Abstract
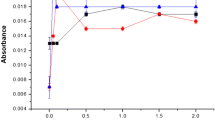
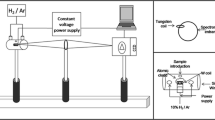
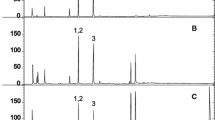
A method for direct analysis of tea and coffee samples by using electrothermal atomic absorption spectrometry is described. Coffee and tea from different sources were analyzed without digestion step. For slurry analyses the samples were ground, sieved at 105 μm and then suspended in 0.2% v/v HNO3 and 10% v/v Triton X-100 medium. For liquid phase aluminium determination the samples were prepared in the same way and only the liquid is introduced directly into the graphite furnace. Calibration was performed by aqueous standards for both cases and the determinations were carried out in the linear range between 50 and 250 μg L−1. The characteristic mass of aluminium and the detection limit were 45 pg and 2 μg L−1, respectively. Using a typical 0.1% m/v coffee slurry sample, the relative standard deviation of measurements (n=15) for repeatability was about 8.2%.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received December 27, 1998. Revision March 18, 1999.
Rights and permissions
About this article
Cite this article
Magalhães, C., Lima, É., Krug, F. et al. Direct Analysis of Coffee and Tea for Aluminium Determinationby Electrothermal Atomic Absorption Spectrometry. Mikrochim Acta 132, 95–100 (1999). https://doi.org/10.1007/s006040050048
Issue Date:
DOI: https://doi.org/10.1007/s006040050048