Abstract
Background
Resection is recommended for low-grade gliomas, but often it is not performed if the tumor is suspected of invading the primary motor cortex. The study aim is to assess what influence preoperative navigated transcranial magnetic stimulation (nTMS) has on the treatment strategy and clinical outcome for suspected low-grade gliomas in presumed motor eloquent location.
Methods
This paper reports on all our patients with gliomas in the primary motor cortex that were non-enhancing on MRI, since we began using nTMS (n = 11). For the comparison group, we identified the 11 most recent such patients just before we started using nTMS.
Results
Exact delineation of motor functional versus non-functional cortical tissue was provided by nTMS in all cases, also within the area of altered FLAIR signal. In 6 out of 11 cases, the nTMS mapping result changed the treatment plan towards early and more extensive resection. Only one nTMS patient had another seizure within the follow-up period, whereas four patients in the comparison group had further seizures. In the nTMS group, 1 of 4 patients with pre-op neurological deficits improved by one year; whereas the comparison group had increased neurological deficits in 3 of the 8 patients not having surgery. The median (range) change of tumor volume from baseline to 1 year was −83 % (−67 % to −100 %) in the nTMS group, but +12 % (+40 % to −56 %) in the comparison group (p < 0.001).
Conclusions
nTMS provides accurate motor mapping results also in infiltrative gliomas and enables more frequent and more extensive surgical resection of non-enhancing gliomas in or near the primary motor cortex. The substantial differences observed here in neurological and oncological outcomes suggest that further comparative research is warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gliomas are brain tumors originating from glia, the supporting cells of the central nervous system and WHO grades them I-IV, according to increasing malignancy [13, 20]. Low-grade gliomas were until recently viewed as benign because patients could live with them for years, so they were “treated” with a wait-and-watch approach [2, 9, 27]. Now, however, grade II gliomas are also viewed as malignant, because inevitably they will become anaplastic and kill the patient [3, 21]. Early resection has been shown to increase survival time over waiting-and-watching [11]. So the recommended first-line treatment for grade II gliomas is undelayed aggressive resection, when possible [1, 11, 25, 31].
Yet, surgical resection can cause motor deficits when gliomas occur in or near the primary motor cortex, and although leading experts and guidelines still recommend resection in such cases [4, 7, 10, 31], many neurosurgeons feel the risk is too high for non-enhancing gliomas and prefer the traditional wait-and-watch approach or non-surgical treatment in such cases [26, 33, 34]. A recent nationwide survey of German academic neurosurgery departments reported that 50 % of all respondents routinely practice a wait-and-watch approach for low-grade gliomas in general [30].
Navigated transcranial magnetic stimulation (nTMS) can be used to map the motor cortex preoperatively [23]. The aim of this report is to assess the difference in treatment strategy for non-enhancing gliomas in or near the motor cortex when the patient receives nTMS mapping.
Methods
Study design and sample
The study is designed as an observational comparative effectiveness study. The prospective nTMS group includes all patients with non-enhancing gliomas suspected of invading the primary motor cortex, seen at our department since we began using nTMS in October 2007 through the end of 2010 (n = 11). To form a comparison group, we performed a post hoc analysis of prospectively collected data of our last 11 patients before we introduced nTMS who had non-enhancing gliomas suspected of invading the primary motor cortex (September 2001 to April 2007). To facilitate comparison, we limit ourselves to the first year from the initial presentation. Collection of data was approved by the university hospital’s ethics commission (reference # EA4/007/06).
Preoperative diagnostics
All patients underwent brain mapping with nTMS (eXimia; Nexstim; Helsinki, Finland), as described previously [23]. Briefly, magnetic stimulation was applied transcranially to points of the brain in and around the tumor, and the motor response was recorded by EMG. This enabled us to map the cortical areas essential for motor function.
Influence on surgical planning
The nTMS results were presented to the surgeon only after the treatment strategy had been stated based on the anatomical MRI’s alone. The surgeon then revised the surgical plan if desired and evaluated the influence of the nTMS results on the surgical planning, using a 0–6 descriptive categorical ranking questionnaire, as previously described [24].
Surgery
Surgery was performed in a standard fashion under general anesthesia, using neuronavigation, somatosensory electric potential phase reversal, and direct electrical stimulation (DES), as described previously in greater detail [23, 32]. The nTMS mapping result was confirmed by DES in all cases.
Clinical data
Motor status was graded on the BMRC scale (British Medical Research Council scale for muscular strength). The tumor location was determined by the Department of Radiology on the basis of MRI. Tumor volume was measured from the FLAIR-weighted MRIs. Tumor histology was determined post-operatively by the Department of Pathology on the basis of resected tissue samples.
