Abstract
Beak and feather disease virus (BFDV) infections are often fatal to both captive and wild parrot populations. Its recent discovery in a wild population of native red-fronted parakeets has raised concerns for the conservation of native parrots, all of which are threatened or endangered. The question of a recent introduction versus a native genotype of the virus poses different conservation-management challenges, and thus, a clear understanding of the molecular phylogeny of BDFV is a crucial step towards integrated management planning. This study represents the first comprehensive attempt to screen New Zealand’s endangered and threatened psittacines systematically for BFDV. We sampled and screened kakapos (Strigops habroptilus), kakas (Nestor meridionalis), keas (N. notabilis), Chatham parakeets (Cyanoramphus forbesi), Malherbe’s parakeets (Cyanoramphus malherbi), yellow-crowned parakeets (C. auriceps) and red-fronted parakeets (Cyanoramphus novaezelandiae), as well as eastern rosellas (Platycercus eximius), an introduced species that is now common throughout the North Island, for BFDV. Out of all species and populations sampled (786 individuals), we found 16 BFDV-positive red-fronted parakeets from Little Barrier Island/Hauturu, seven eastern rosellas from the Auckland region, and eight yellow-crowned parakeets from the Eglinton Valley in the South Island. The full genomes of the viral isolates from the red-fronted parakeets share 95–97 % sequence identity to those from the invasive eastern rosellas and 92.7–93.4 % to those isolates from the South Island yellow-crowned parakeets. The yellow-crowned parakeet BFDV isolates share 92–94 % sequence identity with those from eastern rosellas. The low level of diversity among all BFDV isolates from red-fronted parakeets could suggest a more recent infection among these birds compared to the yellow-crowned parakeets, whereas the diversity in the eastern rosellas indicates a much more established infection. Pro-active screening and monitoring of BFDV infection rates in aviaries as well as in wild populations are necessary to limit the risk of transmission among threatened and endangered parrot populations in New Zealand.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Psittacine beak and feather disease (PBFD) is a potentially fatal viral disease affecting parrots. PBFD was first described in Australian psittacine species in the 1970s; however, records from as early as 1887 associated the decline of Psephotus spp. in Southern Australia with feather deformities that severely impaired the flight of these birds [37, 41]. Since PBFD was first described, the disease has spread throughout the world, affecting both wild and captive populations of a broad range of psittacine species [2, 3, 6, 15, 22, 41, 42, 44, 49]. The global spread of PBFD has been largely attributed to the international trafficking of parrots for the pet trade [6, 40, 54].
PBFD affects the lymphoid tissue, causing immunodeficiency and a deterioration in body condition [17, 47]. Symptoms of this disease include weight loss, development of morphologically abnormal feathers, feather loss, and anaemia [23, 43]. PBFD-associated bacterial or fungal infections can subsequently lead to death [17, 47]. Clinical signs are dependent on the dose and route of infection, as well as the individual’s age and species [35]. In healthy adult birds, an initial and vigorous immune response often clears the virus [35]. In contrast, in neonates the peracute form of this disease can cause sudden death [46]. The acute form in nestlings and fledglings is characterized by changes in growing feathers such as dystrophy, necrosis or haemorrhage. More overt signs are diarrhoea, weakness and depression, typically followed by death within 1–2 weeks [46]. Transmission occurs predominantly through environmental contamination including faeces, crop secretions and feather dust [2, 45].
The causative agent of PBFD is beak and feather disease virus (BFDV), a circular single-stranded DNA (ssDNA) virus belonging to the genus Circovirus in the family Circoviridae [56]. This genus includes avian circoviruses, such as columbid circovirus, gull circovirus, canary circovirus, duck circovirus, starling circovirus, finch circovirus, and raven circovirus, and two that infect pigs – porcine circovirus-1 and -2 [20, 38, 53, 55, 57, 59, 62]. Circoviruses and recently described cycloviruses [27, 48] are the smallest known autonomously replicating viruses, with 1.7- to 2-kb ambisense genomes encoding two genes (coat protein [cp] and replication-associated protein [rep]). Their replication-associated proteins have similarities to the plant-infecting geminiviruses and nanoviruses. Recombination is common amongst ssDNA viruses. Due to the recombinant nature of ssDNA viruses, these circoviruses evolve rapidly and could easily move across hosts and thus spread among different species of parrots.
