Abstract
Background
Delirium is an acute and reversible deterioration of mental state. Postoperative delirium (POD) can develop after surgical procedures and is associated with impaired health status and worse recovery. So far, there is little data about postoperative delirium after brain surgery. The aim of this study was to evaluate frequency, risk factors, and prognostic value of POD in predicting short-term postoperative outcomes after brain tumor surgery.
Methods
Five-hundred and twenty-two patients who underwent elective brain tumor surgery in 2010–2017 were included in this prospective study. Patients were monitored for POD using the Confusion Assessment Method for the ICU (CAM-ICU) for 2 to 7 days after the surgery. At hospital discharge, outcomes were evaluated using the Glasgow Outcome Scale (GOS).
Results
POD was diagnosed in 22 (4.2%) patients. Risk factors of POD were low level of hemoglobin, poor functional status at time of admission, low education level and older age (65 years and older). POD incidence was not associated with brain tumor laterality, location, extent of resection, histological diagnosis, or affected brain lobe. POD was associated with greater risk for unfavorable outcomes at hospital discharge (OR = 5.3; 95% CI [2.1–13.4], p = 0.001).
Conclusions
POD is not a common complication after elective brain tumor surgery. Older age, poor functional status, low education level and anemia are associated with greater POD risk. Extent of surgical intervention and brain tumor location are not associated with POD risk. POD is associated with worse outcome at hospital discharge.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Delirium is an acute and reversible deterioration of mental abilities characterized by disturbed level of consciousness and/or cognitive dysfunction [17]. Postoperative delirium (POD) is a common complication after surgery with incidence rate ranging between 3 and 77% [6, 36, 38, 39]. It is highly prevalent in elderly patients [20]. Prevention, detection, and management of POD are gaining increasing importance due to the aging population [27]. POD is associated with longer hospitalization time, greater health-care costs, and increased complication rate and adverse outcomes [17]. Therefore, early recognition, prevention, and appropriate management of POD could have important clinical implications for patient recovery after surgery. It could also translate into long-term health benefits as POD was linked to cognitive decline later in life [13]. Most published studies to date have studied POD after cardiovascular, orthopedic, and spinal surgeries or in elderly patient populations [1, 6, 33, 35, 36, 44]. So far, there is little data about POD after elective cranial surgery as only a few studies attempted to evaluate POD in neurosurgical patients [11, 41]. Therefore, incidence, risk factors, and clinical significance of POD after brain surgery remain to be further investigated.
Numerous studies attempted to identify risk factor of POD. It is well-established that type of surgery and anesthesia, and patient age are associated with POD risk across patient populations [1, 17, 20, 33, 35, 36, 44]. Less studied predictors of POD are C-reactive protein [43] and interleukin-6 [4] concentrations, histories of mental disorders [14], intraoperative blood loss, and medication use [23]. Risk factors of POD after cranial neurosurgical procedures remain understudied.
The aim of this study was to evaluate incidence and risk factors of POD after elective cranial surgery for brain tumor and the association of POD with outcomes at hospital discharge.
Patients and methods
Patients and procedures
The study was approved by the Regional Bioethics committee at the Lithuanian University of Health Sciences, Kaunas, Lithuania. Written informed consent was obtained from all study patients before enrollment in the study.
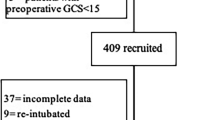
The study sample was comprised of two prospective cohorts of brain tumor patients who underwent elective cranial surgery (craniotomy or biopsy) for brain tumor at the Department of Neurosurgery of Lithuanian University of Health Sciences, Kaunas, Lithuania. A priori exclusion criteria covered patient’s inability to communicate with the assessor due to inability to be fluent in Lithuanian. The first cohort of brain tumor patients was recruited in 2010–2011 (N = 232) and the second cohort in 2015–2017 (N = 290).
