Abstract
Objective
Cryptococcal infection in CNS is frequently seen in HIV patients and those with other immunosuppressed conditions. However, cryptococcal granuloma in CNS in immunocompetent patient is rare. We present one new case of cryptococcoma and review literature to illustrate diagnosis and treatment of these lesions.
Methods
We conducted literature search in Pubmed search engine of the National Center for Biotechnology Information.
Results
Seventeen cases of CNS cryptoccoma in immunocompetent patients, including ours, have been reported to date. Of them, two patients had lesions inside spinal cord, and C. neoformans var. gattii was identified in three cases. All patients were symptomatic with normal immunocompetency although two patients had type 2 diabetes mellitus and one had torsades de pointes. Eight patients received surgical treatment and all were given antifungal agents except one suspected of teniasis.
Conclusions
With literature reports and our experiences, we suggest that ring shaped enhancement of mass lesion with or without cystic changes in MR scan may indicate cryptococcoma, but definitive diagnosis relies on pathology study of lesion specimen. Open surgery and anti-fungal therapy should be scheduled, and outcome of cryptococcoma is largely determined by its locations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Central nervous system cryptococcal infection frequently occurs in patients with HIV infection and other immunocompromised situations. It is caused by C. neoformans, encapsulated yeast that is ubiquitous and transmitted primarily through airway inhalation. Typical meningeal infection in these patients has been well documented in the literature [16, 33, 34]. However, cryptoccocal infection in immunocompetent patients is rare, and CNS cryptococcomas in such patients has seldom been reported. We present a new case of intracranial cryptococcoma in a 49-year-old woman with normal immunity and review published articles that report similar cases [1, 3, 6–8, 11, 13–15, 18, 21, 22, 24, 26, 28, 36] to elucidate the clinical features, diagnosis as well as treatment of CNS cryptococcoma in immunocompetent patients.
Literature review
We conducted a literature search in the PubMed search engine of the National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov/). Our search was based on the following terms: “cryptococcoma,” “cryptococcal abscess,” “cryptococcal cyst,” “cryptococcal granuloma” and “cryptococcal infection,” all confined by “central nervous system.” Target reports had to be in English, and the authors had to state clearly that the patients were not immunosuppressed. In addition, reports from before 1985 were excluded because it was not until the First International AIDS Conference in 1985 in Atlanta that AIDS aroused worldwide attention. Also, cases in reference articles of retrieved reports were included if they complied with the above requirements. In total, 17 cases including ours were selected for the literature analysis.
Analysis
A summary of analyzed articles is shown in Table 1. Our results showed a slightly higher prevalence of the disease in men than in women (10 men versus 7 women), with age ranging from 11 to 72 years (average 43.8 years). Cryptococcoma occured in the spinal cord in two patients, while other patients harbored intracranial lesions. Six patients (35.3%) had more than one brain lesion.
The most common symptoms were headaches or vomiting in 11 cases (64.7%) [movement disorders in 7 cases (41.2%), consciousness or mental changes in 7 cases (41.2%) and cranial nerve dysfunction in 5 cases (29.4%)]. Seizures were present in two cases (11.8%), including one with the sole presentation of epilepsy (5.9%). Only one patient had fever. One patient presented with sellar region symptoms of polyuria and decreased libido. Two patients had type 2 diabetes mellitus with one complicated by Parkinson's disease. Cryptococcoma was found in one patient treated for hypertension and torsades de pointes. There was also one woman who was pregnant in our series.
Cryptococcoma appeared in the cerebrum in ten cases (58.8%), including four cases of basal ganglia lesions (23.5%), followed by brainstem involvement in four (23.5%) and the cerebellum in five cases (29.4%). Two patients harbored ventricular foci, and another two had a spinal cord mass at the T12 level. Only one patient had granuloma in the pituitary gland.
