Abstract
This review (with 142 references) summarize the state of the art in molecularly imprinting technology as applied to the surface of carbon nanotubes (CNTs) which result in so-called CNTs@MIPs. These nanomaterials offer a remedy to the flaws of traditional MIPs, such as poor site accessibility for templates, slow mass transfer and template leakage. They also are flexible in that different materials can be integrated with CNTs. Given the advantages of using CNT@MIPs, this technology has experienced rapid expansion, not the least because CNT@MIPs can be produced at low cost and by a variety of synthetic approaches. We summarize methods of, and recent advances in the synthesis of CNT@MIPs, and then highlight some representative applications. We also comment on their potential future developments and research directions.

ᅟ
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The omnipresence of molecular recognition process indicates its essential role in nature, this exquisite process is exploited in enzymatic catalysis, antibody-antigen recognition, cell communication and some other biological processes [1]. Via biomolecular recognition process, numerous purposes have been achieved such as immunoassay and enzyme catalysis. But the usage has been greatly circumscribed due to the biomolecule’s restricted availability, intolerance towards rigid chemical conditions and inferior performance compared to intracellular functioning. To circumvent the limitations above, much research has been carried out to obtain equivalents to biological recognition system.
Mimicking the mechanism of biomolecular recognition, but with high stability, ease of preparation and low cost, molecularly imprinted polymers (MIPs) have been demonstrated itself as the most promising synthetic materials bearing selective molecular recognition sites [2]. In the 1970’s, molecular imprinting technology (MIT) was first introduced by Wulff et al. using covalent approach [3]. This technique has been advanced and soon developed [4] since the emerging of non-covalent approach which is marked by the synthesis of an acrylic-based imprinted polymer by Mosbach in 1981 [5].
According to Nicholls [6], “molecular imprinting is a technique which involves the formation of binding sites in a synthetic polymer matrix that are of complementary functional and structural character to its ‘substrate’ molecule”. The monomers were first compounded with the analytes (or templates) and then polymerized. There are cavities created in the polymers, which are sterically and chemically complementary to the templates. Upon removal of the templates, the specific binding cavities are able to discriminate the templates from their analogs.
According to the mechanism of interaction between the monomers and templates, the molecular imprinting methods can be divided in to four categories: pre-organized approach (covalent bonding), self-assembly approach (non-covalent interactions), hybrid molecularly imprinting method and metal chelating method [7]. Attributed to the specific bonding, covalent imprinting polymers show high selectivity and homogeneous binding sites distribution. Nevertheless, they were confined to a narrow range of use because of the restrictive choices of templates [8]. On the other hand, profited from less restricted binding sites, MIPs resulting from self-assembly approach have been frequently developed and could be applied for a wider scope [4]. Some comprehensive reviews collecting and comparing different synthesizing methods and approaches of MIPs are recommended for reading [8–10].
Although conventionally prepared bulk MIPs exhibit high selectivity, some inherent defects are suffered from coarse post-treatment (as grounding and sieving of MIP [11]). These post-treatments reduce the polymer yield and produce irregular polymer fragments which narrow the usage in chromatography or solid-phase extraction. As another main congenital imperfection of bulk MIPs, limited mass transfer efficiency become the main issue in the macromolecule imprinting or biosensor field [12]. Different polymerization methodologies are shown in Fig. 1.
Polymerization methodologies. M Monomers; I Initiator; D Dispersing phase; S Surfactant, IN Insoluble nanoseeds [10]
To solve the above-mentioned problems, surface imprinting technology was developed. Because the imprinting sites are located on or near the surface of the substrates, polymers synthesized by surface imprinting method show high mass transfer efficiency, high sensitivity and short response time. Furthermore, new properties (e.g. electrical, magnetic and optical [13, 14]) could be incorporated into the materials by changing or modifying the solid supports. In recent years, many new solid supports have been studied, such as monodispersed polystyrene [15], chitosan [16–19], silica beads [20–29], Fe3O4 nanoparticles [30], graphene [31–34], halloysite nanotubes [35–37] and quantum dots [38–45], some of which suffer disadvantages like lacking in mechanical strength and small specific surface area.
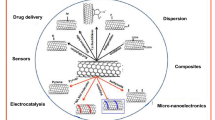
Belonging to the fullerene family, first described by Iijima [46], carbon nanotubes (CNTs) had been then expanded into multiwalled carbon nanotubes (MWNTs) and single-walled carbon nanotubes (SWNTs). Multiwalled carbon nanotubes are ideal support materials because of high strength, stability under acidic conditions, lack of swelling and large surface areas [47]. Taking these advantages, CNTs@MIPs should be superior, in some aspects, to other similar materials. Schematic protocol for the preparation of CNTs@MIPs is given in Fig. 2.
This review reports the principle of synthesizing CNTs@MIPs. Different particulars in the syntheses were also discussed. Applications in the field of SPE and sensor are highlighted. In the closing paragraph, we attempt to explore the potential development of CNTs@MIPs.
Properties of CNTs
Many studies have been carried out to use CNTs as analytical tools. Some of the most salient properties of CNTs are summarized below for facilitating further research. There are also some inclusive reviews recommended for reading in case of further interest [48, 49].
-
(1)
Nonpolar bonds and high aspect ratios (length to diameter ratio) lead to the insolubility of CNTs in water and their spontaneous aggregation. Meanwhile, in a few kinds of organic solvents, such as DMF (dimethyl formamide), DMAc (dimethyl acetamide) and dimethyl pyrrolidone, they show passable dispersibility [50]. Their insolubility and inclination to aggregation diminish the actual surface/area ratio [51]. High surface/area ratio is one of the useful properties of CNTs, hence, intensified research on their disaggregation has been conducted.
-
(2)
As many other organic materials, CNTs can be functionalized covalently [52, 53]. In most case, CNTs are chemically inert. But defects on the walls and small bond angle of the orifices makes carbon atoms in the defects and orifices more prone to be oxidized [54]. Under extreme chemical conditions, hydroxyl or carboxyl groups can be grafted onto the pipe walls and pipe orifices.
-
(3)
High electron mobility and electrical conductivity are also remarkable properties of CNTs. The arc-produced MWNTs are not be damaged under the current densities of J > 107 A·cm−2 [55]. These properties have been exploited to develop electrochemical sensors. These characteristics are contributed by the π orbital electrons delocalized across their hexagonal atomic arrays. Indeed, the arrangement of the hexagonal arrays determine the electronic capability of CNTs. As a result, CNTs can be metallic or semiconducting.
-
(4)
Owing to their high surface area to volume ratio and excellent physisorption performance, CNTs had been applied to solid-phase extraction (SPE), sensing, and hydrogen storage devices for years [56], and the absorption behaviors of CNTs towards many chemicals have been studied [57–60].
Synthesis of molecularly imprinted polymers on the surface of CNTs
To date, non-covalent approach is prevailing in the analytical molecular imprinting field, so the following discussion is specifically concerned with non-covalent approach. In general, synthesis steps of CNTs@MIPs include: First, commingle the templates (i.e. analytes) and functional monomers in solution, after incubation, the templates and functional monomers interact via non-covalent bonds. Then, initiated by an initiator and helped by crosslinkers, the monomers are polymerized around the templates. After polymerization, the materials are washed repeatedly by large amount of eluent (solvents that can dissolve the templates) to remove the templates, thus there are special cavities exposed. They can specifically rebind the template molecules [61]. To create high performance MIP, it is quite significant to select the templates, monomers and other reagents and apply efficient polymerization methods.
Selecting the templates
MIPs were often prepared for designated analytes. Binding sites in the cavities are determined by the structure and functional groups of the analyte. The norm of selecting templates contains the necessity, availability, cost etc. Most important, the chemical properties of templates should be considered. First, there should be certain chemical groups on the template for interacting with functional monomers. In non-covalent approach, because of the weak interplay, templates with multiple binding sites should be favored for strengthening self-assembling. Templates of commercialized MIPs contain aromatic rings or groups capable of hydrogen bond (clenbuterol [62], beta-agonists [63] and tobacco-specific nitrosamine [64]). Second, the templates should be stable under the reaction conditions, otherwise the imprinted molecules would be random by-product of the templates. Finally, there should not be any groups in the templates which might inhibit or participate in the polymerization.
For most cases, the molecules imprinted and rebound are the same, but when the templates were hard to obtain, inappropriate for the syntheses [65, 66] or hard to be completely removed from MIPs, structural substitutes (i.e. dummy templates) can be used in the syntheses. When using MIPs as SPE materials, for avoiding templates leakage, the dummy templates could be used. This approach shows its significance in the trace or ultra trace detection field because any template leakage should be different from the analyte and could be easily detected. The substitutes should conform to the target analytes in respect of shape, size and characteristic groups. According to the structure of dummy templates, this technology can be divided into fragment imprinting [67] and interval immobilization [68], progress in this field has been reviewed by Chen [9].