Parrots are one of the most threatened bird groups worldwide, with 26 % of the 350 known species at risk of global extinction [7]. New Zealand is home to eight extant species, all of which are endemic and classified as threatened in the IUCN Red List of Threatened Species (Version 2010.4. http://www.iucnredlist.org). While predation by introduced mammalian species has been identified as the main driver of population declines of New Zealand native parrots [6, 8, 54], the potential damaging effect of infectious diseases, such as PBFD, among the already depauperate New Zealand parrot fauna has been highlighted [32]. To date in New Zealand, BFDV has been reported in captive lorikeets (Trichoglossus spp., Lorius chlorocerus, Psitteuteles goldiei, Eos reticulata), long-billed corellas (Cacatua tenuirostris) and budgerigars (Melopsittacus undulatus), as well as in wild populations of two introduced Australian parrots, the Eastern Rosella (Platycercus eximius) and Sulphur-crested Cockatoo (Cacatua galerita) [15, 47]. In 2008, BFDV was detected for the first time in a wild population of an endemic New Zealand parrot species, the Red-fronted Parakeet (Cyanoramphus novaezelandiae), on Little Barrier Island/Hauturu [32, 33]. Full-genome analysis of the BFDV isolates obtained from red-fronted parakeets indicated that they share less than 84–92 % full-genome pairwise identity to all other isolates from around the world [33, 61]. However, full virus genomes have never been characterised previously for isolates obtained from infected, introduced parrots in New Zealand. Hence, there is a lack of baseline data for both introduced and endemic parrots in New Zealand that would allow us to understand the evolutionary relationships amongst these isolates and in relation to those from the rest of the world. Characterisation of full virus genomes is necessary to assess whether native New Zealand parrots have been infected by wild populations of Australian parrots (eastern rosellas and sulphur-crested cockatoos), or whether native New Zealand parrots carry a unique BFDV genotype that may have been transmitted to introduced Australian parrots. An understanding of disease transmission among parrots in New Zealand has implications for the management of this emerging, infectious disease.
To address this question and to investigate BFDV comprehensively (at the molecular level) in New Zealand’s threatened and endangered parrots, we screened seven native New Zealand species of parrots for BFDV: the critically endangered Kakapo (Strigops habroptila) and Malherbe’s Parakeet (Cyanoramphus malherbi), the endangered Kaka (Nestor meridionalis) and Chatham Parakeet (Cyanoramphus forbesi), and the ‘vulnerable’ Kea (Nestor notabilis), Yellow-crowned Parakeet (Cyanoramphus auriceps) and Red-fronted Parakeet (IUCN Red List of Threatened Species. Version 2010.4., http://www.iucnredlist.org). Wild populations of native parrots were sampled at 16 locations throughout the North and South Islands and 11 offshore islands (Fig. 1, Table 1). Additionally, we tested native birds at five captive-breeding locations for BFDV. Finally, we also sampled introduced eastern rosellas, an Australian parrot species that was introduced to New Zealand around 1910 [17] and is now common and widespread in the North Island and parts of the South Island [63]. We caught and sampled eastern rosellas at two locations in the Auckland region and from the North Shore Society for the Prevention of Cruelty to Animals (SPCA) Bird Rescue Centre.
From samples that tested positive for BFDV, we characterised the full genomes of the virus isolates and compared these with other global isolates using standard phylogenetic methods to infer how the transmission of this disease may have occurred in New Zealand.
Materials and methods
Sample collection, isolation of genomic DNA and screening for BFDV by polymerase chain reaction (PCR)
We sampled a total of 788 birds to test for BFDV (Table 1 and Fig. 1). Wild birds were caught using mistnets, with the exception of kakapos, which were caught by hand. Blood samples (100-200 μl each) were collected by venipuncture of the brachial vein and stored in Queens lysis buffer (10 mM Tris, 10 mM NaCl, 10 mM disodium-EDTA, 1 % n-lauroylsarcosine, pH 8.0), also called Seutin’s buffer [50]. In some cases we were only able to collect body feathers (e.g., Cyanoramphus malherbi selected for translocation). Depending on the sample, either 2 mm of the quill tip (calamus) cut from feather sample or 10 μl of blood in Queens lysis buffer was added to 10 μl of ‘lysis solution’ from the Extract-n-Amp™ Blood PCR Kit (Sigma-Aldrich, USA) and incubated for 10 minutes at room temperature. Ninety microlitres of this kit’s ‘neutralisation solution’ was subsequently added to yield crude total DNA. One microlitre of the crude extract was used as template in the subsequent PCR.
PCR was performed using KAPA Blood PCR Kit Mix B (KAPA Biosystems, South Africa) according to the manufacturer’s protocols. The primers used were 5’-TTAACAACCCTACAGACGGCGA and 5’-GGCGGAGCATCTCGCAATAAG, which amplify a 605-bp segment of the rep gene of BFDV [47]. The PCR program had an initial step of 94 °C for 5 min, which was followed by 25 cycles of 94 °C for 30 s, 56 °C for 30 s and 72 °C for 45 s, with a final 1-min extension step at 72 °C and cooling to 4 °C for 10 min. DNA from BFDV-infected red-fronted parakeets reported from Little Barrier Island were used as positive controls [33]. The PCR products were resolved on a 1 % agarose gel and stained with SYBR®Safe DNA stain.
Isolation and cloning of full BFDV genomes using rolling-circle amplification
BFDV viral genomes were amplified (from BFDV PCR-positive samples) using RCA (TempliPhi™, GE Healthcare, USA) as described previously [33, 51, 60, 61]. In brief, 1 μl of the total DNA extracted using Extract-n-Amp™ solutions was mixed with 4 μl of the TempliPhi™ sample buffer, heated for 2 min at 94 °C, and cooled to room temperature. Five microlitres of reaction buffer and 0.2 μl of TempliPhi™ enzyme mix (containing ϕ29 DNA polymerase) were added, and the nonspecific amplification reaction was allowed to continue at 30 °C for 20 h. The resulting concatemers were digested with BamHI to release unit-length genomes. The digested DNA was separated on a 0.7 % agarose gel, and the ~2-kb bands were excised, recovered using a DNA Clean & Concentrator™-5 (Zymo Research, USA) and ligated into a BamHI-digested pUC19 plasmid vector. The cloned virus genomes were sequenced by primer walking at Macrogen Inc., Korea.