Eligible patients were approached at hospital admission and invited to participate in the study. The study investigator prospectively recorded demographic characteristics (age and gender) and past histories of psychiatric and somatic disorders. Preoperative functional status was assessed using the Barthel Index (BI) (cohort 1) [28], Karnofsky Performance Index (KPI) (cohort 2) [29], and Modified Rankin Scale (mRS) (cohort 2) [42]. Anxiety and depressive symptom severity was assessed using the Hospital Anxiety and Depression Scale (HADS) [37]. Before surgery, blood samples were drawn for complete blood count and evaluation of high-sensitivity C-reactive protein (hs-CRP), interleukin 6 (IL-6), and N-terminal pro-b-type natriuretic peptide (NT-proBNP) concentrations. Type of brain tumor surgery (stereotactic biopsy or craniotomy) was prospectively recorded. Extent of surgical tumor resection was recorded from operative reports to postoperative CT scans. POD was evaluated for two consecutive days (cohort 1) or seven consecutive days (cohort 2) after brain tumor surgery using the Confusion Assessment Method for the ICU (CAM-ICU) [15]. In-hospital complications were prospectively recorded during the hospitalization period by the study investigators. At hospital, discharge outcomes were assessed using the Glasgow Outcome Scale (GOS) [21]. Assessments were performed by trained study investigators (ST, AS, AB, IN, MS, and AP).
All patients included in the study received standard preoperative, intraoperative, and postoperative care according to clinical practice guidelines at our department.
Anesthesia
Craniotomy for tumor resection procedures was performed under the general anesthesia, while stereotactic tumor biopsy procedures were typically performed under the local anesthesia combined with intravenous sedation. Standard intraoperative monitoring according to the American Society of Anesthesiologists was applied in all cases. General anesthesia was induced with fentanyl (1–2 mcg/kg), propofol (2–2.5 mg/kg), and rocuronium (0.6 mg/kg). Following confirmed tracheal intubation, anesthesia was maintained with sevoflurane (up to 1.0 minimum alveolar concentration) and intermittent boluses of fentanyl and rocuronium according to clinical demand. One percent lidocaine was used for skin infiltration in cases of stereotactic tumor biopsy procedures. Intravenous sedation was produced with fentanyl (1 mcg/kg) and continuous infusion of propofol that was titrated to the desired effect.
Instruments
Preoperative functional status was assessed using the BI in the first cohort, and using the KPI and mRS in the second cohort. The BI was used to evaluate impairment of daily living activities, such as dressing, bathing, feeding, grooming, moving from bed to chair and back, bladder and bowels control, toilet use, and mobility [28]. Global BI score ranges from 0 to 100 points, with higher score indicating greater level of functional independence. The KPI similarly to BI was used to asses patient’s functional status by indicating a score from 100 (normal functioning) to 0 (dead); the scale is divided into subgroups of 10, and each group indicates a different level of independency [29]. The mRS scale is a functional outcome scale where all aspects of physical and cognitive abilities are combined in the single handicap grade, which ranges from 0 (no symptoms at all) to 6 (dead) [42]. All study patients were categorized as having poor functional status or good functional status. Functional status was considered as good if a patient was able to care for oneself and carry out normal daily functions. Patients who were dependent upon others were considered as having poor functional status. Scoring details are shown in the Table 1.
Presence of POD was prospectively evaluated using the CAM-ICU rating scale. The CAM-ICU is a physician rating scale that evaluates four symptoms: (1) acute onset of change or fluctuations of mental status, (2) inattention, (3) disorganized thinking, and (4) altered level of consciousness. A patient is screened positive if she/he has features (1) and (2), plus either feature (3) or (4). Diagnosis of POD is based on one positive test. The CAM-ICU is a reliable instrument and is commonly used for assessment of delirium in critically ill patients and after major surgery [43].
Depressive and anxiety symptoms were evaluated by administering the HADS to patients before surgery. The HADS is a self-rating scale that is designed to evaluate current depressive and anxiety symptoms in patients with somatic disorders. It includes two 7-item subscales of depression and anxiety with total scores in each subscales ranging from 0 to 21. Higher ratings correspond to greater symptom severity.