Selecting the other reagents
Methacrylic acid (MAA), 2-or 4-vinylpyridine (2-VP or 4-VP) and several silylating agents are widely used as functional monomer, tailor-made functional monomers were also reported [69, 70]. Apparently, monomers capable of strong interactions with templates were favored. Computational design and nuclear magnetic resonance was exploited for selecting functional monomers [71, 72]. The strength of interactions can be simulated and easily tested between various kinds of functional monomers and templates. In the work of M.B. Gholivand et al. [73], they tested 18 kinds of functional monomers and various kinds of solvents using molecular modeling. NMR of functional monomers and template mixture can also assist evaluating the interactions [36]. These methods increase the time and effort required for synthesis and charcterization. Recently, C. Baggiani et al. reported a theory that the MIP binding properties is corresponding with NIP (nonimprinted polymer) binding properties [74]. Using this theory, a huge amount of work might be economized.
The choice of crosslinkers is also very significant since the binding capacity is affected by different crosslinkers [75]. In the work of C. Michailof et al. [76], pentaerythritol triacrylate (PETRA), ethylene glycol dimethacrylate (EDMA) and divinylbenzene (DVB) were compared for synthesizing MIPs for caffeic acid. The binding effect can be impacted by the content and proportion of crosslinkers [77].
Choosing of porogen is very critical since the assembly between the functional monomers and templates was correlated with the chemical and physical properties of porogen. In one hand, the porogen should be able to dissolve the templates and the monomers. In the other, the porogen should not interfere the interplay between the functional monomers and templates. In free radical polymerization approach, a non to medium polar solvent is used as porogen, most importantly, this solvent should be aprotic (e.g. toluene, chloroform, DMF and DMSO). So, under protic conditions, metal chelating method is preferred because metal coordination interaction is still effective in protic solvents. Recently, there were studies reported of using methanol, ethanol even water as porogen [78, 79], however, no such articles has been found in the CNTs@MIPs field. In sol–gel approach, porogen is not so restricted, alcohol, ACN and water were used as porogen [80–82].
Some works are reported in Table 1 for comparison of different functional monomers, crosslinkers, initiators and porogen solvents. And some of the pioneering advances made to date are illustrated in Fig. 3.
Steps and methods of synthesizing CNTs@MIPs
Generally, surface imprinting means creating thin MIP films on suitable substrates, which exploit the concept of core-shell system. Using this system, the thickness of MIP shell can be accurately controlled by regulating the time span of syntheses and amounts of monomers. High absorbance is obtained by thick MIP shell and when frequent mass transfer was needed, thin MIP shell can be employed.
Functionalization of CNTs
First, the MWNTs should be functionalized to improve their reaction activity and dispersity. The methods can be divided into covalent and non-covalent functionalization. Covalent approach involves serial stages: (1) The crude MWNTs were treated with strong acids or strong oxidants to remove impurities as amorphous carbon and metal catalyst, and to obtain shorter MWNTs with carboxylic groups. (2) MWNTs-COCl was subsequently obtained by treating with thionyl chloride. (3) Allyl alcohol or allylamine was used for producing MWNTs-CH=CH2 [84, 90]. These groups enhance the solubility and dispersity of MWNTs, and also increase the jointing strength between MWNTs and MIP films.
The first to graft MIP films on the surface of functionalized CNTs was reported by Lee et al. [99]. CNTs were decorated with acrylated Tween 20 by hydrophobic interactions. Then they were incubated with photoinitiator for polymerization. This method has a main flaw that the polymerization takes place in solution because the photoinitiator might be dissociated in solution. So they modified their method by introducing 3-chloropropyl groups then photoactive iniferter to the surface of Tween 20 decorated CNTs [100]. With iniferter bounded to CNTs, this problem was basically solved. A 9-vinylanthracene functionalized CNTs@MIPs were reported, this method was facile and environment-friendly [101]. These foregoing approaches are functionalizing CNTs through non-covalent adsorption.
Materials incorporated with CNTs@MIPs
In our knowledge, various kinds of nanoparticles were incorporated on MWNTs (e.g. gold, SiO2 and Fe3O4 nanoparticles [13, 80, 102]). Gold nanoparticles are easy to prepare and possess a large surface area, most important, they were added for improving the electroconductivity of MIPs. So gold nanoparticles incorporating was common in the area of MIP based sensors [95, 96, 103, 104]. SiO2 often form into shells which provided a biocompatible and hydrophilic surface, and prevented oxidation of Fe3O4. Furthermore, silanol groups simplifies chemical modification on the surface of Fe3O4@SiO2 [80, 105, 106]. Once formed, the Fe3O4 nanoparticles become magnetic, which enables the entire CNTs@MIPs easily separated under an external magnetic field [14, 86, 93, 102, 107]. Table 2 shows some works using different kinds of materials incorporated on MWNTs.
Polymerization methods
Free radical polymerization and sol–gel process are two main developed methods to obtain CNTs@MIPs, some of the typical work were also summarized by Table 2. In the free radical polymerization method, polymerization of vinyl or acrylic monomers are initiated and accelerated by free radicals which are attained by thermal or photic decomposition of initiators. While azo-initiators, especially azo-bisisobutyronitrile (AIBN), were commonly used, photo-initiators allow preparing MIPs at mild temperature, which enables temperature-sensitive templates to be imprinted [108]. Novel technologies (as controlled/living free radical polymerization (CLRP), microwave-assisted heating and ionic liquid as porogen [109–111]), considered advantageous, were applied in traditional MIPs synthesis, but they were not reported in the CNTs@MIPs field yet.
Free radical polymerization remains the most commonly applied method for the preparation of CNTs@MIPs. The radicals decomposed from initiators allow reaction with various monomers and diversified templates, which expand the application area in molecular imprinting. But the high catalytic activity of these radicals can result in unnecessary subsidiary reaction, and the rigid condition of porogen confines the imprinting of many water-soluble molecules.
In sol–gel process, silicon alkoxide monomers are hydrolyzed and then polycondensated to produce an imprinted silica polymer network, these reactions are catalyzed by acids or bases [112]. In this fashion, the pH of pre-polymerization mixtures can be adjusted based on the requirements of templates, protic solvents such as water and alcohol should be used, and nitrogen is no longer required. Enjoying the versatile process and mild condition, a wider range of molecules can be imprinted in sol–gel process.
Electropolymerization was commonly used in the area of MIP-based sensors. This method was comprehensively reviewed by several teams, their works are recommended for reading [72, 113, 114], some of the typical research papers are as listed [115, 116].
Polymerization methods can be also sorted into “grafting-to” and “grafting-from”, according to the growing pattern of the polymers [117, 118]. As the name implies, “grafting-to” method is physically coupling macromolecular polymers on the materials or chemically coupling polymers on reactive groups of the materials, “grafting-from” method concerns covalently attaching initiators on the substrates and then grafting polymers in situ. Exploiting grafting-from method, polymerization in the solution can be remarkably reduced, thus the synthetic efficiency and regularity of the polymers is greatly enhanced. In the field of molecular imprinting, little research has been done exploiting the “grafting-from” methods [119]. But we foresee that these theories will be an inspiration for the synthesis of MIPs.
Applications
As observed from Fig. 4, the number of research articles about CNTs@MIPs augmented over the years due to their excellent properties and great versatility, illustrating their potential in multiple fields.
Solid phase extraction (SPE)
Most of the trace analytes existing in environmental or biological samples are critical for human health. Industrial materials unlawfully added into food products [120] or food contaminants [83] were considered as toxic or carcinogenic; the residues of some widely used antibiotics [88] in the environment and food chain may cause the spread of drug resistant microbes; some of these chemicals are accumulated through the food chain. These exigencies encourage researchers to develop fast and sensitive analytical methods applicable in aqueous samples.
Given its rapidity (when compared to traditional methods), conventional SPE has become a major procedure for sample treatment. It can precontrate target compounds but cannot eliminate most of the interferences. In view of its high selectivity, MIP-based SPE can determine even low-concentration analytes without intricate pretreatments and diminish most interferences.
On-line solid phase extraction
Though ease of commercialization and rapidity while analysis is enjoyed, the fabrication of MIP-based on-line SPE devices is more complicated than off-line ones. So the progress in recent years is not significant as expected. The devices were mainly produced in the format of columns [120] and steel frits [83].
