Abstract
Purpose
A noninvasive method to predict the progress or treatment response of meningiomas is desirable to improve the tumor management. Studies showed that apparent diffusion coefficient (ADC) pretreatment values can predict treatment response in brain tumors. The aim of this study was to analyze changes of intratumoral ADC values in patients with meningiomas undergoing conservative or radiosurgery.
Method
MR images of 51 patients with diagnose of meningiomas were retrospectively reviewed. Twenty-five patients undergoing conservative or radiosurgery treatment, respectively, were included in the study. The follow-up data ranged between 1 and 10 years. Based on ROI analysis, the mean ADC values, ADC10%min, and ADC90%max were evaluated at different time points during follow-up.
Results
Baseline ADC values in between both groups were similar. The ADCmean values, ADC10%min, and ADC90%max within the different groups did not show any significant changes during the follow-up times in the untreated (ADCmean over 10 years period: 0.87 ± 0.05 × 10−3 mm2/s) and radiosurgically treated (ADCmean over 4 years period: 1.02 ± 0.12 × 10−3 mm2/s) group. However, statistically significant difference was observed when comparing the ADCmean and ADC90%max values of untreated with radiosurgically treated (p < 0.0001) meningiomas. Also, ADC10%min revealed statistically significant difference between the untreated and the radiosurgery group (p < 0.05).
Conclusions
ADC values in conservatively managed meningiomas remain stable during the follow-up. However, meningiomas undergoing radiosurgery reveal significant change of the mean ADC values over time, suggesting that ADC may reflect a change in the biological behavior of the tumor. These observations might suggest the value of ADC changes as an indicator of treatment response.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Meningiomas are the most common intracranial tumors and account for up to 30% of all primary intracranial tumors in adults [21, 35]. They are typically slow-growing tumors that arise from the meningothelial cells of the arachnoid. Histological grading of meningiomas is based on the current WHO classification. The majority of lesions are benign WHO grade I lesions, representing approximately 90% of cases. The histological subtypes of grade I meningiomas differ from the more aggressive meningiomas, WHO grade II (atypical) and WHO grade III (anaplastic), 5–7% and 1–3% of cases, in their number of mitoses, cellularity, nuclear-to-cytoplasmic ratio, histological patterns, and their relatively low risk of recurrence or aggressive growth pattern [17, 34, 35].
Stereotactic radiosurgery (SRS) is an important and well-established modality in the treatment armamentarium of meningiomas either as stand-alone therapy or in combination with microsurgery [4, 5, 19]. Radiosurgery in meningiomas has shown to be effective and associated with low toxicity rates. Long-term results have shown good clinical outcomes as well as tumor control rates for meningiomas [3, 20, 22, 25].
Magnetic resonance imaging (MRI) is the modality of choice for the investigation of meningiomas. Although typical meningiomas have characteristic imaging features, there are multiple atypical variants that may be diagnostically challenging, and the value of MRI in predicting WHO grades in meningiomas is limited [34]. Standard MRI sequences (T1, T2) have limited value with regard to assess the biological behavior particularly of slowly growing tumor, e.g., during or following radiation or chemotherapy [23].
Diffusion-weighted magnet resonance imaging (DWI) provides information on water diffusivity, which is expressed by the apparent diffusion coefficient (ADC) [15]. It has been shown that DWI can be used to distinguish benign from malignant tumors and to differentiate meningiomas from other tumors such as hemangiopericytomas, which appear similar to meningiomas on T1- and T2-weighted sequences [14, 23, 26, 33]. DWI provides information regarding the microstructure of a tumor, and it has been shown that an increase of cell density leads to restricted water diffusion and to decreased ADC [9, 28,29,30]. In gliomas it has been shown that ADC can be used as a predictor of treatment response or as an early response assessment [11, 18]. As meningiomas may show delayed volume changes following treatment, a measure of tumor cell density and ADC could be important for treatment evaluation [12, 27]. Few studies in meningiomas have investigated ADC values and reported lower ADC values in WHO grade II/III compared with grade I meningiomas [26]; other studies did not find such a correlation [24].