Functional outcome at hospital discharge was evaluated using the GOS with possible outcomes ranging from 1 (death) to 5 (good recovery). The GOS is widely used for research purposes in neurosurgical patients [8]. Patients were dichotomized as having good discharge outcome (GOS score of 4 or 5) or poor discharge outcome (GOS score from 1 to 3).
Blood samples
All participants had their blood samples collected on the first day of admission at the neurosurgical clinic. Venous blood samples were drawn for evaluation of complete blood count, NT-proBNP, hs-CRP, and IL-6 concentrations. Blood was centrifuged and serum was stored frozen at − 40 °C. Serum concentration of NT-proBNP and IL-6 was assessed using the radio-immunoassay method (Roche Cobas analyzer, Roche Diagnostics, UK). Normal serum concentration of NT-proBNP was < 157 ng/L (cohort 1) or < 125 ng/L (cohort 2) and normal values of IL-6 were from 0 to 7 pg/mL (cohort 1) or from 0 to 5.9 pg/mL (cohort 2). In cohort 1, serum hs-CRP concentrations were assessed using the chemiluminescent immunoassay method (Beckman Coulter Unicel DXC 600) and by immunoturbidimetric assay method in cohort 2 (Roche/Hitachi cobas analyzer, Roche Diagnostics, Germany) with normal values of ≤ 3.0 mg/L. Normal red blood cell (RBC) count ranged from 3.9 to 5.1 × 10^12/L, hemoglobin (Hb) from 119 to 146 g/L, and platelet (PLT) count from 166 to 308 × 10^9/L. Values were dichotomized according to their normal ranges. NT-proBNP, hs-CRP, and IL6 concentrations were studied in a proportion of our cohort.
Statistical analyses
Continuous data are presented as mean ± standard deviation and categorical data as number (percent). All statistical analyses were performed using the IBM SPSS 21.0 statistical software (International Business Machines Corporation, Armonk, New York).
Demographic and clinical characteristics between the two study cohorts were compared using the independent sample T test (or Mann-Whitney U test for values with not normal distribution) for continuous data or Pearson Chi-squared test for categorical data. Logistic regression analysis was used to evaluate the association of different clinical characteristics with POD adjusting for patient age, gender, and histological brain tumor diagnosis.
Finally, we evaluated the association of POD with discharge outcome by comparing GOS scores in patients with POD vs. without POD. Logistic regression analysis was used to evaluate the association of POD with unfavorable discharge outcome adjusting for patient age, gender, and histological brain tumor diagnosis.
Results
A total of 522 individual patients were enrolled in the study (232 patients from cohort 1 and 290 patients from cohort 2). Twenty-two or 4.2% (95%CI [0.02–0.06]) of patients were diagnosed with POD. Incidence of POD was similar between the two cohorts (3.9% in first cohort vs. 4.5% in the second cohort, p = 0.828).
Patients diagnosed with POD were significantly older relative to patients without POD (66.91 ± 9.61 years vs. 56.81 ± 15.04 years, respectively, p = 0.002). When patients were dichotomized in two groups as a function of age, we found that POD was more prevalent in patients aged 65 or more years, relative to patients younger than 65 years (72.7% vs. 36.2%, X2 = 11.,966, p = 0.001). Education status was also associated with POD risk, as greater proportion of patients diagnosed with POD had lower than secondary school education relative to patients without POD (X2 = 10.608, p = 0.005). Preoperative functional status was also associated with POD risk as a greater proportion of patients diagnosed with POD had poor preoperative functional status when compared to patients without POD (50% vs. 10.6%, respectively, X2 = 16.025, p = 0.0005). Patients affected with POD, when compared to patients without POD, also had lower preoperative Hb concentration (p = 0.014), RBC count (p = 0.001), and PLT count (p = 0.0025) (Table 2). However, when Hb concentration and RBC/PLT counts were dichotomized to normal and low concentration categories, only reduced Hb concentration was associated with POD incidence (X2 = 6507, p = 0.04). NT-proBNP concentration was greater in patients with POD relative to patients without POD (679.78 ± 1132.45 mg/L vs. 182.34 ± 423.38 mg/L, respectively, p = 0.001).