An SPE microcolumn for Sudan IV online detection was achieved by Zhang and his team, using APTES-MWNTs as functionalized MWNTs, PTMOS and MTMOS as functional monomers, TEOS as crosslinker, acetone as porogen solvent [120]. The enrichment ratio of the CNTs@MIPs is up to 741-fold and the MIPs shows good recognition ability for Sudan IV. However, beyond the pH range of 6.0–6.8, the performance of imprinted column showed a notable decrease. Using “grafting-from” method, the author claimed that APTES assists the polymerization taking place on the MWNTs, but no experimental evidence was given. They also implied experimental results would be better with binary functional monomers, which was proved by many other papers [81, 121, 122].
Yu et al. obtained a molecular imprinted polypyrrole (MIPPy)/MWNTs steel frit by electropolymerization for online SPME (solid phase micro extraction) coupled with HPLC analysis of ochratoxin A [83]. The schematic diagram of this device was shown in Fig. 5. The results demonstrated a significant enhancement of selective binding for OTA compared with a C18-SPE cartridge. Using red wine volume of 30*100 μL for preconcentration, the detection and quantification limits are down to ppt scale. Nevertheless, the reproducibility is not good as traditional methods because this method needs multiple sample injection.
MIPPy/CNT-μSPP-PE-HPLC-FLD instrumentation [79]
Off-line solid phase extraction
There are many forms of off-line MIP SPE, e.g. fiber [88], cartridge [81] and syringe [123]. Different synthetic methods are listed in the following text by reviewing several representative works, such as electropolymerization [88], binary functional monomers [81], ion imprinting [124], sol–gel process [80] and free radical polymerization [87, 125].
An MIPPy/MWNTs/Pt fiber for SPME of fluoroquinolones (FQs) was made by Liu et al. [88]. The device was shown in Fig. 6 The MWNTs were deposited electrophoretically onto a Pt wire, then pyrrole was electropolymerized. This fiber can simultaneously extract six types of FQs.
Schematic diagram of EE-SPME extraction device based on molecularly imprinted conductive polymer/MWCNTs coating on Pt wire [119]
An CNTs@MIPs cartridge was synthesized by Zhang and his team using acryloyl-β-CD, MAA as functional monomers for the examination of erythromycin abuse in chicken breeding [81]. The MIPs using binary functional monomers took shorter time to reach adsorption saturation than traditional MIPs. Because the hydrophobic cavities of acryloyl-β-CD forms complexes with the water-insoluble erythromycin, the binary monomers MIPs exhibited more specific selectivity and better adsorption capacity. However, the performance of the SPE cartridge had not been compared with traditional SPE.
A Ga(III) (a heavy metal widely used in IT technology) ion-imprinted MWNTs column and a gold ion-imprinted column was synthesized recently [124, 126]. These methods were accurate and precise enough and performed good selectivity and cleanliness of the matrices but its linear range was not wide enough for various kinds of usage. The flaws should be blamed for the narrow linear range of atomic absorption spectrometry, not for the absorption capacities of MIPs.
A CNTs@MIPs column for selective removal and determination of triclosan was obtained by Gao et al. through sol–gel process [80]. In environmental water samples, after applied with their method, the peak of triclosan augmented distinctly and the peaks of other compounds were almost eliminated, while under direct injection, the peak of triclosan can not be seen (the results were presented in Fig. 7).
Extracts from the original chromatograms of river water samples. a Samples spiked with TCS at the concentration of 0.5 μg L−1, elution of (b) CNTs@NIPs and (c) CNTs@TCS-MIPs washed with a mixture of ethanol/6 M HCl (1:1, v/v) after CNTs@TCS-MIPs and CNTs@NIPs adsorbing 100 mL of river water spiked samples [76]
There is also primary form that directly blending SPE material with sample solutions. Though this blending approach is not advanced as other forms, it is quite simple if the imprinted materials can be separated from the sample solutions by a straightforward way. Our team [87, 125] and some other researchers [127] have been working on CNTs@MIPs with magnetic property, which can be easily separated from the sample solution by an external magnet. We synthesized MIPs on the surface of magnetic CNTs for directly determining gatifloxacin in serum samples [87], and the simultaneous extraction of four FQs from egg samples [125].
Chemical sensors
Sensitive and highly selective sensors were required for determining low content, hazardous substances or indicative, online-monitoring-needed compounds in complex matrices. Contributed by the premium electric properties of CNTs, high selectivity and robustness of MIPs, chemical sensors were held to be the most promising CNTs@MIPs applications (in the Web of Science database, the number of papers concerning CNTs@MIPs sensors was bigger than any other applications of CNTs@MIPs). The superiority of CNTs@MIPs over MIP sensors and CNTs sensors were also demonstrated by many publications [104, 128, 129].
CNTs@MIPs sensors were fabricated, in the main, by a simple drop-dry method. In particular, CNTs@MIPs were first synthesized, and templates were removed. Then the MIP suspension was dropped and dried on the surface of polished electrodes. Though it was believed that the stability and electronic activity of drop-dry sensors might not be good as electropolymerized or “grafting-from” ones [127], the reusability was yet quite outstanding because of the robustness of the imprinted cavities, and the sensors were functional after the electrodes were eluted by washing solutions [89].
In this part, we also chose some typical works in this area to elaborate on different methods of MIP-based sensor fabrication.
To the best of our knowledge, the first article reported CNTs@MIPs based biosensors were achieved by Zhang et al. [130]. Although preparing and working mechanisms of this sensor are not exactly conformed to general ones, but this initiative work has inspired considerable successors. The authors covalently attached the cofactor (NAD+) of glucose dehydrogenase to biopolymer CS, and the CNTs were added, ultimately, a CNTs-macrocomplexes film electrode detecting glucose was achieved. Because of the intricacy of the mechanism, sensitivity, stability and linear range of the sensor were moderate compared with up-to-date sensors.
Choong et al. electropolymerized an imprinted polypyrrole (PPy) film on carbon nanotubes which directly grow on a silicon substrate (the SEM images are shown in Fig. 8) [85]. The advantages of this method are remarkable: the thickness of the PPy film can be adjusted according to the molecular weight of the templates. And the binding capacity can be modulated by simply changing the CNTs height, density (number of nanotubes per area) and the thickness of the PPy. Cai et al. developed a similar but more elaborate constructed molecular imprinting sensors made for human ferritin and human papillomavirus derived E7 protein with sub pg L-1 sensitivity [131]. Though too complicated to be practical, this work offered us an innovative design in the field of MIP sensors [132].
a and (b) Schematic illustration of the concept of high density, surface molecular imprinting technique using CNTs array. SEM images of a sparse CNTs array (c) before and (d) after caffeine imprinting electropolymerization. SEM images of a dense CNTs array (e) before and (f) after caffeine imprinting electropolymerization [81]
A sensor based on free radical polymerized CNTs@MIPs for parathion-methyl (a highly toxic pesticide) determining was prepared by Zhang et al. [133]. This sensor was successfully applied in the detection of parathion-methyl in pear and cucumber samples with recoveries ranging between 94.9 and 106.2 %, and RSD less than 5 % (n = 5). However, so many processes were employed in the real sample pre-treatment, that the practicability of this sensor should be doubted. Based on free radical polymerization, an electrode coated with CNTs@MIPs and gold NPs for the detection of chloramphenicol were fabricated [104]. It is worth mentioning that in the concentration range of 2–40 mg L−1, the recoveries and RSDs of chloramphenicol sensor were quite close to HPLC method (92.5–105 % vs 95.5–110 %, 3.2–12.5 % vs 0.9–14.8 %).
Several CNTs@MIPs real sample detecting electrodes were fabricated by Prasad et al. using state-of-the-art technologies, such as tailor-made functional monomer for insulin detection [69], tailor-made functional monomer and crosslinker for bovine serum albumin (BSA) detection [108]. For the insulin sensor, the excellent performance (for blood serum samples, the linear range is between 0.068 and 5.682 nmol L−1 with RSD of 0.9 %, n = 3) should be attributed to the fact that the tailor-made functional monomer restricts non-specific proteins adsorption. Tailor-made functional monomer and crosslinker were used for the water compatibility of the MIPs. They also fabricated a dopamine electrode using “grafting-from” method [98]. When synthesizing dopamine electrode, the MWNTs were coated by iniferter (initiator transfer agent terminator) first, because of the immobilized iniferter, polymerization occurs on the surface of CNTs.
Synthesized through electrochemical copolymerization, based on Ru(bpy)3 2+-Au nanoparticles, a molecularly imprinted electrochemiluminescence (ECL) sensor was proposed by Wu et al. to selectively determine isoniazid (a wonder drug for tuberculosis but has serious adverse effects when overdosing) [129]. For tablet samples, the analytical data were consistent with China Pharmacopoeia method (RSD = 3.2 % while the RSD of Pharmacopoeia method is 3.6 %) and the sensor was also successfully applied to determine isoniazid in human urine and pharmaceutical samples with the recovery of 96–102.5 %. It is worth mentioning that untreated urine samples can be analyzed.
