Abstract
Aims
To determine the effect of the use of intermittently scanned continuous glucose monitoring (isCGM) on acute diabetes-related complications in adult type 1 diabetes patients.
Methods
Six hundred and forty-two adult type 1 diabetes patients with isCGM were identified from electronic health records in Siun sote region in Eastern Finland. A retrospective real-world analysis was conducted combining hospital admission and prehospital emergency service data to compare incidences of hypoglycemia requiring emergency medical support (EMS) involvement or hospital admission and diabetic ketoacidosis (DKA) before and after the start of isCGM. Data were collected from January 2015 to April 2020. Primary outcome was the rate of hypoglycemia requiring EMS involvement or hospital admission and DKA events. HbA1c was recorded at the start of isCGM and was compared with the last known HbA1c during the use of isCGM. The isCGM used in the study did not contain alarm functions.
Results
Altogether 220 hypoglycemic events were identified during the study period. Incidence rate of hypoglycemic events decreased after the start of isCGM (72 events, incidence rate 50 events/1000 person-years) compared with the time before the start (148 events, incidence rate 76 events/1000 person-years) (p = 0.043). The incidence rate of DKA decreased after the start of isCGM compared with time before isCGM use (4 and 15 events/1000 person-years, respectively; p = 0.002). The change in mean HbA1c was − 0.28% (− 3.1 mmol/mol) between baseline and the last HbA1c measurement (p < 0.001).
Conclusions
In addition to lowering HbA1c in type 1 diabetes patients, isCGM is also effective in preventing acute diabetes-related complications such as hypoglycemia requiring EMS involvement or hospital admission and DKA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hypoglycemia and diabetic ketoacidosis (DKA) are acute and potentially life-threatening complications of diabetes. When aiming to achieve tight glycemic control to avoid chronic complications, the rate of hypoglycemia is often elevated [1]. ADA workgroup on hypoglycemia defined hypoglycemia to cover all episodes of an abnormally low plasma glucose concentration that expose the individual to potential harm [2]. Hypoglycemia can be divided as mild (self-treated) or severe (requiring external help). In the Danish–British multicenter study, type 1 diabetes patients experienced on average two mild hypoglycemic events weekly and 37% reported severe hypoglycemia in a year [3].
DKA is characterized by lack of insulin action and hyperglycemia. DKA most often affects patients with type 1 diabetes. According to a systematic literature review, incidence of DKA varies from 0 to 263 per 1000 patient-years [4]. Being a severe condition, DKA is usually treated in hospital in contrast to hypoglycemia. An observational study showed that hospital admissions represent the minority of severe hypoglycemic events (37.9%), and many of the hypoglycemia episodes are treated at home by emergency medical assistance of ambulance services [5].
Achieving good glycemic control has been traditionally done by self-monitoring blood glucose (SMBG). In the past few years, new technology is introduced to replace SMBG since there is growing evidence that both real-time continuous glucose monitoring (rtCGM) [6] and intermittently scanned continuous glucose monitoring (isCGM) [7] improve treatment outcomes (in terms of lower HbA1c levels) in type 1 diabetes patients.
Real-world studies also suggest lower rates of DKA after initiation of rtCGM [8] or isCGM [9, 10]. Use of rtCGM reduced severe hypoglycemic events in RCT settings compared with self-monitoring blood glucose (SMBG) [11, 12]. However, results are contradictory whether isCGM reduces severe hypoglycemic events [9, 13] which can be due to the nature of the studies and classification of severe hypoglycemia.
Commonly, real-world studies rely on hospital admission [9] or questionnaire data [13] which can lead to possible underestimation of severe hypoglycemia and may dilute potential effect of isCGM if patients are not transported to hospital or do not recall all hypoglycemic events. To be more objective, in the present study, we combined electronic health record-based hospital admission and the use of emergency medical service data in isCGM users, aiming to investigate potential benefit of isCGM to reduce acute complications in type 1 diabetes patients.