POD incidence was not associated with patient gender (p = 0.762), marital status (p = 0.791), history of mental illness (p = 0.447), presence of other somatic disorders (p = 0.079), depressive (p = 0.75) and anxiety (p = 0.42) symptoms severity, hs-CRP concentration (p = 0.734), IL-6 (p = 0.193) concentration, and brain tumor laterality (p = 0.837), location (p = 0.901), extent of resection (p = 0.709), histological diagnosis (p = 0.513), or affected brain lobe (p = 0.621).
In univariate logistic regression analyses, greater POD risk was associated with advanced patient age (65 years or more years) (OR = 4.7; 95% CI [1.8–12.2], p = 0.002), lesser than secondary education (OR = 4.2; 95% CI [1.6–10.8], p = 0.003), poor functional status before surgery (OR = 2.8; 95% CI [1.5–5.7], p = 0.001), low concentrations of Hb (OR = 5.3 95% CI [1.3–21.8], p = 0.021), and elevated concentration of NT-proBNP (OR = 4.4; 95% CI [1.1–17.6], p = 0.034). In multivariate analyses, variables that were significantly associated with POD incidence were tested together with patient’s age, gender, and histological diagnosis entered as covariates. Multivariate logistic regression analysis revealed that secondary education (OR = 3.5; 95% CI [1.3–9.1], p = 0.011), poor functional status before surgery (OR = 4.7; 95% CI [1.9–11.8], p = 0.001), and low concentration of Hb (OR = 5 95% CI [1.1–22.5], p = 0.036) remained independently associated with greater POD risk. Also, age of 65 years or greater was associated with POD risk after taking into account patient gender and histological brain tumor diagnosis (OR = 4.6; 95% CI [1.7–12.1], p = 0.002). Elevated NT-proBNP concentration was not associated with POD risk in final analyses (p = 0.191). Summary of predictor analysis is shown in Table 3.
GOS score at hospital discharge was significantly lower in patients who were diagnosed with POD when comparted to patients without POD (3.4 ± 1.14 vs. 4.32 ± 0.80, p = 0.0005; Table 4, Fig. 1). The mean length of hospital stay was 14 ± 10.26 days, with minimal stay of 7 days. POD was associated with greater risk of unfavorable functional outcome at hospital discharge in univariate logistic regression analyses (OR = 6; 95% CI [2.4–14.7], p = 0.0005), and in multivariate logistic regression analysis (OR = 5.3; 95% CI [2.1–13.4], p = 0.0005), independently from patient age, gender, and histological brain tumor diagnosis.
Discussion
POD was diagnosed in 4.2% of patients. Advanced patient age, lesser education, preoperative functional impairment, and anemia were associated with elevated POD risk independently from patient gender and histological brain tumor diagnosis. POD increased the risk for poor discharge outcome independently from patient age, gender, and histological brain tumor diagnosis.
The incidence rate of POD was 4.2% in our mixed sample of brain tumor patients. It was lower than median POD incidence rate of 11% (range from 3 to 77%) in patients undergoing major elective vascular and orthopedic surgery [17, 34]. Studies investigating POD in neurosurgical patients are scant since neurological disorders are usually considered as exclusion criteria in POD studies [11, 41]. Our findings are in agreement with recent study in 554 glioblastoma patients that reported incidence rate of POD of 7% [18]. Another study found that 10% of neurosurgical patients developed POD and POD incidence rate was lower in neurosurgical patients when compared to patients undergoing other major surgical procedures [41]. However, a study of patients after craniotomy for brain tumor found much higher incidence rate of POD (30%) [11]. Greater delirium rate in the latter study can be explained that in addition to delirium the authors also considered agitation, which also temporarily affects cognitive abilities but is different construct than POD. In contrast to previous studies, our study was performed in stable patients at the hospital ward (as opposed to ICU setting) and included patients operated for various brain tumors using craniotomy and stereotactic biopsy procedures. There is a need for further studies investigating the incidence rate of POD across hospital settings (ward vs. ICU), surgical procedures, and neurosurgical disorders. Summary of existing studies of POD in neurosurgical patients is provided in Table 5.
