Abstract
Objective
To review current management strategies for olfactory groove meningioma (OGM)s and the recent literature comparing endoscopic endonasal (EEA) with traditional transcranial (TCA) approaches.
Methods
A PubMed search of the recent literature (2011–2016) was performed to examine outcomes following EEA and TCA for OGM. The extent of resection, visual outcome, postoperative complications and recurrence rates were analyzed using percentages and proportions, the Fischer exact test and the Student’s t-test using Graphpad PRISM 7.0Aa (San Diego, CA) software.
Results
There were 444 patients in the TCA group with a mean diameter of 4.61 (±1.17) cm and 101 patients in the EEA group with a mean diameter of 3.55 (± 0.58) cm (p = 0.0589). GTR was achieved in 90.9% (404/444) in the TCA group and 70.2% (71/101) in the EEA group (p < 0.0001). Of the patients with preoperative visual disturbances, 80.7% (21/26) of patients in the EEA cohort had an improvement in vision compared to 12.83%(29/226) in the TCA group (p < 0.0001). Olfaction was lost in 61% of TCA and in 100% of EEA patients. CSF leaks and meningitis occurred in 25.7% and 4.95% of EEA patients and 6.3% and 1.12% of TCA patients, respectively (p < 0.0001; p = 0.023).
Conclusions
Our updated literature review demonstrates that despite more experience with endoscopic resection and skull base reconstruction, the literature still supports TCA over EEA with respect to the extent of resection and complications. EEA may be an option in selected cases where visual improvement is the main goal of surgery and postoperative anosmia is acceptable to the patient or in medium-sized tumors with existing preoperative anosmia. Nevertheless, based on our results, it seems more prudent at this time to use TCA for the majority of OGMs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Olfactory groove meningiomas (OGMs) account for 4–13% of all intracranial meningiomas [1, 2, 8, 33]. Surgical removal is the treatment of choice in larger tumors with frontal lobe compression. Gross total resection with removal or fulguration of the surrounding dura mater and involved bone reduces the risk of recurrence. Options for removal of these tumors include variations of the pterional or bifrontal craniotomy, endonasal endoscopic and supraorbital minicraniotomy [4].
The endonasal endoscopic approach (EEA) for OGM is the most controversial of the approaches, and critics argue that the limited view between the orbits and difficult reconstruction anatomy behind the frontal sinus result in a suboptimal extent of resection and a high rate of postoperative cerebrospinal fluid (CSF) leakage [4]. In a review of the literature from 2000 to 2010, Komotar et al. [6] found that gross total resection (GTR) was only achieved in 63.2% using an endonasal endoscopic approach, whereas transcranial approaches (TCA) achieved a GTR in 92.8% [23]. Likewise, the CSF leak rate was 31.6% in the EEA group and 6% in the TCA group, and anosmia was present universally in the endonasal patients. However, this study included only a small number of EEA cases and was performed during the early phases of the development of this procedure. For this reason, we elected to revisit this question and review the more current literature to compare outcomes of TCA with EEA to see if experience and novel reconstruction techniques have improved on outcome.
Methods
This study was approved by the Institutional Review Board (IRB 6060088601). The methodology of our systematic review process was based on the PRISMA guidelines [30]. We performed a systematic review of recent literature using the PubMed database for the period between January 2011 and June 2016 to compare the TCA with the EEA for resection of OGMs. The following key words were queried singly and in combination: meningioma, olfactory groove, endoscopic, transcranial and approaches. All publications in the English language examining outcomes after surgery during this period for OGMs were selected. As the previous review by Komotar et al. [30] included reports between 2000 and 2010, during the early phase of the endoscopic approach to OGM, we did not include reports prior to 2010. Editorials, commentaries, review articles, articles of technical note about the approaches and anatomic studies were excluded because they did not include original data. Also excluded were articles containing mixed data (for instance, OGM with planum and tuberculum sella meningioma) that have described outcomes together for the entire group and those studies that completely lack data regarding outcome. The date of last search was June 2016.