Magnetic resonance imaging was applied to 12 patients in our cohort; the remaining patients underwent computed tomography only. Major MR features included hypointensity in T1 and hyperintensity in T2 sequences with peripheral edema and ring-shaped enhancement after gadolinium injection (66.7%, 8 in 12 cases). Cystic appearances were prevalent (65.0%, 11 of 17 patients). Calcification was found in one patient. In addition to regular magnetic resonance sequences, diffusion weighted images (DWI) and apparent diffusion coefficient maps (ADC) showed different patterns in various patients, and the fluid attenuated inversion recovery (FLAIR) technique used in four patients was hypertense in two and hypotense in the other two patients. Magnetic resonance spectroscopy (MRS) was applied in just one case and was void of choline signal.
Nine patients (52.9%) were misdiagnosed as having tumors, vascular lesions, tuberculoma or teniasis, and there were no data concerning the initial diagnosis in the other eight cases. Open surgery was conducted in eight cases, and six patients received stereotactic or needle biopsy (including one for both biopsy and craniotomy), with the diagnosis of one case confirmed in autopsy. The remaining three cases were diagnosed by India ink stain or not specified. Microbiology study identified C. neoformans var. gattii (CNVG) in three patients’ specimens. Antifungal treatment was enforced in all cases except the one with autopsy, and multiple antifungal agents were administered in eight cases. A common combination of agents included amphotericin B (AMB) plus flucytosine (5FC) plus fluconazole (FLC, four of eight cases) and AMB plus either 5FC or FLC (two of four cases, respectively). AMB alone was given to four patients and FLC to just one patient. The remaining four patients received unspecified antifungal (three of four cases) or dexmethasone therapy (the one with autopsy). The duration of antifungal treatment is not explicitly available based on case descriptions in most cases, and the exact criteria for stopping drugs remain a mystery.
Follow-up ranges from 2 days to 2 years, averaging 5.6 months for the 15 patients with follow-up information. Five patients died, and 11 patients enjoyed improved or baseline conditions with unknown outcome in one patient.
Illustrative case
A 49-year-old previously healthy female complained of ongoing headache, dizziness and vomiting for 1 month. No close contact with pigeons or other avian species was present. However, she kept a dog as a pet in her home and often had close contact. Upon physical examination, she was alert with normal temperature, but left congruous homonymous hemianopia was observed. She had no difficulty with motility of her extremities, and pathological signs or meningeal irritation was negative.
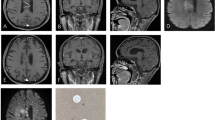
Laboratory test results were notable only for a slightly increased red blood cell count of 6.04 × 1,012/l. Preoperative MRI (see Fig. 1) with gadolinium enhancement showed a large, lobulated 5 × 4 × 4.5-cm parasinoidal, right occipital mass in the T1-weighted scan, with multiple hypointense centers among ring-shaped enhancement surrounded by perifocal white matter edema. The right lateral ventricle was almost occluded and the midline shifted. Meningeal enhancement was not remarkable. Based on her age, clinical presentation, past medical history as well as radiological findings, a preoperative diagnosis of high grade glioma was highly suspected.
The patient underwent a total resection of the mass in the operating room via right occipital craniotomy. Pathological study (see Fig. 2) of the specimen identified numerous Cryptococcus neoforman organisms and granuloma, which was confirmed by Gomori methenamine silver stain and mucicarmine stain. No malignant cells were observed. Upon the diagnosis of cryptococcoma, complete blood cell count and HIV assay were repeated. The percentage of CD3- and CD8-positive T cells decreased a little: 62.10% for CD3 T cells (normal range 66.9%–88.1%) and 17.7% for CD8 T cells (normal range 20.4%–30.7%). Either the total number or percentage of blood lymphocytes was within normal range. Also, she was HIV negative both in the preoperative screening test and the repeated test afterwards. Postoperative India ink stain of the CSF specimen revealed no sign of C.neoformans.