Santos et al. made an imprinted sensor for caffeine using sol–gel process. Compared with previous works [134–136], the recoveries were excellent (98.0–103.8 %) for untreated real drink samples [137]. An indium tin oxide electrode modified by CNTs@MIPs via sol–gel technology was prepared by Zhang et al. for the detection of L-histidine [128]. It was successfully employed to detect L-histidine in human blood serum (the recovery is 96.7–101.4 %). The authors did not specify what treatment they used in the serum sample. If raw serum were used, this method is quite promising in clinical test.
Conclusions and perspectives
The current status, synthetic approaches and highlighted applications of CNTs@MIPs have been described in this review. In conclusion, CNTs@MIPs retain the affinity and selectivity of antibodies, enzymes or biological receptors, while adding several benefits including high mechanical strength, large specific surface area and chemical stability. Much work has been performed to resolve the existing problems hindering the development of CNTs@MIPs and the commercial potential has been proved by considerable MIP products during past years.
Although remarkable achievements have been attained in CNTs@MIPs, there are still substantial development challenges and opportunities. We attempt to tackle some important exploration initiatives as follows: (1) to improve the dispersibility of the CNTs in organic phase or aqueous phase, reaching high efficiency of molecular imprinting process; (2) to design and exploit new, convenient polymerization methods (e.g. CLRP, microwave-assisted heating) for molecular imprinting on the surface of CNTs for higher binding capacity and selectivity; (3) to explain the recognition mechanism and the role of CNTs at the molecular level with the aid of advanced equipment, especially the imprinting mechanism of sol–gel method; (4) to broaden imprinting targets from small molecules to proteins in order to make CNTs@MIPs a truly practical tool for molecular recognition and a promising commercial product for real applications.
Abbreviations
- 2 or 4-VP:
-
2-or 4-vinylpyridine
- AA:
-
Acrylic acid
- AEP:
-
(p-acryloylaminophenyl)-{(4-aminophenyl)-diethyl ammonium}-ethylphosphate
- AIBN:
-
Azo-bisisobutyronitrile
- APS:
-
Ammonium persulfate
- APTMS:
-
Aminopropyltrimethoxysilane
- β-CD:
-
Beta-cyclodextrin
- BDC:
-
N,N-diethyldithiocarbamate
- BSA:
-
Bovine serum albumin
- CLRP:
-
Controlled/living free radical polymerization
- CNTs:
-
Carbon nanotubes
- CNTs@MIPs:
-
Molecularly imprinted polymers on the surface of carbon nanotubes
- CS:
-
Chitosan
- CSDT:
-
Chitosan derivative
- DMAc:
-
Dimethyl acetamide
- DMF:
-
Dimethyl formamide
- DMSO:
-
Dimethyl sulfoxide
- DVB:
-
Divinylbenzene
- ECL:
-
Electrochemiluminescence
- EE:
-
Ethoxyethanol
- EGDMA or EDMA:
-
Ethylene glycol dimethacrylate
- FQs:
-
Fluoroquinolones
- Ga(III):
-
Gallium(III)
- MAA:
-
Methacrylic acid
- MIM:
-
Molecularly imprinted membrane
- MIPPy:
-
Molecular imprinted polypyrrole
- MIPs:
-
Molecularly imprinted polymers
- MIT:
-
Molecular imprinting technology
- MTMOS:
-
Methyltrimethoxysilane
- MWNTs:
-
Multiwalled carbon nanotubes
- NNMBA:
-
N,N-methylenebisacrylamide
- NPA:
-
4-nitrophenyl acrylate
- OTA:
-
Ochratoxin A
- PAMAM:
-
Polyamide amine
- PEG:
-
Polyethylene glycol
- PETRA:
-
Pentaerythritol triacrylate
- PPy:
-
Polypyrrole
- PTMOS:
-
phenyltrimethoxysilane
- SPE:
-
Solid-phase extraction
- SPME:
-
Solid phase micro extraction
- SWNTs:
-
Single-walled carbon nanotubes
- TCP:
-
2,4,6-Trichlorophenol
- TEOS:
-
Tetraethylorthosilicate
- THF:
-
Tetrahydrofuran
- μ-SPP:
-
Micro-solid phase preconcentration
References
Hinterdorfer P, Dufrene YF (2006) Detection and localization of single molecular recognition events using atomic force microscopy. Nat Methods 3(5):347–355. doi:10.1038/nmeth871
Ye L, Mosbach K (2008) Molecular imprinting: synthetic materials as substitutes for biological antibodies and receptors†. Chem Mater 20(3):859–868. doi:10.1021/cm703190w
Wulff G, Sarhan A (1972) Über die Anwendung von enzymanalog gebauten Polymeren zur Racemattrennung. Angew Chem 84(8):364. doi:10.1002/ange.19720840838
Vlatakis G, Andersson LI, Muller R, Mosbach K (1993) Drug assay using antibody mimics made by molecular imprinting. Nature 361(6413):645–647. doi:10.1038/361645a0
Arshady R, Mosbach K (1981) Synthesis of substrate-selective polymers by host-guest polymerization. Die Makromol Chem 182(2):687–692. doi:10.1002/macp.1981.021820240
Nicholls IA, Rosengren JP (2001) Molecular imprinting of surfaces. Bioseparation 10(6):301–305. doi:10.1023/a:1021541631063
Wulff G (2002) Enzyme-like catalysis by molecularly imprinted polymers. Chem Rev 102(1):1–28. doi:10.1021/cr980039a
Alexander C, Andersson HS, Andersson LI, Ansell RJ, Kirsch N, Nicholls IA, O’Mahony J, Whitcombe MJ (2006) Molecular imprinting science and technology: a survey of the literature for the years up to and including 2003. J Mol Recognit 19(2):106–180. doi:10.1002/jmr.760
Chen L, Xu S, Li J (2011) Recent advances in molecular imprinting technology: current status, challenges and highlighted applications. Chem Soc Rev 40(5):2922–2942. doi:10.1039/c0cs00084a
Díaz-Díaz G, Antuña-Jiménez D, Carmen Blanco-López M, Jesús Lobo-Castañón M, Miranda-Ordieres AJ, Tuñón-Blanco P (2012) New materials for analytical biomimetic assays based on affinity and catalytic receptors prepared by molecular imprinting. TrAC Trends Anal Chem 33:68–80. doi:10.1016/j.trac.2011.09.011
Tan CJ, Tong YW (2007) Molecularly imprinted beads by surface imprinting. Anal Bioanal Chem 389(2):369–376. doi:10.1007/s00216-007-1362-4
Ge Y, Turner AP (2008) Too large to fit? Recent developments in macromolecular imprinting. Trends Biotechnol 26(4):218–224. doi:10.1016/j.tibtech.2008.01.001
Wang H, Zhao H, Quan X, Chen S (2011) Electrochemical determination of tetracycline using molecularly imprinted polymer modified carbon nanotube-gold nanoparticles electrode. Electroanalysis 23(8):1863–1869. doi:10.1002/elan.201100049
Zhang Z, Yang X, Chen X, Zhang M, Luo L, Peng M, Yao S (2011) Novel magnetic bovine serum albumin imprinted polymers with a matrix of carbon nanotubes, and their application to protein separation. Anal Bioanal Chem 401(9):2855–2863. doi:10.1007/s00216-011-5373-9
Nagata T, Goji S, Akamatsu K, Nawafune H, Matsui J (2012) Monodispersed molecularly imprinted polymer beads with enhanced atrazine retention ability synthesized with polymeric diluents. Anal Lett 45(9):977–985. doi:10.1080/00032719.2012.670786
Fan L, Zhang Y, Li X, Luo C, Lu F, Qiu H (2012) Removal of alizarin red from water environment using magnetic chitosan with Alizarin Red as imprinted molecules. Colloids Surf, B 91:250–257. doi:10.1016/j.colsurfb.2011.11.014
Xia YQ, Guo TY, Song MD, Zhang BH, Zhang BL (2006) Selective separation of quercetin by molecular imprinting using chitosan beads as functional matrix. React Funct Polym 66(12):1734–1740. doi:10.1016/j.reactfunctpolym.2006.08.001
Song X, Li C, Xu R, Wang K (2012) Molecular-ion-imprinted chitosan hydrogels for the selective adsorption of silver(I) in aqueous solution. Ind Eng Chem Res 51(34):11261–11265. doi:10.1021/ie3010989
Dai C, Liu C, Wei J, Hong H, Zhao Q (2010) Molecular imprinted macroporous chitosan coated mesoporous silica xerogels for hemorrhage control. Biomaterials 31(30):7620–7630. doi:10.1016/j.biomaterials.2010.06.049
