized as viremic/not viremic at the different cutoffs (TND, < LoD, < 200 c/mL, and < 1,000 c/mL). To assess associations with viral suppression, we included the following variables: gender; age (≤ 25 years,the lower quartile, vs. older); CD4 T cell counts (below vs. above 500 cells/µL); viremia (below vs. above 100,000 c/mL); B/non-B HIV subtype; presence or absence of (i) non-nucleoside reverse transcriptase inhibitor (NNRTI), (ii) nucleoside reverse transcriptase inhibitor (NRTI), or (iii) protease-inhibitor (PI) mutations; use of an NNRTI-based first-line ART vs. other regimens at enrollment; and ART regimen change vs. no change, before virological outcome assessment. Variables with p = 0.2 or lower were retained for adjusted analyses.
The study was approved by the institutional ethical committee (CEPIAL - CAAE 022980012.6.0000.0059) and was supported in part by grants FAPESP 2013/19441-7 and FAPESP PPSUS2016/14813-1.
Results
Patient characteristics
Between January 2014 and April 2016, pre-ART genotypic resistance testing was conducted using unique samples from 622 HIV-1-infected individuals. After checking the treatment database, 26 patients were excluded from the study because there was documentation that they had received ART previously; the remaining 596 ART-naïve individuals were included in the analyses. Most of the cases were recent diagnoses at the time of genotypic resistance testing. The median time from diagnosis to testing was 33 days (IQR: 22–68), with only 38 cases (6.3%) and 14 cases (2.4%) with durations longer than one and two years, respectively. The characteristics of the study population are summarized in Table 1.
Transmitted drug resistance mutations
The viruses in 65 out of the 596 cases harbored at least one TDR (10.9%, 95% CI: 8.6-13.6) in the HIV-1 polymerase. Mutations associated with NNRTI resistance, present in 41 cases (6.9%), were the most common, followed by mutations associated with NRTI resistance (22; 3.7%) and PI resistance (13; 2.2%). Dual-class mutations were observed in six (1%) cases, two NRTI + NNRTI, two NRTI + PI and two NNRTI + PI, and triple-class mutations were observed in two (0.34%) sequences. The NRTI drugs used in the currently recommended combination, tenofovir (TDF) and lamivudine (3TC), were predicted to be fully active in 97.5% and 98.8% of the cases, respectively. Integrase resistance was not evaluated. Table 2 shows TDR prevalence estimates from previous studies that used similar methodology [18, 19], showing a small but nonsignificant increase (p = 0.4) in the proportion of sequences with TDR.
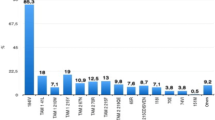
Using the clinical Stanford mutation list (GRI-HIVdb), which includes additional mutations not in the CPR/WHO list, at least one mutation associated with NNRTI resistance was detected in 113 (19%) sequences, while mutations associated with NRTI resistance and major mutations associated with PI resistance were detected in 30 (5.3%) and 13 (2.2%) of the sequences, respectively. Overall, males had a similar proportion of sequences with TDR (10.9%) than females (11.3%, p = 0.92). Figure 1 shows the observed mutations from the 596 sequences.
Proportion of sequences with mutations in the protease and reverse transcriptase (RT) at each resistance-associated codon according to antiretroviral drug class, including a) protease inhibitors, b) nucleoside RT inhibitors (NRTI), and c) non-nucleoside RT inhibitors (NNRTI). Some mutations in RT are shown that are not on the CPR/WHO list, including A62 V and V75I, associated with NRTI resistance, and several minor (accessory) mutations associated with resistance to the NNRTI class (V90I, A98G, V106I, V179I/T/D), as well as one major but polymorphic NNRTI mutation: E138A (according to the Stanford HIV Resistance Database)
HIV-1 subtypes
Subtype B was the predominant subtype (72.8%), followed by subtypes C (14.3%), and F (8.1%). Two cases of subtype AG, one subtype D, and 26 cases of different recombinant patterns (4.3%) with mostly BF mosaics were also identified (Table 1). Women tended to have a higher proportion of HIV-1 F or F mosaics then men (22.5% vs. 10.5%, p = 0.003). We observed some differences in the rate of TDR within subtypes. At least one mutation was observed in 10.7% of all subtype B (46/428), 4.7% of subtype C, 20.8% of subtype F, and 18.2% of BF mosaic sequences. Using subtype B as the reference, subtype F had comparatively more mutations (p = 0.003). The proportion of mutations associated with NNRTI resistance was similar (7.4% vs. 5.5%, p = 0.66) with the observed difference due to mutations associated with NRTI (10.4 vs. 3%, p = 0.011) and PI (6.3% vs. 1.9%, p = 0.055) resistance.