All the included studies were reviewed and scrutinized for study design, methodology, patient characteristics and primary findings by an independent researcher. The total number of patients for each study was extracted and divided into cohorts according to treatment strategy. Selected studies were classified as either TCA or EEA. The TCA group was comprised of patients undergoing craniotomy for excision of OGMs. No distinction was made based on the type of TCA. All TCA approaches—subfrontal (unilateral and bilateral), fronto-temporal, anterior interhemispheric and lateral supraorbital—were all included under the TCA category. Patient characteristics, preoperative symptoms, extent of resection, visual and olfactory outcome, postoperative complications, mean follow-up and recurrences were extracted and analyzed for each of these groups individually and then compared. The extent of resection was documented as either gross total resection (GTR) or subtotal resection (STR). Simpson grades I and II were categorized under GTR and Simpson grades III and IV as STR. This method was followed as most (70%) of the papers reviewed presented their data as either GTR or STR without specifying the exact Simpson grading in their series. In studies where patients have undergone multiple surgeries, the percentages of GTR and STR mentioned in those series were considered. Visual outcomes were classified into “improved,” “stable” and “worsened” groups and compared between TCA and EEA cohorts. The assessment of postoperative visual function consisted of formal visual field testing in some reports and subjective patient assessment in others, but these were considered together for this analysis. The interpretation of these results is limited by an unquantified level of bias. As this review comprised case series and case reports only, an assessment of bias was made only on the outcome level rather than at the individual study level.
Statistical analysis
Data from the individual studies were combined by cohort. Percentages and proportions were used to compare the data. Statistical analyses of categorical variables were performed with Fisher exact test and Student’s t-test (for unpaired data) using Graphpad PRISM 7.0Aa (San Diego, CA). A p value < 0.05 was considered statistically significant.
Results
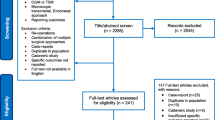
We identified 109 studies related to OGMs. After removing the duplicates and studies published prior to December 2010, 44 studies remained. Of these, 21 studies were excluded because they were purely anatomical, did not contain original data, did not separately mention the outcomes between TCA and EEA or were just technical notes. In the end, 23 studies were selected for review. Of the 23, there were 13 TCAs, 8 EEAs and 2 that compared TCA with EEA [4, 9]. The data from these last two studies [4, 9] were separated into TCA and EEA groups and included in the main cohort, thereby giving us a total of 15 transcranial and 10 endoscopic studies with data for analysis and comparison (Fig. 1).
The details of each group with regard to the first author, year of publication, patient characteristics, outcome and complications have been tabulated and presented in Tables 1 and 2. Of the 15 reports in the TCA group, 12 were retrospective, 2 were prospective and 1 was a prospective observational study. Among the ten EEA reports, five were retrospective, and five were case reports.
TCA cohort
There were 444 patients in the TCA group; 39.85% (169/424) of them were males. The mean age at presentation was 56.3 years. The most common presenting symptoms were anosmia, present in 249 of the 434 reported cases (57.37%), followed by headache (194/384, 50.5%), visual disturbances (197/431, 45.7%), behavioral changes (180/415, 43.37%) and seizures (60/321, 18.69%). There were five patients (5/431) with incidental meningiomas (1.16%), and 10.05% of patients had prior surgery (19/189). The mean tumor diameter was 4.61 (±1.17) cm.
EEA cohort
There were 101 patients in the EEA group of which 28.7% were males. The mean age was 51.7 years. The most common presenting symptom in this cohort was visual disturbances, seen in 26 of the 77 reported cases (33.7%), followed by behavioral changes (23/80–28.7%), headache (22/82–26.8%) and anosmia (21/82–25.6%). Incidental meningiomas were seen in 18.2% (15/82) and seizures were present in 17.5% (14/80) of patients at presentation. The mean tumor diameter was 3.55 (±0.58) cm (p = 0.0589).
Outcomes
The outcome with regard to extent of resection, postoperative improvement in symptoms, postoperative complications, perioperative mortality and postoperative recurrence is presented in Table 4 and Fig. 2.
Extent of resection
Gross total resection (GTR) included Simpson’s grades I and II. GTR was achieved in 90.9% (404/444) in the TCA group and 70.2% (71/101) in the EEA group (p < 0.0001).
Subtotal resection (STR) was 29.7% (30/101) in the EEA group and 9% (40/444) in the TCA group (p < 0.0001).
Visual and olfactory outcomes
Of the patients with preoperative visual disturbances, 80.7% (21/26) of patients in the EEA cohort had an improvement in vision compared to 12.8% (29/226) in the TCA group (p < 0.0001). None of the patients had postoperative worsening of vision in the EEA group while postoperative visual worsening was seen in 6.63% (15/226) patients in the TCA cohort (p = 0.37).
In the TCA group, olfaction was lost in 61.9% (156/252) of the patients in whom it was reported. Olfaction is not reported in most EEA reports since it is assumed to be lost in all patients. Therefore, statistics cannot be performed on this data point.
Postoperative complications
CSF leak was found to be significantly higher in the EEA cohort (26/101: 25.74%) compared to the TCA cohort (28/444: 6.3%; p < 0.0001) as was the chance of meningitis (TCA 5/444: 1.12%, EEA 5/101: 4.95%; p = 0.023).