Advanced age was the strongest predictor of POD in our study as nearly two thirds of patients affected with POD were 65 years and older. Advanced age increased the risk of POD by nearly 5-fold. Other studies also reported advanced age as an independent and important risk factor of POD after neurosurgical [18] and not neurosurgical [2, 17] procedures. Age is also considered as a risk factor in POD management guidelines [3, 20] and POD was specifically studied in elderly populations across numerous studies [12, 31].
Other important risk factors of POD in our cohort were lower education level and poor preoperative functional status. To the best of our knowledge, the observed association between lower education level and greater POD risk is new to the literature. Education is associated with better cognitive reserve [40] that has a protective role against clinical impairment in patients with CNS disorders [26]. Also, better education and greater cognitive reserve are associated with better cognitive functions that were shown to decrease the risk of POD [2, 19, 40]. Patients with lower education status should be considered at increased risk for POD that is an important risk factor for future cognitive decline [13].
Poor functional status is widely recognized independent risk factor for POD [5, 30]. Half of our patients who developed POD had poor functional status before surgery when compared to only 10% of patients unaffected by POD. Similar findings were reported by other groups that used the American Society of Anesthesiologists classification system that incorporates functional status assessment [12]. Brain tumor patients presenting with impaired functional ability before surgery should be considered at greater risk for POD.
Others previously found that POD risk was associated with patient gender [11, 17]. However, these findings were not replicated in our cohort and can be attributed to gender differences across different brain tumor diagnoses.
In contrast to some previous studies, we did not find an association of history of mental illness, and depressive and anxiety symptom severity with POD risk [12, 14, 17, 30]. These findings can be explained by low identified incidence of mental disorders in our cohort, poor recognition of mental disorders in brain tumor patients, and high co-morbidity of mental symptoms with certain brain tumors [16]. Mental disorders were not prospectively assessed in our patients and were based on patient self-reports; depressive/anxiety symptoms were also based on self-reports. Furthermore, the majority of study patients were early in their disease progression and it is possible that mental disorders and symptoms develop later in the disease course when patients deal with established brain tumor diagnosis.
When investigating precipitating clinical factors, we found that low Hb level, RBC count, and PLT count were associated with increased POD risk. After multiple adjustments, only low levels of Hb remained an independent predictor of POD. The association between anemia and greater POD risk was previously reported by several other studies [17, 30, 32]. Low level of Hb is associated with smaller blood loss reserve and reduced brain oxygenation that can increase POD risk [10, 24].
Increased concentration of NT-proBNP, hs-CRP, and IL6 was shown to predict worse outcome and greater mortality in non-cardiac patients [7, 22], including patients with brain tumors [9]. Elevated NT-proBNP concentration was associated with greater POD risk in univariate analyses; however, the association was not significant after adjusting for age, gender, and histological diagnosis. Only one previous study investigated the possible association between NT-proBNP and POD risk and also did not find an association [25].
Others found that minimally invasive surgeries were associated with reduced POD risk when compared to major surgical interventions [2, 17, 31]. Frontal craniotomy was documented as POD risk factor [4]. We did not find an association of extent of surgical intervention (craniotomy vs. biopsy) and brain tumor location with POD risk. We recorded the scale of the operation rather than the type of the operation; therefore, gross total tumor resection cannot be considered as more invasive approach relative to subtotal resection. As opposed to other surgical field, in neurosurgery, there are smaller differences between minimally invasive approaches and standard approaches in terms of procedure duration and anesthesia. For example, certain less invasive approaches may take longer than standard surgical approaches and can paradoxically increase the risk of POD.