Results
Study sample
The two study groups were quite similar (Table 1). Based on anatomical MRI, all tumors were suspected of invading the precentral gyrus. All tumors had well defined brain–tumor interfaces with the FLAIR signal abnormality only slightly exceeding the T1 hyperintense area.
Treatment course
The treatment course during the first year is presented in Fig. 1. Nearly all nTMS patients had surgery within the first month. Only three historical patients (# 3, 4, 10) received surgery during the first year. Chemotherapy was not used more in the historical group than the nTMS group (n = 2 in each group), and radiotherapy was also not used strikingly more in the historical group (n = 3 during 1 month each) than in the nTMS group (n = 1 during one month). Three patients in the historical group (# 1, 5, 8) never received any form of oncological treatment at all during the first year.
Course of treatment. The x-axis show the time from initial presentation; yhe y-axis shows the subject numbers. S surgery, b biopsy, r radiotherapy, c chemotherapy. The timeline is only accurate to the nearest month, but the sequence of letters does properly indicate the sequence of treatments when more than one occurred in the same month
Navigated transcranial magnetic stimulation (nTMS)
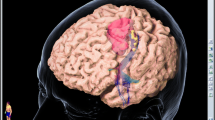
The MRIs and nTMS mapping are shown in Fig. 2. nTMS demonstrated that there was no motor function within the solid tumor mass (the T1 hypointense area) in any case (Fig. 2 – middle column). In six cases (# 1, 2, 5, 8, 10, 11), the MRI FLAIR area was slightly larger than the T1 hypointense area, and nTMS identified motor function within this excess FLAIR area in all 5 cases where it was performed.
Imaging and key information on the nTMS cases. The figure shows the pre-op MRI, the pre-op TMS map, and the post-op MRI for all 11 nTMS cases. Each row of text and figures is for one patient. The text in each row presents the case number, sex, and age of the patient; the hemisphere, location, and volume of the tumor; and the extent of surgical resection achieved. The left column of figures is the pre-op MRI. The central column of figures is the TMS map. Colored pins indicate points where TMS resulted in a motor response; gray pins indicate points where TMS did not result in a motor response. We have outlined the MRI T1 area (solid tumor) in red and MRI FLAIR area (tumor invasion) in yellow. The right column of figures is the post-op MRI which is a matching slice to the pre-op image. Pre-op and post-op MRIS are without contrast enhancement. Two cases (#2, 9) had a second surgery at 7 months using a new nTMS mapping (images not shown). On the cortical level, the DES “stop signal” always coincided with the resection margins determined preoperatively with nTMS
Influence on surgical planning
The addition of the nTMS data changed the surgeon’s approach in four cases (36 %) towards a more extensive resection (cases #3, #4, #5, #11) and in two cases (18 %) the surgical indication was changed (cases #3, #6)—meaning that without the nTMS data, the surgeons would have opted for watchful waiting, which is not uncommon if the tumor is predominantly located within an area which normally carries essential function [12, 30].
Surgery
The solid tumor mass was completely resected in 10 of 11 nTMS cases (Fig. 2, right column). In the six cases where the MRI FLAIR area noticeably exceeded the T1 hypointense area (# 1, 2, 5, 8, 10, 11), the resection usually extended beyond the solid tumor mass into this presumed tumor infiltration zone, except where nTMS and DES had confirmed function. Three historical controls (#3, 4, 10) had surgery, but only a partial resection was achieved: about 5 % of T1 in control #3, 70 % in control #4, and 60 % in control #10.
Pathology
All cases and controls were initially believed to have low-grade gliomas, because of the lack of contrast enhancement on MRI, and this was confirmed histologically in the majority of patients in each group (Table 1). However, 4 of 10 nTMS cases with early surgery and 1 of the 3 controls who had surgery were determined to already have grade III gliomas, despite the lack of contrast enhancement. The nTMS case who first had surgery at 11 months was also grade III by that time, despite an early biopsy reporting grade II. Three historical controls had no laboratory assessment at all within the first year (Table 1).
Motor outcomes
In each group, 10 of 11 patients had the same motor status one year after the initial presentation as they did initially. The remaining nTMS patient (#6) reported at one-year follow-up a slight loss of occupationally necessary fine motor skills (BMRC 5), and the remaining historical control (#10) had BMRC 4 due to surgery.