Sequence analysis
BFDV genomes were assembled using DNAMAN (version 5.2.9; Lynnon Biosoft), and the genomes were aligned to all other BFDV genomes available in GenBank (see Supplementary Table 1 for details) using the ClustalW-based sub-alignment tool available in MEGA (gap open penalty = 10; gap extension penalty = 5; [18] with manual editing. Maximum-likelihood phylogenies were inferred using PHYML [14], with 1000 non-parametric bootstrap replicates and the model GTR + G4 (determined using MODELTEST [19]).
Recombination amongst the New Zealand BFDV isolates was analysed using RDP4 [31], using the RDP [29], GENECONV [34], Bootscan [30], Maxchi [52], Chimera [39], Siscan [11] and 3Seq [4] methods. Events that were detected by more than three methods with significant p-values coupled with phylogenetic evidence of recombination were considered credible recombination events.
Results
PBFD screening of parrots in New Zealand
Of the 753 samples from seven native New Zealand parrots, 16 red-fronted parakeets from Little Barrier Island, ~10 km off the coast of the North Island, were found to be BFDV positive, with a prevalence of 10.5 % (95 % CI: 6.1 % –16.4 %). In addition, seven out of 32 eastern rosellas sampled in the Auckland region, North Island, also tested positive for BFDV, indicating a prevalence of 22.9 % (95 % CI 9.9 %–42.3 %) (see Table 1 and Fig. 1 for details). Between November 2011 and February 2012, we sampled 30 yellow-crowned parakeets from the Eglinton region following reports of birds with feather loss. Among these, we found eight samples to be positive for BFDV, thereby suggesting a prevalence of 26.7 % (95 % CI 12.3 %–45.9 %).
Full-genome sequence analysis of BFDV isolates
Full-genome characterisation of BFDV from 16 positive red-fronted parakeet samples from Little Barrier Island (GenBank accession numbers: GQ396652-GQ396656, GU936287-GU936297, and JF519618) revealed that all of the BFDV isolates from the island share >99 % sequence pairwise identity, whereas the eight yellow-crowned parakeet isolates (JQ782201-JQ782208) share >98 % sequence identity (Fig. 2). On the other hand, the seven eastern rosella BFDV isolates (GenBank accession numbers: FJ519619, GU936287, and JQ782196-JQ782200) share 93.8–97.7 % sequence pairwise identity (Fig. 2).
Maximum-likelihood phylogenetic relationships of the full genomes of red-fronted parakeet, yellow-crowned parakeet and eastern rosella BFDV isolates from New Zealand, with all publicly available full-length BDFV genomes (for more details of isolates, see Supplementary Table 1). The panel on the far right shows the pairwise nucleotide identities of A. BFDV-A1 [NZ-80-2008] B. BFDV-A2 [NZ-R18-2010], BFDV-A6 [NZ-R26-2010] and C. BFDV-O1 [NZ-YFP01-2011] to all other isolates. The maximum-likelihood tree was constructed using the model GTR+G4 in PHYML [14] with 1000 full maximum-likelihood bootstrap replicates (color figure online)
BFDV isolates from the two native species on the North and South Island share between 92.7–93.4 % pairwise identity, and this, coupled with the maximum-likelihood phylogenetic analysis, reveals that the origins of BFDV in these two populations are different (Fig. 2). The BFDV isolates from red-fronted parakeets on Little Barrier Island share ~94.9–97.9 % pairwise identity to the isolates from eastern rosellas. Pairwise identity and ML phylogenetic analysis indicate that the isolates from the eastern rosellas and those from the red-fronted parakeets share a common ancestor (Fig. 2). On the other hand, the isolates from the yellow-crowned parakeets share 92–94 % pairwise identity with those from the eastern rosellas, and 84–94 % pairwise identity to all the BFDV isolates from around the world (total GenBank entries: 113, including the samples from this study) (Fig. 2). The maximum-likelihood phylogenetic tree (Fig. 2) indicated that the 16 red-fronted parakeet and seven eastern rosella BFDV isolates from New Zealand form a well-supported monophyletic clade belonging to the BFDV A strain (genotypes BFDV-A1-A6; see Varsani et al. [61] for species, strain and genotype demarcation). The yellow-crowned parakeet isolates form a clade with BFDV-I isolates from South Africa and Europe and represent a new BFDV strain, which we have labelled as BFDV-O. All of the isolates from yellow-crowned parakeets fall under the same genotype – BFDV-O1.