When evaluating short-term impact of POD for patient outcomes, we found that patients with POD had significantly lower GOS score at hospital discharge when compared to patients without POD. POD was associated with greater risk of unfavorable discharge outcome independently from patient age, gender, and brain tumor histological diagnosis. Other authors also found an association between POD and greater mortality and morbidity risk and greater postoperative cognitive impairment [4, 13, 43]. Greater mortality and morbidity, higher complication rate, is also associated with POD in neurosurgical patients as shown in previous studies [11, 18, 41]. These findings highlight the need of POD studies that would help to improve prevention, detection, and management of POD in neurosurgical patients.
The study has limitations. Due to low prevalence rate of POD, investigations of certain POD risk factors were limited. Therefore, multi-centered studies and meta-analyses of POD in patients undergoing cranial surgery are encouraged. We used the CAM-ICU instead of CAM that can be considered as more suitable for hospital ward environment. However, the CAM-ICU is more suitable for delirium screening process in neurological patients, especially in non-verbalizing patients. Our study included two separate cohorts of brain tumor patients with a 4-year difference between them and certain surgical and anesthesia techniques could have changed or improved during this time window. Due to mixed sample size, the number of patients with certain diagnostic entities was small. For example, none of our patients affected with POD had metastatic brain tumor; therefore, POD prevalence in this patient population could not be investigated. Also, numerous other potentially important POD risk factors, such as type of anesthesia and concurrent medication use, and concurrent somatic and mental disorders were not prospectively evaluated. Short follow-up period is another limitation of our study that prevented us from assessment of long-term sequelae of POD. Further studies exploring long-term clinical significance of POD in brain tumor patients are encouraged.
Conclusion
POD was diagnosed in 4.2% of patients. Advanced patient age, lesser education, preoperative functional impairment, and anemia increased the POD risk independently from patient gender and histological brain tumor diagnosis. POD increased the risk for poor discharge outcome independently from patient age, gender, and histological brain tumor diagnosis. POD should be considered an important complication after brain tumor surgery. Further studies exploring optimal assessment, prevention, and treatment strategies of POD in neurosurgical patients are strongly encouraged.
References
Ahmed S, Leurent B, Sampson E (2014) Risk factors for incident delirium among older people in acute hospital medical units: a systematic review and meta-analysis. Age Ageing 43(3):326–333
Aitken S, Blyth F, Naganathan V (2017) Incidence, prognostic factors and impact of postoperative delirium after major vascular surgery: a meta-analysis and systematic review. Vasc Med 22(5):387–397
Aldecoa C, Bettelli G, Bilotta F et al (2017) European Society of Anaesthesiology evidence-based and consensus-based guideline on postoperative delirium. Eur J Anaesthesiol 34(4):192–214
Androsova G, Krause R, Winterer G, Schneider R (2015) Biomarkers of postoperative delirium and cognitive dysfunction. Front Aging Neurosci 7:1–16
Bettelli G, Neuner B (2017) Postoperative delirium: a preventable complication in the elderly surgical patient. Monaldi Arch Chest Dis 87(2):31–33
Brauer C, Morrison R, Silberzweig S, Siu A (2000) The cause of delirium in patients with hip fracture. Arch Intern Med 160(12):1856–1860
Bunevicius A, Kazlauskas H, Raskauskiene N, Mickuviene N, Ndreu R, Corsano E et al (2015) Role of N-terminal pro-B-type natriuretic peptide, high-sensitivity C-reactive protein, and Inteleukin-6 in predicting a poor outcome after a stroke. Neuroimmunomodulation 22(6):365–372