Methods
Design and participants
The present retrospective study was carried out in the area of the Joint Municipal Authority for North Karelia Social and Health Services (Siun sote), which provides social service, healthcare services (both primary and specialized health care), and emergency service for the inhabitants (n = 166,400) living in its catchment area. FreeStyle Libre isCGM (later isCGM) was introduced to patients in 2016. From 2016 to 2020, Siun sote-funded isCGM was limited to type 1 diabetes patients fulfilling special criteria as described in our previous article [14]. These criteria included (1) suboptimal HbA1c despite optimized treatment with repeated hypoglycemic events, (2) planning to become or being pregnant, (3) fear of hypoglycemia, (4) fear of needles, leading to inability to perform SMBG, (5) end-stage renal disease and dialysis treatment, (6) inability to execute SMBG because of amputation or disability, and (7) difficulties in carrying out SMBG because of occupation. If at least one of these criteria was fulfilled, isCGM could be initiated.
Identification of patients with type 1 diabetes in the Siun sote region was done using ICD-10 codes (E10.1–E10.9), and the data were collected from the electronic health records as described in our previous article [15]. A total of 1764 patients with type 1 diabetes were identified, of whom 805 patients were domiciled in the Siun sote region, were alive at the end of 2020, and were 18 years or more at the time of the start of isCGM. Patients who were continuously using isCGM were identified from the electronic health records. The starting date of continuous use of isCGM was identified from the electronic health records, and if the use of isCGM had ended, the registered end date was also collected. Patients who started isCGM prior to the end of 2019 were included in the analysis (Fig. 1).
Information on HbA1c levels and admissions to hospital for severe hypoglycemia (ICD code E10.00) and DKA (ICD code E10.1) was collected from the electronic health records. In addition to acute complications that were retrieved from the EHR, severe hypoglycemic events requiring emergency medical services (EMS) were retrieved from the records of the ambulance service. All visits of ambulance service to the patients in the study population were manually validated by an experienced diabetologist (JMu) to identify cases of severe hypoglycemia requiring EMS service (later hypoglycemic event) and were included in the analysis if one or more following conditions were satisfied: (1) First measured plasma glucose by paramedics was 70 mg/dL (3.9 mmol/l) or less, (2) primary unconsciousness withdrawn by glucose administration, (3) glucose administration by paramedics, or (4) glucose administration by another person. If the patient was transported to hospital by emergency medical service, only one event was reported from either EHR or records of the ambulance service. Data were collected from the beginning of 2015 to the end of April 2020. EMS electronic database was altered in the end of April 2020, so it was chosen as an endpoint of the study. Self-reported hypoglycemia was not registered and thus was not included in the analysis. DKA event at the point of diabetes diagnosis was excluded from the analysis. The type of isCGM in this study was FreeStyle Libre 1 which does not have alarm functions, and it requires user action to get the real-time glucose information. FreeStyle Libre 2 system which has optional alarms was not used in this study because it was not available in Siun sote region during the study period.
Before initiation of isCGM, patients received 1‒2-hour introduction from a nurse specialized in diabetes care. No special interventions were introduced to the patients after starting isCGM, and they received standard clinical care during the time using isCGM usually consisting of 1–2 visits to a nurse or a diabetologist annually. The data from isCGM were used to optimize treatment during the visits. For this study, sensor metrics were not available and were not used in the analysis.
Due to the retrospective register-based nature of the study, the approval to conduct the study was acquired from the Institutional Review Board of Siun sote.
Statistical analyses
Descriptive statistics, such as means, medians, and standard deviations, were used to describe the data. Incidence rate, i.e., number of events divided by 1000 person-years, was used for the occurrence of hypoglycemic events and DKA before and after the start of isCGM. Baseline of the analysis was the time of isCGM initiation. Dependent samples t test was used to test difference in HbA1c values between baseline and the last measurement during the follow-up. Poisson’s regression with generalized estimating equation (GEE) was used to examine differences in incidence rates before and after initiation of isCGM. Number of events was used as an independent variable with natural logarithm of person-years as an offset variable, and subject id was used as a subject effect variable as each individual served as their own control. Also, gender, classified HbA1c, and classified age were added to the model separately with time and their interaction to see whether the time effect was different in separate classes. Subgroup analyses were done as post hoc analyses from these models when there was interaction between time and other explanatory factors. HbA1c classes < 7%, 7–9%, and > 9% (< 53, 53–75, and > 75 mmol/mol) were used in the subgroup analyses. The R language and environment for statistical computing (version 4.0.3) and IBM SPSS Statistics for Widows (version 27.0) were used in statistical analyses. P values less than 0.05 were regarded as statistically significant, and 95% confidence intervals (Cis) were used for statistics.