Gu JY, Zhang H, Yuan G, Chen LR, Xu XJ (2011) Surface-initiated molecularly imprinted polymeric column: in situ synthesis and application for semi-preparative separation by high performance liquid chromatography. J Chromatogr A 1218(45):8150–8155. doi:10.1016/j.chroma.2011.09.019
Zhao D, Jia J, Yu X, Sun X (2011) Preparation and characterization of a molecularly imprinted polymer by grafting on silica supports: a selective sorbent for patulin toxin. Anal Bioanal Chem 401(7):2259–2273. doi:10.1007/s00216-011-5282-y
Zhang W, Qin L, He XW, Li WY, Zhang YK (2009) Novel surface modified molecularly imprinted polymer using acryloyl-beta-cyclodextrin and acrylamide as monomers for selective recognition of lysozyme in aqueous solution. J Chromatogr A 1216(21):4560–4567. doi:10.1016/j.chroma.2009.03.056
Xia YQ, Guo TY, Zhao HL, Song MD, Zhang BH, Zhang BL (2009) Protein recognition onto silica particles using chitosan as intermedium substrate. J Biomed Mater Res, Part A 90A(2):326–332. doi:10.1002/jbm.a.32084
Moreira FTC, Dutra RAF, Noronha JPC, Sales MGF (2011) Myoglobin-biomimetic electroactive materials made by surface molecular imprinting on silica beads and their use as ionophores in polymeric membranes for potentiometric transduction. Biosens Bioelectron 26(12):4760–4766. doi:10.1016/j.bios.2011.05.045
Hua Z, Zhou S, Zhao M (2009) Fabrication of a surface imprinted hydrogel shell over silica microspheres using bovine serum albumin as a model protein template. Biosens Bioelectron 25(3):615–622. doi:10.1016/j.bios.2009.01.027
Fukazawa K, Li Q, Seeger S, Ishihara K (2013) Direct observation of selective protein capturing on molecular imprinting substrates. Biosens Bioelectron 40(1):96–101. doi:10.1016/j.bios.2012.06.033
Fukazawa K, Ishihara K (2009) Fabrication of a cell-adhesive protein imprinting surface with an artificial cell membrane structure for cell capturing. Biosens Bioelectron 25(3):609–614. doi:10.1016/j.bios.2009.02.034
Chang L, Li Y, Chu J, Qi J, Li X (2010) Preparation of core-shell molecularly imprinted polymer via the combination of reversible addition-fragmentation chain transfer polymerization and click reaction. Anal Chim Acta 680(1–2):65–71. doi:10.1016/j.aca.2010.09.017
Barahona F, Turiel E, Cormack PAG, Martin-Esteban A (2010) Chromatographic performance of molecularly imprinted polymers: core-shell microspheres by precipitation polymerization and grafted MIP films via iniferter-modified silica beads. J Polym Sci, Part A: Polym Chem 48(5):1058–1066. doi:10.1002/pola.23860
Dramou P, Xiao D, He H, Liu T, Zou W (2013) Loading behavior of gatifloxacin in urine and lake water on a novel magnetic molecularly imprinted polymer used as extraction sorbent with spectrophotometric analysis. J Sep Sci 36(5):898–906. doi:10.1002/jssc.201200831
Wen T, Xue C, Li Y, Wang Y, Wang R, Hong J, Zhou X, Jiang H (2012) Reduced graphene oxide-platinum nanoparticles composites based imprinting sensor for sensitively electrochemical analysis of 17 beta-estradiol. J Electroanal Chem 682:121–127. doi:10.1016/j.jelechem.2012.07.015
Mao Y, Bao Y, Gan S, Li F, Niu L (2011) Electrochemical sensor for dopamine based on a novel graphene-molecular imprinted polymers composite recognition element. Biosens Bioelectron 28(1):291–297. doi:10.1016/j.bios.2011.07.034
Liu Y, Zhu L, Zhang Y, Tang H (2012) Electrochemical sensoring of 2,4-dinitrophenol by using composites of graphene oxide with surface molecular imprinted polymer. Sens Actuators, B 171:1151–1158. doi:10.1016/j.snb.2012.06.054
Chang L, Wu S, Chen S, Li X (2011) Preparation of graphene oxide-molecularly imprinted polymer composites via atom transfer radical polymerization. J Mater Sci 46(7):2024–2029. doi:10.1007/s10853-010-5033-z
Pan J, Hang H, Dai X, Dai J, Huo P, Yan Y (2012) Switched recognition and release ability of temperature responsive molecularly imprinted polymers based on magnetic halloysite nanotubes. J Mater Chem 22(33):17167–17175. doi:10.1039/C2jm32821f
Pan J, Wang B, Dai J, Dai X, Hang H, Ou H, Yan Y (2012) Selective recognition of 2,4,5-trichlorophenol by temperature responsive and magnetic molecularly imprinted polymers based on halloysite nanotubes. J Mater Chem 22(8):3360–3369. doi:10.1039/C1jm14825g
Pan J, Yao H, Xu L, Ou H, Huo P, Li X, Yan Y (2011) Selective recognition of 2,4,6-trichlorophenol by molecularly imprinted polymers based on magnetic halloysite nanotubes composites. J Phys Chem C 115(13):5440–5449. doi:10.1021/jp111120x
Zhang W, He XW, Chen Y, Li WY, Zhang YK (2012) Molecularly imprinted polymer anchored on the surface of denatured bovine serum albumin modified CdTe quantum dots as fluorescent artificial receptor for recognition of target protein. Biosens Bioelectron 31(1):84–89. doi:10.1016/j.bios.2011.09.042
Zhang W, He XW, Chen Y, Li WY, Zhang YK (2011) Composite of CdTe quantum dots and molecularly imprinted polymer as a sensing material for cytochrome c. Biosens Bioelectron 26(5):2553–2558. doi:10.1016/j.bios.2010.11.004
Wang HF, He Y, Ji TR, Yan XP (2009) Surface molecular imprinting on Mn-Doped ZnS quantum dots for room-temperature phosphorescence optosensing of pentachlorophenol in water. Anal Chem 81(4):1615–1621. doi:10.1021/ac802375a
Mao Y, Bao Y, Han D, Li F, Niu L (2012) Efficient one-pot synthesis of molecularly imprinted silica nanospheres embedded carbon dots for fluorescent dopamine optosensing. Biosens Bioelectron 38(1):55–60. doi:10.1016/j.bios.2012.04.043
Liu J, Chen H, Lin Z, Lin JM (2010) Preparation of surface imprinting polymer capped Mn-Doped ZnS quantum dots and their application for chemiluminescence detection of 4-nitrophenol in tap water. Anal Chem 82(17):7380–7386. doi:10.1021/ac101510b
Lin HY, Ho MS, Lee MH (2009) Instant formation of molecularly imprinted poly(ethylene-co-vinyl alcohol)/quantum dot composite nanoparticles and their use in one-pot urinalysis. Biosens Bioelectron 25(3):579–586. doi:10.1016/j.bios.2009.03.039
Lian HT, Liu B, Chen YP, Sun XY (2012) A urea electrochemical sensor based on molecularly imprinted chitosan film doping with CdS quantum dots. Anal Biochem 426(1):40–46. doi:10.1016/j.ab.2012.03.024
Ge S, Zhang C, Yu F, Yan M, Yu J (2011) Layer-by-layer self-assembly CdTe quantum dots and molecularly imprinted polymers modified chemiluminescence sensor for deltamethrin detection. Sens Actuators, B 156(1):222–227. doi:10.1016/j.snb.2011.04.024
Iijima S (1991) Helical microtubules of graphitic carbon. Nature 354(6348):56–58. doi:10.1038/354056a0
Hirsch A (2002) Functionalization of single-walled carbon nanotubes. Angew Chem Int Ed Engl 41(11):1853–1859. doi:10.1002/1521-3773(20020603)41:11<1853::AID-ANIE1853>3.0.CO;2-N
Sun YP, Fu K, Lin Y, Huang W (2002) Functionalized carbon nanotubes: properties and applications. Acc Chem Res 35(12):1096–1104. doi:10.1021/ar010160v
Dai HJ (2002) Carbon nanotubes: synthesis, integration, and properties. Acc Chem Res 35(12):1035–1044. doi:10.1021/ar0101640
Kim B, Lee Y-H, Ryu J-H, Suh K-D (2006) Enhanced colloidal properties of single-wall carbon nanotubes in α-terpineol and Texanol. Colloids Surf, A 273(1–3):161–164. doi:10.1016/j.colsurfa.2005.08.024
Vaisman L, Wagner HD, Marom G (2006) The role of surfactants in dispersion of carbon nanotubes. Adv Colloid Interface Sci 128–130:37–46. doi:10.1016/j.cis.2006.11.007
Bianco A, Kostarelos K, Prato M (2011) Making carbon nanotubes biocompatible and biodegradable. Chem Commun 47(37):10182–10188. doi:10.1039/c1cc13011k
