Abstract
The rising temperatures and decreasing rainfall are expected to have negative effects on ecosystem services by causing significant shrinkage or shift of forest distribution particularly in Mediterranean basin. In this study, it was aimed to determine the distribution of oriental beech (Fagus orientalis Lipsky.) by modelling the current and future potential locations of the habitats. With Maximum Entropy (MaxEnt) approach, we predicted its distribution under current and future conditions (RCP 4.5 and RCP 8.5) in Turkey. Modelling was performed by using eight bioclimatic variables that show significant relationship to the current distribution of oriental beech and were widely used in the literature. The fitted model had high quality (93.5% AUC) and is biological meaningful. The prediction of warmer condition in future showed that the populations on the southern slopes of the North Anatolian Mountains were expected to shrink and that there would be reduction in the populations found in the main distribution area in the Istranca Mountains and the Northern Anatolia, especially in populations in the transition zone of Central Anatolia. Also, oriental beech would lose its isolated-marginal populations in the southeast of Turkey. The results highlight the importance of a genetic conservation programme for beech population in Turkey. Otherwise, the genetic pools seem to extinct under climate change. Furthermore, the paper is intended to provide a starting point for a monitoring of oriental beech at the edge of its distribution, to observe its climatic migration.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Oriental beech (Fagus orientalis Lipsky.), of which a significant portion of geographical distribution is mostly in Turkey, is also distributed in Bulgaria, Greece, Crimea, the Caucasus, and Iran. It is an economically and ecologically important species (Ertekin et al. 2015). The most suitable growth conditions are the north-facing slopes of the Northern Anatolian orogenic belt located in the Black Sea region and the Istranca Mountains in Thrace (Yaltırık 1982; Ertekin et al. 2015).
Including the Anatolian Peninsula, the entire Mediterranean basin is considered as one of the most vulnerable regions to climate change in the world (IPCC 2007; Ozdemir et al., 2011; Kovats et al. 2014). According to UNDP (2019), Turkey is included among the “countries at risk” that are highly vulnerable to climate change (UNDP 2019). Global climate change will rise to occasion with the rise of temperatures and a decrease in precipitation in the Mediterranean Basin (Giorgi and Lionello 2008). The rising temperatures are expected to have negative effects on ecosystem services such as water conservation and production, climate regulation, soil retention and flood control by causing significant changes in ecosystems.
Talu et al. (2011) state that climate change will affect Turkey through climate-related natural disasters such as frequent forest fires, droughts, floods, desertification and erosion as well as an increase in ecological degradation rate and depletion of water resources. Toros (2012) underlines that the rise in temperature during warm periods will be higher than the rise in temperature during cold periods. (Tayanc et al., 2009) emphasizes that there is a significant warming trend since 1993 in Turkey, and that the maximum temperatures in history have been recorded in recent years, while Sen (2013) reports that the average summer temperature in the 2000s in Turkey is about 1.5 °C higher compared to the temperature 30–40 years ago. Demircan et al. (2017) predicts that the snow will melt early in the spring due to this rise in temperature. In a study conducted by Erlat and Turkes (2011), it was stated that there was an overall reduction in the annual number of frost days in many stations around Turkey during the period between 1950 and 2010. Sen (2013) reported that no significant change was observed in the precipitation during the period, when climate records were kept, except north-eastern Turkey where precipitation increased regionally. In spite of that, future projections indicate that the annual total precipitation will decrease, especially in Central Anatolia and South-eastern Anatolia. Dalfes et al. (2007) stated that during the period between 1951 and 2004, winter precipitation considerably decreased in the western provinces of Turkey, whereas autumn precipitation increased in the stations located in the northern part of Central Anatolia. According to García Ruiz et al. (2011), precipitation decreased in the western coastline of Turkey between 1950 and 2002, despite the positive trend observed in the north of Turkey. Yalcin (2012) reported that the water potential of Turkey decreased down to 163.79 billion m3 from 178.15 billion m3 in the period between 1989 and 2009 depending on the decrease observed in the runoff.
According to future climate projections for the period between 2071 and 2100, it was stated that Turkey’s average annual temperature would increase all across the country. The average temperature was expected to rise up to 6 °C, especially in the Aegean region (Dalfes et al. 2007). Sen (2013) stated that the average summer temperature will rise around 1.0 to 2.5 °C in the mid-twenty-first century and 5 to 5.0 °C by the end of the twenty-first century. Moreover, winter temperature increase was estimated to be higher in the eastern half of the country during this period. In the same period, it was stated that the precipitation would decrease along the Aegean and Mediterranean coastlines as well as Turkey’s Black Sea coastline, while little or no change would be observed in Central Anatolia (Dalfes et al. 2007). While significant decrease in precipitation was expected to be observed in the south-western coast of Turkey, more precipitation was expected to occur in the coastal region of the Caucasus, and less winter precipitation in the Mediterranean coast (Yalcin 2012). Talu et al. (2011) estimate that there will be a greater decrease in summer precipitation in the northern half of Turkey compared to the southern half.
Forests are one of the ecosystems most affected by climate change. Adaptation of trees, which can survive for centuries or even for thousands of years, to rapid change in the climate is not that easy (Lindner et al., 2010). For this reason, local extinctions and loss of important functions and services are expected (Keenan 2012). Several dendroclimatological studies show that the most important factor of annual ring growth in Turkey is the precipitation occurring in in May and June (Akkemik 2000; Akkemik et al. 2008). In addition to this limiting effect of precipitation, high temperatures experienced in the same period increase the severity of drought and limit radial growth (Kose et al. 2012, 2017; Guner et al. 2017; Akkemik et al. 2005, 2008; Touchan et al. 2007). Over the last two decades, researches on the effects of climate change, species adaptation and migration have increased significantly.