The occurrence of other complications such as epilepsy (TCA 19/444: 4.27%, EEA 2/101: 1.98%), hydrocephalus (TCA 9/444: 2.02%, EEA 4/101: 3.96%), infection (TCA 9/444: 2.02%, EEA 4/101: 3.96%) and stroke (28/444: 6.30%, 3/101: 2.97%) was not found to be significantly different between the two cohorts.
While there was no perioperative mortality in the EEA cohort, there was a 2.47% (11 of the 444 patients) perioperative mortality in the TCA cohort (p = 0.23).
Recurrences
Recurrences were documented in 7/90 patients (7.77%) in the EEA cohort compared to 31/441 (7.02%) in the TCA cohort (p = 0.8224). However, the TCA cohort had a significantly longer mean follow-up (65.39 ± 29.19 months) compared to the EEA cohort (22.6 ± 17.01 months) (p = 0.0030).
Discussion
Our study demonstrates that, despite improvement of the endoscopic technique, the results of the TCA are still superior to the EEA for treating OGMs. If preservation of neurological function is considered a primary goal, then TCA would be the preferred approach in most patients with OGM and preserved preoperative olfaction since EEA universally leads to loss of olfaction. Moreover, those patients with exiting preoperative anosmia tend to have larger tumors extending beyond the mid-orbit and up the back wall of the frontal sinus; TCA is also preferable for these patients to achieve GTR or Simpson grade I or II with low morbidity. However, visual outcome may be better with EEA; thus, there may be a subgroup of patients with predominantly visual symptoms and appropriately sized and located tumors that may be amenable to EEA. However, loss of olfaction is almost inevitable, and risk of CSF leak is still on the order of 25%, so careful discussion must be had with the patients prior to embarking on an EEA. There is only a single case report in the literature where, in a patient with a small, unilaterally positioned OGM, the surgeons were able to preserve olfaction with the EEA. However, this is an atypical case, and in general EEA results in anosmia [54].
Historical context
The traditional approach to OGMs has been the transcranial approach. The subfrontal approach (either bifrontal or unifrontal), pterional and fronto-temporal approaches, lateral supraorbital and trans-glabellar approaches, and anterior interhemispheric approach have all been utilized to approach and resect OGMs. These approaches may involve extensive bone removal, transgression of the frontal sinus with increased risk of CSF leak and meningitis, various degrees of brain retraction, narrow working angle and the late visualization of critical neurovascular structures [34, 36, 39, 43, 47, 50]. However, complete removal of tumors is likely, and tumors that extend beyond the lamina papyracea or up to the back wall of the frontal sinus generally require a craniotomy for complete removal.
Over the last 10 years, EEA has been increasingly utilized to remove OGMS with several potential advantages over TCA. EEA provides wide visualization of the midline skull base, complete resection of all abnormal bone and dura under the tumor, early devascularization of the meningioma and direct visualization of the neurovascular structures of the suprasellar and infrachiasmatic region from below. This allows a safe tumor resection with minimal neurovascular manipulation, easy decompression of the bilateral medial optic canals, lack of brain retraction and no scar on the skin [7, 10, 12, 16, 17, 25]. The disadvantages of EEA, however, are the increased risk of CSF leakage, which necessitates repeat surgeries for repair and a potentially higher risk of postoperative meningitis and abscess, and the inevitability of anosmia, which is not a fait accompli when performing a craniotomy, particularly in the smaller tumors that presumably would be the best candidates for EEA [25]. The other important limitation of EEA is the inability to see laterally beyond the lamina papyracea and the difficulty in reaching high up behind the frontal sinus [23, 25].
Our literature review has demonstrated that 101 patients in 10 published series have undergone EEA resection of OGMs in the last 6 years (2011–2016). This is a significant increase from the previous 10 years (2000–2010) when 3 series were published totaling 19 patients [23]. This indicates a definite trend toward increasing utilization of EEA for resection of OGMs over the last few years. However, it is unclear whether the literature supports such an expansion in the use of EEA.
Patient selection
When we compared the initial symptoms of patients chosen for TCA versus EEA, we found a higher percentage of patients in the TCA cohort had loss of smell, headache and behavioral disturbances compared to patients in the EEA cohort, and this difference was statistically significant (p < 0.05) (Table 3). These symptoms are markers for larger tumors. The incidental meningiomas were more common in the endoscopic cohort. This difference could be due to the fact that patients with large symptomatic tumors were more likely to be operated on via TCA, while patients with a relatively smaller incidental meningioma might prefer to avoid a craniotomy. However, the smaller tumors are the very same tumors where olfaction can be preserved through a TCA and is inevitably lost through EEA.