Singh P, Campidelli S, Giordani S, Bonifazi D, Bianco A, Prato M (2009) Organic functionalisation and characterisation of single-walled carbon nanotubes. Chem Soc Rev 38(8):2214–2230. doi:10.1039/b518111a
Liu J, Rinzler AG, Dai H, Hafner JH, Bradley RK, Boul PJ, Lu A, Iverson T, Shelimov K, Huffman CB, Rodriguez-Macias F, Shon YS, Lee TR, Colbert DT, Smalley RE (1998) Fullerene pipes. Science 280(5367):1253–1256. doi:10.1126/science.280.5367.1253
Frank S, Poncharal P, Wang ZL, Heer WA (1998) Carbon nanotube quantum resistors. Science 280(5370):1744–1746. doi:10.1126/science.280.5370.1744
Raffaelle RP, Landi BJ, Harris JD, Bailey SG, Hepp AF (2005) Carbon nanotubes for power applications. Mater Sci Eng B 116(3):233–243. doi:10.1016/j.mseb.2004.09.034
Li LL, Lin R, He H, Jiang L, Gao MM (2013) Interaction of carboxylated single-walled carbon nanotubes with bovine serum albumin. Spectrochim Acta, Part A 105:45–51. doi:10.1016/j.saa.2012.11.111
Jiang L, Liu TB, He H, Pham-Huy LA, Li LL, Pham-Huy C, Xiao DL (2012) Adsorption behavior of pazufloxacin mesilate on amino-functionalized carbon nanotubes. J Nanosci Nanotechnol 12(9):7271–7279. doi:10.1166/jnn.2012.6562
Chen Z, Pierre D, He H, Tan SH, Chuong PH, Hong H, Huang JL (2011) Adsorption behavior of epirubicin hydrochloride on carboxylated carbon nanotubes. Int J Pharm 405(1–2):153–161. doi:10.1016/j.ijpharm.2010.11.034
Xiao DL, Dramou P, He H, Lien APH, Li H, Yao YY, Chuong PH (2012) Magnetic carbon nanotubes: synthesis by a simple solvothermal process and application in magnetic targeted drug delivery system. J Nanopart Res 14(7):984–996. doi:10.1007/s11051-012-0984-4
Huang J, Xing X, Zhang X, He X, Lin Q, Lian W, Zhu H (2011) A molecularly imprinted electrochemical sensor based on multiwalled carbon nanotube-gold nanoparticle composites and chitosan for the detection of tyramine. Food Res Int 44(1):276–281. doi:10.1016/j.foodres.2010.10.020
Blomgren A, Berggren C, Holmberg A, Larsson F, Sellergren B, Ensing K (2002) Extraction of clenbuterol from calf urine using a molecularly imprinted polymer followed by quantitation by high-performance liquid chromatography with UV detection. J Chromatogr A 975(1):157–164. doi:10.1016/s0021-9673(02)01359-6
Kootstra PR, Kuijpers CJPF, Wubs KL, van Doorn D, Sterk SS, van Ginkel LA, Stephany RW (2005) The analysis of beta-agonists in bovine muscle using molecular imprinted polymers with ion trap LCMS screening. Anal Chim Acta 529(1–2):75–81. doi:10.1016/j.aca.2004.09.053
Xia Y, McGuffey JE, Bhattacharyya S, Sellergren B, Yilmaz E, Wang L, Bernert JT (2005) Analysis of the tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol in urine by extraction on a molecularly imprinted polymer column and liquid chromatography/atmospheric pressure ionization tandem mass spectrometry. Anal Chem 77(23):7639–7645. doi:10.1021/ac058027u
Yan H, Cheng X, Yang G (2012) Dummy molecularly imprinted solid-phase extraction for selective determination of five phthalate esters in plastic bottled functional beverages. J Agric Food Chem 60(22):5524–5531. doi:10.1021/jf300660m
Yin YM, Chen YP, Wang XF, Liu Y, Liu HL, Xie MX (2012) Dummy molecularly imprinted polymers on silica particles for selective solid-phase extraction of tetrabromobisphenol A from water samples. J Chromatogr A 1220:7–13. doi:10.1016/j.chroma.2011.11.065
Nemoto K, Kubo T, Nomachi M, Sano T, Matsumoto T, Hosoya K, Hattori T, Kaya K (2007) Simple and effective 3D recognition of domoic acid using a molecularly imprinted polymer. J Am Chem Soc 129(44):13626–13632. doi:10.1021/ja0741426
Tominaga Y, Kubo T, Kaya K, Hosoya K (2009) Effective recognition on the surface of a polymer prepared by molecular imprinting using ionic complex. Macromolecules 42(8):2911–2915. doi:10.1021/ma802880z
Prasad BB, Madhuri R, Tiwari MP, Sharma PS (2010) Imprinting molecular recognition sites on multiwalled carbon nanotubes surface for electrochemical detection of insulin in real samples. Electrochim Acta 55(28):9146–9156. doi:10.1016/j.electacta.2010.09.008
Prasad BB, Prasad A, Tiwari MP (2013) Highly selective and sensitive analysis of gamma-aminobutyric acid using a new molecularly imprinted polymer modified at the surface of abrasively immobilized multi-walled carbon nanotubes on pencil graphite electrode. Electrochim Acta 102:400–408. doi:10.1016/j.electacta.2013.04.043
Courtois J, Fischer G, Schauff S, Albert K, Irgum K (2005) Interactions of bupivacaine with a molecularly imprinted polymer in a monolithic format studied by NMR. Anal Chem 78(2):580–584. doi:10.1021/ac0515733
Whitcombe MJ, Chianella I, Larcombe L, Piletsky SA, Noble J, Porter R, Horgan A (2011) The rational development of molecularly imprinted polymer-based sensors for protein detection. Chem Soc Rev 40(3):1547–1571. doi:10.1039/c0cs00049c
Gholivand MB, Khodadadian M (2011) Rationally designed molecularly imprinted polymers for selective extraction of methocarbamol from human plasma. Talanta 85(3):1680–1688. doi:10.1016/j.talanta.2011.06.066
Baggiani C, Giovannoli C, Anfossi L, Passini C, Baravalle P, Giraudi G (2012) A connection between the binding properties of imprinted and nonimprinted polymers: a change of perspective in molecular imprinting. J Am Chem Soc 134(3):1513–1518. doi:10.1021/ja205632t
Pichon V, Haupt K (2006) Affinity separations on molecularly imprinted polymers with special emphasis on solid-phase extraction. J Liq Chromatogr Relat Technol 29(7–8):989–1023. doi:10.1080/10826070600574739
Michailof C, Manesiotis P, Panayiotou C (2008) Synthesis of caffeic acid and p-hydroxybenzoic acid molecularly imprinted polymers and their application for the selective extraction of polyphenols from olive mill waste waters. J Chromatogr A 1182(1):25–33. doi:10.1016/j.chroma.2008.01.001
Cai W, Gupta RB (2004) Molecularly-imprinted polymers selective for tetracycline binding. Sep Purif Technol 35(3):215–221. doi:10.1016/S1383-5866(03)00143-6
Pardeshi S, Dhodapkar R, Kumar A (2013) Quantum chemical density functional theory studies on the molecular structure and vibrational spectra of Gallic acid imprinted polymers. Spectrochim Acta A Mol Biomol Spectrosc 116:562–573. doi:10.1016/j.saa.2013.07.067
Panahi R, Vasheghani-Farahani E, Shojaosadati SA (2007) Separation of l-lysine from dilute aqueous solution using molecular imprinting technique. Biochem Eng J 35(3):352–356. doi:10.1016/j.bej.2007.01.027
Gao R, Kong X, Su F, He X, Chen L, Zhang Y (2010) Synthesis and evaluation of molecularly imprinted core-shell carbon nanotubes for the determination of triclosan in environmental water samples. J Chromatogr A 1217(52):8095–8102. doi:10.1016/j.chroma.2010.10.121
Zhang Z, Yang X, Zhang H, Zhang M, Luo L, Hu Y, Yao S (2011) Novel molecularly imprinted polymers based on multi-walled carbon nanotubes with binary functional monomer for the solid-phase extraction of erythromycin from chicken muscle. J Chromatogr B Anal Technol Biomed Life Sci 879(19):1617–1624. doi:10.1016/j.jchromb.2011.03.054
Hu YF, Zhang ZH, Zhang HB, Luo LJ, Yao SZ (2012) Sensitive and selective imprinted electrochemical sensor for p-nitrophenol based on ZnO nanoparticles/carbon nanotubes doped chitosan film. Thin Solid Films 520(16):5314–5321. doi:10.1016/j.tsf.2011.11.083
Yu JCC, Lai EPC (2006) Molecularly imprinted polypyrrole modified carbon nanotubes on stainless steel frit for selective micro solid phase pre-concentration of ochratoxin A. React Funct Polym 66(7):702–711. doi:10.1016/j.reactfunctpolym.2005.10.021