Results
A total of 642 patients met the inclusion criteria. Baseline characteristics are displayed in Table 1. Mean age was 47 years. HbA1c level at the start of isCGM was 8.7% (71.6 mmol/mol). Median follow-up before and after the initiation of isCGM was 2.87 and 2.45 years, respectively. Six hundred and thirty-one patients had at least one HbA1c measurement done after the start of isCGM. Median time from the start of isCGM to the last HbA1c measurement was 2.05 years. The change in mean HbA1c levels was − 0.28% (− 3.1 mmol/mol) between baseline and the last HbA1c measurement during the follow-up (p < 0.001). 8.7% of the patients had continuous subcutaneous insulin infusion (CSII) at the start of isCGM.
Altogether 213 hypoglycemic events requiring EMS were identified, and in 51 events (23.8%), patient was transported to the emergency department. In addition, 7 patients were admitted to hospital due to hypoglycemia without EMS involvement. Eighty-three patients encountered hypoglycemic event before isCGM initiation and 43 patients after the start of isCGM, and the frequencies of hypoglycemic events per patient varied from 0 to 11 and from 0 to 12, respectively. Of these 220 hypoglycemic events, 148 occurred prior to isCGM start and 72 after the start of isCGM, incidence rate being significantly lower after the start: 76 and 50 events/1000 person-years, respectively (p = 0.043) (Fig. 2).
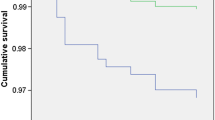
In the study cohort, a total of 35 ketoacidosis events were identified. From these, 29 occurred prior to the start of isCGM and 6 after start of isCGM representing significant reduction in incidence from 15 to 4 events/1000 person-years, respectively (p = 0.002) (Fig. 1).
When hypoglycemic and DKA events were combined as an acute complication, the reduction in these events was significant (177 vs 78 events; 90 and 54 events/1000 person-years when comparing events before and after isCGM start) (p = 0.008) (Fig. 1).
In subgroup analysis stratified by baseline HbA1c levels (HbA1c < 7%, 7–9%, and > 9% (< 53, 53–75, and > 75 mmol/mol)), the reduction in hypoglycemic events after the start of isCGM did not reach statistical significance. Patients with highest HbA1c (> 9% (> 75 mmol/mol)) showed a trend in lower rate of hypoglycemic events although it did not reach statistical significance (p = 0.064). Patients whose HbA1c level was > 9% (> 75 mmol/mol) showed significant reduction in DKA events after the start of isCGM (31 vs 5 events/1000 person-years before and after the start of isCGM, respectively) (p = 0.002) (Table 2).
Age at the start of isCGM was not correlated with the change in hypoglycemic or DKA events. Furthermore, there was no difference between genders in acute complications. If the patient had CSII at the onset of isCGM, there was not a significant change in hypoglycemic (p = 0.330) or ketoacidosis events (p = 0.670).
Discussion
In the present study, we observed a significant reduction in severe hypoglycemia requiring EMS involvement or hospital admission and DKA after the initiation of isCGM. Based on our knowledge, this is the first study combining prehospital emergency medical support and hospital admission data directly from electronic health records (instead of questionnaires) to identify severe hypoglycemic and DKA events in type 1 diabetes patients using isCGM. In addition, our study demonstrated significant reductions in both severe hypoglycemia requiring EMS or hospital admission and hospitalizations for DKA after start of isCGM in a real-world setting. Also, the significant reduction of HbA1c is in line with previous RCTs [16] and real-world studies [7, 14, 17], as well as a recent meta-analysis [18], showing that isCGM is efficient in lowering HbA1c in patients with type 1 diabetes although our findings were modest compared to the findings from Evans et al. (− 0.28% (− 3.1 mmol/mol) vs − 0.56% (− 6.1 mmol/mol)) [18] which may be due to the longer follow-up in our study.