Extent of resection
The goal of meningioma surgery is safe gross total tumor resection irrespective of the approach. The gross total resection (Simpson’s grade I and II) rate was significantly better in patients who underwent TCA compared to the EEA approach. Koutourousiou et al. [25] in their paper on endoscopic endonasal resection of olfactory groove meningiomas rightly pointed out that most series mention Simpson grade I and II resections together. In their series, they achieved a Simpson grade I resection in 66.7% of patients (in patients planned for GTR), in line with most traditional open approaches where Simpson grade I resection varies from 23.8% to 84.6% [2, 5, 18]. In our review, we also found that a significantly higher number of patients in the endoscopic group had STR (Simpson grade III and IV) compared to open approaches. This seems to be a concern with the endoscopic approach. Neurosurgeons are either able to achieve Simpson grade I excision or they are left with a residual tumor (adherent to the brain and neurovascular structures) leading to subtotal resection. There is no scope for a Simpson grade 2 resection with EEA. This was highlighted in a study by Almeida et al. [9]. In their series, the transcranial group had 20% grade I, 70% grade II and 10% grade IV excision, and the endoscopic group had 70% grade I, 0% grade II and 30% grade IV excision. The reason for the lack of grade 2 resection is that the ability to coagulate the dural tail is lost in EEA based on the limited field of view, especially if the dural tail extends laterally over the skull base. Hence, tumors with a dural tail extending laterally may not be good candidates for EEA, whereas those with no tail may be more appropriate.
Size influencing GTR
For suitable candidates, EEA is capable of achieving a grade I excision because of the inevitable removal of the bony base. Koutourousiou et al. [25] were able to achieve a grade I excision in 90.5% of the tumors measuring <40 mm and only 45.8% in tumors >40 mm. The lack of an arachnoid plane and large tumors with lateral and anterior tumor extension are seen to limit the extent of resection in the endoscopic endonasal approach. However, tumors <40 mm are precisely the tumors that have intact preoperative olfaction and where olfaction can be preserved after TCA [25]. Moreover, smaller tumors are less likely to invade the cribriform plate and less in need of bone removal [25]. For TCA, on the other hand, size does not seem to significantly influence GTR. Studies on open TCA of small (<35 mm) meningiomas (Puppa et al. [41]) showed a GTR of 100%, and TCA for giant (>60 mm) OGMs (Tomasello et al. [53]) showed a GTR of 94.4%. In this review, we found GTR of 90.9% in patients who underwent TCA with the mean tumor diameter being 46 mm.
Outcomes
Vision
In this review, we found a significant improvement in postoperative visual outcome in patients who underwent endoscopic resection. Further, there was no deterioration of vision following EEA, while worsening was seen in 6.63% of patients with TCA. Technically, both approaches attempt to achieve decompression of the optic apparatus. While the transcranial approach may involve opening up the optic canal for early identification of the optic nerve [27, 35], manipulation of the optic apparatus may be required to reach and remove tumors located below the chiasm. The better outcomes achieved with EEA could be attributed to the technical advantage of being able to decompress from below with minimal manipulation of the optic apparatus [3, 11, 14, 22, 24],. However, the pitfall with trying to make a direct comparison is that the microsurgical series have tumors of different sizes compared to the endoscopic group where most of the tumors are <40 mm. A selection bias was also found whereby surgeons are sub-selecting smaller OGMs for EEA, especially the tumors that are posteriorly located along the olfactory groove and are causing visual compromise. These are the patients whose vision would benefit most from an endoscopic decompression of the chiasm from below, even with a sub-total resection of the tumor. In addition, most of the microsurgical series reviewed had data spanning 2 decades. Whether the refinements in microsurgical techniques during this time could have made a difference to the visual outcome in patients undergoing TCA is possible but remains speculative. Further, the degree of visual compromise at presentation, duration of symptoms and patient age may all contribute to the outcome [28, 31, 44].
Olfaction
Loss of olfaction can cause profound psychological problems affecting occupation and quality of life [48, 52]. In this review, olfaction was lost in 61.9% (156/252) of the patients in the TCA group. Unfortunately, most EEA studies do not document olfactory function after resection of OGM since it is assumed to be lost in all patients with removal of the cribriform plate. Transcranial approaches seem to have a better chance of preserving olfaction than endoscopic approaches. In a study by Banu et al. [4], which compared the supraorbital approach with endoscopic and combined approaches for OGMs, anosmia occurred in 100% of patients who underwent EEA and combined approaches, while only 57.1% of the supraorbital patients developed postoperative anosmia. A study by Jang et al. [19] found that preservation of olfaction was better in patients without preoperative olfactory dysfunction, size <40 mm (<40 mm: 78.6%, >40 mm: 42.3%; p = 0.035) and with fronto-lateral approaches. Although a single case has been reported of olfactory preservation by EEA to remove a small unilateral OGM, this is highly atypical [19].