Because of the higher proportion of subtype F in women and the possibility of previous undocumented ART exposure from mother-to-child transmission prophylaxis, we analyzed men separately. Although smaller, the difference pattern persisted for NRTI mutations (10.8% vs. 2.6%, p = 0.08). Similar significant differences were also observed when F genomes were compared to all the other subtypes and recombinant pol genomes, with more mutations associated with NRTI resistance (11.3% vs. 2.7%, p < 0.001) and PIs (5.6% vs. 1.7% p = 0.03) but not associated with NNRTI resistance (7.0% vs. 6.9%, p = 0.95).
Subtype C viruses tended to have fewer TDR mutations, but the differences were generally not significant, except when NRTI mutation rate were compared to those of subtype F (0% vs. 14.8%, p < 0.007).
First-line ART regimens
Data on first-line ART was available for 566 (95%) of the 596 patients. ART was initiated at a median of 99 days (IQR: 45-227) after HIV diagnosis and 49 days (IQR: 14-147) after genotyping sampling, with 54 initiating therapy before the availability of test results. During the study, the most common regimen, tenofovir disoproxil fumarate, lamivudine and efavirenz (TDF/3TC/EFV), was used by 72% of patients following the Brazilian guidelines. Alternative regimens included TDF plus 3TC and atazanavir/ritonavir (TDF/3TC/ATZ/r) in 14% of the patients, zidovudine/3TC and lopinavir/ritonavir (AZT/3TC/LPV/r) in 5%, TDF/3TC/LPV/r in 3%, and other combinations in 6%. Patients used one (70%) to five (0.2%) regimens during the observation period. Although we were not able to document the reason for therapy change, only 3.2% of patients using an NNRTI-based first-line regimen had an NNRTI TDR mutation identified, compared to 11.2% of those initiating a PI-based regimen (p = 0.001). Moreover, most patients (80%) on an NNRTI-based regimen that harbored an NNRTI TDR changed regimens. However, patients on NNRTI-based regimens were less likely to change than those on other ART combinations (17.6% vs. 33.6%, p < 0.001). First-line LPV/r-based regimens were the most likely to be changed, with 50% patients changing regimens during the observation period compared to only 25% for the other regimens (p = 0.01).
Patients with T CD4 counts below 200 cells/µL tended to modify their ART more frequently than those with higher T CD4 counts (32% vs. 20%, p = 0.04).
Virological Outcome
The VL results after 48 weeks of ART were available in public databases for 421 (75%) of the 566 treated patients. Viral suppression below 1,000 c/mL was attained by 96.4%, below 200 c/mL in 94.8%, and below LoD (40 c/mL) in 91.7% of the patients. Target RNA not detected (TND), the most stringent metric used, suggesting full suppression, was documented in 65.3% of the cases. In a missing-equals-failure analysis, 72% of the 566 treated cases had documented viremia below 1,000 c/mL and 70.5% below 200 c/mL. At more stringent metrics (< LoD or TND), suppression levels were associated with CD4 > 500 cells/µL (TND, p < 0.001 and < 40 c/mL, p = 0.014), VL < 100,000 c/mL (TND, p < 0.001 and < 40 c/mL, p = 0.02), absence of mutations associated with NNRTI resistance (TND, p = 0.013 and < 40 c/mL, p = 0.012), and a younger age (≤ 25 years old) (TND, p = 0.007 and < 40 c/mL, p = 0.059). There was no significant association with gender, ART change, type of first-line regimen, non-B subtype, non-NNRTI TDR, or the combination of all TDR and non-TDR mutations associated with NNRTI resistance. To explore predictors of viral suppression, we performed multivariable unadjusted and adjusted analyses using age, CD4 count, VL at sample collection, subtype, and presence of mutations associated with NRTI resistance, NNRTI, or PI. After adjustment, CD4 > 500 cells/µL, the absence of TDR mutations associated with NNRTI resistance, VL < 100,000 c/mL, and age < 26 years old remained independently associated with TND (full suppression) (Table 3).