Other neurological outcomes
Only 1 of 6 nTMS patients with a seizure history had further seizures (the patient who had delayed surgery); whereas, 6 of 9 controls with a seizure history had further seizures (Table 2). The proportion of patients with other neurological deficits was initially similar in the two groups but had better outcomes in the nTMS group (Table 2). Surgery in the nTMS group led to no patient becoming worse (according to BMRC grading) and 1 of 4 patients with pre-operative deficits having an improvement. By contrast, 5 of the 8 historical controls without surgery had increased neurological deficits, while there was no change in the 3 controls with partial resections.
Postop refers to the first year from the initial visit. Y yes, dash none, FMS fine motor skills, BMRC British Medical Research Council scale for muscular strength, R right, L left
Oncological outcomes
The median (range) change of tumor volume from baseline to 1 year for the 11 nTMS cases was –83 % (−67 % to −100 %). By contrast, the median (range) change of tumor volume at 1 year for the 11 controls was +12 % (+40 % to −56 %). Despite the small sample size, this difference between the two groups (Fig. 3) was so substantial that it was also statistically highly significant (p < 0.001 by Mann Whitney rank sum). Additionally, one control had progressed from grade II to grade III, while none of the nTMS patients had progressed.
Discussion
Although low-grade gliomas are not “aggressive” (i.e., fast-growing and fast-spreading), they are “malignant”, because inevitably they will become anaplastic and kill the patient. In the meanwhile, their presence in the patient’s brain can cause epileptic seizures, deterioration of motor function, and other neurological deficits. For these reasons, it is now widely recognized that low-grade gliomas should be surgically resected [1, 11, 25, 31], especially since some non-enhancing gliomas may already be high-grade [8, 18, 21, 26, 29]. Yet research has documented that 50 % of neurosurgeons do not operate on non-enhancing gliomas in general [30], and the rate could be worse when the tumor occurs in the primary motor cortex where resection can cause permanent functional deficits [12]. Here we have shown that nTMS can help to adopt the state-of-the art treatment strategy, i.e., early and aggressive resection of LGGs guided by DES, by proving – preoperatively – which cortical areas are functionally essential.
Several main lessons can be drawn from this study. First, without preoperative nTMS mapping, surgeons in this series have been hesitant to resect a non-enhancing glioma suspected of invading the primary motor cortex. Consequently, the patient’s tumor will grow, and their neurological status will deteriorate. Second, nTMS allows to delineate motor functional from non-functional cortical tissue even within the area of altered FLAIR signal. Third, although the resection should still be guided by intraoperative DES [5, 6], preoperative nTMS functional mapping is what provides the necessary pre-operative guidance, reassurance, and planning [24], that enables neurosurgeons to make the decision to schedule a patient with a LGG within the primary motor cortex for surgery. In this series, the strategy was changed from watchful waiting to resection in two cases after the nTMS mapping results were added to the risk-benefit balancing.
DES was always available here and could have been used, but without preoperative nTMS, surgeons at our department often never even attempted surgery, and when they did, resection was never complete because they were reluctant to aggressively resect to the functional borders, even though the gold standard of DES was being performed by a highly experienced team [14–17, 22]. Fourth, surgical resection can lead to immediate improvements in the patient’s neurological health and usually succeeds at eliminating the entire solid tumor mass, which should improve their life-expectancy [11, 28, 31]. Any remaining risk of motor deficits or other complications from surgery are outweighed by the neurological and oncological risks of not resecting the glioma.
Our study is limited by its use of a retrospective comparison group from previous years. The outcome differences we observed could be due in part to other factors that changed over the years, especially the increased awareness of the need for prompt surgery. Nonetheless, the functional nTMS cartography with the exact delineation of resectable versus non-resectable cortical tissue, even within the area of altered FLAIR signal, had a significant effect on treatment decisions leading to earlier and more extensive resections. This confirms previous observations about the impact of nTMS on the surgical strategy [19, 24]. Further carefully designed clinical trials and multicenter patient registries are now warranted to double-verify how much of the treatment effect observed here is truly attributable to the preoperative use of nTMS.