Previous studies of BFDV in captive parrots in New Zealand – cockatoos, budgerigars and lorikeets – reported three distinct lineages identified on the basis of a partial sequence (547 bp) of the rep gene [47]. Our analysis of all partial sequences of rep deposited in GenBank (n = 162) indicates that all of the New Zealand BFDV isolates for which we have determined the full genomes (n = 31) share 90–93 % sequence identity to the three lineages previously identified by Ritchie et al. [47] (Fig. 3). The analysis of all available partial sequences (404 bp) of the cp domain deposited in GenBank (n = 191) reveal that the 16 BFDV isolates from the red-fronted parakeets and the seven eastern rosella isolates are generally similar in this region and share <92 % sequence identity to all other cp sequences in GenBank. The cp sequences of the yellow-crowned parakeets, on the other hand, share ~95 % identity to the isolates from Africa and Europe, similar to the full genome analysis (Fig. 4). We find no host-specificity link of the New Zealand eastern rosella isolates to other rosella cp sequences from the Czech Republic (Fig. 4). The analysis of rep and cp amplicons provides further unique genotype support for the 23 BFDV-A isolates from red-fronted parakeets (n = 16), and eastern rosellas (n = 7), and it additionally provides support for the unique BFDV-O genotype for the isolates from the yellow-crowned parakeets (Fig. 5).
A. Maximum-likelihood phylogenetic relationships of partial rep (~574 nt) sequences of BFDV isolate sequences deposited in GenBank. The ML phylogenetic tree was constructed using PhyML [14]. B, C, D and E highlight the clades with New Zealand sequences (color figure online)
A. Maximum-likelihood phylogenetic relationships of partial cp (~404 nt) sequences of BFDV isolate sequences deposited in GenBank. The trees were constructed using PhyML [14] (model GTR+G4). B Highlights the clade with yellow-crowned parakeet sequences from New Zealand, C highlights the clade with eastern and northern rosella samples from Europe, and D highlights the clade with eastern rosella and red-fronted parakeet sequences from New Zealand (color figure online)
Recombination analysis
Evidence of recombination was found in two eastern rosella (BFDV-A) samples from the Auckland region. BFDV-A5 [NZ-D134148-2010] and BFDV-O5 [NZ-RSPCA2-2010] have a recombinant region of unknown origin in the cp gene. A second recombinant region was found in BFDV-A5 [NZ-RSPCA2-2010] in the C-terminal portion of the Rep from a BFDV-O isolate from a yellow-crowned parakeet. A recent global analysis of BFDV isolates (87 full genomes) by Varsani et al., [61] indicates, similar to what has been reported by Lefeuvre et al. [26], that there are two significant recombination hotspots, one in the C-terminal portion of the coat protein, and the second in the intergenic region of the genome. The two events of recombination we found within the New Zealand eastern rosella samples have recombination breakpoints within these recombination hotspots. It is clear in the case of the eastern rosellas that recombination is playing a role in the evolution of BFDV-A and that more sampling and determination of full genome sequences will improve the resolution in determining the origin of the BFDV-A variants on the North Island of New Zealand. Nonetheless, the recombination analysis does indicate that there are unsampled BFDV variants in the ecosystem that are ancestral to BFDV-A and BFDV-O.
Discussion
The full genome analysis of the BFDV isolates carried out in this study reveals that the BFDV-A and BFDV-O strains have only been sampled in New Zealand. The low level of divergence of the BFDV-A1 and BFDV-O1 isolates in the red-fronted and yellow-crowned parakeet isolates possibly indicates a much more recent outbreak within the Little Barrier Island and Eglinton populations compared to the isolates from eastern rosellas, which display a great diversity, thereby indicating a more established viral population within this species. The close similarity of the red-fronted parakeet BFDV isolates to those of eastern rosellas suggests a common viral source within New Zealand; however, this is significantly different to that of the yellow-crowned parakeets. Recombination analysis indicated that it is not uncommon within New Zealand BFDV isolates, and this might be a factor driving the evolution of BFDV and detection of unique genotypes. The virus may have been introduced to New Zealand by trafficking infected birds, both legally and illegally. However, nowadays, in countries with strict import restrictions on exotic parrots, such as New Zealand, it is the illegal trade that poses an ongoing and insidious risk for introduction of novel virus strains.
Full-genome analysis of BFDV isolates, as we have conducted in this study, has the advantage of revealing the evolutionary history of BFDV, taking recombination into account, and potentially the source of BFDV infection in various wild and captive settings. Further, full-genome analysis has potential use in tracking legal and illegal trade in parrots and phylogeographical analysis. For example, an African grey parrot in Portugal, infected with BFDV-I4 genotype (Fig. 2), was almost certainly trafficked as an infected individual from a breeding facility in South Africa. Similarly, the birds infected with the BFDV-C2 genotype in the United States came from a breeding facility in South Africa that breeds African grey parrots, white cockatoos and white-bellied caiques (Pionites leucogaster) (Fig. 2) [52]. Finally, a clear example of cross-species transmission within a breeding facility is demonstrated by the BFDV-F1 genotype found infecting seven parrot species (all exotic) in Thailand (Fig.2).
Given that circoviruses have very high mutation rates [24] and are also highly recombinant [26, 61], they have the ability to explore sequence space rapidly. Therefore, it is essential to characterise full genomes of BFDV in infected individuals and conduct a variety of downstream bioinformatic analyses [61] to answer questions about interspecies host transmission and introduction of the virus into new environments.