Discussion
Transmitted drug resistance mutations (TDR) are potential obstacles to therapy success. Some studies report a deleterious impact on the response to first-line therapy [22], while others report limited or no impact [23, 24] in treatment response, especially if patients used non-NNRTI-based regimens [25]. TDR has been observed worldwide [1], and the rates of prevalence are trending down from the relatively high rates of the late 1990s, as documented recently by the CASCADE collaboration in Europe [26], with stable rates in recent years in developed nations [1]. This scenario contrasts with the increasing rates, especially of mutations conferring resistance to the NNRTI drug class, in developing countries where ART has been introduced more recently [1, 27]. Due to HIV diversity, the Stanford CPR mutations list, adopted by WHO, allows comparability of estimates from different regions. The first objective of our study was to assess the prevalence of mutations and compare it to previous studies conducted by us using a similar methodology, and with the literature, to provide a reliable estimate of mutation prevalence (Table 2). In line with the current recommendation for immediate treatment, this study included mostly recently diagnosed patients. Rates of TDR are comparable to those observed in most studies in the region. This finding strongly suggests a stable, or slowly increasing, intermediate level of resistance, mostly to the NNRTI class. A recent meta-analysis of studies in Latin America estimates similar rates and suggests an increasing level of NNRTI TDR [28].
Brazil has changed the recommended first regimen in April 2017 from an efavirenz-based regimen to incorporating dolutegravir as the preferred drug for use with TDF and 3TC. Both of the currently recommended NRTIs were predicted to be fully active against most of the variants (> 97%) evaluated in this study. As the prevalence of resistance to integrase inhibitors among untreated individuals has been shown to be low and the impact of circulating TDR to NRTI antiretroviral drugs used in the dolutegravir-based regimens is low, resistance to this combination should not be a concern at the moment. However, about half a million people were already using ART in Brazil before the new recommendations were issued. Moreover, in many parts of the world, especially in resource-limited settings, NNRTI-based regimens are allowing an impressive scaling up of treatment in the region. The recent finding associating dolutegravir use with neural birth defects [29] resulted in the return to NNRTI-based regimens for women in some places.
Another objective of this study was to address the impact of TDR on virological outcome after one year of ART. This evaluation must consider that test results were sent to clinical services upon availability, and therapy was instituted or changed at the physicians’ discretion. Accordingly, the use of NNRTI-based regimens was significantly less frequent in cases with TDR to NNRTI, and most changed their regimen after receiving the genotype test results. Rates of suppression after 48 weeks of therapy were high overall: over 90% for most metrics. This percentage is higher than that reported at the national level [11], where pre-treatment genotype testing is not available, but this comparison cannot be used directly to support the indication of pre-treatment genotyping.
Presence of TDR to NNRTI, lower T CD4 counts, and higher viremia at entry were significantly associated with virological failure applying the LoD (40 copies/mL) VL threshold, but only TDR to NNRTI remained significant in adjusted analysis. As some studies suggest that full viral suppression may be beneficial to patients [14, 15], we also evaluated the impact of TDR and other variables on the RNA target-not-detected (TND) rate, which is a more stringent metric that is not commonly used in clinical settings. With this criterion, we observed significantly lower rates of viral suppression in adjusted analysis in the presence of TDR to NNRTI, T CD4 cell counts lower than 500 cells/µL, viremia above 100,000 c/mL, and older age (Table 3).
Many patients (22%) changed their antiretroviral regimen in the first year. The reasons for the changes were not documented, and although some seemed to have changed due to the genotype report, adverse effects appears to be common factors [30]. Although we could not evaluate this issue in this study, the lopinavir-based regimen, which is no longer recommended in Brazil, was the most common ART associated with regimen change, suggesting intolerance as a major issue. A third point to highlight in the study is the increase in subtype C in the region. The current study detected 14.3% (IQR: 11.3-16.8) HIV-1 C in the pol region (15.5% if C mosaics are considered), which is twice the proportion observed in a previous study (7.1% CI 95%: 4.2-11.1) [18], with C genomes replacing HIV-1 F as the second most prevalent subtype in this area. The fact that fewer mutations were observed in subtype-C-infected individuals might have contributed to the relative stability in TDR estimates in the region.
There are many limitations to our study. Although genotype testing was provided to all public services within the state, logistical issues limited the access to many services, especially in more remote areas. Moreover, even within a given service, some physicians may order tests more often than others, reflecting different clinical care standards. The use of genotype testing to support therapy allows the overall impact on the virological suppression rate in treated individuals to be evaluated, but not the specific impact of TDR in treatment response; therefore, the actual direct impact of the test in the success of therapy cannot be inferred from our study.