Kan X, Zhao Y, Geng Z, Wang Z, Zhu JJ (2008) Composites of multiwalled carbon nanotubes and molecularly imprinted polymers for dopamine recognition. J Phys Chem C 112(13):4849–4854. doi:10.1021/jp077445v
Choong CL, Bendall JS, Milne WI (2009) Carbon nanotube array: a new MIP platform. Biosens Bioelectron 25(3):652–656. doi:10.1016/j.bios.2008.11.025
Chen HJ, Zhang ZH, Luo LJ, Yao SZ (2012) Surface-imprinted chitosan-coated magnetic nanoparticles modified multi-walled carbon nanotubes biosensor for detection of bovine serum albumin. Sens Actuators, B 163(1):76–83. doi:10.1016/j.snb.2012.01.010
Xiao D, Dramou P, Xiong N, He H, Li H, Yuan D, Dai H (2013) Development of novel molecularly imprinted magnetic solid-phase extraction materials based on magnetic carbon nanotubes and their application for the determination of gatifloxacin in serum samples coupled with high performance liquid chromatography. J Chromatogr A 1274:44–53. doi:10.1016/j.chroma.2012.12.011
Liu X, Wang X, Tan F, Zhao H, Quan X, Chen J, Li L (2012) An electrochemically enhanced solid-phase microextraction approach based on molecularly imprinted polypyrrole/multi-walled carbon nanotubes composite coating for selective extraction of fluoroquinolones in aqueous samples. Anal Chim Acta 727:26–33. doi:10.1016/j.aca.2012.03.054
Tan F, Deng M, Liu X, Zhao H, Li X, Quan X, Chen J (2011) Evaluation of a novel microextraction technique for aqueous samples: porous membrane envelope filled with multiwalled carbon nanotubes coated with molecularly imprinted polymer. J Sep Sci 34(6):707–715. doi:10.1002/jssc.201000791
Yen CP, Chin NP, Wei HC, Chuan HK (2010) Detection of uric acid based on multi-walled carbon nanotubes polymerized with a layer of molecularly imprinted PMAA. Sens Actuators, B 146(2):466–471. doi:10.1016/j.snb.2009.11.035
Diaz-Diaz G, Carmen Blanco-Lopez M, Jesus Lobo-Castanon M, Miranda-Ordieres AJ, Tunon-Blanco P (2011) Preparation and characterization of a molecularly imprinted microgel for electrochemical sensing of 2,4,6-trichlorophenol. Electroanalysis 23(1):201–208. doi:10.1002/elan.201000481
Yufang H, Zhaohui Z, Huabin Z, Lijuan L, Shouzhuo Y (2012) A sensitive and selective sensor-coated molecularly imprinted sol-gel film incorporating beta-cyclodextrin-multi-walled carbon nanotubes and cobalt nanoparticles-chitosan for oxacillin determination. Surf Interface Anal 44(3):334–341. doi:10.1002/sia.3807
Hu Y, Zhang Z, Zhang H, Luo L, Yao S (2012) Selective and sensitive molecularly imprinted sol-gel film-based electrochemical sensor combining mercaptoacetic acid-modified PbS nanoparticles with Fe3O4@Au-multi-walled carbon nanotubes-chitosan. J Solid State Electrochem 16(3):857–867. doi:10.1007/s10008-011-1434-4
Kan X, Liu T, Zhou H, Li C, Fang B (2010) Molecular imprinting polymer electrosensor based on gold nanoparticles for theophylline recognition and determination. Microchim Acta 171(3–4):423–429. doi:10.1007/s00604-010-0455-5
Li H, Xie C, Li S, Xu K (2012) Electropolymerized molecular imprinting on gold nanoparticle-carbon nanotube modified electrode for electrochemical detection of triazophos. Colloids Surf, B 89:175–181. doi:10.1016/j.colsurfb.2011.09.010
Lian W, Huang J, Yu J, Zhang X, Lin Q, He X, Xing X, Liu S (2012) A molecularly imprinted sensor based on beta-cyclodextrin incorporated multiwalled carbon nanotube and gold nanoparticles-polyamide amine dendrimer nanocomposites combining with water-soluble chitosan derivative for the detection of chlortetracycline. Food Control 26(2):620–627. doi:10.1016/j.foodcont.2012.02.023
Moreira FTC, Dutra RAF, Noronha JPC, Cunha AL, Sales MGF (2011) Artificial antibodies for troponin T by its imprinting on the surface of multiwalled carbon nanotubes: Its use as sensory surfaces. Biosens Bioelectron 28(1):243–250. doi:10.1016/j.bios.2011.07.026
Prasad BB, Kumar D, Madhuri R, Tiwari MP (2011) Sol-gel derived multiwalled carbon nanotubes ceramic electrode modified with molecularly imprinted polymer for ultra trace sensing of dopamine in real samples. Electrochim Acta 56(20):7202–7211. doi:10.1016/j.electacta.2011.04.090
Lee E, Park DW, Lee J-O, Kim DS, Lee BH, Kim BS (2008) Molecularly imprinted polymers immobilized on carbon nanotube. Colloids Surf, A 313:202–206. doi:10.1016/j.colsurfa.2007.04.093
Lee HY, Kim BS (2009) Grafting of molecularly imprinted polymers on iniferter-modified carbon nanotube. Biosens Bioelectron 25(3):587–591. doi:10.1016/j.bios.2009.03.040
Zhang XL, Zhang Y, Yin XF, Du BB, Zheng C, Yang HH (2013) A facile approach for preparation of molecularly imprinted polymers layer on the surface of carbon nanotubes. Talanta 105:403–408. doi:10.1016/j.talanta.2012.10.062
Hu Y, Li J, Zhang Z, Zhang H, Luo L, Yao S (2011) Imprinted sol–gel electrochemical sensor for the determination of benzylpenicillin based on Fe3O4@SiO2/multi-walled carbon nanotubes-chitosans nanocomposite film modified carbon electrode. Anal Chim Acta 698(1–2):61–68. doi:10.1016/j.aca.2011.04.054
Fu XC, Wu J, Nie L, Xie CG, Liu JH, Huang XJ (2012) Electropolymerized surface ion imprinting films on a gold nanoparticles/single-wall carbon nanotube nanohybrids modified glassy carbon electrode for electrochemical detection of trace mercury(II) in water. Anal Chim Acta 720:29–37. doi:10.1016/j.aca.2011.12.071
Zhao H, Chen Y, Tian J, Yu H, Quan X (2012) Selectively electrochemical determination of chloramphenicol in aqueous solution using molecularly imprinted polymer-carbon nanotubes-gold nanoparticles modified electrode. J Electrochem Soc 159(6):J231–J236. doi:10.1149/2.116206jes
Lu F, Li H, Sun M, Fan L, Qiu H, Li X, Luo C (2012) Flow injection chemiluminescence sensor based on core-shell magnetic molecularly imprinted nanoparticles for determination of sulfadiazine. Anal Chim Acta 718:84–91. doi:10.1016/j.aca.2011.12.054
Gao R, Su X, He X, Chen L, Zhang Y (2011) Preparation and characterisation of core-shell CNTs@MIPs nanocomposites and selective removal of estrone from water samples. Talanta 83(3):757–764. doi:10.1016/j.talanta.2010.10.034
Kong X, Gao R, He X, Chen L, Zhang Y (2012) Synthesis and characterization of the core-shell magnetic molecularly imprinted polymers Fe3O4@MIPs adsorbents for effective extraction and determination of sulfonamides in the poultry feed. J Chromatogr A 1245:8–16. doi:10.1016/j.chroma.2012.04.061
Prasad BB, Prasad A, Tiwari MP (2013) Multiwalled carbon nanotubes-ceramic electrode modified with substrate-selective imprinted polymer for ultra-trace detection of bovine serum albumin. Biosens Bioelectron 39(1):236–243. doi:10.1016/j.bios.2012.07.080
Yang M, Zhang Y, Lin S, Yang X, Fan Z, Yang L, Dong X (2013) Preparation of a bifunctional pyrazosulfuron-ethyl imprinted polymer with hydrophilic external layers by reversible addition-fragmentation chain transfer polymerization and its application in the sulfonylurea residue analysis. Talanta 114:143–151. doi:10.1016/j.talanta.2013.03.078
Hu Y, Li Y, Liu R, Tan W, Li G (2011) Magnetic molecularly imprinted polymer beads prepared by microwave heating for selective enrichment of beta-agonists in pork and pig liver samples. Talanta 84(2):462–470. doi:10.1016/j.talanta.2011.01.045