Head MRI of the patient. A Preoperative T1-weighted scan shows a right occipital mass with hypointense centers. Note the midline shift and occlusion of the lateral ventricles. B Preoperative T2-weighted scan reveals brain edema surrounding the mass and infiltration of CSF in the left occipital lobe. C to E Preoperative gadolinium-enhanced MR scan shows a large, lobulated 5 × 4 × 4.5-cm parasinoidal, right occipital mass in T1-weighted scan, with multiple hypointense centers among ring-shaped enhancement. F One month after surgery, gadolinium-enhanced MR reveals complete resection of the mass and restoration of intracranial structures
Pathology of the specimen. A Numerous clear gelatinous capsules surrounding a pale blue nucleus are characteristic of C. neoformans in hematoxylin-eosin (HE) stain. B Granuloma is observed as granulocytes aggregate around C.neoformans organisms in HE stain. Note fibril hypertrophy in the foci. C Gomori methenamine silver stain shows budding yeasts with narrow-based buds and chains of organisms consistent with Cryptococcus neoformans. D Mucicarmine stain reveals purple organisms with poorly stained center and numerous budding yeasts. (Magnification × 100)
Intravenous AMB therapy was started at 1 mg per day for the first day, then dosage of the agent was elevated by a scale of 2-5 mg per day to maintainance dosage of 25 mg per day for about 20 days. Toxicity of AMB was monitored on a weekly basis. The patient tolerated the whole process well. Headache, dizziness and nausea or vomiting disappeared after the operation. Postoperative CT scan confirmed stable intracranial conditions and restoration of midline and ventricular systems. One month after surgery (also approximately after antifungal therapy), the follow-up MR scan (see Fig. 1F) showed total resection of the original mass and significantly reduced surrounding edema, and her symptom of hemianopia resolved.
Discussion
Intracranial cryptococcal infection is a rare situation in otherwise immunocompetent patients. The incidence of intracranial cryptococcoma that is characterized by accumulation of Cryptococcus and inflammatory cells is even smaller. In a report of 40 cases of intracranial fungal granuloma, only 3 were positive for cryptococcosis [5].
The infection is generally believed to be caused by C. neoformans var. neoformans (CNVN). However, three cases of intracranial cryptococcoma have been reported to be caused by CNVG [3, 18, 24]. Although its source has yet to be identified, an association of CNVG and eucalyptus trees has been demonstrated, and CNVG is a threat to human beings as well as dogs and cats [12]. It is reported that patients infected with CNVG are more likely to develop a CNS mass [2, 20, 30]. This can be explained by the fact that metabolites released by CNVG can inhibit the migration and function of leukocytes and promote survival as well as localized replication of the pathogen, facilitating formation of cryptococcoma [4, 35]. In this cohort, only three cases were identified for CNVG infection, and the true prevalence of CNVG may be distorted because none the other cases were genotyped for a variant of C. neoformans. However, identification of the C. neoformans subtype might be necessary because CNVG is more resistant to treatment and can cause more long-term sequelae [20].
Major symptoms and signs can be attributed to intracranial hypertension and compression of adjacent structures, and blurred by the non-specific location of the lesion and absence of an immunosuppressed history, the diagnosis of CNS cryptococcoma can be quite challenging to neurosurgeons. In this article, the minimum misdiagnosis rate was 52.9%, rendering tumor a priority for the differential dignosis. By summarizing magnetic resonance features of cryptococcoma in the patients, we found that hypotensity in T1 and hypertensity in T2 sequences with peripheral edema and ring-shaped enhancement after gadolinium presented in over 66% of cases with MR scans. Although this feature may be insufficient to distinguish cryptococcoma from neoplasms, the ring-shaped enhancement in a mass lesion, especially when with cystic changes (see Fig. 1), may at least suggest the possibility of cryptococcoma. Unfortunately, more advanced techniques, including DWI, ADC, FLAIR and MRS, showed no explicit features for the differential diagnosis of cryptococcoma [6, 8, 13, 14, 28]. Proton MRS and a combination of 201Tl SPECT and 99mTcO4-SPECT have revealed encouraging results for the differential diagnosis between neoplasmic and tumor-like lesions [9, 32], but cryptococcoma cannot be distinguished from other tumor-like lesions.