Conclusion
TDR rates are stable in the region and may potentially compromise NNRTI-based regimens. Genotype testing may not be needed before the current integrase-based combination but may be important to patients considering NNRTI-based regimens. High rates of suppression were attained after one year of therapy in this setting with the availability of alternative regimens that allowed ART switching to circumvent the potential deleterious effect of TDR or intolerance.
As viral suppression defined by the 200 c/mL threshold has been correlated to transmission protection (https://www.preventionaccess.org/ consensus) it may be a more appropriate goal for therapy than the 1,000 c/mL standard. The use of more-stringent metrics suggests that high viremia, low CD4 T cell counts, older age and, even with genotype testing, mutations conferring resistance to the NNRTI antiretroviral class may be associated with lower rates of suppression at week 48, but longer follow-up is necessary to assess its relevance for long-term clinical outcomes.
References
Günthard HF, Calvez V, Paredes R, Pillay D, Shafer RW, Wensing AM, Jacobsen DM, Richman DD (2018) Human Immunodeficiency Virus Drug Resistance: 2018 recommendations of the international antiviral society-USA Panel. Clin Infect Dis
Lundgren JD, Mocroft A, Gatell JM, Ledergerber B, D’Arminio Monforte A, Hermans P, Goebel FD, Blaxhult A, Kirk O, Phillips AN, EuroSIDA Study Group (2002) A clinically prognostic scoring system for patients receiving highly active antiretroviral therapy: Results from the EuroSIDA study. J Infect Dis 185:178–187
Teeraananchai S, Kerr SJ, Amin J, Ruxrungtham K, Law MG (2017) Life expectancy of HIV-positive people after starting combination antiretroviral therapy: a meta-analysis. HIV Med 18:256–266
Hermankova M, Ray SC, Ruff C, Powell-Davis M, Ingersoll R, D’Aquila RT, Ingersoll R, D’Aquila RT, Quinn TC, Siliciano JD, Siliciano RF, Persaud D (2001) HIV-1 drug resistance profiles in children and adults with viral load of > 50 copies/ml receiving combination therapy. JAMA 286:196–207
Marschner IC, Collier AC, Coombs RW, D’Aquila RT, DeGruttola V, Fischl MA, Hammer SM, Hughes MD, Johnson VA, Katzenstein DA, Richman DD, Smeaton LM, Spector SA, Saag MS (1998) Use of changes in plasma levels of human immunodeficiency virus type 1 RNA to assess the clinical benefit of antiretroviral therapy. J Infect Dis 177:40–47
Bennett DE, Bertagnolio S, Sutherland D, Gilks CF (2008) The World Health Organization’s global strategy for prevention and assessment of HIV drug resistance. Antivir Ther 13:1–13
Asboe D, Aitken C, Boffito M, Booth C, Cane P, Fakoya A et al (2011) BHIVA Guidelines Subcommittee et al British HIV Association guidelines for the routine investigation and monitoring of adult HIV-1-infected individuals
Hoen B, Bonnet F, Delaugerre C, Delobel P, Goujard C, L’Hénaff M, Persiaux R, Rey D, Rouzioux C, Taburet AM, Morlat P French HIV expert group (2013) French 2013 guidelines for antiretroviral therapy of HIV-1 infection in adults. J Int AIDS Soc 17:19034
Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. https://aidsinfo.nih.gov/guidelines. Accessed 17 July 2018
Protocolo Clínico e Diretrizes Terapêuticas para Manejo da Infecção pelo HIV em Adultos/Ministério da Saúde, Secretaria de Vigilância em Saúde, Departamento de Vigilância, Prevenção e Controle das Infecções Sexualmente Transmissíveis, do HIV/Aids e das Hepatites Virais.—Brasília: Ministério da Saúde, 2018. http://www.aids.gov.br/pt-br/pub/2013/protocolo-clinico-e-diretrizes-terapeuticas-para-manejo-da-infeccao-pelo-hiv-em-adultos. Accessed 17 Jan 2018