According to our findings, the use of isCGM significantly lowers also the rate of severe hypoglycemic events requiring EMS involvement or hospital admission. In a RCT setting [16], isCGM users spend less time in hypoglycemic range, and in real-world setting, Nathanson et al. [13] found that there were less self-reported hypoglycemic events and marginally but significantly lower HbA1c when comparing isCGM users to control using SMBG in a 2-year follow-up. In a nationwide audit to clinicians in the UK [19], the use of isCGM significantly reduced absolute number of paramedic callouts and hospital admissions due to hypoglycemia when comparing 12 months before starting isCGM and seven-and-a-half-month follow-up. In addition, a real-world study showed that the number of hospital admissions due to severe hypoglycemia reduced significantly after start of isCGM without deterioration of HbA1c levels [20]. These findings are in line with our study. However, there are also studies that failed to demonstrate a positive effect of isCGM in preventing patients from severe hypoglycemia. For example, a previous prospective, open-label RCT did not found a significant difference in severe hypoglycemia between isCGM and SMBG in a 6-month follow-up [21]. A prospective observational study by Stimson et al. showed an increase in hypoglycemia admissions after initiation of isCGM during 12-month follow-up [22]. In a RCT comparing rtCGM and isCGM, the rate of self-reported severe hypoglycemic events was lower in the rtCGM group but not in the isCGM group in 6-month follow-up [23].
One possible reason for conflicting findings concerning severe hypoglycemia in isCGM users could be that patients do not recall or do not want to report severe hypoglycemic events. Pedersen-Bjergaard et al. showed that changes in EU driver’s licence legislation led to significant reduction in self-reporting severe hypoglycemic events in type 1 diabetes patients [24]. Another possible explanation is that hospital admissions represent only a part of severe hypoglycemia. A study by Villani et al. [5] found that 38% of patients requiring emergency medical service were transported to hospital which was more frequent than in our study. In our study, only 23.8% of patients requiring EMS for hypoglycemia were transported to hospital, which is similar to the proportions observed in previous studies [25, 26]. When patients are not admitted to a hospital, the possible beneficial effect of isCGM can be underestimated in studies relying on hospital admission data only.
The role of sensor alarm function has been discussed when comparing rtCGM and isCGM [23]. RtCGM devices have had alarm functions for a while, but first generation of isCGM does not provide alarms. Our study does not support an assumption that the alarm function is the key factor in reducing severe hypoglycemia because isCGM in our study was the FreeStyle Libre 1 without alarms. This might be because some patients suffer from alarm fatigue as described by Shivers et al. [27] and turn off other than mandatory hypoglycemic alarms of rtCGM.
Our study showed a significant reduction in the rate of DKA. Real-world studies have also shown that the use of both rtCGM8 and isCGM [7, 9, 28] reduces admission due to DKA. Tyndall et al. showed that the start of isCGM reduces significantly hospital admissions caused by DKA in 900 type 1 diabetes patients in a median follow-up of 245 days although the rate of DKA was small (10 events before and 2 after start of isCGM) [7]. In a nationwide cohort, France Roussel et al. found that the use of isCGM led to significantly lower incidence of DKA [9]. They identified a total of 33,165 patients with type 1 diabetes and hospitalizations because DKA fell 56.2% in follow-up of 12 months in comparison with 12 months before the start of isCGM. This effect was maintained over two-year period in a follow-up study of the original analysis [29]. In a study of 46 type 1 diabetes patients, Hayek et al. demonstrated that the use of isCGM was associated with a significant reduction in DKA [28]. These findings are in line with our data. Although the number of DKA events was small in our study, the reduction is significant. In FUTURE study by Charleer et al., the use of isCGM did not lead to significant reduction in hospital admissions due to DKA [20]. These findings conflict with our present findings. Weinstock et al. found that the frequency of DKA is increased with higher HbA1c levels [30]. In our study, HbA1c level at the start of isCGM (8.7% or 71.6 mmol/mol) was higher than in the FUTURE study where the HbA1c at the baseline was 7.8% (62 mmol/mol) which can affect the results. This effect might be due to the intensification of insulin therapy after the start of isCGM as the group with highest HbA1c levels showed the greatest drop in ketoacidosis admissions. The reduction of DKA is important because it has been shown that recurrent DKA is associated with increased mortality [31]. The dispersion of results concerning isCGM and acute diabetes-related complications can be because starting criteria for isCGM differ between different study populations, and selection bias can affect the results also in our study.