The impact of the inevitability of the loss of olfaction on approach selection is significant. In patients with intact preoperative olfaction, EEA is less appealing since anosmia will occur. These patients tend to have smaller tumors that might be more anatomically suitable for EEA. However, the larger tumors with preoperative anosmia may extend too far laterally for EEA. Hence, only medium-sized tumors with impaired olfaction are best suited for EEA. Stereotactic radiosurgery (SRS) is another option to consider in the management of OGMs. SRS can be used either up front in a select group of patients with appropriately sized tumors or as an adjuvant for residual tumors after partial resection. Control rates of 95% with preservation of olfaction in 100% have been reported [15]. These data further support the concept of TCA to preserve olfaction even at the risk of leaving a small amount of invaded dura at the skull base since SRS may control further tumors growth without causing anosmia.
Complications
CSF leak
In this review, patients with EEA had a significantly higher rate of postoperative CSF leak and meningitis compared to patients undergoing TCA. Koutourousiou et al. [25] in their study of endoscopic endonasal surgery for OGMs reported a postoperative CSF leak rate of 30%. They found the configuration of the free surface of the meningioma to be the single significant factor associated with increased CSF leak rates. The importance of the learning curve was also highlighted in that study, with the CSF leak rates decreased to 20% in their most recent 15 cases. Banu et al. [4] similarly reported CSF leak in 16.6% of their EEA cases.
The fact that even this more updated literature review, taken from years when the nasoseptal flap was in use in all advanced centers for skull base reconstruction, still shows such a high rate of CSF leak indicates that the anterior cranial fossa remains a difficult area to close. While CSF leak rates are declining for other pathologies in other locations, the leak rate remains fairly high for OGMs. The posterior wall of the frontal sinus and the area of the crista galli are difficult to cover with a nasoseptal flap.
The learning curve
We compared this review of literature done for the time period between 2011 and 2016 with another review that compared open and endoscopic approaches for OGM between 2000 and 2010 published by Komotar et al. [23] (Tables 4 and 5). As shown in Table 5, there is little difference in either the TCA or the EEA cohorts between the two time periods, with the exception of EEA volume. The number of patients who have undergone endonasal endoscopic resection of OGM has gone up more than five fold in the last 6 years. There is a small increase in the percentage of GTR from 63.2% to 70.2% with a slight reduction in STR from 36.8% to 29.7%. The CSF leak rates were also marginally reduced from 31.6% to 25.7% in the present review but continue to be significantly higher than for the TCA approaches. The lack of substantial improvement in the outcomes for OGMs should give pause to those strongly supporting EEA for OGMs.
Limitations
The various limitations of this study are:
-
1.
Heterogeneity between the two cohorts. The mean tumor diameter was greater in the TCA cohort. However, this would only favor better results in the EEA group, which was not the case. The EEA cohort had limited follow-up (mean, 22.6 months) than the TCA group (mean, 65.39 months). Longer follow-up is necessary to evaluate the long-term outcome and recurrence rates in the endoscopic cohort.
-
2.
The study involves published literature from multiple centers and multiple surgeons. Study heterogeneity was substantial with various study designs, methodologies and patient populations. This heterogeneity prevented true meta-analysis or multivariable statistics.
-
3.
The outcome reporting bias within individual studies adds to the limitation of this review. Inadequate or incomplete data among the published literature compound the problem. It is important for researchers/clinicians/editors to make it mandatory to incorporate a minimum data set when publishing a report for a particular pathology. For instance, in patients undergoing surgery for OGM, age, tumor size, preoperative anosmia/visual disturbance, prior surgery, Simpson’s grading, postoperative anosmia/visual outcomes, CSF leaks, mortality, follow-up duration and recurrence rates should be the minimum data that need to be presented. This would help in a thorough comparison between various approaches in the future.
Conclusion
In the resection of OGM, the recent literature continues to favor TCA for its higher gross total resection rates with lower postoperative CSF leaks irrespective of tumor size. SRS may also be an option for smaller tumors without significant mass effect or small residual tumors after surgery, and olfaction preservation is quite high. EEA may be an option in selected cases where visual improvement is the main goal of surgery and postoperative anosmia is acceptable to the patient or in medium-sized tumor with existing preoperative anosmia. Nevertheless, based on our results, it seems more prudent at this time to use TCA for the majority of OGMs.