Definitive diagnosis for intracranial cryptococcosis relies on culture and India ink stain. Culture of the organism remains the gold standard for its diagnosis, with a reported positive rate of almost 100% [10]. However, Regina et al. suggest the polymerase chain reaction has a higher sensitivity of 92.9% compared to 76.8% for culture [25]. The positive rate of the India ink test ranges from 5.2% [23] to 91% [29]. Various factors, including the number of CFU/ml of CSF, different forms of C. neoformans, medium composition, temperature, prior use of anti-fungal agents and pH value may be responsible for the detection rate of C. neoformans [25]. In immunocompetent patients with intracranial cryptococcoma, however, the positive rate of the India ink test has yet to be elucidated and is possibly lower than that of ordinary forms of cryptococcosis, e.g., cryptococcal meningitis, etc. Several reasons account for this: (1) the incidence of cryptococcoma is relatively much lower; (2) yeasts are often confined to localized lesions within the parenchyma, diminishing the number of CFU/ml in CSF [31]; (3) incorrect preoperative diagnosis eliminates the need for a lumber puncture and examination of the CSF (like in the present case), and then postoperative pathology further diminishes the significance of the CSF test after a conclusive diagnosis is made. Only two cases in this review were identified with India ink stain, and over 82% of patients were diagnosed with pathological study (14 in 17 cases); thus, pathology is the major method for diagnosing cryptocococma. The negative finding of C. neoformans in CSF in the present case further raises doubt about the usefulness of India ink stain to monitor the effect of antifungal therapy. Also, it seems that a decline of CSF Cryptococcus antigen is correlated to fungal clearance, but may not be used as an index of cure [17, 19].
CNS cryptococcoma should be treated with antifungal agents and surgery when necessary and possible. In all eight patients undergoing open surgery, locations in the peripheral lobes allowed relatively easy access for total resection by open surgery. For other patients, the most frequent locations would be either the basal ganglia or brainstem zone, which is much more difficult for surgical excision, not to mention related postoperative complications. As a matter of fact, considering the compressive effect and high misdiagnosis rate, the indication for surgical treatment of cryptococcoma is similar to that of CNS tumors with the exception of tumor in the basal ganglia or brainstem area. Neoplasms in these places should be treated surgically because other treatment modalities such as chemotherapy are not effective on their own, and surgery brings more benefit than harm to patients with lesions in these sites. However, for infectious masses like cryptococcoma, the benefit of open surgery is dwarfed by the relative efficacy of antifungal agents. Neurosurgeons weigh the risks and benefits of surgery to determine the most appropriate plans for each patient. Nonetheless, cryptococcoma with a relatively easy surgical route should be totally resected to reduce the pathogen load of patients. If it is not safe to conduct a surgical removal for patients with deeply seated lesions, stereotactic biopsy may help. In the 17 cases, 9 patients harbored foci in the basal ganglia, brainstem or ventricles [3, 11, 13, 14, 18, 22, 26, 28, 36]; of them, 4 patients received stereotactic biopsy [3, 13, 18, 36]. In addition, another two patients without deep lesions also underwent the procedure [21, 24]. Of the six patients with biopsy, three died after intensive treatment [13, 18, 24]. Although the death rate for patients with versus those without deep lesions in the biopsy group is 50% to 50%, we believe that the diagnosis can be confirmed once a proper specimen is obtained through stereotactic biopsy and hence may help physicians prescribe proper medications. However, the decompressive effect of biopsy is unknown and probably is less than that of a craniotomy surgery, because the latter is straightforward and helpful for total removal with gross pathologic observations.