Oriental beech forests in Turkey are about 2.05 million hectares, which corresponds to 8.5% (OGM 2015) of the country’s total forest area. The altitude range of the oriental beech in the Balkans is 10 to 800 m (m.a.s.l.) above sea level. In Turkey, it goes up to 1500 to 1700 m in the Black Sea valleys and up to 2000 m in the Aegean mountains (Atay 1982; Atalay 1992; Ata 1995; Ozel and Ertekin 2011; Ertekin et al. 2015). Apart from Northern Anatolia, where it is mainly distributed, oriental beech residual forests are also available in the Eastern Mediterranean region of Turkey including Adana, Osmaniye, Hatay and Kahramanmaras (Yılmaz, 2010). Although oriental beech forests generally form mix stands with Caucasian fir (Abies nordmanniana (Steven) Spach), nordmann fir (A. nordmanniana (Stev. Spach. subsp. bornmuelleriana (Mattf.) Coon & Cullen), Ida Mountain fir (A. equitrojani Aschers et Sinten), Scotspine (Pinus sylvestris L.), Anatolian black pine (P. nigra Arnold subsp. pallasiana (Lamb.) Holmboe) and oriental spruce (Picea orientalis (L.) Link.) at high altitudes in Turkey. However, oriental beech also occurs in large areas in pure stands (Atalay 1992; Ata 1995; Ertekin et al. 2015). The distribution patterns of the F. orientalis in the south-eastern Balkans suggest that F. orientalis may occur on drier and warmer sites than F. sylvatica (Kandemir and Kaya 2009).
Modelling the possible future distribution areas of species such as climate change-sensitive oriental beech (Martin-Benito et al. 2018; Kose and Guner 2012) may be a significant guide in terms of forest protection and management. Species distribution models are among the most effective and widely used methods in understanding future distribution of species (Dyderski et al. 2018; Kramer et al. 2010). These models help to identify zones similar to the current distribution areas for species, in case the environmental variables change, by using the current distribution data of the species and various environmental variables (Pearson 2007; Li and Wang 2013; Elith and Leathwick 2009).
In this study, it is aimed to support high-accuracy silvicultural decisions in conservation and management practices by examining possible changes due to climate change in oriental beech forests towards the end of this century. For this aim, modelling was carried out with Maximum Entropy (MaxEnt) software using 8 different bioclimatic variables according to Representative Concentration Pathway (RCP) 4.5 and RCP 8.5 global climate change scenarios in 2050 and 2070 from WorldClim database. Specifically, we pursue the following objectives: (i) to model the present distribution of F. orientalis, and (ii) to project the future distribution of F. orientalis under different climate change scenarios.
2 Material and method
2.1 Study region and occurrence data
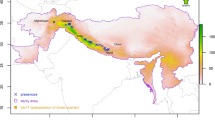
The current study focused on the distribution of oriental beech in the borders of Turkey. Oriental beech occurrence data were obtained from forest management plans of the General Directorate of Forestry of Turkey (GDF, traceable reference of occurrence data) which are much more sensitive spatial data than European Forest Genetic Resources Programme (EUFORGEN). In this study, we used 70% training and 30% test data set to validate the model across the entire dataset. The spatial data of the oriental beech used in the study and the location of the study area are given in Fig. 1.
2.2 Climate data
For our investigation, we used the WorldClim 1.4 (WorldClim 2019, 2.5-min spatial resolution). The climate data comprised the period from 1960 to 1990. The data is widely used in modelling studies for species distribution (Panagos et al. 2017). In this platform, 19 bioclimatic variables are available which represent the types of seasonal trends pertinent to the physiological constraints of different species (Hijmans et al. 2005; O’Donnell and Ignizio 2012). All these bioclimatic variables and distributions related to beech were tested before the models were introduced. Highly correlated variables to best express the natural distribution of oriental beech were identified and screening was conducted among the bioclimatic variables. The eight most important bioclimatic variables, which are frequently used and widely used in species distribution modelling studies and also directly affect the natural distribution of oriental beech, were used in this study to optimize the future distribution of eastern beech in the literature (Graham 2003; Garcia et al. 2013; Haghshenas et al. 2016a, b; Qin et al. 2017; Yaman et al. 2021). In the study, eight variables are used as seen Table 1.
Hadley Global Environment Model 2-Earth System (HadGEM2-ES) 2.5-min spatial resolution featured climate model was used in this study also from the WorldClim 1.4 dataset. This global circulation model was selected because it fits best our assumptions for the climatic change in Turkey (see supplementary data 1 (S1a and S1b) and mentioned climatic assumptions in Sect. 1). We prefer the usage of one global circulation model instead of ensemble. In our case, we expect this to simplify the interpretation of results. That is why, we have applied four climatic situations to observe under which conditions range loss of beech starts. The most extreme and intermediate scenario were selected from HadGEM2-ES: the former achieving 8.5 W/m2 by 2100 (RCP 8.5), while the latter an increase of 4.5 W/m2 (RCP 4.5), also in the same deadline. These scenarios represent the concentration of greenhouse gases and pollutants resulting from human activity, adopted by the Fifth Assessment Report (IPCC 2014). Two time slots were studied for each RCP: 2050 (average for 2041–2060) and 2070 (average for 2061–2080).
2.3 Statistical model
The distribution of beech was fitted by Maximum Entropy. The approach uses the principle of Maximum Entropy on presence only data to estimate a set of functions that relate environmental variables and habitat suitability in order to approximate the species’ niche and potential geographic distribution (Phillips et al. 2006). It is a discriminative modelling technique, meaning it fits species occurrences relative to available habitat in a model as uniform as possible between two probability densities (Elith et al. 2011). Although Maximum Entropy was designed to use current only data, it also performs well when compared to presence absence procedures that utilize both real and pseudo absence data (Elith et al. 2006). Validation of the models created with receiver operating characteristic (ROC) curve, which was widely used in a wide variety of modelling studies, and area under curve (AUC), which expressed the area under this curve as well as the level of success of the model, was performed (Metz 1978; Alonzo and Pepe 2002; Bugday 2018; Laaribya et al. 2021). In ecological modelling studies, also the Jackknife test is applied in order to reveal the predictive sensitivity of the model (Efron 1982). In this validation method, the models with AUC values of 0.5 and above are taken into consideration. Here, the success of the model is defined by its proximity to the number 1. In the literature, the fact that the AUC value is 0.65–0.75 indicates a medium-scale model success, while values over 0.75 indicate an acceptable model success, and 0.95 and above indicate that the model success is excellent (Hanley and McNeil 1982; Rice and Harris 2005; Tharwat 2018).