References
Adappa ND, Lee JYK, Chiu AG, Palmer JN (2011) Olfactory groove meningioma. Otolaryngol Clin N Am 44(4):965–980 ix
Aguiar PHP d, Tahara A, Almeida AN, Simm R, Silva, Maldaun MVC, Panagopoulos AT, Zicarelli CA, Silva PG (2009) Olfactory groove meningiomas: approaches and complications. J Clin Neurosci 16(9):1168–1173
al-Mefty O, Holoubi A, Rifai A, Fox JL (1985) Microsurgical removal of suprasellar meningiomas. Neurosurgery 16(3):364–372
Banu MA, Mehta A, Ottenhausen M, Fraser JF, Patel KS, Szentirmai O, Anand VK, Tsiouris AJ, Schwartz TH (2016) Endoscope-assisted endonasal versus supraorbital keyhole resection of olfactory groove meningiomas: comparison and combination of 2 minimally invasive approaches. J Neurosurg 124(3):605–620
Bassiouni H, Asgari S, Stolke D (2007) Olfactory groove meningiomas: functional outcome in a series treated microsurgically. Acta Neurochir 149(2):109–121 discussion 121
Bitter AD, Stavrinou LC, Ntoulias G, Petridis AK, Dukagjin M, Scholz M, Hassler W (2013) The role of the Pterional approach in the surgical treatment of olfactory groove Meningiomas: a 20-year experience. J Neurol Surg B Skull Base 74(2):97–102
Cappabianca P, Cavallo LM, Esposito F, De Divitiis O, Messina A, De Divitiis E (2008) Extended endoscopic endonasal approach to the midline skull base: the evolving role of transsphenoidal surgery. Adv Tech Stand Neurosurg 33:151–199
Ciurea AV, Iencean SM, Rizea RE, Brehar FM (2012) Olfactory groove meningiomas: a retrospective study on 59 surgical cases. Neurosurg Rev 35(2):195–202 discussion 202
de Almeida JR, Carvalho F, Vaz Guimaraes Filho F et al (2015) Comparison of endoscopic endonasal and bifrontal craniotomy approaches for olfactory groove meningiomas: a matched pair analysis of outcomes and frontal lobe changes on MRI. J Clin Neurosci 22(11):1733–1741
de Divitiis E, Cavallo LM, Cappabianca P, Esposito F (2007) Extended endoscopic endonasal transsphenoidal approach for the removal of suprasellar tumors: part 2. Neurosurgery 60(1):46–58 discussion 58–
de Divitiis O, de Divitiis E (2012) Anterior cranial fossa meningiomas: a new surgical perspective. World Neurosurg 77(5–6):623–624
Dehdashti AR, Ganna A, Witterick I, Gentili F (2009) Expanded endoscopic endonasalapproach for anterior cranial base and suprasellar lesions. Neurosurgery 64(4):677–689
Di Maio S, Ramanathan D, Garcia-Lopez R, Rocha MH, Guerrero FP, Ferreira M, Sekhar LN (2012) Evolution and future of skull base surgery: the paradigm of skull base meningiomas. World Neurosurg 78(3–4):260–275
Fahlbusch R, Schott W (2002) Pterional surgery of meningiomas of the tuberculum sellae and planum sphenoidale: surgical results with special consideration of ophthalmological and endocrinological outcomes. J Neurosurg 96(2):235–243
Gande A, Kano H, Bowden G, Mousavi SH, Niranjan A, Flickinger JC, Lunsford DL (2014) Gamma knife radiosurgery of olfactory groove meningiomas provides a method to preserve subjective olfactory function. J Neuro-Oncol 116:577–583
Gardner PA, Kassam AB, Thomas A, Snyderman CH, Carrau RL, Mintz AH, Prevedello DM (2008) Endoscopic endonasal resection of anterior cranial base meningiomas. Neurosurgery 63(1):36–52 discussion 52–4
Gardner PA, Vescan A, de Almeida JR, Janjua A, Kassam AB, Prevedello DM, Carrau RL, Snyderman CH (2012) Endoscopic endonasal approach for olfactory groove meningiomas. In: Endoscopic Approaches to the Skull Base. S. Karger AG, Basel, pp 76–86
Hentschel SJ, DeMonte F (2003) Olfactory groove meningiomas. Neurosurg Focus 14(6):e4
Jang W-Y, Jung S, Jung T-Y, Moon K-S, Kim I-Y (2013) Preservation of olfaction in surgery of olfactory groove meningiomas. Clin Neurol Neurosurg 115(8):1288–1292
Khan OH, Krischek B, Holliman D, Klironomos G, Kucharczyk W, Vescan A, Gentili F, Zadeh G (2014) Pure endoscopic expanded endonasal approach for olfactory groove and tuberculum sellae meningiomas. J Clin Neurosci 21(6):927–933
Kim YH, Han DH, Park CK, Lee CH (2011) An endoscopic endonsal transethmoidal approach to olfactory groove meningioma. Cent Eur Neurosurg 72(04):205–208
Kitano M, Taneda M, Nakao Y (2007) Postoperative improvement in visual function in patients with tuberculum sellae meningiomas: results of the extended transsphenoidal and transcranial approaches. J Neurosurg 107(2):337–346