Teira R, Vidal F, Muñoz-Sánchez P, Geijo P, Viciana P, Ribera E, Domingo P, Castaño M, Martínez E, Roca B, Puig T, Estrada V, Deig E, Galindo MJ, de la Fuente B, Lozano F, Montero M, Muñoz-Sanz A, Sanchez T, Terrón A, Romero-Palacios A, Lacalle JR, Garrido M, Suárez-Lozano I, VACH Study Group (2017) Very low level viraemia and risk of virological failure in treated HIV-1-infected patients. HIV Med 18:196–203
Berta P, Marta G, Sonia P, Angelina C, Angeles C, Alvaro M, Iria RO, Andres T, Jose DP, Eva P (2016) Any impact of blips and low-level viraemia episodes among HIV-infected patients with sustained virological suppression on ART? J Antimicrob Chemother 71:1051–1055
Doyle T, Smith C, Vitiello P, Cambiano V, Johnson M, Owen A, Phillips AN, Geretti AM (2012) Plasma HIV-1 RNA detection below 50 copies/ml and risk of virologic rebound in patients receiving highly active antiretroviral therapy. Clin Infect Dis 54:724–732
Laprise C, de Pokomandy A, Baril JG, Dufresne S, Trottier H (2013) Virologic failure following persistent low-level viremia in a cohort of HIV-positive patients: results from 12 years of observation. Clin Infect Dis 57:1489–1496
Hermans LE, Moorhouse M, Carmona S, Grobbee DE, Hofstra LM, Richman DD, Tempelman HA, Venter WDF, Wensing AMJ (2018) Effect of HIV-1 low-level viraemia during antiretroviral therapy on treatment outcomes in WHO-guided South African treatment programmes: a multicentre cohort study. Lancet Infect Dis 18:188–197
Alencar CS, Sabino EC, Carvalho SM, Leao SC, Carneiro-Proietti AB, Capuani L, Oliveira CL, Carrick D, Birch RJ, Gonçalez TT, Keating S, Swanson PA, Hackett J Jr, Busch MP (2013) NHLBI Retrovirus Epidemiology Donor Study-II (REDS-II), International Component. HIV genotypes and primary drug resistance among HIV-seropositive blood donors in Brazil: role of infected blood donors as sentinel populations for molecular surveillance of HIV. J Acquir Immune Defic Syndr 63:387–392
Ferreira JL, Rodrigues R, Lança AM, de Almeida VC, Rocha SQ, Ragazzo TG, Estevam DL, Brigido LF (2013) Transmitted drug resistance among people living with HIV/aids at major cities of Sao Paulo State, Brazil. Adv Virol 15:878237
Guimarães PM, Ferreira JL, Coelho LP, Cavalcanti Jde S, Lopes GI, Matsuda EM, Almeida FJ, Almeida VC, Campeas AE, Junior LC, Brígido LF, São Paulo Savage Workgroup (2015) Transmitted Drug Resistence Among Recently Diagnosed Adults and Children in São Paulo, Brazil. AIDS Res Hum Retroviruses 31:1219–1224
HIV Drug Resistance Report 2017. http://www.who.int/hiv
Arruda MB, Boullosa LT, Cardoso CC, da Costa CM, Alves CR, de Lima ST,Kaminski HT, Aleixo AW, Esposito AO, Cavalcanti AM, Riedel M, Couto-Fernandez JC, Ferreira SB, de Oliveira IC, Portal LE, Wolf HH, Fernandes SB, de M C Pardini MI, Feiteiro MV, Tolentino FM, Diaz RS, Lopes GI, Francisco RB, Véras NM, Pires AF, Franchini M, Mesquita F, Tanuri A HIV-BResNet Brazilian network for HIV Drug Resistance Surveillance (2018) (HIV-BresNet): a survey of treatment-naive individuals. J Int AIDS Soc 21
Wittkop L, Günthard HF, de Wolf F, Dunn D, Cozzi-Lepri A, de Luca A, Kücherer C, Obel N, von Wyl V, Masquelier B, Stephan C, Torti C, Antinori A, García F, Judd A, Porter K, Thiébaut R, Castro H, van Sighem AI, Colin C, Kjaer J, Lundgren JD, Paredes R, Pozniak A, Clotet B, Phillips A, Pillay D, Chêne G, EuroCoord-CHAIN study group (2011) Effect of transmitted drug resistance on virological and immunological response to initial combination antiretroviral therapy for HIV (EuroCoord-CHAIN joint project): a European multicohort study. Lancet Infect Dis 11:363–371
Margot NA, Wong P, Kulkarni R, White K, Porter D, Abram ME, Callebaut C, Miller MD (2017) Commonly transmitted HIV-1 drug resistance mutations in reverse transcriptase and protease in antiretroviral treatment-naive patients and response to regimens containing tenofovir disoproxil fumarate or tenofovir alafenamide. J Infect Dis 215:920–927