In order to be able to model the existing and potential distribution areas, MaxEnt 3.4.1 software (Steven et al. 2020) was used, and map displays and the calculation of area gain or loss and species shift (Table 2) were made with ArcGIS 10.3 software (ESRI 2011).
3 Results
As a result of the modelling done within the scope of this study, validation area under curve (AUC) value was found to be 0.935 (93.5%). This value indicates that modelling is done precisely and model quality is quite high (Fig. 2).
As a result of the Jackknife test applied in this study, Bio 2 (mean diurnal range (mean of monthly (max temp–min temp))) was defined as the most significant variable with a rate of 90.3%. This was followed by Bio 14 (precipitation of driest month) with 78.2% and Bio 18 (precipitation of warmest quarter) with 72.9%, respectively (Fig. 3).
The model results obtained based on the current and future (the years of 2050 and 2070) projections carried out in this study were mapped and displayed as below (Figs. 4, 5 and 6).
According to the RCP 4.5 scenario for the years of 2050 and 2070, it is predicted that oriental beech will lose its isolated and marginal populations found in Adana, Osmaniye, Hatay and Kahramanmaras provinces of eastern Mediterranean region of Turkey, and also its populations found on Istranca Mountains in Thrace will shrink and reduce. Also, it is predicted that in the Eastern Black Sea region, which is the main distribution area of the species, the populations will continue to exist on the northern slope of the mountains, where the humidity and precipitation economy are more advantageous; therefore, it will remain its existence in a narrower lane than its current distribution (Figs. 5 and 6).
According to the RCP 8.5 scenario for the years of 2050 and 2070, it is predicted that the oriental beech will lose its isolated and marginal populations in Adana, Osmaniye, Hatay and Kahramanmaras provinces, and also there will be reduction and significant spatial losses in populations inserted from the Black Sea towards Central Anatolia in Tokat, Corum and Amasya provinces as well as Istranca Mountains in Thrace. Along with the narrowing of the populations on the Istranca Mountains, it is understood that there will be a reduction in the populations found in Marmara region, especially in Balikesir province (Figs. 7 and 8).
Accordingly, current and future (years of 2050 and 2070) spatial distribution amounts and percentage changes are given in Table 2. Overall, it was found that there would be a reduction in terms of distribution of this species in the future, and that the species would lose its distribution in the south of Turkey. The total habitat suitability area decreased gradually from 2050 to 2070 under the moderate emission scenario (RCP 4.5) and the severe emission scenario (RCP 8.5) (Table 2).
As a result of the modelling, according to the projection obtained by using the RCP 4.5 scenario for 2050, it was predicted that the lowest vertical distribution of oriental beech species in Turkey would be around the coastlines in Zonguldak-Eregli in Western Blacksea Region and Istanbul-Sile (0 m) in Marmara Region. The highest distribution of oriental beech was projected to be around Bursa-Uludag (2000 m) and Bolu-Dortdivan (1950 m). According to the projection obtained by using the RCP 8.5 scenario for 2050, it was predicted that the lowest vertical distribution of Fagus orientalis in Turkey would be around the coastlines in Zonguldak-Eregli and Sinop (0 m), whereas the vertically highest distribution of oriental beech would be around Bursa-Uludag (2000 m) and Artvin-Borcka (1980) m.
As a result of the modelling, according to the projection obtained by using the RCP 4.5 scenario for 2070, it was predicted that the lowest vertical distribution of oriental beech species in Turkey would be around the coastlines in Sakarya-Karasu and Bartin-Kurucasile (0 m). The highest distribution of the oriental beech was projected to be around Artvin-Borcka (2010 m) and Bursa-Uludag (2000 m). According to the projection obtained by using the RCP 8.5 climate change scenario for 2070, it was predicted that the lowest vertical distribution of beech in Turkey would be around the coastlines in Zonguldak-Eregli and Kastamonu-Inebolu (0 m), whereas the vertically highest distribution of oriental beech would be around the areas located on the North of Bolu-Seben (2070 m) and around Bursa-Uludag (1900 m).
Until 2070 in Turkey, it is predicted that the horizontal distribution of oriental beech will become very limited in the southern parts, that it will expand in the central and north-eastern direction and that the density of oriental beech will increase in these areas. According to the RCP 8.5 scenario for 2050 and 2070, it is predicted that oriental beech will be extinct in the southern parts. According to WorldClim 2050 and 2070 climate change scenarios, the spatial distribution of beech in Turkey will be between 41°53′N latitude and 26°49′E longitude, and 39°51′N latitude and 41°48′E longitude.
4 Discussion and conclusions
It is well known that global climate change strongly affects plant distribution and survival. In this study, which aimed to model the distribution of oriental beech in Turkey, a total of eight bioclimatic variables were used. Evaluations were made based on two different climate scenarios, RCP 4.5 and RCP 8.5, which were obtained from WorldClim. According to these two different climate scenarios, it is predicted that the natural distribution area of oriental beech, which is one of the important primary forest tree species in in Turkey and has important silvicultural functions for forest ecosystems, will reduce; that it will decrease to a great extent in the south and southwest locations of Turkey until 2070 and that it will completely lose its isolated marginal populations in Amanos mountains. Moreover, it is predicted that there will be reductions in its populations found in the Istranca Mountains, in the South of Marmara region and in the area between Central Black Sea and Black Sea regions.
Considering the MaxEnt’s projections based on optimistic, moderate and severe scenarios for 2070, it is also foreseen by Dagtekin et al. (2019) that there will be a severe reduction in the geographical distribution of oriental beech, and the species will continue its existence, especially in the Caucasus. The results of MaxEnt showed that it could not catch the distribution on the Amanos Mountains for the current model. In opposition to MaxEnt outputs, random forest (RF) gave the result that there would be a small expansion in the southern Caspian Sea. This supports the idea of this region becoming a shelter region for the future (Dagtekin et al. 2019).