Bunevicius A, Deltuva V, Tamasauskas S, Smith T, Laws E, Bunevicius R et al (2016) Preoperative low tri-iodothyronine concentration is associated with worse health status and shorter five year survival of primary brain tumor patients. Oncotarget 8(5):8648–8656
Bunevicius A, Deltuva V, Laws E, Iervasi G, Tamsauskas A, Bunevicius R (2017) Preoperative N-terminal pro-B-type natriuretic peptide concertation and prognosis of brain tumor patients: a 5-year follow up study. Sci Rep 7(1):1–8
Casati A, Fanelli G, Pietropaoli P, Proietti R, Tufano R, Montanini S et al (2007) Monitoring cerebral oxygen saturation in elderly patients undergoing general abdominal surgery. Eur J Anaesthesiol 24(1):59–65
Chen L, Xu M, Li G, Cai W, Zhou J (2014) Incidence, risk factors and consequences of emergence agitation in adult patients after elective craniotomy for brain tumor: a prospective cohort study. PLoS One 9(12):1–15
Choi Y, Kim D, Kim T, Lim T, Kim S, Yoo J (2017) Early postoperative delirium after hemiarthroplasty in elderly patients aged over 70 years with displaced femoral neck fracture. Clin Interv Aging 12:1835–1842
Crocker E, Beggs T, Hassan A et al (2016) Long-term effects of postoperative delirium in patients undergoing cardiac operation: a systematic review. Ann Thorac Surg 102(4):1391–1399
Elsamadicy AA, Adogwa O, Lydon E, Sergesketter A, Kaakati R, Mehta AI et al (2017) Depression as an independent predictor of postoperative delirium in spine deformity patients undergoing elective spine surgery. J Neurosurg Spine 27(2):209–214
Ely EW, Margolin R, Francis J, May L, Truman B, Dittus R et al (2001) Evaluation of delirium in critically ill patients: validation of the confusion assessment method for the intensive care unit (CAM-ICU). Crit Care Med 29(7):1370–1379
Filley C, Kleinschmidt-Demasters B (1995) Neurobehavioral presentations of brain neoplasms. West J Med 163(1):19–25
Fineberg SJ, Nandyala SV, Oglesby M, Patel AA, Singh K, Marquez-Lara A et al (2013) Incidence and risk factors for postoperative delirium after lumbar spine surgery. Spine 38(20):1790–1796
Flanigan P, Jahangiri A, Weinstein D et al (2018) Postoperative delirium in glioblastoma patients: risk factors and prognostic implications. Neurosurgery. https://doi.org/10.1093/neuros/nyx606
Fong TG, Hshieh TT, Wong B, Tommet D, Jones RN, Schmitt EM et al (2015) Neuropsychological profiles of an elderly cohort undergoing elective surgery and the relationship between cognitive performance and delirium. J Am Geriatr Soc 63(5):977–982
Inouye SK, Robinson T, Blaum C et al (2014) American geriatrics society abstracted clinical practice guideline for postoperative delirium in older adults. J Am Geriatr Soc 63(1):142–150
Jennett B (1975) Assessment of outcome after severe brain damage: a practical scale. Lancet 305(7905):480–484
Jeong JH, Seo YH, Ahn JY, Kim KH, Seo JY, Kim MJ et al (2016) The prognostic value of serum levels of heart-type fatty acid binding protein and high sensitivity C-reactive protein in patients with increased levels of amino-terminal pro-B type natriuretic peptide. Ann Lab Med 36(5):420–426
Jiang X, Chen D, Lou Y, Li Z (2016) Risk factors for postoperative delirium after spine surgery in middle- and old-aged patients. Aging Clin Exp Res 29(5):1039–1044
Kobayashi K, Imagama S, Ando K et al (2017) Risk factors for delirium after spine surgery in extremely elderly patients aged 80 years or older and review of the literature: Japan Association of Spine Surgeons with Ambition Multicenter Study. Glob spine J 7(6):560–566
Krzych ŁJ, Szurlej D, Kołodziej T, Machej L, Węglarzy A, Błach A et al (2018) Diagnostic accuracy of pre−operative NT−proBNP level in predicting short−term outcomes in coronary surgery: a pilot study. Kardiol Pol 69(11):1121–1127
Lenehan M, Summers M, Saunders N, Summers J, Vickers J (2014) Relationship between education and age-related cognitive decline: a review of recent research. Psychogeriatrics 15(2):154–162
Madeira T, Peixoto-Plácido C, Goulão B et al (2016) National survey of the Portuguese elderly nutritional status: study protocol. BMC Geriatr 16(1):1–9