References
Ashby LS (2010) Low-grade glioma: no longer to treat or not to treat, but why to treat. World Neurosurg 73:e5
Cairncross JG, Laperriere NJ (1989) Low-Grade Glioma: To Treat or Not to Treat? Arch Neurol 46:1238–1239
Cavaliere R, Lopers MBS, Schiff D (2005) Low-grade gliomas: an update on pathology and therapy. Lancet Neurol 4:760–770
Chang EF, Clark A, Smith JS, Polley M-Y, Chang SM, Barbaro NM, Parsa AT, McDermott MW, Berger MS (2011) Functional mapping–guided resection of low-grade gliomas in eloquent areas of the brain: improvement of long-term survival. J Neurosurg 114:566–573
Duffau H, Capelle L, Denvil D, Sichez N, Gatignol P, Taillandier L, Lopes M, Mitchell MC, Roche S, Muller JC, Bitar A, Sichez JP, van Effenterre R (2003) Usefulness of intraoperative electrical subcortical mapping during surgery for low-grade gliomas located within eloquent brain regions: functional results in a consecutive series of 103 patients. J Neurosurg 98:764–778
Duffau H (2006) New concepts in surgery of WHO grade II gliomas: functional brain mapping, connectionism and plasticity – a review. J Neurooncol 79:77–115
Duffau H (2012) The challenge to remove diffuse low-grade gliomas while preserving brain functions. Acta Neurochir 154:569–574
Ginsberg LE, Fuller GN, Hashmi M, Leeds NE, Schomer DF (1998) The Significance of Lack of MR Contrast Enhancement of Supratentorial Brain Tumors in Adults: Histopathological Evaluation of a Series. Surg Neurol 49:436–440
Grier JT, Batchelor T (2006) Low-Grade Gliomas In Adults. Oncologist 11:681–692
Jackson RJ, Fuller GN, Abi-Said D, Lang FF, Gokaslan ZL, Shi WM, Wildrick DM, Sawaya R (2001) Limitations of stereotactic biopsy in the initial management of gliomas. Neuro Oncol 3:193–200
Jakola AS, Myrmel KS, Kloster R, Torp SH, Lindal S, Unsgård G, Solheim O (2012) Comparison of a Strategy Favoring Early Surgical Resection vs a Strategy Favoring Watchful Waiting in Low-Grade Gliomas. JAMA 308:1881–1888
Jakola AS, Unsgård G, Myrmel KS, Kloster R, Torp SH, Lindal S, Solheim O (2012) Low grade gliomas in eloquent locations - implications for surgical strategy, survival and long term quality of life. PLoS One 7:e51450
Kleihues P, Louis DN, Scheithauer BW, Rorke LB, Reifenberger G, Burger PC, Cavenee WK (2002) The WHO Classification of Tumors of the Nervous System. J Neuropathol Exp Neurol 61:215–225
Kombos T, Suess O, Ciklatekerlio O, Brock M (2001) Monitoring of Intraoperative Motor Evoked Potentials to Increase the Safety of Surgery in and around the Motor Cortex. J Neurosurg 95:608–614
Kombos T, Kopetsch O, Suess O, Brock M (2003) Does Preoperative Paresis Influence Intraoperative Monitoring of the Motor Cortex? J Clin Neurophysiol 20:129–134
Kombos T, Picht T, Suess O (2008) Electrical Excitability of the Angular Gyrus. J Clin Neurophysiol 25:340–345
Kombos T, Picht T, Derdilopoulos A, Suess O (2009) Impact of Intraoperative Neurophysiological Monitoring on Surgery of High-Grade Gliomas. J Clin Neurophysiol 26:422–425
Kondziolka D, Lunsford LD, Martinez AJ (1993) Unreliability of contemporary neurodiagnostic imaging in evaluating suspected adult supratentorial (low-grade) astrocytoma. J Neurosurg 79:533–536
Krieg SM, Shiban E, Buchmann N, Gempt J, Foerschler A, Meyer B, Ringel F (2012) Utility of presurgical navigated transcranial magnetic brain stimulation for the resection of tumors in eloquent motor areas. J Neurosurg 116:994–1001
Kros JM (2011) Grading of Gliomas: The Road from Eminence to Evidence. J Neuropathol Exp Neurol 70:101–109
Lang FF, Gilbert MR (2006) Diffusely Infiltrative Low-Grade Gliomas in Adults. J Clin Oncol 24:1236–1245
Picht T, Wachter D, Mularski S, Kuehn B, Brock M, Kombos T, Suess O (2008) Functional Magnetic Resonance Imaging and Cortical Mapping in Motor Cortex Tumor Surgery: Complementary Methods. Zentralbl Neurochir 69:1–6
Picht P, Schmidt S, Brandt S, Frey D, Hannula H, Neuvonen T, Karhu J, Vajkoczy P, Suess O (2011) Preoperative Functional Mapping for Rolandic Brain Tumor Surgery: Comparison of Navigated Transcranial Magnetic Stimulation to Direct Cortical Stimulation. Neurosurgery 69:581–589
Picht T, Schulz J, Hanna M, Schmidt S, Suess O, Vajkoczy P (2012) Assessment of the Influence of Navigated Transcranial Magnetic Stimulation on Surgical Planning for Tumors in or Near the Motor Cortex. Neurosurgery 70:1248–1257
Piepmeier JM (2009) Current concepts in the evaluation and management of WHO grade II gliomas. J Neurooncol 92:253–259
Pouratian N, Asthagiri A, Jagannathan J, Shaffey ME, Schiff D (2007) Surgery Insight: the role of surgery in the management of low-grade gliomas. Nat Clin Pract Neurol 3:628–639