Many studies conducted emphasize that the density of Fagus sylvatica L. and Picea abies (L.) Karst. (Köble Seufert, 2001; EUROSTAT 2018), which are the most common species of Boreal and European forests, and are economically valuable, especially for their role in lumber industry, will decrease due to climate change in European forests (Kellomaki et al. 2001; Hanewinkel et al. 2013; Ruiz-Labourdette et al. 2013; Forest Europe 2015), and it is stated that species with more thermophilic character will be found in the distribution areas of these species (Kullman 2008).
In a study conducted by Thurm et al. (2018), it is stated that there will be a 56% decrease in potential distribution area of F. sylvatica, and this result supports the previous studies assuming a large loss of habitat suitability for F. sylvatica (Maiorano et al. 2013; Meier et al. 2012). In addition, Haghshenas et al (2016a, b) emphasized oriental beech may be sensitive to increasing temperatures in the Caspian forests, north of Iran. However, this result contradicts with the studies of Dyderski et al. (2018) and Falk and Hempelmann (2013) stating that there will be an increase in the distribution. The determining factor of gaining or losing in the distribution area of a species under global warming is closely related to the latitude centre and temperature adaptation of the current distribution. This situation confirms the hypothesis of the gain in distribution area of the southern tree species is stronger than the northern tree species (Thurm et al. 2018). Other studies focusing on mountainous areas show that climate change causes most of the studied plant species to shift upwards (Lenoir et al. 2008; Penuelas and Boada 2003; Rigling et al. 2013; Walther et al. 2005).
In a study conducted by Yalcin (2012) on the natural distribution area of oriental beech species in Turkey, it is stated that the change in suitable areas would be dramatic, most of the existing suitable areas would disappear by the end of 2050, and based on optimistic predictions, only 3–7% of current stands would remain suitable by 2080. Yalcin (2012) states that especially in Kastamonu province, the protected areas would remain along the Kure Mountains, for example most of the oriental beech forests in Kure Mountains National Park would be preserved. Also, it was emphasized that there would be some gain along the Ilgaz Mountains, another compact area would remain at high altitudes above 1000 m of Yenice forests; however, most of the existing beech distribution around the Uludag Mountains would be lost, and that the areas at high altitudes, where forest cover was not currently available, would become suitable for the oriental beech species in the future (Yalcin 2012).
In another study conducted in Georgia and the Western Caucasus, it was found that the effect of drought was quite low in oriental beech trees grown at high altitudes, while high temperatures in spring and summer increased the development of annual rings. It was predicted that the increase in temperature foreseen in the climate change scenarios could accelerate the growth of the oriental beech, which grew in the middle and upper altitudes in this region. The study results showed that the region could act as a potential shelter, especially considering the more arid regions surrounding the Western Caucasus, and emphasized that the naturally aged forests in this region must be preserved (Martin-Benito et al. 2018).
In our study, in severe emission scenario conditions in both 2050 and 2070 periods, fewer area losses were detected in the distribution area of oriental beech than in the moderate emission scenario, interestingly. As it known, oriental beech, under Euxinian climate (a type of climate typical from northern aspects of the Black Sea Region) characterized by a totally different precipitation pattern (substantial summer rain) and higher humidity (López-Tirado et al. 2021), makes its widest and most suitable spread in Turkey in the middle and high parts of the mountains running parallel to the Black Sea coast, and especially in the pure and mixed forests, it forms in the northern mountains (Kandemir et al. 2016). The fact that the oriental beech shows less habitat loss in the severe emission scenario may be based on the positive effect of possible increased humidity due to the effect of increased temperature. The effect of Bio18 (Precipitation of Warmest Quarter) in modelling can be considered as a separate basis for explaining this situation. The Amanos Mountains are distinctive for this species, because the oriental beech of this is home to some relict populations (Avci 2018). However, Dagtekin et al. (2019) stated that they described a region of high importance such as Amanos Mountains as accurately as possible with the results of random forest (RF) community classifier. Future climatic conditions are expected to cause the current distribution of oriental beech to shift higher altitudes due to more mild and rainy climatic conditions. The dendroclimatological studies in the region support the model findings and designate that the oriental beech grown in the middle and upper altitudes may increase the growth by benefiting from the rising spring–summer temperatures, and that this region may be a potential shelter area in the future (Martin-Benito et al. 2018; Dagtekin et al. 2019).
Due to the fact that the percentage of empty seeds in the populations in the most southern part of the Fagus crenata distribution in Japan is high, it has been determined that the regeneration ability of the forest has weakened and the diameter increase has decreased in the last 50 years due to global warming. For this reason, it was assumed that those populations could be replaced with other species (Mizunaga et al. 2005). Similarly, Yilmaz (2010) pointed out that some unprecedented characteristics were observed in the oriental beech residual forests in response to summer drought in southern Anatolia, the empty seed rate was low and that the fragility of these forests significantly differed from the populations in the main distribution area.
The effects of climate change may vary depending on the ecological and geographical characteristics of a species. Oriental beech is a taxon to the areas that are moist in terms of ecological preferences and have summer rainfalls (Alsos et al. 2012). However, this species occupies a narrow ecological niche being sensitive to late spring frosts and summer drought (Peters 1997). Consequently, it has been identified as especially vulnerable to climate change effects (Kose and Guner 2012; Haghshenas et al. 2016a, b). Preservation of genetic diversity of a species is of a great importance for adapting to climate change. Genetic diversity loss is likely due to the narrowing of the species’ distribution areas (Alsos et al. 2012). Especially oriental beech may experience loss of genetic diversity due to the large distribution loss (Yalcin, 2012). Therefore, existing marginal populations are important tools to preserve genetic diversity in the future. They have already adapted to the optimum climatic conditions defined in relation to the main distribution of the species (Barbati et al. 2018). Establishing seed orchards with seed origin and/or clonal route is important as an ex situ protection measure with the production materials of marginal and edge populations. These orchards can be critical tools for afforestation works to be carried out in the future.
We recognize that species distribution models driven entirely by climate variables can only explain part of a species distribution. This can be seen also by the comparison of the real current distribution to the potential current distribution. Besides the anthropogenic influence, biotic effects and soil conditions have to be taken into account, especially on local level (Conedera et al. 2021). For our study, however, it was important to detect the large-scale patterns and these are mainly determined by the climate. Large-scale dieback caused by biotic pests, as we known from Chestnut blight fungus, also in Mediterranean region, is rather rare in oriental beech ecosystems. However, under increasing temperature, the impact of alien species on beech can increase and is very hard to predict.