A noninvasive radiobiological method to predict treatment response in meningiomas would be desirable to improve tumor management. Therefore, the purpose of this study was to analyze whether intratumoral ADC values change over time in untreated compared with radiosurgically treated meningiomas.
Materials and methods
Patients
We retrospectively analyzed MRI of 51 patients with meningiomas or suspected meningiomas that fulfilled the imaging criteria for these tumors (26 untreated and were conservatively managed whereas 25 were treated with radiosurgery). The follow-up data ranged between 1 and 10 years. This study was approved by the regional ethics committee.
Radiosurgery parameters
We analyzed the radiosurgery treatments of 25 patients. Thirteen radiosurgery procedures were performed with a Gamma Knife Perfexion (Elekta, Stockholm, Sweden) system, and 12 tumors were treated with a TrueBeam Novalis STx (Varian, CA, USA) system. One patient was treated with a hypofractionated scheme (5 × 5 Gy); all other patients underwent a single-dose radiosurgery treatment. The selection criteria for radiosurgical treatment was decided by an interdisciplinary tumor board decision and depended mainly on the location, size of the tumor, as well as patient’s condition and age.
MR imaging acquisition
MRI data were acquired with 1.5T Avanto or Espree scanner (Siemens, Erlangen, Germany) with twelve-channel head array coil. Our study MRI examination included sagittal T1 MPRAGE with contrast agent (repetition time (TR)/echo time (TE)/inversion time (TI) = 2200/4.9/900 ms, 1-mm slice thickness, 20% gap between slices, flip angle 8o, acquisition time (TA) 3:20). Diffusion-weighted images were acquired by using the diffusion-weighted single-shot echo-planar imaging (EPI) sequence (TR/TE: 3400/89 ms; 5-mm slice thickness; 1.5-mm gap between slices; 2 averages; TA 1:38 min; obtained with b values of 0 and 1000 s/mm2 in the read, phase, and slice directions).
Data analysis
Quantification of ADC values
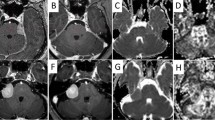
Comparing ADC with T1-weighted sequences care was taken to place the ROI within visually tumor margins avoiding the surrounding normal brain (Fig. 1). A global ROI was then set on the adjacent slice above and below, if the tumor was large enough. The ADC values of these three ROIs were then averaged (ADCmean) and extracted using PMOD software (PMOD Technologies LLC, Zürich, Switzerland). Global ROIs were used to derive ADC90%max values using a cut-off at 90%, representing the highest 10% values of the ADC range. ADC10%min values was calculated with a cut-off at 10%, representing the lowest 10% of the ADC values.
Statistics
Descriptive statistics were used to characterize the patient population. Data are presented as mean ± SD. To test differences between untreated and treated meningiomas, we used the unpaired t test (SPSS version 24, IBM, New York, USA).
Results
Clinical features
The mean age of the 51 patients was 58 ± 12 years (37 female). Twenty-six patients remained untreated (mean age 58 ± 11 years, 18 female) and were conservatively managed whereas 25 were treated with radiosurgery (mean age 57 ± 13 years, 19 female). The follow-up data ranged between 1 and 10 years. One patient was treated with a hypofractionated scheme (5 × 5 Gy); all other patients underwent a single-dose radiosurgery treatment.
ADC
The mean ADC values, ADC10%min, and ADC90%max within the different groups did not show any significant changes during the follow-up times in the untreated (over 10 years period, Table 1) and radiosurgically treated (over 4 years period, Table 2) group when comparing baseline to follow-up values. However, statistically significant difference was observed when comparing the baseline mean ADC of untreated and radiosurgically treated meningiomas (p < 0.01), as well as mean ADC at follow-up (p < 0.01) (Fig. 2). ADC90%max values revealed statistically significant difference between the groups at baseline (p < 0.01) as well as at follow-up (p < 0.0001) (Fig. 3). ADC10%min showed no changes either at baseline or at follow-up.