Zu Knyphausen F, Scheufele R, Kücherer C, Jansen K, Somogyi S, Dupke S, Jessen H, Schürmann D, Hamouda O, Meixenberger K, Bartmeyer B (2014) First line treatment response in patients with transmitted HIV drug resistance and well defined time point of HIV infection: updated results from the German HIV-1 seroconverter study. PLoS One. e95956
Clutter DS, Fessel WJ, Rhee SY, Hurley LB, Klein DB, Ioannidis JP, Silverberg MJ, Shafer RW (2016) Response to therapy in antiretroviral therapy-naive patients with isolated nonnucleoside reverse transcriptase inhibitor-associated transmitted drug resistance. J Acquir Immune Defic Syndr 72:171–176
Ashley O, Norbert B, Anders S, de Carmen M, Matthew P, Robert Z, Marie-Laure C, Maria P, Anne MBK, John G, Dimitrios P, Kholoud P, For CASCADE Collaboration in EuroCoordM (2018) Temporal trends of transmitted HIV drug resistance in a multinational seroconversion cohort. AIDS 32:161–169
http://www.unaids.org/sites/default/files/media_asset/2017-Global-AIDS-Monitoring_en.pdf. Accessed 17 Jan 2018
Avila-Rios S, Sued O, Rhee SY, Shafer RW, Reyes-Teran G, Ravasi G (2016) Surveillance of HIV transmitted drug resistance in Latin America and the Caribbean: a systematic review and meta-analysis. PLoS One 29(11–16):e0158560
http://www.who.int/medicines/publications/drugalerts/Statement_on_DTG_18May_2018final.pdf
Cesar C, Shepherd BE, Krolewiecki AJ, Fink VI, Schechter M, Tuboi SH, Wolff M, Pape JW, Leger P, Padgett D, Madero JS, Gotuzzo E, Sued O, McGowan CC, Masys DR, Cahn PE, Caribbean, Central and South America Network for HIV Research (CCASAnet) Collaboration of the International Epidemiologic Databases to Evaluate AIDS (IeDEA) Program (2018) Rates and reasons for early change of first HAART in HIV-1-infected patients in 7 sites throughout the Caribbean and Latin America. PLoS One 1:e0158560
Acknowledgements
We acknowledge the patients and their families, health care workers and laboratory personal from the participating sites, and data management personal.
MIHR: Monitoring Incident & Recent HIV infection working group: Daniela Rodrigues Colpas, Gabriela Bastos Cabral, Giselle Ibete Silva López Lopes, Norberto Camilo Campos, Isadora Coutinho Ribeiro, Cintia Mayumi Ahagon, Guilherme Penteado Teixeira, João Leandro de Paula Ferreira, Ivana Barros de Campos, Mariana Carvalho, Rafaella Gomes, Flavia Gennari Pinheiro, Luiz Carlos Pereira Jr, Karen Morejon, Álvaro Furtado da Costa, Érika Maria do Nascimento Kalmar, Fábio Luis Nascimento Nogui, Fábio Luis Nascimento Nogui, Patrícia Rady Müller, Silvia Pereira Goulart, Suzana Toledo da Silva Leme.
Funding
FAPESP (award number 2013/19441-7 and PPSUS 2016/14813-1).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
No potential conflicts of interest were identified by the authors.
Ethical approval
The research involved no intervention on humans or animals, and patients in this observational study agreed to be included and signed an informed consent statement. Additional MIRH-participating institutions include Regional de Santo André do Instituto Adolfo Lutz /SP, Ambulatório de Moléstia Infecciosa, Jundiaí; Hospital das Clínicas da Faculdade de Medicina, Ribeirão Preto/SP; Instituto de Infectologia Emilio Ribas/SP.
Additional information
Handling Editor: Li Wu.
Rights and permissions
About this article
Cite this article
Coelho, L.P.O., Matsuda, E.M., Nogueira, R.S. et al. Prevalence of HIV-1 transmitted drug resistance and viral suppression among recently diagnosed adults in São Paulo, Brazil. Arch Virol 164, 699–706 (2019). https://doi.org/10.1007/s00705-018-04122-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-018-04122-8