Our research represents the first comprehensive attempt to screen New Zealand’s endangered and threatened parrots for systematically BFDV. The finding of a new viral genotype of BFDV in a wild population of native red-fronted parakeets on Little Barrier Island [32, 33], a species listed as ‘vulnerable’ by the IUCN, and in yellow-crowned parakeets raises concerns for the conservation of New Zealand’s most endangered parrots, in particular the Kakapo and Malherbe’s Parakeet. On Little Barrier Island, we found that 10.5 % (95 % CI: 6.1 %–16.4 %) of 153 red-fronted parakeets sampled tested positive for BFDV. In both the yellow-crowned parakeets and the eastern rosellas, a high prevalence (23–27 %) was detected. Except for samples from yellow-crowned parakeets, none of the South Island birds sampled (n = 338 from 6 species) were BFDV positive, including the samples from kakapos and Malherbe’s parakeets (0–1.1 % prevalence with a 95 % CI). Studies by Ha et al. [15] found BFDV in birds as far south as the Hutt Valley on the lower North Island but did not detect BFDV in any birds sampled from the South Island. Therefore, this report represents the first report and molecular analysis of BFDV in endemic birds on the South Island. Thus, to date, BFDV has not been detected in any free-living parrots on any of the southern and far northern offshore islands (e.g., in the Auckland Islands, the Chatham Islands or the Kermadec Islands).
In New Zealand, translocations of wild and/or captive-bred birds from one area or island to another is a common method used in the conservation management of threatened bird species [12, 13]. Many predator-free offshore islands, for example, are used as ‘safe havens’ to establish new populations of threatened species. If translocated individuals are infected with BFDV, this management practice risks introduction of this disease to previously ‘BFDV-naïve’ bird populations. Severe population declines can reduce genetic diversity within species due to genetic drift and inbreeding [9, 21]. Previous studies of threatened New Zealand species that have undergone substantial declines have shown that these birds are often immuno-compromised and hence more susceptible to novel pathogens [16, 58]. As BFDV infection can result in substantially higher mortality rates, PBFD increases the risk of extinction of threatened and endangered parrots. This has been documented in the critically endangered Mauritius Parakeet (Psittacula echo), where 83 % of the BFDV-infected individuals died [25, 28].
With a population of around 120 birds, the endemic Kakapo is most at risk from disease-caused extinction. Suitable habitats for kakapos are limited to the very few offshore islands with appropriate vegetation that are free of and beyond the dispersal range of introduced mammalian predators. Pest-free Little Barrier Island is being considered as a potential site; however, the discovery of BFDV in red-fronted parakeets will have direct implications for the management and recovery of kakapos at this site.
Currently in New Zealand, captive breeding facilities for both native and exotic parrots risk geographic and host range expansion of BFDV. Captive-breeding facilities may create opportunity for disease transmission, as these facilities are a frequent source of founder stock and/or supplement for wild populations. As such, they present a direct risk of disease transfer from captive to wild individuals. Since the discovery of BFDV-infected red-fronted parakeets on Little Barrier Island, the Department of Conservation has implemented a rigorous BFDV screening programme for both its captive breeding stock and any bird reared by captive parents. Captive birds are screened annually, and all individuals being translocated are tested prior to release.
This study highlights the need for continued, systematic BFDV screening in captive-breeding facilities as well as wild populations of parrots in New Zealand. In particular, individuals destined to be translocated and parrot populations on offshore islands where endangered birds might be placed for species recovery need to be screened immediately prior to translocation, given that exposure to pathogens could compromise the viability of newly established populations [36] and/or the long-term persistence of critically endangered species [1, 5, 10].
Genbank accession numbers of circoviruses sequences determined in this study