Anti-fungal drugs should be administered once diagnosis is confirmed. Although current guidelines advocate combination therapy of AMB and 5FC [27], treatment for cryptococcoma with or without surgery varies. Popular prescriptions in this review include triple or mono anti-mycotic agents, and still some of the patients received therapy based on Saag’s recommendation. Most cases do not present clear plans for the duration of anti-fungal treatment and criteria to stop it, although two of the cases did mention cryptococcal antigen titers and their relation to the administration of anti-mycotic agents [3, 28]. Based on the extremely limited data, we could not make a certain recommendation for the administration of anti-mycotic agents. It is probably workable to adopt remedies based on various experiences at each institution. However, with desired outcomes in nearly 65% of all cases (11 in 17 patients), it is difficult to compare and select the best antifungal therapy for all. More cases, if not random controlled trials, with definite follow-up and outcome description are needed to form better anti-mycotic recommendations for cryptococcoma.
It is intriguing to see that all five patients who died were treated non-surgically, and reasons for death were respiratory arrest, septic shock and cryptococcal pneumonia. Of 11 cases with favorable outcome, lesions generally were located in peripheral brain lobes, but 4 of the 5 patients who died had lesions in the brainstem or basal ganglia. Hence, the location of the lesion may be one factor that determines the outcome of cryptococcoma in immunocompetent patients.
Conclusions
CNS cryptococcoma is rare in immunointact individuals. It mimics symptoms and signs of CNS tumors and can be difficult to diagnose preoperatively. Ring-shaped enhancement of mass lesions with or without cystic changes in the MR scan may indicate cryptococcoma, but definitive diagnosis relies on pathological study of the lesion specimen. Open surgery and anti-fungal therapy should be scheduled, and the outcome of cryptococcoma is largely determined by its location.
Abbreviations
- ADC:
-
Apparent diffusion coefficient map
- AIDS:
-
Acquired immune deficiency syndrome
- AMB:
-
Amphotericin B
- CFU:
-
Colony formation unit
- CNS:
-
Central nervous system
- CNVN:
-
C. neoformans var. neoformans
- CNVG:
-
C. neoformans var. gattii
- CSF:
-
Cerebral spinal fluid
- CT:
-
Computed tomography
- DWI:
-
Diffusion weighted image
- FLAIR:
-
Fluid attenuated inversion recovery
- FLC:
-
Fluconazole
- MRI:
-
Magnetic resonance imaging
- MRS:
-
Magnetic resonance spectroscopy
- SPECT:
-
Single photon emission computed tomography
- HIV:
-
Human immunodeficiency virus
- 5FC:
-
Flucytosine
References
Caldemeyer KS, Mathews VP, Edwards-Brown MK, Smith RR (1997) Central nervous system cryptococcosis: parenchymal calcification and large gelatinous pseudocysts. AJNR Am J Neuroradiol 18:107–109
Chen S, Sorrell T, Nimmo G, Speed B, Currie B, Ellis D, Marriott D, Pfeiffer T, Parr D, Byth K (2000) Epidemiology and host- and variety-dependent characteristics of infection due to Cryptococcus neoformans in Australia and New Zealand. Australasian Cryptococcal study group. Clin Infect Dis 31:499–508. doi:10.1086/313992
Colom MF, Frasés S, Ferrer C, Jover A, Andreu M, Reus S, Sánchez M, Torres-Rodríguez JM (2005) First case of human cryptococcosis due to Cryptococcus neoformans var. gattii in Spain. J Clin Microbiol 43:3548–3550. doi:10.1128/JCM.43.7.3548-3550.2005