ADC90%max values revealed a statistically significant difference between the ADC values in conservatively treated meningiomas and radiosurgically treated meningiomas at a baseline (*p < 0.01) and at b follow-up (**p < 0.0001). When comparing the ADC90%max values from baseline to follow-up, no significant changes within the groups were detected
Tumor volume
Tumor volume remained stable in 8 (31%) and increased in 18 (69%) patients on the conservatively managed meningiomas (Table 1). Of those meningiomas which underwent radiosurgery, 14 revealed a reduced tumor volume in the last follow-up (56%) whereas 4 increased slightly in size (16%, Table 2). The remaining ten radiosurgically treated meningioma volumes stayed stable over time (n = 7, 28%).
Discussion
With DWI water diffusion in tissue can be measured and quantified by ADC [13]. ADCmean, ADC10%min, and ADC90%max have been widely examined in several types of tumors with varying results [6, 7, 26,27,28,29]. ADC values may be influenced by cellularity, proliferation, nucleic volume and size, permeability of cell membrane, cell size, composition of extracellular matrix, microvessel density, cell density, and tumor grade [1, 23, 28]. This allows to distinguish between brain tissue, edema, and tumor and provides information about cell density [7]. Within the last years, ADC has been increasingly shown to be a useful biological marker for treatment response in neuro-oncology, mainly in gliomas [8, 11, 16, 18].
Different studies have looked whether ADC values correlate with the tumor grading in meningiomas [1, 17, 31]. Others studied if ADC could differentiate fibrous tumor/hemangiopericytoma from angiomatous meningioma [14] or if ADC could be used as a preoperative predictor for progression or recurrence in meningiomas [12]. They found that parasagittal and parafalcine meningiomas with preoperative high DWI signal and lower ADC values had higher risks for progression or recurrence.
In our study, we analyzed if ADC values change in meningiomas over time and therefore compared a series of conservatively managed versus radiosurgically treated meningiomas. Since we hypothesize that a change in cell density highly corresponds with a change in ADC values [29], either an increase or decrease in ADC values would reflect a change in the biological tumor behavior. To evaluate this, we measured mean ADC values as well as ADC10%min and ADC90%max values in untreated and treated meningiomas. In our series, ADCmean values increased after radiosurgery. These results are in line with the existing literature possibly indicating that ADC could be used as a marker for treatment response and used to distinguish tumor recurrence versus treatment related (radiogenic) changes [2, 10, 32]. In the future, it would be interesting to evaluate the ADC values of those treated meningiomas that grew in the follow- up period, decreased in size, or remained in their original size. Due to our small series in our study, this subgroup analysis could not be carried out; however, further studies are planned to address this question.
Conclusion
Radiosurgically treated meningiomas reveal significant change of the mean ADC values over time whereas ADC values in conservatively treated meningiomas remain stable during follow-up, suggesting that ADC may reflect a change in the biological behavior of the tumor. Thus, especially in meningiomas where volume change after radiosurgery is not frequent, ADC analysis could be a useful tool to measure early treatment response; however, additional long-term studies are needed to address this issue.