BFDV [NZ_R18_2010]: JF519619, BFDV [NZ_L53_2010]: JF519618, BFDV [NZ_B51_2008]: GU936287, BFDV [NZ_B195_2008]:GU936288, BFDV [NZ_B157_2008]: GU936289, BFDV [NZ_B326_2008]: GU936290, BFDV [NZ_B79_2008]: GU936291, BFDV [NZ_B162a_2008]: GU936292, BFDV [NZ_B133_2008]: GU936293, BFDV [NZ_B125_2008]: GU936294, BFDV [NZ_B192_2008]: GU936295, BFDV [NZ_B127_2008]: GU936296, BFDV [NZ_B162b_2008:] GU936297, BFDV [NZ_R9_2010]: JQ782196, BFDV [NZ_R24_2010]: JQ782197, BFDV [NZ_R22_2010]: JQ782198, BFDV [NZ_R25_2010]: JQ782199, BFDV [NZ_R26_2010]: JQ782200, BFDV [NZ_ycp01_2011]: JQ782201, BFDV [NZ_ycp28F_2012]: JQ782202, BFDV [NZ_ycp24_2012]: JQ782203, BFDV [NZ_ycp23_2012]: JQ782204, BFDV [NZ_ycp21_2012]: JQ782205, BFDV [NZ_ycp19_2012]: JQ782206, BFDV [NZ_ycp25_2012]: JQ782207, BFDV [NZ_ycp20_2012]: JQ782208
References
Altizer S, Harvell D, Friedle E (2003) Rapid evolutionary dynamics and disease threats to biodiversity. Trends Ecol Evol 18:589–596
Bassami MR, Ypelaar I, Berryman D, Wilcox GE, Raidal SR (2001) Genetic diversity of beak and feather disease virus detected in psittacine species in Australia. Virology 279:392–400
Bert E, Tomassone L, Peccati C, Navarrete MG, Sola SC (2005) Detection of beak and feather disease virus (BFDV) and avian polyomavirus (APV) DNA in psittacine birds in Italy. J Vet Med Ser B Infect Dis Vet Public Health 52:64–68
Boni MF, Posada D, Feldman MW (2007) An exact nonparametric method for inferring mosaic structure in sequence triplets. Genetics 176:1035–1047
Cleveland S, Hess GP, Dobson AP, Laurenson MK, McCallum HI, Roberts MG, Woodroffe R (2002) The role of pathogens in biological conservation. The ecology of wildlife diseases. Oxford University Press, Oxford, pp 139–150
Clout MN, Merton DV (1998) Saving the Kakapo: the conservation of the world’s most peculiar parrot. Bird Conserv Int 8:281–295
Collar NJ, Gonzaga LP, Krabbe N, Madroño Nieto A, Naranjo LG, Parket TA, Wege DC (1992) Threatened birds of the Americas: the ICBP/IUCN Red Data Book. International Council for Bird Preservation, Cambridge
Elliott GP, Dilks PJ, O’Donnell CFJ (1996) The ecology of yellow-crowned parakeets (Cyanoramphus auriceps) in Nothofagus forest in Fiordland, New Zealand. N Z J Zool 23:249–265
Frankham R, Ballou JD, Brisjoe DA (2002) Introduction to conservation genetics. Cambridge University Press, Cambridge
Gartrell BD, Alley MR, Mack H, Donald J, McInnes K, Jansen P (2005) Erysipelas in the critically endangered kakapo (Strigops habroptilus). Avian Pathol 34:383–387
Gibbs MJ, Armstrong JS, Gibbs AJ (2000) Sister-scanning: a Monte Carlo procedure for assessing signals in recombinant sequences. Bioinformatics 16:573–582
Gibbs MJ, Smeianov VV, Steele JL, Upcroft P, Efimov BA (2006) Two families of Rep-like genes that probably originated by interspecies recombination are represented in viral, plasmid, bacterial, and parasitic protozoan genomes. Mol Biol Evol 23:1097–1100
Grant AD, Kearvell J (2001) Orange-fronted parakeet (Cyanoramphus malherbi) recovery plan, 1999–2005. Department of Conservation, Wellington
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Ha HJ, Anderson IL, Alley MR, Springett BP, Gartrell BD (2007) The prevalence of beak and feather disease virus infection in wild populations of parrots and cockatoos in New Zealand. N Z Vet J 55:235–238
Hale KA, Briskie JV (2007) Decreased immunocompetence in a severely bottlenecked population of an endemic New Zealand bird. Anim Conserv 10:2–10
Hamel J (1970) Hybridisation of eastern and crimson rosellas in Otago. Notornis 17:126–129
He CQ, Ding NZ, Fan W, Wu YH, Li JP, Li YL (2007) Identification of chicken anemia virus putative intergenotype recombinants. Virology 366:1–7
Horlen KP, Dritz SS, Nietfeld JC, Henry SC, Hesse RA, Oberst R, Hays M, Anderson J, Rowland RR (2008) A field evaluation of mortality rate and growth performance in pigs vaccinated against porcine circovirus type 2. J Am Vet Med Assoc 232:906–912
Johne R, Fernandez-de-Luco D, Hofle U, Muller H (2006) Genome of a novel circovirus of starlings, amplified by multiply primed rolling-circle amplification. J Gen Virol 87:1189–1195
Keller LF, Waller DM (2002) Inbreeding effects in wild populations. Trends Ecol Evol 17:230–241
Kiatipattanasakul-Banlunara W, Tantileartcharoen R, Katayama K, Suzuki K, Lekdumrogsak T, Nakayama H, Doi K (2002) Psittacine beak and feather disease in three captive sulphur-crested cockatoos (Cacatua galerita) in Thailand. J Vet Med Sci 64:527–529
Kock RA, Woodford MH, Rossiter PB (2010) Disease risks associated with the translocation of wildlife. Rev Sci Tech 29:329–350
Koonin EV, Senkevich TG, Dolja VV (2006) The ancient virus world and evolution of cells. Biol Direct 1:29
Kundu S, Faulkes CG, Greenwood AG, Jones CG, Kaiser P, Lyne OD, Black SA, Chowrimootoo A, Groombridge JJ (2012) Tracking viral evolution during a disease outbreak: The rapid and complete selective sweep of a circovirus in the endangered Echo parakeet. J Virol. doi:10.1128/JVI.06504-06511
Lefeuvre P, Lett JM, Varsani A, Martin DP (2009) Widely conserved recombination patterns among single-stranded DNA viruses. J Virol 83:2697–2707