Dong ZM, Murphy JW (1995) Effects of the two varieties of Cryptococcus neoformans cells and culture filtrate antigens on neutrophil locomotion. Infect Immun 63:2632–2644
Dubey A, Patwardhan RV, Sampth S, Santosh V, Kolluri S, Nanda A (2005) Intracranial fungal granuloma: analysis of 40 patients and review of the literature. Surg Neurol 63:254–260. doi:10.1016/j.surneu.2004.04.020
Gologorsky Y, DeLaMora P, Souweidane MM, Greenfield JP (2007) Cerebellar cryptococcoma in an immunocompetent child. Case report. J Neurosurg 107:314–317. doi:10.3171/ped.2007.107.4.314
Gültaşli NZ, Ercan K, Orhun S, Albayrak S (2007) MRI findings of intramedullary spinal cryptococcoma. Diagn Interv Radiol 13:64–67
Ho TL, Lee HJ, Lee KW, Chen WL (2005) Diffusion-weighted and conventional magnetic resonance imaging in cerebral cryptococcoma. Acta Radiol 46:411–414. doi:10.1080/02841850510021201
Hourani R, Horska A, Albayram S, Brant LJ, Melhem E, Cohen KJ, Burger PC, Weingart JD, Carson B, Wharam MD, Barker PD (2006) Proton magnetic resonance spectroscopic imaging to differentiate between nonneoplastic lesions and brain tumours in children. J Magn Reson Imaging 23:99–107. doi:10.1002/jmri.20480
Imwidthaya P, Poungvarin N (2000) Cryptococcosis in AIDS. Postgrad Med J 76:85
Kesler R, Maertens P (1999) Pontine cryptococcoma in a nonimmunocompromised individual: MRI characteristics. J Neuroimaging 9:118–121
Kidd SE, Hagen F, Tscharke RL, Huynh M, Bartlett KH, Fyfe M, Macdougall L, Boekhout T, Kwon-Chung KJ, Meyer W (2004) A rare genotype of Cryptococcus gattii caused the cryptococcosis outbreak on Vancouver Island (British Columbia, Canada). Proc Natl Acad Sci USA 101:17258–17263. doi:10.1073/pnas.0402981101
Kocaeli H, Hakyemez B, Bekar A, Yilmazlar S, Abas F, Yilmaz E, Korfali E (2008) Unusual complications and presentations of intracranial abscess: experience of a single institution. Surg Neurol 69:383–391. doi:10.1016/j.surneu.2007.03.011
Krishnan AV, Corbett A (2004) Intracranial and dermatological cryptococcal infection in an immunocompetent man. J Clin Neurosci 11:765–767. doi:10.1016/j.jocn.2003.10.021
Lai PH, Wang JS, Chen WL, Pan HB, Yang CF (2001) Intramedullary spinal cryptococcoma: a case report. J Formos Med Assoc 100:776–778
Leal AL, Faganello J, Fuentefria AM, Boldo JT, Bassanesi MC, Vainstein MH (2008) Epidemiological profile of cryptococcal meningitis patients in Rio Grande do Sul, Brazil. Mycopathologia 166:71–75. doi:10.1007/s11046-008-9123-2
Lu H, Zhou Y, Yin Y, Pan X, Weng X (2005) Cryptococcal antigen test revisited: Significance for cryptococcal meningitis therapy monitoring in a tertiary Chinese hospital. J Clin Microbiol 43:2989–2990. doi:10.1128/JCM.43.6.2989-2990.2005
McMahon JH, Grayson ML (2008) Torsades de pointes in a patient receiving fluconazole for cerebral cryptococcosis. Am J Health Syst Pharm 65:619–623. doi:10.2146/ajhp070203
Milefchik EN, Leal ME, Larsen RA (1994) Correlation of CSF cryptococcal antigen titer with quantitative cultures. Int Conf AIDS 10:146
Mitchell DH, Sorrell TC, Allworth AM, Heath CH, McGregor AR, Papanaoum K, Richards MJ, Gottlieb T (1995) Cryptococcal disease of the CNS in immunocompetent hosts: influence of cryptococcal variety on clinical manifestations and outcome. Clin Infect Dis 20:611–616
Nadkarni TD, Menon RK, Desai KI, Goel A (2005) A solitary cryptococcal granuloma in an immunocompetent host. Neurol India 53:365–367