Komotar RJ, Starke RM, Raper DMS, Anand VK, Schwartz TH (2012) Endoscopic endonasal versus open transcranial resection of anterior midline skull base meningiomas. World Neurosurg 77(5–6):713–724
Koutourousiou M, Fernandez-Miranda JC, Stefko ST, Wang EW, Snyderman CH, Gardner PA (2014) Endoscopic endonasal surgery for suprasellar meningiomas: experience with 75 patients. J Neurosurg 120(6):1326–1339
Koutourousiou M, Fernandez-Miranda JC, Wang EW, Snyderman CH, Gardner PA (2014) Endoscopic endonasal surgery for olfactory groove meningiomas: outcomes and limitations in 50 patients. Neurosurg Focus 37(4):E8
Liu JK, Christiano LD, Patel SK, Tubbs RS, Eloy JA (2011) Surgical nuances for removal of olfactory groove meningiomas using the endoscopic endonasal transcribriform approach. Neurosurg Focus 30(5):E3
Mahmoud M, Nader R, Al-Mefty O (2010) Optic canal involvement in tuberculum sellae meningiomas. Operative. Neurosurgery 67:ons108–ons119
Margalit N, Shahar T, Barkay G, Gonen L, Nossek E, Rozovski U, Kesler A (2013) Tuberculum sellae meningiomas: surgical technique, visual outcome, and prognostic factors in 51 cases. J Neurol Surg B Skull Base 74(4):247–258
Mielke D, Mayfrank L, Psychogios MN, Rohde V (2014) The anterior interhemispheric approach: a safe and effective approach to anterior skull base lesions. Acta Neurochir 156(4):689–696
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264–269 W64
Mortini P, Barzaghi LR, Serra C, Orlandi V, Bianchi S, Losa M (2012) Visual outcome after fronto-temporo-orbito-zygomatic approach combined with early extradural and intradural optic nerve decompression in tuberculum and diaphragma sellae meningiomas. Clin Neurol Neurosurg 114(6):597–606
Musluman AM, Yilmaz A, R TC, Cavusoglu H, Kahyaoglu O, Aydin Y (2012) Unilateral frontal interhemispheric transfalcial approaches for the removal of olfactory groove meninjiomas. Turk Neurosurg 22(2):174–182
Nakamura M, Struck M, Roser F, Vorkapic P, Samii M (2007) Olfactory groove meningiomas. Neurosurgery 60(5):844–852
Nakamura M, Struck M, Roser F, Vorkapic P, Samii M (2008) Olfactory groove meningiomas: clinical outcome and recurrence rates after tumor removal through the frontolateral and bifrontal approach. Neurosurgery 62(6 Suppl 3):1224–1232
Nozaki K, Kikuta K-I, Takagi Y, Mineharu Y, Takahashi JA, Hashimoto N (2008) Effect of early optic canal unroofing on the outcome of visual functions in surgery for meningiomas of the tuberculum sellae and planum sphenoidale. Neurosurgery 62(4):839–846
Obeid F, Al-Mefty O (2003) Recurrence of olfactory groove meningiomas. Neurosurgery 53(3):534–542 discussion 542–3
Padhye V, Naidoo Y, Alexander H et al (2012) Endoscopic endonasal resection of anterior skull base meningiomas. Otolaryngol Head Neck Surg 147(3):575–582
Pallini R, Fernandez E, Lauretti L, Doglietto F, D'Alessandris QG, Montano N, Capo G, Meglio M, Maira G (2015) Olfactory groove meningioma: report of 99 cases surgically treated at the Catholic University School of Medicine, Rome. World Neurosurg 83(2):219–231 e1–3
Pamir MN, Ozduman K, Belirgen M, Kilic T, Ozek MM (2005) Outcome determinants of pterional surgery for tuberculum sellae meningiomas. Acta Neurochir 147(11):1121–1130 discussion 1130
Pepper J-P, Hecht SL, Gebarski SS, Lin EM, Sullivan SE, Marentette LJ (2011) Olfactory groove meningioma: discussion of clinical presentation and surgical outcomes following excision via the subcranial approach. Laryngoscope 121(11):2282–2289
Puppa Della A, d’Avella E, Rossetto M, Volpin F, Rustemi O, Gioffrè G, Lombardi G, Rolma G, Scienza R (2015) Open transcranial resection of small (<35 mm) meningiomas of the anterior midline skull base in current microsurgical practice. World Neurosurg 84(3):741–750
Refaat MI, Eissa EM, Ali MH (2015) Surgical management of midline anterior skull base meningiomas: experience of 30 cases. Turk Neurosurg 25(3):432–437
Samii M, Ammirati M (1992) Olfactory groove meningiomas. In: Surgery of Skull Base Meningiomas. Springer, Berlin Heidelberg, pp 15–26
Schick U, Hassler W (2005) Surgical management of tuberculum sellae meningiomas: involvement of the optic canal and visual outcome. J Neurol Neurosurg Psychiatry 76(7):977–983