Monitoring populations at the limits of their bioclimatic suitability will provide more information about species responses to climate change. A network of such monitoring stations should be immediately set up. Monitoring the bioclimatic suitability of the populations at the borders of distribution will provide more information on the species’ responses to climate change. Based on the results of this study, the distribution for oriental beech, genetic monitoring can start to save local population which might extinct in future. Establishing a network of such monitoring stations is a priority. In future studies, the application of both different modelling methods and the combination of different parameters will further improve dispersion modelling studies. At the same time, Dagtekin et al. (2020) stated that comparing the model results with the distribution data obtained from different sources such as pollen data, when applying species distribution models, helps to understand the quality and accuracy of the models. Consequently, modelling the past distribution areas and possible future distribution areas of species is an important guide in terms of conservation and sustainable management of forests. In the next process, it is thought that comparative studies can be made using different modelling methods including soil and topographic parameters.
5 Supplementary information
6 Ethics approval
Not applicable.
7 Consent for publication
All the authors consented to publish the paper.
8 Conflict of interests
The authors declare no competing interests.
Availability of data and material
The data belongs to the General Directory of Forestry, Turkey. That is why, we are not allowed to distribute the data ourselves, yet, it is available upon request to the General Directoryof Forestry, Turkey. Contactinformation: Republic of Turkey, General Directorate of Forestry Address: Beştepe Mahallesi Sogutozu Caddesi No:8/1 06,560 Yenimahalle/Ankara/TURKEY.
Code availability
Not applicable.
References
Kose N, Guner HT, Harley GL, Guiot J 2017 Spring temperature variability over Turkey since 1800CE reconstructed from a broad network of tree-ring data. Climate of the Past 13:1-15. https://doi.org/10.5194/cp-13-1-2017
Akkemik Ü (2000) Dendroclimatology of umbrella pine (Pinus pinea L.) in Istanbul. Turkey Tree-Ring Bulletin 56:17–20
Akkemik Ü, Dagdeviren N, Aras A (2005) A preliminary reconstruction (A.D. 1635–2000) of spring precipitation using oak tree rings in the west. Int J Biometeorol 49:297–302. https://doi.org/10.1007/s00484-004-0249-8
Akkemik Ü, D’Arrigo R, Cherubini P, Kose N, Jacoby G (2008) Tree-ring reconstructions of precipitation and streamflow for north-western Turkey. Int J Climatol 28:173–183. https://doi.org/10.1002/joc.1522
Alonzo TA, Pepe MS (2002) Distribution-free ROC analysis using binary regression techniques. Biostatistics 3(3):421–432. https://doi.org/10.1093/biostatistics/3.3.421
Alsos IG, Ehrich D, Thuiller W, Eidesen PB, Tribsch A, Schönswetter P, Lagaye C, Taberle TP, Brochmann C 2012 Genetic consequences of climate change for northern plants. Proceedings Biological sciences / The Royal Society, 279(1735), 2042 51 https://doi.org/10.1098/rspb.2011.2363
Ata C 1995 Si1viculture techniques (in Turkish). Zonguldak Karaelmas University, Bartın Faculty of Forestry, No: 3–4, Bartın, Turkey, 449 p.
Atalay İ 1992 Kayın (Fagus orientalis Lipsky.) Ormanlarının Ekolojisi ve Tohum Transferi Yönünden Bölgelere Ayrılması. Orman Bakanlığı, Orman Ağaçları ve Tohumları Islah Araştırma Müdürlüğü, Yayın No: 5, Ankara, Turkey, 209 p.
Atay İ 1982 Natural Regeneration Methods II, Dogal Gençleştirme Yöntemleri II. Istanbul University, Faculty of Forestry, Pub. No: 320, Istanbul, Turkey, 160 p.
Avci M 2018 Türkiye’nin Bitki Çeşitliliği ve Coğrafi Açıdan Değerlendirmesi (Ed. Ü. Akkemik) Türkiye’nin Doğal-Egzotik Ağaç ve Çalıları. Orman Genel Müdürlüğü Yayınları, Ankara. s: 30–44.
Barbati A, Scarascia G, Ayan S, Blasi E, Calama R, Canaveira P, Cicatiello C, Collalti A, Corona P, Del Rio M, Ducci F, Perugini L 2018 State of Mediterranean forests 2018. Chapter 8: Adaptation and mitigation, p. 128–146, Published by the FAO of the United Nations and Plan Bleu, Regional Activity Center of UN Environment / Mediterranean Action Plan, ISBN FAO: 978–92–5–131047–2 ISBN Plan Bleu: 978–2–912081–52–0, Rome. http://www.fao.org/3/CA2081EN/ca2081en.PDF
Bugday E (2018) Application of artificial neural network system based on ANFIS using GIS for predicting forest road network suitability mapping. Fresenius Environ Bull 27(3):1656–1668
Conedera M, Krebs P, Gehring E, Wunder J, Hülsmann L, Abegg M, Maringer J 2021 How future-proof is Sweet chestnut (Castanea sativa) in a global change context? In: Forest Eco Manag 494, S. 119320. https://doi.org/10.1016/j.foreco.2021.119320.
Dagtekin D, Şahan EA, Denk T, Köse N, Dalfes HN (2020) Past, present and future distributions of oriental beech (Fagus orientalis)under climate change projections. PLoS ONE 15(11):e0242280. https://doi.org/10.1371/journal.pone.0242280
Dagtekin D, Sahan EA, Denk T, Kose N, Dalfes HN 2019 Predicting Late Quaternary distribution and future refugia of oriental beech (Fagus orientalis Lipsky) in Asia minor. International Conference on Climate Change & Forestry (ICCCF’2019) 12–15 2019, TOD Publication Number: 49, Conference Proceeding, 177–182. Antalya.