Nucci A, Maciel Júnior JA, Queiroz Lde S, Montenegro MA, De Carvalho RB (1999) Pseudocystic form of neurocryptococcosis in pregnancy. Case report. Arq Neuropsiquiatr 57:678–682. doi:10.1590/S0004-282X1999000400023
Odhiambo FA, Murage EM, Ngare W, Ndinya-Achola JO (1997) Detection rate of Cryptococcus neoformans in cerebrospinal fluid specimens at Kenyatta National Hospital, Nairobi. East Afr Med J 74:576–578
Oliveira Fde M, Severo CB, Guazzelli LS, Severo LC (2007) Cryptococcus gattii fungemia: report of a case with lung and brain lesions mimicking radiological features of malignancy. Rev Inst Med Trop Sao Paulo 49:263–265
Paschoal RC, Hirata MH, Hirata RC, Melhem Mde S, Dias AL, Paula CR (2004) Neurocryptococcosis: diagnosis by PCR method. Rev Inst Med trop S. Paulo 46:203–207
Popovich MJ, Arthur RH, Helmer E (1990) CT of intracranial cryptococcosis. AJR Am J Roentgenol 154:603–618
Saag MS, Graybill RJ, Larsen RA, Pappas PG, Perfect JR, Powderly WG, Sobel JD, Dismukes WE (2000) Practice guidelines for the management of cryptococcal disease. Infectious Diseases Society of America. Clin Infect Dis 30:710–718. doi:10.1086/313757
Saigal G, Post MJ, Lolayekar S, Murtaza A (2005) Unusual presentation of central nervous system cryptococcal infection in an immunocompetent patient. AJNR Am J Neuroradiol 26:2522–2526
Sato Y, Osabe S, Kuno H, Kaji M, Oizumi K (1999) Rapid diagnosis of cryptococcal meningitis by microscopic examination of centrifuged cerebrospinal fluid sediment. J Neurol Sci 164:72–75. doi:10.1016/S0022-510X(99)00047-7
Speed B, Dunt D (1995) Clinical and host differences between infections with the two varieties of Cryptococcus neoformans. Clin Infect Dis 21:28–34
Sunhee CL, Dennis WD, Arturo C (1996) Pathology of cryptococcal meningoencephalitis: Analysis of 27 patients with pathogenetic implications. Hum Pathol 27:839–847. doi:10.1016/S0046-8177(96)90459-1
Terada H, Kamata N (2003) Contribution of the combination of 201Tl SPECT and 99mTcO 4- SPECT to the differential diagnosis of brain tumours and tumour-like lesions: A preliminary report. J Neuroradiol 20:91–94
Thakur R, Sarma S, Kushwaha S (2008) Prevalence of HIV-associated cryptococcal meningitis and utility of microbiological determinants for its diagnosis in a tertiary care center. Indian J Pathol Microbiol 51:212–214. doi:10.4103/0377-4929.41689
Wadhwa A, Kaur R, Bhalla P (2008) Profile of central nervous system disease in HIV/AIDS patients with special reference to cryptococcal infections. Neurologist 14:247–251. doi:10.1097/NRL.0b013e3181678a7a
Wright L, Bubb W, Davidson J, Santangelo R, Krockenberger M, Himmelreich U, Sorrell T (2002) Metabolites released by Cryptococcus neoformans var. neoformans and var. gattii differentially affect human neutrophil function. Microbes Infect 4:1427–1438. doi:10.1016/S1286-4579(02)00024-2
Yu YQ, Jiang XX, Gao YJ (1995) MRI of a pituitary cryptococcoma simulating an adenoma. Neuroradiology 37:449–450. doi:10.1007/BF00600089
Acknowledgements
We thank Dr. Ke Yang in the Department of Pathology for help during collection of pathological data and figures.
Conflict of interest
This report is original and has no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Q., You, C., Liu, Q. et al. Central nervous system cryptococcoma in immunocompetent patients: a short review illustrated by a new case. Acta Neurochir 152, 129–136 (2010). https://doi.org/10.1007/s00701-009-0311-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-009-0311-8