Sun X, He J, Cai G, Lin A, Zheng W, Liu X, Chen L, He X, Zhang Y (2010) Room temperature ionic liquid-mediated molecularly imprinted polymer monolith for the selective recognition of quinolones in pork samples. J Sep Sci 33(23–24):3786–3793. doi:10.1002/jssc.201000337
Walcarius A, Collinson MM (2009) Analytical chemistry with silica sol-gels: traditional routes to new materials for chemical analysis. Annu Rev Anal Chem 2:121–143. doi:10.1146/annurev-anchem-060908-155139
Suriyanarayanan S, Cywinski PJ, Moro AJ, Mohr GJ, Kutner W (2012) Chemosensors based on molecularly imprinted polymers. Top Curr Chem 325:165–265. doi:10.1007/128_2010_92
Malitesta C, Mazzotta E, Picca RA, Poma A, Chianella I, Piletsky SA (2012) MIP sensors–the electrochemical approach. Anal Bioanal Chem 402(5):1827–1846. doi:10.1007/s00216-011-5405-5
Lian W, Liu S, Yu J, Li J, Cui M, Xu W, Huang J (2013) Electrochemical sensor using neomycin-imprinted film as recognition element based on chitosan-silver nanoparticles/graphene-multiwalled carbon nanotubes composites modified electrode. Biosens Bioelectron 44:70–76. doi:10.1016/j.bios.2013.01.002
Shekarchizadeh H, Ensafi AA, Kadivar M (2013) Selective determination of sucrose based on electropolymerized molecularly imprinted polymer modified multiwall carbon nanotubes/glassy carbon electrode. Mater Sci Eng C 33(6):3553–3561. doi:10.1016/j.msec.2013.04.052
Minko S (2008) Grafting on solid surfaces: “Grafting to” and “Grafting from” methods. In: Stamm M (ed) Polymer surfaces and interfaces. Springer, Berlin, pp 215–234. doi:10.1007/978-3-540-73865-7_11
Lépinay S, Kham K, Millot M-C, Carbonnier B (2012) In-situ polymerized molecularly imprinted polymeric thin films used as sensing layers in surface plasmon resonance sensors: mini-review focused on 2010–2011. Chem Pap 66(5):340–351. doi:10.2478/s11696-012-0134-6
Prasad BB, Pandey I, Srivastava A, Kumar D, Tiwari MP (2013) Multiwalled carbon nanotubes-based pencil graphite electrode modified with an electrosynthesized molecularly imprinted nanofilm for electrochemical sensing of methionine enantiomers. Sens Actuators, B 176:863–874. doi:10.1016/j.snb.2012.09.050
Zhang Z, Zhang H, Hu Y, Yao S (2010) Synthesis and application of multi-walled carbon nanotubes-molecularly imprinted sol-gel composite material for on-line solid-phase extraction and high-performance liquid chromatography determination of trace Sudan IV. Anal Chim Acta 661(2):173–180. doi:10.1016/j.aca.2009.12.024
Zeng H, Wang Y, Liu X, Kong J, Nie C (2012) Preparation of molecular imprinted polymers using bi-functional monomer and bi-crosslinker for solid-phase extraction of rutin. Talanta 93:172–181. doi:10.1016/j.talanta.2012.02.008
Cai X, Li J, Zhang Z, Yang F, Dong R, Chen L (2014) Novel Pb2+ ion imprinted polymers based on ionic interaction via synergy of dual functional monomers for selective solid-phase extraction of Pb2+ in water samples. ACS Appl Mater Interfaces 6(1):305–313. doi:10.1021/am4042405
Wei Y, Qiu LH, Yu JCC, Lai EPC (2007) Molecularly imprinted solid phase extraction in a syringe needle packed with polypyrrole-encapsulated carbon nanotubes for determination of ochratoxin a in red wine. Food Sci Technol Int 13(5):375–380. doi:10.1177/1082013207085914
Zhang Z, Zhang H, Hu Y, Yang X, Yao S (2010) Novel surface molecularly imprinted material modified multi-walled carbon nanotubes as solid-phase extraction sorbent for selective extraction gallium ion from fly ash. Talanta 82(1):304–311. doi:10.1016/j.talanta.2010.04.038
Xiao D, Dramou P, Xiong N, He H, Yuan D, Dai H, Li H, He X, Peng J, Li N (2013) Preparation of molecularly imprinted polymers on the surface of magnetic carbon nanotubes with a pseudo template for rapid simultaneous extraction of four fluoroquinolones in egg samples. Analyst 138(11):3287–3296. doi:10.1039/c3an36755j
Ebrahimzadeh H, Moazzen E, Amini MM, Sadeghi O (2013) Novel ion imprinted polymer coated multiwalled carbon nanotubes as a high selective sorbent for determination of gold ions in environmental samples. Chem Eng J 215:315–321. doi:10.1016/j.cej.2012.11.031
Madrakian T, Ahmadi M, Afkhami A, Soleimani M (2013) Selective solid-phase extraction of naproxen drug from human urine samples using molecularly imprinted polymer-coated magnetic multi-walled carbon nanotubes prior to its spectrofluorometric determination. Analyst 138(16):4542–4549. doi:10.1039/c3an00686g
Zhang Z, Hu Y, Zhang H, Luo L, Yao S (2010) Layer-by-layer assembly sensitive electrochemical sensor for selectively probing L-histidine based on molecular imprinting sol-gel at functionalized indium tin oxide electrode. Biosens Bioelectron 26(2):696–702. doi:10.1016/j.bios.2010.06.062
Wu B, Wang Z, Xue Z, Zhou X, Du J, Liu X, Lu X (2012) A novel molecularly imprinted electrochemiluminescence sensor for isoniazid detection. Analyst 137(16):3644–3652. doi:10.1039/c2an35499c
Zhang M, Mullens C, Gorski W (2007) Coimmobilization of dehydrogenases and their cofactors in electrochemical biosensors. Anal Chem 79(6):2446–2450. doi:10.1021/ac061698n
Cai D, Ren L, Zhao H, Xu C, Zhang L, Yu Y, Wang H, Lan Y, Roberts MF, Chuang JH, Naughton MJ, Ren Z, Chiles TC (2010) A molecular-imprint nanosensor for ultrasensitive detection of proteins. Nat Nanotechnol 5(8):597–601. doi:10.1038/nnano.2010.114
Prasad BB, Srivastava A, Pandey I, Tiwari MP (2013) Electrochemically grown imprinted polybenzidine nanofilm on multiwalled carbon nanotubes anchored pencil graphite fibers for enantioselective micro-solid phase extraction coupled with ultratrace sensing of D- and L-methionine. J Chromatogr B Anal Technol Biomed Life Sci 912:65–74. doi:10.1016/j.jchromb.2012.10.010
Zhang D, Yu D, Zhao W, Yang Q, Kajiura H, Li Y, Zhou T, Shi G (2012) A molecularly imprinted polymer based on functionalized multiwalled carbon nanotubes for the electrochemical detection of parathion-methyl. Analyst 137(11):2629–2636. doi:10.1039/c2an35338e
Yang S, Yang R, Li G, Qu L, Li J, Yu L (2010) Nafion/multi-wall carbon nanotubes composite film coated glassy carbon electrode for sensitive determination of caffeine. J Electroanal Chem 639(1–2):77–82. doi:10.1016/j.jelechem.2009.11.025
Alizadeh T, Ganjali MR, Zare M, Norouzi P (2010) Development of a voltammetric sensor based on a molecularly imprinted polymer (MIP) for caffeine measurement. Electrochim Acta 55(5):1568–1574. doi:10.1016/j.electacta.2009.09.086
Sun JY, Huang KJ, Wei SY, Wu ZW, Ren FP (2011) A graphene-based electrochemical sensor for sensitive determination of caffeine. Colloids Surf B: Biointerfaces 84(2):421–426. doi:10.1016/j.colsurfb.2011.01.036
Santos WJR, Santhiago M, Yoshida IVP, Kubota LT (2012) Electrochemical sensor based on imprinted sol–gel and nanomaterial for determination of caffeine. Sens Actuators, B 166–167:739–745. doi:10.1016/j.snb.2012.03.051
Acknowledgments
This work was supported by the National Natural Science Foundation of China(Grant NO. 81402899), the Open Project of Key Laboratory of Modern Toxicology of the Ministry of Education (Grant NO. NMUMT201404), and the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hao Dai and Deli Xiao equally contributed to this work and should be considered co-first authors.
Rights and permissions
About this article
Cite this article
Dai, H., Xiao, D., He, H. et al. Synthesis and analytical applications of molecularly imprinted polymers on the surface of carbon nanotubes: a review. Microchim Acta 182, 893–908 (2015). https://doi.org/10.1007/s00604-014-1376-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-014-1376-5