Dalfes HN, Karaca M, Şen ÖL, Güven Ç (Ed.) (2007) Climate change scenarios for Turkey. Climate change & Turkey impacts, sectoral analyses, socio economic dimensions. Ankara: United Nations Development
Demircan M, Gürkan H, Eskioğlu O, Arabacı H, Coşkun M 2017 Climate change projections for Turkey three models and two scenarios. Turkish J Water Sci and Manag, 1 1 22 43. https://doi.org/10.31807/tjwsm.297183
Dyderski MK, Paź S, Frelich LE, Jagodziński AM (2018) How much does climate change threaten European forest tree species distributions? Glob Change Biol 24:1150–1163. https://doi.org/10.1111/gcb.13925
Efron B 1982 The Jackknife, the bootstrap, and other resampling plans. SIAM, monograph #38, CBMS-NSF.
Elith J, Leathwick JR (2009) Species distribution models: ecological explanation and prediction across space and time. Annu Rev Ecol Evol Syst 40:677–697. https://doi.org/10.1146/annurev.ecolsys.110308.120159
Elith J, Graham CH, Anderson RP, Dudik M, Ferrier S, Guisan A, Hijmans RJ, Huettmann F, Leathwick JR, Lehmann A, Li J, Lohmann LG, Loiselle BA, Manion G, Moritz C, Nakamura M, Nakazawa Y, McC Overton J, Peterson AT, Phillips SJ, Richardson KS, Scachetti-Pereira R, Schapire RE, Sobero´n J, Williams S, Wisz MS, Zimmermann NE, (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151. https://doi.org/10.1111/j.2006.0906-7590.04596.x
Elith J, Phillips SJ, Hastie T, Dudík M, Chee YE, Yates CJ 2011 A statistical explanation of MaxEnt for ecologists Divers Distrib 17 1 43 57. https://doi.org/10.1111/j.1472-4642.2010.00725.x
Erlat E, Turkes M 2011 Analysis of observed variability and trends in numbers of frost days in Turkey for the period 1950–2010 Int J Climatol. 32: 1889–1898. https://doi.org/10.1002/joc.2403
Ertekin M, Kırdar E, Ayan S 2015 Effects of tree ages, exposures and elevations on some seed characteristics of oriental beech (Fagus orientalis Lipsky.), Journal of South-east European Forestry, 6 1 15 23. https://doi.org/10.15177/seefor.15-03
ESRI 2011 ArcGIS desktop: release 10. Redlands, CA: Environmental Systems Research Institute.
F Europe 2015 State of Europe’s forests 2015: Europe’s status & trends in sustainable forest management in Europe Madrid
EUROSTAT 2018 Industrial roundwood by species: export in Euro.
Falk W, Hempelmann N 2013 Species favourability shift in Europe due to climate change: a case study for Fagus sylvatica L. and Picea abies (L.) Karst. Based on an ensemble of climate models. J. Climatol. 1–18. https://doi.org/10.1155/2013/787250
Garcia K, Lasco R, Ines A, Lyon B, Pulhin F (2013) Predicting geographic distribution and habitat suitability due to climate change of selected threatened forest tree species in the Philippines. Appl Geogr 44:12–22. https://doi.org/10.1016/j.apgeog.2013.07.005
García Ruiz JM, López Moreno JI, Vicente Serrano SM, Lasanta Martínez T, Beguería S (2011) Mediterranean water resources in a global change scenario. Earth-Sci Rev 105(34):121–139. https://doi.org/10.1016/j.earscirev.2011.01.006
Giorgi F, Lionello P 2008 Climate change projections for the Mediterranean region. Global and Planetary Change 63, 90–104. https://doi.org/10.1016/j.gloplacha.2007.09.005
Graham MH (2003) Confronting multicollinearity in ecological multiple regression. Ecology 84:2809–2815. https://doi.org/10.1890/02-3114
Guner HT, Kose N, Harley GL (2017) A 200-year reconstruction of Kocasu River (Sakarya River Basin, Turkey) streamflow derived from a tree-ring network. Int J Biometeorol 61:427–437. https://doi.org/10.1007/s00484-016-1223-y
Haghshenas M, Marvi Mohadjer MR, Attarod P, Pourtahmasi K, Feldhaus J, Moein Sadeghi SM (2016a) Climate effect on tree-ring widths of Fagus orientalis in the Caspian forests, northern Iran. For Sci Technol 12(4):176–182. https://doi.org/10.1080/21580103.2016.1144542
Haghshenas M, Mohadjer MR, Attarod P, Pourtahmasi K, Feldhaus J, Sadeghi SM (2016b) Climate effect on tree-ring widths of Fagus orientalis in the Caspian forests, northern Iran. Forest Sci Technol 12(4):176–182
Hanewinkel M, Cullmann DA, Schelhaas M-J, Nabuurs GJ, Zimmermann NE (2013) Climate change may cause severe loss in the economic value of European forest land. Nat Clim Change 3:203–207. https://doi.org/10.1038/nclimate1687
Hanley JA, McNeil BJ (1982) The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 143(1):29–36. https://doi.org/10.1148/radiology.143.1.7063747
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978. https://doi.org/10.1002/joc.1276
IPCC (2007) Climate change 2007, vol 4. Published for the intergovernmental panel on climate change, Cambridge University Press, Cambridge
IPCC 2014 Climate change 2014: synthesis report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC, Geneva.
Kandemir G, Kaya Z 2009 EUFORGEN Technical Guidelines for genetic conservation and use of oriental beech (Fagus orientalis). Bioversity International, Rome, Italy. 6 pages.
Kandemir GE, Tayanc Y, Cengel B, Velioglu E 2016 Molecular phylogeny of beech (Fagus) populations in Turkey. Journal of Forestry Research 2016/2, A, 1:4, 69–79. https://doi.org/10.17568/oad.84722
Keenan RJ (2012) Adaptation of forests and forest management to climate change: an editorial. Forests 3(1):75–82. https://doi.org/10.3390/f3010075
Kellomaki S, Rouvinen I, Peltola H, Strandman H, Steinbrecher R (2001) Impact of global warming on the tree species composition of boreal forests in Finland and effects on emissions of isoprenoids. Glob Chang Biol 7:531–544. https://doi.org/10.1046/j.1365-2486.2001.00414.x
Köble R, Seufert G (Eds.) 2001 Novel maps for forest tree species in Europe.