Li LL, Kapoor A, Slikas B, Bamidele OS, Wang CL, Shaukat S, Masroor MA, Wilson ML, Ndjango JBN, Peeters M, Gross-Camp ND, Muller MN, Hahn BH, Wolfe ND, Triki H, Bartkus J, Zaidi SZ, Delwart E (2010) Multiple diverse circoviruses infect farm animals and are commonly found in human and Chimpanzee feces. J Virol 84:1674–1682
Malham J, Kovac E, Reuleaux A, Linnebjerg JF, Tollington S, Raisin C, Marsh P, McPherson S (2008) Results of screening echo and ringneck parakeets for psittacine beak and feather disease in mauritius march 2008. Report to the Mauritian Wildlife Foundation, National Parks and Conservation Service of Mauritius; International Zoo Veterinary Group, Durrell Wildlife Conservation Trust, IBL Aviation, Shipping and Other Services; Chester Zoo; and the World Parrot Trustand Other Services; Chester Zoo; and the World Parrot Trust
Martin D, Rybicki E (2000) RDP: detection of recombination amongst aligned sequences. Bioinformatics 16:562–563
Martin DP, Posada D, Crandall KA, Williamson C (2005) A modified bootscan algorithm for automated identification of recombinant sequences and recombination breakpoints. AIDS Res Hum Retroviruses 21:98–102
Martin DP, Lemey P, Lott M, Moulton V, Posada D, Lefeuvre P (2010) RDP3: a flexible and fast computer program for analyzing recombination. Bioinformatics 26:2462–2463
Ortiz-Catedral L, McInnes K, Hauber ME, Brunton DH (2009) First report of beak and feather disease virus (BFDV) in wild Red-fronted Parakeets (Cyanoramphus novaezelandiae) in New Zealand. Emu 109:244–247
Ortiz-Catedral L, Kurenbach B, Massaro M, McInnes K, Brunton DH, Hauber ME, Martin DP, Varsani A (2010) A new isolate of beak and feather disease virus from endemic wild red-fronted parakeets (Cyanoramphus novaezelandiae) in New Zealand. Arch Virol 155:613–620
Padidam M, Sawyer S, Fauquet CM (1999) Possible emergence of new geminiviruses by frequent recombination. Virology 265:218–225
Paré JA, Robert N (2007) Circoviruses. Infectious diseases of wild birds. Blackwell Publishing, Melbourne, pp 194–205
Parker KA, Brunton DH, Jakob-Hoff R (2006) Avian translocations and wildlife disease; implications for New Zealand conservation. Pac Conserv Biol 12:155–162
Pass DA, Perry RA (1984) The pathology of psittacine beak and feather disease. Aust Vet J 61:69–74
Phenix KV, Weston JH, Ypelaar I, Lavazza A, Smyth JA, Todd D, Wilcox GE, Raidal SR (2001) Nucleotide sequence analysis of a novel circovirus of canaries and its relationship to other members of the genus Circovirus of the family Circoviridae. J Gen Virol 82:2805–2809
Posada D, Crandall KA (2001) Evaluation of methods for detecting recombination from DNA sequences: computer simulations. Proc Nat Acad Sci USA 98:13757–13762
Rahaus M, Wolff MH (2003) Psittacine beak and feather disease: a first survey of the distribution of beak and feather disease virus inside the population of captive psittacine birds in Germany. J Vet Med Ser B Infect Dis Vet Public Health 50:368–371
Raidal SR, Sabine M, Cross GM (1993) Laboratory diagnosis of psittacine beak and feather disease by hemagglutination and hemagglutination inhibition. Aust Vet J 70:133–137
Raidal SR, Cross GM (1994) The hemagglutination spectrum of psittacine beak and feather disease virus. Avian Pathol 23:621–630
Ritchie BW, Niagro FD, Lukert PD, Steffens WL, Latimer KS (1989) Characterization of a new virus from cockatoos with psittacine beak and feather disease. Virology 171:83–88
Ritchie BW, Niagro FD, Latimer KS, Lukert PD, Steffens WL, Rakich PM, Pritchard N (1990) Ultrastructural, protein-composition, and antigenic comparison of psittacine beak and feather disease virus purified from 4 genera of psittacine birds. J Wildl Dis 26:196–203
Ritchie BW, Niagro FD, Latimer KS, Steffens WL, Pesti D, Lukert PD (1991) Hemagglutination by psittacine beak and feather disease virus and use of hemagglutination inhibition for detection of antibodies against the virus. Am J Vet Res 52:1810–1815
Ritchie BW (1995) Circoviridae. Avian viruses, function and control. Wingers Publishing Inc, Lake Worth, pp 223–252
Ritchie PA, Anderson IL, Lambert DM (2003) Evidence for specificity of psittacine beak and feather disease viruses among avian hosts. Virology 306:109–115
Rosario K, Marinov M, Stainton D, Kraberger S, Wiltshire EJ, Collings DA, Walters M, Martin DP, Breitbart M, Varsani A (2011) Dragonfly cyclovirus, a novel single-stranded DNA virus discovered in dragonflies (Odonata: Anisoptera). J Gen Virol 92:1302–1308
Sanada Y, Sanada N, Kubo M (1999) Electron microscopical observations of psittacine beak and feather disease in an umbrella cockatoo (Cacatua alba). J Vet Med Sci 61:1063–1065
Seutin G, White BN, Boag PT (1991) Preservation of avian blood and tissue samples for DNA analyses. Can J Zool (Revue Canadienne De Zoologie) 69:82–90