References
Aslan K, Gunbey HP, Tomak L, Incesu L (2018) The diagnostic value of using combined MR diffusion tensor imaging parameters to differentiate between low- and high-grade meningioma. Br J Radiol 91:20180088
Chuang CC, Chang CS, Tyan YS, Chuang KS, Tu HT, Huang CF (2012) Use of apparent diffusion coefficients in evaluating the response of vestibular schwannomas to Gamma Knife surgery. J Neurosurg 117(Suppl):63–68
Cohen-Inbar O, Lee CC, Schlesinger D, Xu Z, Sheehan JP (2016) Long-term results of stereotactic radiosurgery for skull base meningiomas. Neurosurgery 79:58–68
Cohen-Inbar O, Lee CC, Sheehan JP (2016) The contemporary role of stereotactic radiosurgery in the treatment of meningiomas. Neurosurg Clin N Am 27:215–228
Davidson L, Fishback D, Russin JJ, Weiss MH, Yu C, Pagnini PG, Zelman V, Apuzzo ML, Giannotta SL (2007) Postoperative Gamma Knife surgery for benign meningiomas of the cranial base. Neurosurg Focus 23:E6
Driessen JP, Caldas-Magalhaes J, Janssen LM, Pameijer FA, Kooij N, Terhaard CH, Grolman W, Philippens ME (2014) Diffusion-weighted MR imaging in laryngeal and hypopharyngeal carcinoma: association between apparent diffusion coefficient and histologic findings. Radiology 272:456–463
Eis M, Els T, Hoehn-Berlage M, Hossmann KA (1994) Quantitative diffusion MR imaging of cerebral tumor and edema. Acta Neurochir Suppl (Wien) 60:344–346
Ellingson BM, Malkin MG, Rand SD, Connelly JM, Quinsey C, LaViolette PS, Bedekar DP, Schmainda KM (2010) Validation of functional diffusion maps (fDMs) as a biomarker for human glioma cellularity. J Magn Reson Imaging 31:538–548
He W, Xiao X, Li X, Guo Y, Guo L, Liu X, Xu Y, Zhou J, Wu Y (2019) Whole-tumor histogram analysis of apparent diffusion coefficient in differentiating intracranial solitary fibrous tumor/hemangiopericytoma from angiomatous meningioma. Eur J Radiol 112:186–191
Huang CF, Chiou SY, Wu MF, Tu HT, Liu WS, Chuang JC (2010) Apparent diffusion coefficients for evaluation of the response of brain tumors treated by Gamma Knife surgery. J Neurosurg 113(Suppl):97–104
Jain R, Scarpace LM, Ellika S, Torcuator R, Schultz LR, Hearshen D, Mikkelsen T (2010) Imaging response criteria for recurrent gliomas treated with bevacizumab: role of diffusion weighted imaging as an imaging biomarker. J Neuro-Oncol 96:423–431
Ko CC, Chen TY, Lim SW, Kuo YT, Wu TC, Chen JH (2019) Prediction of recurrence in parasagittal and parafalcine meningiomas: added value of diffusion-weighted magnetic resonance imaging. World Neurosurg 3:S1878–8750(18)32940–1. https://doi.org/10.1016/j.wneu.2018.12.117
Le Bihan D (1995) Molecular diffusion, tissue microdynamics and microstructure. NMR Biomed 8:375–386
Liu L, Yin B, Geng DY, Li Y, Zhang BY, Peng WJ (2014) Comparison of ADC values of intracranial hemangiopericytomas and angiomatous and anaplastic meningiomas. J Neuroradiol 41:188–194
Maier SE, Sun Y, Mulkern RV (2010) Diffusion imaging of brain tumors. NMR Biomed 23:849–864
Nagane M, Kobayashi K, Tanaka M, Tsuchiya K, Shishido-Hara Y, Shimizu S, Shiokawa Y (2014) Predictive significance of mean apparent diffusion coefficient value for responsiveness of temozolomide-refractory malignant glioma to bevacizumab: preliminary report. Int J Clin Oncol 19:16–23
Nagar VA, Ye JR, Ng WH, Chan YH, Hui F, Lee CK, Lim CC (2008) Diffusion-weighted MR imaging: diagnosing atypical or malignant meningiomas and detecting tumor dedifferentiation. AJNR Am J Neuroradiol 29:1147–1152
Nowosielski M, Recheis W, Goebel G, Guler O, Tinkhauser G, Kostron H, Schocke M, Gotwald T, Stockhammer G, Hutterer M (2011) ADC histograms predict response to anti-angiogenic therapy in patients with recurrent high-grade glioma. Neuroradiology 53:291–302