Kose N, Guner HT (2012) The effect of temperature and precipitation on the intra-annual radial growth of Fagus orientalis Lipsky in Artvin, Turkey. Turk J Agric for 36:501–509. https://doi.org/10.3906/tar-1109-4
Kose N, Akkemik Ü, Dalfes HN, Özeren MS, Tolunay D (2012) Tree-ring growth of Pinus nigra Arn. subsp. pallasiana under different climate conditions throughout western Anatolia. Dendrochronologia 30:295–301. https://doi.org/10.1016/j.dendro.2012.04.003
Kovats RS, Valentini R, Bouwer LM, Georgopoulou E, Jacob D, Martin E, Rounsevell M, Soussana JF (2014) Europe. In: Climate change 2014: impacts, adaptation, and vulnerability. Part B: Regional aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Barros, V.R., C.B. Field, D.J. Dokken, M.D. Mastrandrea, K.J. Mach, T.E. Bilir, M. Chatterjee, K.L. Ebi, Y.O. Estrada, R.C. Genova, B. Girma, E.S. Kissel, A.N. Levy, S. MacCracken, P.R. Mastrandrea, and L.L. White (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 1267–1326. https://www.ipcc.ch/site/assets/uploads/2018/02/WGIIAR5-hap23_FINAL.pdf, accessed 20.11.2019
Kramer K, Degen B, Buschbom J, Hickler T, Thuiller W, Sykes M, de Winter W 2010 Modelling exploration of the future of European beech Fagus sylvatica L. under climate change—range, abundance, genetic diversity and adaptive response. Forest Ecology and Management, 259 11 2213–2222. https://doi.org/10.1016/j.foreco.2009.12.023
Kullman L (2008) Thermophilic tree species reinvade subalpine Sweden—early responses to anomalous late Holocene climate warming. Arctic, Antarctic, Alpine Res 40:104–110. https://doi.org/10.1657/1523-0430(06-120)[KULLMAN]2.0.CO;2
Laaribya S, Alaoui A, Ayan S, Benabou A, Labbaci A, Ouhaddou H, Bijou M 2021 Prediction by Maximum Entropy of potential habitat of the cork oak (Quercus suber L.) in Maamora Forest, Morocco, FORESTIST, 71 2 63 69. https://doi.org/10.5152/forestist.2021.20059
Lenoir J, Gégout JC, Marquet PA, de Ruffray P, Brisse H 2008. A significant upward shift in plant species optimum elevation during the 20th century. Science 320, 1768–1771. https://doi.org/10.1126/science.1156831
Li X, Wang Y (2013) Applying various algorithms for species distribution modelling. Integrative Zoology 8(2):124–135. https://doi.org/10.1111/1749-4877.12000
Lindner M, Maroschek M, Netherer S, Kremer A, Barbati A, Garcia Gonzalo J, Seidl R, Delzon S, Corona P, Kolstrom M, Lexer MJ, Marchetti M (2010) Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. Forest Ecology and Management 259(4):698–709. https://doi.org/10.1016/j.foreco.2009.09.023
López-Tirado J, Vessella F, Stephan J, Ayan S, Schirone B, Hidalgo PJ (2021). Effect of climate change on potential distribution of Cedrus libani A. Rich in the twenty‑first century: an Ecological Niche Modeling assessment. New Forests, 52: 363-376https://doi.org/10.1007/s11056-020-09798-y
Maiorano L, Cheddadi R, Zimmermann NE, Pellissier L, Petitpierre B, Pottier J, Laborde H, Hurdu BI, Pearman PB, Psomas A, Singarayer JS, Broennimann O, Vittoz P, Dubuis A, Edwards ME, Binney HA, Guisan A (2013) Building the niche through time: using 13,000 years of data to predict the effects of climate change on three tree species in Europe. Global Ecol Biogeogr 22:302–317. https://doi.org/10.1111/j.1466-8238.2012.00767.x
Martin-Benito D, Pederson N, Kose N, Doğan M, Bugmann H, Mosulishvili M, Bigler C (2018) Pervasive effects of drought on tree growth across a wide climatic gradient in the temperate forests of the Caucasus. Global Ecology and Biogeography 27:1314–1325. https://doi.org/10.1111/geb.12799
Meier ES, Lischke H, Schmatz DR, Zimmermann NE (2012) Climate, competition and connectivity affect future migration and ranges of European trees. Global Ecol. Biogeogr. 21:164–178. https://doi.org/10.1111/j.1466-8238.2011.00669.x
Metz CE 1978 Basic principles of ROC analysis. In Seminars in nuclear medicine.8 4 283 298). 1978, WB Saunders. https://doi.org/10.1016/S0001-2998(78)80014-2
Mizunaga H, Sako S, Nakao Y, Shimano Y (2005) Factors affecting the dynamics of the population of Fagus crenata in the Takakuma Mountains, the southern limit of its distribution area. J For Res. 10:481–486. https://doi.org/10.1007/s10310-005-0165-8
O’Donnell MS, Ignizio DA 2012 Bioclimatic predictors for supporting ecological applications in the conterminous United States: U.S. Geological Survey Data Series 691, 10 p.
OGM (2015) Türkiye orman varlığı. https://www.ogm.gov.tr/ekutuphane/Yayinlar/T% C3%BCrkiye%20Orman%20Varl%C4%B1%C4%9F%C4%B1–2016–2017.pdf
Ozdemir H, Unal A, Kindap T, Turuncoglu UU, Durmusoglu ZO, Khan M, Tayanc M, Karaca M (2011) Quantification of the urban heat island under a changing climate over Anatolian Peninsula. Theor Appl Climatology 108(1–2):31–38. https://doi.org/10.1007/s00704-011-0515-8
Ozel HB, Ertekin M 2011 Growth models in investigating oriental beech (Fagus orientalis Lipsky.) juvenilities growth performance in the Western Black Sea in Turkey Devrek-Akçasu Case Study. Romanian Biotechnol Letters 16 1 5850–5857.