Mahoney FI, Barthel DW (1965) Functional evaluation: the Barthel index. Md State Med J 14:61–65
Mor V, Laliberte L, Morris J, Wiemann M (1984) The Karnofsky performance status scale: an examination of its reliability and validity in a research setting. Cancer 53(9):2002–2007
Nazemi A, Gowd A, Carmouche J, Kates S, Albert T, Behrend C (2017) Prevention and management of postoperative delirium in elderly patients following elective spinal surgery. Clin Spine Surg 30(3):112–119
Nishizawa Y, Hata T, Takemasa I et al (2017) Clinical benefits of single-incision laparoscopic surgery for postoperative delirium in elderly colon cancer patients. Surg Endosc 32(3):1434–1440
Oldroyd C, Scholz A, Hinchliffe R, McCarthy K, Hewitt J, Quinn T (2017) A systematic review and meta-analysis of factors for delirium in vascular surgical patients. J Vasc Surg 66(4):1269–1279.e9
Raats J, Steunenberg S, de Lange D, van der Laan L (2016) Risk factors of post-operative delirium after elective vascular surgery in the elderly: a systematic review. Int J Surg 35:1–6
Saporito A, Sturini E (2013) Incidence of postoperative delirium is high even in a population without known risk factors. J Anesth 28(2):198–201
Scholz A, Oldroyd C, McCarthy K, Quinn T, Hewitt J (2015) Systematic review and meta-analysis of risk factors for postoperative delirium among older patients undergoing gastrointestinal surgery. Br J Surg 103(2):e21–e28
Shi C, Yang C, Gao R, Yuan W (2015) Risk factors for delirium after spinal surgery: a meta-analysis. World Neurosurg 84(5):1466–1472
Snaith R, Zigmond A (1986) The hospital anxiety and depression scale. BMJ 292(6516):344–344
Takeuchi M, Takeuchi H, Fujisawa D et al (2012) Incidence and risk factors of postoperative delirium in patients with esophageal cancer. Ann Surg Oncol 19(12):3963–3970
Ushida T, Yokoyama T, Kishida Y et al (2009) Incidence and risk factors of postoperative delirium in cervical spine surgery. Spine 34(23):2500–2504
Valenzuela MJ, Sachdev P (2005) Brain reserve and dementia: a systematic review. Psychol Med 36(04):441–454
van den Boogaard M, Schoonhoven L, van der Hoeven J, van Achterberg T, Pickkers P (2012) Incidence and short-term consequences of delirium in critically ill patients: a prospective observational cohort study. Int J Nurs Stud 49(7):775–783
van Swieten J, Koudstaal P, Visser M, Schouten H, van Gijn J (1988) Interobserver agreement for the assessment of handicap in stroke patients. Stroke 19(5):604–607
Vasunilashorn S, Dillon S, Inouye S et al (2017) High C-reactive protein predicts delirium incidence, duration, and feature severity after major noncardiac surgery. J Am Geriatr Soc 65(8):e109–e116
Wang L, Xu D, Wei X, Chang H, Xu G (2016) Electrolyte disorders and aging: risk factors for delirium in patients undergoing orthopedic surgeries. BMC Psychiatry 16(1):418
Acknowledgments
We would like to thank Dr. Marius Rimaitis, M.D., for his consultations regarding anesthesiology section of the paper.
Funding
The study was financially supported by the Research Council of Lithuania (grant number: MIP-044/2015).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all patients included in the study.
Additional information
Comments
This study indicates that postoperative delirium occurs after surgery for brain tumours, albeit infrequently. Also, demographic rather than tumour-related factors are associated with the occurrence. Since the patients’ performance level was assessed at discharge, this study is unlikely to say much about how postoperative delirium affects long-term performance.
Kirsten Moeller
Denmark
This article is part of the Topical Collection on Brain Tumors
Rights and permissions
About this article
Cite this article
Budėnas, A., Tamašauskas, Š., Šliaužys, A. et al. Incidence and clinical significance of postoperative delirium after brain tumor surgery. Acta Neurochir 160, 2327–2337 (2018). https://doi.org/10.1007/s00701-018-3718-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-018-3718-2