Pinzi V, Biagioli E, Roberto A, Galli F, Rizzi M, Chiappa F, Brenna G, Fariselli L, Floriani I (2017) Radiosurgery for intracranial meningiomas: a systematic review and meta-analysis. Crit Rev Oncol Hematol 113:122–134
Przybylowski CJ, Raper DM, Starke RM, Xu Z, Liu KC, Sheehan JP (2015) Stereotactic radiosurgery of meningiomas following resection: predictors of progression. J Clin Neurosci 22:161–165
Riemenschneider MJ, Perry A, Reifenberger G (2006) Histological classification and molecular genetics of meningiomas. Lancet Neurol 5:1045–1054
Santacroce A, Walier M, Régis J, Liščák R, Motti E, Lindquist C, Kemeny A, Kitz K, Lippitz B, Martínez Álvarez R, Pedersen PH, Yomo S, Lupidi F, Dominikus K, Blackburn P, Mindermann T, Bundschuh O, van Eck AT, Fimmers R, Horstmann GA (2012) Long-term tumor control of benign intracranial meningiomas after radiosurgery in a series of 4565 patients. Neurosurgery 70:32–39
Sasaki M, Eida S, Sumi M, Nakamura T (2011) Apparent diffusion coefficient mapping for sinonasal diseases: differentiation of benign and malignant lesions. AJNR Am J Neuroradiol 32:1100–1106
Schwyzer L, Berberat J, Remonda L, Roelcke U (2015) Susceptibility changes in meningiomas influence the apparent diffusion coefficient in diffusion-weighted MRI. J Neuroradiol 42:332–337
Sheehan JP, Williams BJ, Yen CP (2010) Stereotactic radiosurgery for WHO grade I meningiomas. J Neuro-Oncol 99:407–416
Surov A, Gottschling S, Mawrin C, Prell J, Spielmann RP, Wienke A, Fiedler E (2015) Diffusion-weighted imaging in meningioma: prediction of tumor grade and association with histopathological parameters. Transl Oncol 8:517–523
Surov A, Hamerla G, Meyer HJ, Winter K, Schob S, Fiedler E (2018) Whole lesion histogram analysis of meningiomas derived from ADC values. Correlation with several cellularity parameters, proliferation index KI 67, nucleic content, and membrane permeability. Magn Reson Imaging 51:158–162
Surov A, Meyer HJ, Wienke A (2017) Associations between apparent diffusion coefficient (ADC) and KI 67 in different tumors: a meta-analysis. Part 1: ADCmean. Oncotarget 8:75434–75444
Surov A, Meyer HJ, Wienke A (2017) Correlation between apparent diffusion coefficient (ADC) and cellularity is different in several tumors: a meta-analysis. Oncotarget 8:59492–59499
Surov A, Meyer HJ, Wienke A (2017) Correlation between minimum apparent diffusion coefficient (ADCmin) and tumor cellularity: a meta-analysis. Anticancer Res 37:3807–3810
Tang Y, Dundamadappa SK, Thangasamy S, Flood T, Moser R, Smith T, Cauley K, Takhtani D (2014) Correlation of apparent diffusion coefficient with Ki-67 proliferation index in grading meningioma. AJR Am J Roentgenol 202:1303–1308
Tomura N, Narita K, Izumi J, Suzuki A, Anbai A, Otani T, Sakuma I, Takahashi S, Mizoi K, Watarai J (2006) Diffusion changes in a tumor and peritumoral tissue after stereotactic irradiation for brain tumors: possible prediction of treatment response. J Comput Assist Tomogr 30:496–500
Wang J, Takashima S, Takayama F, Kawakami S, Saito A, Matsushita T, Momose M, Ishiyama T (2001) Head and neck lesions: characterization with diffusion-weighted echo-planar MR imaging. Radiology 220:621–630
Watts J, Box G, Galvin A, Brotchie P, Trost N, Sutherland T (2014) Magnetic resonance imaging of meningiomas: a pictorial review. Insights Imaging 5:113–122
Whittle IR, Smith C, Navoo P, Collie D (2004) Meningiomas. Lancet 363:1535–1543
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional and national research committee (EKNZ 2016-00854) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Due to the retrospective nature of this study, informed consent was not required.