Panagos P, Ballabio C, Meusburger K, Spinoni J, Alewell C, Borrelli P (2017) Towards estimates of future rainfall erosivity in Europe based on REDES and WorldClim datasets. J hydrol 548:251–262. https://doi.org/10.1016/j.jhydrol.2017.03.006
Pearson RG 2007 Species’ distribution modeling for conservation educators and practitioners. Synthesis. American Museum of Natural History. Available at: http://ncep.amnh.org
Penuelas J, Boada M (2003) A global change-induced biome shifts in the Montseny mountains (NE Spain). Glob. Change Biol. 9:131–140. https://doi.org/10.1046/j.1365-2486.2003.00566.x
Peters R (1997) Beech forests: woody species composition, populations and spatial aspects. Beech forests. Springer, Dordrecht, pp 89–130
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum Entropy modeling of species geographic distributions. Ecological Modeling 190(3–4):231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Qin A, Liu B, Guo Q, Bussmann RW, Ma F, Jian Z, Gexi X, Pei S (2017) MaxEnt modeling for predicting impacts of climate change on the potential distribution of Thuja sutchuenensis Franch., an extremely endangered conifer from southwestern China. Global Ecol Conserv 10:139–146. https://doi.org/10.1016/j.gecco.2017.02.004
Rice ME, Harris GT (2005) Comparing effect sizes in follow-up studies: ROC area, Cohen’s d, and r. Law and human behav 29(5):615–620. https://doi.org/10.1007/s10979-005-6832-7
Rigling A, Bigler C, Eilmann B, Feldmeyer-Christe E, Gimmi U, Ginzler C, Graf U, Mayer P, Vacchiano G, Weber P, Wohlgemuth T,Zweifel R, Dobbertin M 2013 Driving factors of a vegetation shift from Scots pine to pubescent oak in dry Alpine forests. Glob. Change Biol. 19, 229–240. https://doi.org/10.1111/gcb.12038
Ruiz-Labourdette D, Schmitz MF, Pineda FD (2013) Changes in tree species composition in Mediterranean mountains under climate change: Indicators for conservation planning. Ecol Indic 24:310–323. https://doi.org/10.1016/j.ecolind.2012.06.021
Sen ÖL 2013 A holistic view of climate change and its impacts in Turkey. Istanbul Policy Center Sabancı University-Stiftung Mercator Initiative. http://ipc.sabanciuniv.edu/en/wp-content/uploads/2012/09/AHolistic-View-of-Climate-Change-and-Its-Impacts-in-Turkey.pdf
Steven J, Phillips, Dudík M, Schapire RE 2020 Maxent software for modelling species niches and distributions (Version 3.4.1). Available from url: http://biodiversityinformatics.amnh.org/open_source/maxent/. Accessed on 2020–4–27.
Talu N, Sinan Ö, Özgün S, Dougherty W, Fencl A 2011 Turkey’s National Climate Change Adaptation Strategy and Action Plan (Draft). (D. Ş. Tapan, Ed.). Ankara: Ministry of Environ and Urban.
Tayanc M, Ulaş İ, Dogruel M (2009) Karaca M (2009) Climate change in Turkey for the last half century. Climatic Change 94:483–502. https://doi.org/10.1007/s10584-008-9511-0
Tharwat A 2018 Classification assessment methods. Applied Computing and Informatics.
Thurm EA, Hernandez L, Baltensweiler A, Ayan S, Razstovits E, Bielak K, Zlatanov TM, Hladnik D, Balic B, Freudenschuss A, Büchsenmeister R, Falk W (2018) Alternative tree species under climate warming in managed European forests. For Ecol Manage 430:485–497. https://doi.org/10.1016/j.foreco.2018.08.028
Toros H 2012 Spatio temporal variation of daily extreme temperatures over Turkey. Int J Climatol, 32(7), 1047 1055. https://doi.org/10.1002/joc.2325
Touchan R, Akkemik Ü, Huges MH, Erkan N (2007) May–June precipitation reconstruction of southwestern Anatolia, Turkey during the last 900 years from tree rings. Quat Res 68:196–202. https://doi.org/10.1016/j.yqres.2007.07.001
UNDP 2019 Turkey UNDP Climate Change Adaptation. Available at: https://www.adaptation-undp.org/explore/western-asia/turkey [Accessed 28 Feb. 2019].
Walther GR, Beißner S, Burga CA (2005) Trends in the upward shift of alpine plants. J Veg Sci 16:541. https://doi.org/10.1111/j.1654-1103.2005.tb02394.x
WorldClim 2019 Web site: www.worldclim.org. Access date: 19/08/2019
Yalcin S 2012 Modelling the current and future ranges of Turkish pine (Pinus brutia) and oriental beech (Fagus orientalis) ın Turkey ın the face of clımate change. Master Thesis, The Graduate School of Natural and Applied Sciences, Middle East Technical University, Ankara.
Yaltırık F (1982) Fagus L. In: Davis PH (ed) Flora of Turkey, vol 7. University Press, Edinburgh, pp 657–658
Yaman B, Özel HB, Yıldız Y, Pulat E, Işık B (2021) Hydrological evaluations and effects of climate on the radial growth of oriental beech (Fagus orientalis Lipsky) in Abdipasa, Bartın. Turkey Forestist 71(2):102–109
Yılmaz M (2010) Is there a future for the isolated oriental beech (Fagus orientalis Lipsky) forests in Southern Turkey? Acta Silv. Lign Hung 6:111–114
Acknowledgements
We are thankful to General Directorate of Forestry, Turkey, for providing current distribution data of Fagus orientalis in Turkey.
Author information
Authors and Affiliations
Contributions
SA and HBO performed background research and designed the study. EB obtained the data and analysed them by support TV. The modelling results were reviewed by EAT. SA wrote the manuscript. All authors discussed the results and commented on the manuscript. The order of the authors is based on the level of their contribution.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
AYAN, S., BUGDAY, E., VAROL, T. et al. Effect of climate change on potential distribution of oriental beech (Fagus orientalis Lipsky.) in the twenty-first century in Turkey. Theor Appl Climatol 148, 165–177 (2022). https://doi.org/10.1007/s00704-022-03940-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00704-022-03940-w