Recht LD, Lew R, Smith TW (1992) Suspected Low-grade Glioma: Is Deferring Treatment Safe? Ann Neurol 31:431–436
Sanai N, Berger MS (2008) Glioma Extent of Resection and Its Impact on Patient Outcome. Neurosurgery 62:753–762
Scott JN, Brasher PMA, Sevick RJ, Rewcastle NB, Forsyth PA (2002) How often are nonenhancing supratentorial gliomas malignant? A population study. Neurology 59:947–949
Seiz M, Freyschlag CF, Schenkel S, Weiss C, Thomé C, Schmieder K, Stummer W, Tuettenberg J (2011) Management of Patients With Low-Grade Gliomas – A Survey Among German Neurosurgical Departments. Cent Eur Neurosurg 72:186–191
Soffietti R, Baumert BG, Bello L, von Deimling A, Duffau H, Frénay M, Grisold W, Grant R, Graus F, Hoang-Xuan K, Klein M, Melin B, Rees J, Siegal T, Smits A, Stupp R, Wick W (2010) Guidelines on management of low-grade gliomas: report of an EFNS-EANO Task Force. Eur J Neurol 17:1124–1133
Suess O, Suess S, Brock M, Kombos T (2006) Intraoperative Electrocortical Stimulation of Brodman Area 4: A 10-Year Analysis of 255 Cases. Head Face Med 2:20
Warnke PC (2010) A 31-Year-Old Woman With a Transformed Low-grade Glioma. JAMA 303:967–976
Whittle IR (2004) The Dilemma of Low Grade Glioma. J Neurol Neurosurg Psychiatry 75(Suppl II):ii31–ii36
Acknowledgments
We would like to thank Michael Hanna, PhD, (Mercury Medical Research & Writing) for providing publication consulting, statistical analysis, and medical writing services. We would also like to thank Adela Castelló, MSc, DPH, (Mercury Medical Research & Writing) for some recommendations on statistics and publishing.
Conflicts of interest
The research reported in this article was supported in part by a grant from the Berlin Cancer Society. Dr. Picht has served as a speaker for Nexstim OY, the manufacturer of the device used in this study. Dr Picht and Dr. Schulz had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
Picht et al. present a study were they assessed the usefulness of preoperative navigated transcranial magnetic stimulation for the surgery of presumed low grade astrocytomas. This topic is interesting as nTMS is a relatively new tool, is not part of the standard armamentarium of most neurosurgical centers and therefore its use is limited and its usefulness still uncertain.
The goal of the study is to define the impact of this technology on the surgical results and on the overall clinical course of patients with presumed WHOII gliomas in or invading the motor cortex. The authors describe their experience with 11 patients and show convincingly good results in terms of extent of resection and low postoperative deficits. Every neurosurgeon knows from experience that lesions in the primary motor cortex are challenging and prone to postoperative motor deficits. Therefore we can appreciate the good results shown by the authors and reasonably deduct that nTMS probably provided an advantage, be it solely the confidence to offer surgery and attempt an extensive resection.
The comparison with an historical group can be useful for the assessment of new technology that is not easily subjected to randomized control trials and the number of patients is low. However, this requires homogenous groups which is not the case here and this is the main flaw of this study. Scientifically, the historical groups are not comparable. The study assesses what the availability of TMS changes in the overall management of these patients. However, this study does not compare surgery with or without TMS nor does it provide comparative data on observation vs Surgery (with or without TMS). Therefore any conclusion about the safety of surgery or the specific contribution of TMS to surgical outcomes cannot be drawn. Furthermore heterogeneity between groups is introduced by the fact that there were grade II and III tumors in both groups but some historical cases do not have a definitive diagnosis.
After all it could well be that surgery with DES alone is equal to nTMS and that the number of patients undergoing surgery in the nTMS group are the result of a change in the surgical philosophy for these lesions overtime.
Ivan Radovanovic
Toronto, Canada
Thomas Picht and Juliane Schulz contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Picht, T., Schulz, J. & Vajkoczy, P. The preoperative use of navigated transcranial magnetic stimulation facilitates early resection of suspected low-grade gliomas in the motor cortex. Acta Neurochir 155, 1813–1821 (2013). https://doi.org/10.1007/s00701-013-1839-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-013-1839-1