Additional information
Comments
Berberat et al are addressing a key issue in Radiosurgery (SRS). We know from large series with long term follow up that Radiosurgery in small WHO I Meningiomas is providing a very advantageous safety efficacy ratio on the long term [2,5]. However, meningiomas are indolent slowly growing tumors. Thus, for years, the stability of the MRI T1 contrast enhanced image is not a convincing demonstration of the capability of radiosurgery to control on the very long term the individual tumor.
Neurosurgeons are lacking a marker of response. In this perspective the contribution of Berberat et al is of interest for intracranial meningiomas management. At the difference of meningiomas managed conservatively meningiomas treated by radiosurgery are revealing “significant change of the mean apparent diffusion coefficient (ADC) values over time”. The authors are speculating that this change of ADC over time “may reflect a change in the biological behavior of the tumor” and “might suggest the value of ADC changes as an indicator of treatment response”. However, till now the authors data are not supporting this hypothesis. Only long-term follow-up of patients presenting with meningiomas treated by radiosurgery with ADC on the occasion of each MR follow up could demonstrate if the change of the ADC over time is, or not, a predictor of long-term control! For future studies we recommend to separate skull meningiomas and convexity meningiomas who have clearly not the same biological behavior after SRS. We also recommend not to mix radiosurgery with stereotactic radiotherapy as long as nowadays the radiobiology of these two approaches are demonstrated to induce different biological responses specially at the level of the microvasculature and immune system. This is an important field of research for the future of SRS in general as long as the same question exists for other tumors like vestibular schwannomas or brain metastases with the additional difficulty of pseudo progression phenomenon which is even making more difficult the assessment of tumor response [1,3,4]!
Jean Marie Regis
Marseille, France
References
1. Hayhurst C, Zadeh G (2012) Tumor pseudoprogression following radiosurgery for vestibular schwannoma. Neuro Oncol 14:87-92. doi: 10.1093/neuonc/nor1171. Epub 2011 Oct 1025.
2. Kondziolka D, Patel AD, Kano H, Flickinger JC, Lunsford LD (2014) Long-term Outcomes After Gamma Knife Radiosurgery for Meningiomas. Am J Clin Oncol. doi:10.1097/coc.0000000000000080
3. Lin NU, Lee EQ, Aoyama H, Barani IJ, Barboriak DP, Baumert BG, Bendszus M, Brown PD, Camidge DR, Chang SM, Dancey J, de Vries EG, Gaspar LE, Harris GJ, Hodi FS, Kalkanis SN, Linskey ME, Macdonald DR, Margolin K, Mehta MP, Schiff D, Soffietti R, Suh JH, van den Bent MJ, Vogelbaum MA, Wen PY (2015) Response assessment criteria for brain metastases: proposal from the RANO group. Lancet Oncol 16:e270-278. doi:10.1016/s1470-2045(15)70057-4
4. Regis J, Delsanti C, Roche PH (2017) Editorial: Vestibular schwannoma radiosurgery: progression or pseudoprogression? J Neurosurg 127:374-379. doi: 310.3171/2016.3177.JNS161236. Epub 162016 Nov 161225.
5. Santacroce A, Walier M, Regis J, Liscak R, Motti E, Lindquist C, Kemeny A, Kitz K, Lippitz B, Martinez Alvarez R, Pedersen PH, Yomo S, Lupidi F, Dominikus K, Blackburn P, Mindermann T, Bundschuh O, van Eck AT, Fimmers R, Horstmann GA (2012) Long-term tumor control of benign intracranial meningiomas after radiosurgery in a series of 4565 patients. Neurosurgery 70:32-39; discussion 39. doi:10.1227/NEU.0b013e31822d408a
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Brain Tumors
Rights and permissions
About this article
Cite this article
Berberat, J., Roelcke, U., Remonda, L. et al. Long-term apparent diffusion coefficient value changes in patients undergoing radiosurgical treatment of meningiomas. Acta Neurochir 163, 89–95 (2021). https://doi.org/10.1007/s00701-020-04567-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-020-04567-4