Schroeder HWS, Hickmann A-K, Baldauf J (2011) Endoscope-assisted microsurgical resection of skull base meningiomas. Neurosurg Rev 34(4):441–455
Skorek A, Liczbik W, Stankiewicz C, Kloc W, Plichta Ł (2014) Personal experience in transnasal endoscopic resection of the olfactory groove meningiomas. What can an otolaryngologist offer to a neurosurgeon? Eur Arch Otorhinolaryngol 271(5):1037–1041
Solero CL, Giombini S, Morello G (1983) Suprasellar and olfactory meningiomas. Report on a series of 153 personal cases. Acta Neurochir 67(3–4):181–194
Spetzler RF, Herman JM, Beals S, Joganic E, Milligan J (1993) Preservation of olfaction in anterior craniofacial approaches. J Neurosurg 79(1):48–52
Sughrue M, Bonney P, Burks J, Hayhurst C, Gore P, Teo C (2016) Results with expanded endonasal resection of skull base meningiomas: technical nuances and approach selection based on an early experience. Turk Neurosurg 26(5):662–670
Symon L (1977) Olfactory groove and suprasellar meningiomas. In: Advances and technical standards in neurosurgery. Springer Vienna, Vienna, pp 67–91
Telera S, Carapella CM, Caroli F, Crispo F, Cristalli G, Raus L, Sperduti I, Pompili A (2012) Supraorbital keyhole approach for removal of midline anterior cranial fossa meningiomas: a series of 20 consecutive cases. Neurosurg Rev 35(1):67–83 discussion 83
Toller SV (1999) Assessing the impact of anosmia: review of a questionnaire's findings. Chem Senses 24(6):705–712
Tomasello F, Angileri FF, Grasso G, Granata F, De Ponte FS, Alafaci C (2011) Giant olfactory groove meningiomas: extent of frontal lobes damage and long-term outcome after the pterional approach. World Neurosurg 76(3–4):311–317 discussion 255–8
Youssef AS, Sampath R, Freeman JL, Mattingly JK, Ramakrishnan VR (2016) Unilateral endonasal transcribriform approach with septal transposition for olfactory groove meningioma: can olfaction be preserved? Acta Neurochir 158(10):1965–1972
Zhang Q, Wang Z, Guo H, Kong F, Chen G, Bao Y, Ling F (2012) Resection of anterior cranial base meningiomas with intra- and extracranial involvement via a purely endoscopic endonasal approach. ORL J Otorhinolaryngol Relat Spec 74(4):199–207
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Comments
The authors performed a systematic review of the recent literature to compare the transcranial approach to endoscopic endonasal approach for resection of olfactory groove meningiomas. The first thing to mention is clearly clarifying what an OGM is. So a tuberculum sella or planum sphenoidale meningioma with a higher risk of visual pathway compression is theoretically excluded. As has been shown previously, in this particular subset (OGMs compared to tuberculum sella meningiomas), there is not much benefit from the endonasal approach. The minimally invasive transcranial approaches [whether fronto-lateral or lateral supraorbital (LSO) craniotomies] are clearly better choices with fewer complications regarding CSF leaks and much better resection rates. The fate of visual improvement, albeit not confirmed by the literature in this study, is dependent on minute microsurgical dissection of the tumor from the visual apparatus. However, many patients with true OGMs who are possible candidates for endonasal approaches (in terms of size and extension) are not symptomatic for visual pathway compression, or, if having larger tumors, are therefore not suitable candidates for an endonasal approach. So the argument for visual improvement via an endonasal technique becomes irrelevant. The olfactory groove is a lost territory for the endonasal approach, and although I am an advocate for expanded endonasal techniques, I feel the endonasal approach is not an appropriate choice for true OGMs.
Amir Dehdashti
NY, USA
Institutional review board (IRB) number: 6060088601
Rights and permissions
About this article
Cite this article
Shetty, S.R., Ruiz-Treviño, A.S., Omay, S.B. et al. Limitations of the endonasal endoscopic approach in treating olfactory groove meningiomas. A systematic review. Acta Neurochir 159, 1875–1885 (2017). https://doi.org/10.1007/s00701-017-3303-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-017-3303-0