Shepherd DN, Martin DP, Lefeuvre P, Monjane AL, Owor BE, Rybicki EP, Varsani A (2008) A protocol for the rapid isolation of full geminivirus genomes from dried plant tissue. J Virol Methods 149:97–102
Smith JM (1992) Analyzing the mosaic structure of genes. J Mol Evol 34:126–129
Stewart ME, Perry R, Raidal SR (2006) Identification of a novel circovirus in Australian ravens (Corvus coronoides) with feather disease. Avian Pathol 35:86–92
Taylor RH (1985) Status, habits and conservation of Cyanoramphus parakeets in the New Zealand region. In: Moors PJ (ed) Conservation of Island birds. International council for bird preservation technical publication no. 3. Cambridge University Press, Cambridge, pp 195–211
Todd D, Weston J, Ball NW, Borghmans BJ, Smyth JA, Gelmini L, Lavazza A (2001) Nucleotide sequence-based identification of a novel circovirus of canaries. Avian Pathol 30:321–325
Todd D, Bendinelli M, Biagini P, Hino S, Mankertz A, Mishiro S, Neil C, Okamoto H, Raidal S, Ritchie BW, Teo GC (2005) Circoviridae. In: Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA (eds) Virus taxonomy, eighth report of the international committee on taxonomy of viruses. Elsevier, London, pp 327–341
Todd D, Scott ANJ, Fringuelji E, Shivraprasad HL, Gavier-Widen D, Smyth JA (2007) Molecular characterization of novel circoviruses from finch and gull. Avian Pathol 36:75–81
Tompkins DM, Mitchell RA, Bryant DM (2006) Hybridization increases measures of innate and cell-mediated immunity in an endangered bird species. J Anim Ecol 75:559–564
Twentyman CM, Alley MR, Meers J, Cooke MM, Duignan PJ (1999) Circovirus-like infection in a southern black-backed gull (Larus dominicanus). Avian Pathol 28:513–516
Varsani A, de Villiers GK, Regnard GL, Bragg RR, Kondiah K, Hitzeroth II, Rybicki EP (2010) A unique isolate of beak and feather disease virus isolated from budgerigars (Melopsittacus undulatus) in South Africa. Arch Virol 155:435–439
Varsani A, Regnard GL, Bragg R, Hitzeroth II, Rybicki EP (2011) Global genetic diversity and geographical and host-species distribution of beak and feather disease virus isolates. J Gen Virol 92:752–767
Woods LW, Latimer KS (2000) Circovirus infection of nonpsittacine birds. J Avian Med Surg 14:154–163
Wright D, Clout M (2001) The Eastern rosella (Platycercus eximius) in New Zealand. DOC science internal series 18. Department of Conservation, Wellington
Acknowledgments
This research was completed with the logistical and financial support of the Brian Mason Scientific and Technical Trust (New Zealand), the University of Canterbury (Science Faculty Early Career Grant), the Department of Conservation, Massey University (Institute of Natural Sciences), the University of Auckland, Motuihe Island Trust, Tawharanui Open Sanctuary Supporters Inc., Auckland Regional Council, and National Council of Science from Mexico (CONACYT). Sample collection was undertaken following methods approved by the DOC (permits AK-15300-RES, AK-20666-FAU, AK-22857-FAU, and 2008070), Massey University Animal Ethics Committee (protocols MUAEC 07/138 and 08/24), and the University of Auckland Animal Ethics Committee (protocol R653). Financial support for the field trip to Rangatira Island in Nov–Dec 2009 was provided through a postdoctoral fellowship by the Foundation for Science, Research & Technology to MM (UOCX0601). B.K. is supported by funding from the Norwegian Agency for Development Cooperation (NORAD). We thank the numerous volunteers who assisted with capture and processing of birds. Special thanks go to Shane McInnes and Liz Whitwell (DOC), who greatly facilitated our work on Little Barrier Island. We thank Alison Botha, Kev Drew, Sophie Fern, Alexandra Sides and Annika Wagenhoff for assistance in the field on Rangatira Island. Abigail Liddy from the Department of Conservation in the Chatham Islands provided tremendous logistical support. We thank Graeme Elliott and Kath Walker for organizing the Adams Island Expedition, and James Armstrong for help with capture and handling of parakeets on Adams Island.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Massaro, M., Ortiz-Catedral, L., Julian, L. et al. Molecular characterisation of beak and feather disease virus (BFDV) in New Zealand and its implications for managing an infectious disease. Arch Virol 157, 1651–1663 (2012). https://doi.org/10.1007/s00705-012-1336-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-012-1336-5


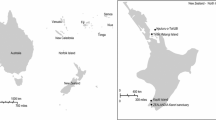



 contains a number of BFDV-positive individuals. Arrows point towards the location of offshore island groups (color figure online)
contains a number of BFDV-positive individuals. Arrows point towards the location of offshore island groups (color figure online)