The strength of the present study is that it combines EMS and hospital electronic health records to cover both prehospital and hospital care of acute diabetes-related complications. As multiple reasons can affect studies relying only on hospital admission and/or questionnaire data concerning severe hypoglycemia, some events could be therefore missed from the analysis and the potential beneficial effect can be diluted. It is also important to point out that we were able to exclude ketoacidosis events at the point of diabetes diagnosis.
Our study has also several limitations. As being entirely register-based study, it lacks control population and the confounding effect of other procedures to reduce acute events cannot be ruled out. Due to the retrospective real-world setting, the EMS and hospital admissions data collection time is not identical before and after starting isCGM. Our study population is also relatively small compared with large register studies and due to this subgroup analysis may lack power to show beneficial effect of isCGM. As the present study was entirely EHR based, we do not have information on the exact indication of use of isCGM in each individual, exact sensor metrics such as active time of sensor use or time in range. The use of only first-generation isCGM can also be seen as a limitation as technology is moving toward rtCGM with alarm functions such as FreeStyle Libre 3. In further studies, it can be difficult to show significant drop in acute complications as more and more type 1 diabetes patients are on rtCGM or isCGM instead of SMBG already.
Conclusions
In summary, this study adds important information of isCGM use not only to lower HbA1c but also to reduce significantly severe hypoglycemia requiring EMS or hospital admission and hospitalizations of DKA. It is important to notice that reduction of these acute complications does not require alarm functions of isCGM. Further studies are needed to show the overall benefit of isCGM use in long term.
Data availability
M.D. Jyrki Mustonen is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Abbreviations
- ADA::
-
American Diabetes Association
- CSII::
-
Continuous subcutaneous insulin infusion
- DKA::
-
Diabetic ketoacidosis
- EMS::
-
Emergency medical support
- isCGM::
-
Intermittently scanned continuous glucose monitoring
- rtCGM::
-
Real-time continuous glucose monitoring
- SMBG::
-
Self-monitoring blood glucose
References
Cryer PE (2014) Glycemic goals in diabetes: trade-off between glycemic control and iatrogenic hypoglycemia. Diabetes 63:2188–2195. https://doi.org/10.2337/db14-0059
Workgroup on Hypoglycemia, American Diabetes Association (2005) Defining and reporting hypoglycemia in diabetes: a report from the American Diabetes Association Workgroup on Hypoglycemia. Diabetes Care 28(5):1245–9. https://doi.org/10.2337/diacare.28.5.1245
Pedersen-Bjergaard U, Pramming S, Heller SR et al (2004) Severe hypoglycaemia in 1076 adult patients with type 1 diabetes: influence of risk markers and selection. Diabetes Metab Res Rev 20(6):479–86. https://doi.org/10.1002/dmrr.482
Farsani SF, Brodovicz K, Soleymanlou N, Marquard J et al (2017) Incidence and prevalence of diabetic ketoacidosis (DKA) among adults with type 1 diabetes mellitus (T1D): a systematic literature review. BMJ Open 7(7):e016587–e016587. https://doi.org/10.1136/bmjopen-2017-016587
Villani M, Nanayakkara N, Ranasinha S et al (2016) Utilisation of emergency medical services for severe hypoglycaemia: an unrecognised health care burden. J Diabetes Complicat 30(6):1081–6. https://doi.org/10.1016/j.jdiacomp.2016.04.015
Lind M, Polonsky W, Hirsch IB et al (2017) Continuous glucose monitoring vs conventional therapy for glycemic control in adults with type 1 diabetes treated with multiple daily insulin injections: the gold randomized clinical trial. JAMA 317(4):379–387. https://doi.org/10.1001/jama.2016.19976
Tyndall V, Stimson RH, Zammitt NN et al (2019) Marked improvement in HbA1c following commencement of flash glucose monitoring in people with type 1 diabetes. Diabetologia 62(8):1349–1356. https://doi.org/10.1007/s00125-019-4894-1
Charleer S, Mathieu C, Nobels F, RESCUE Trial Investigators et al (2018) Effect of continuous glucose monitoring on glycemic control, acute admissions, and quality of life: a real-world study. J Clin Endocrinol Metab 103(3):1224–1232. https://doi.org/10.1210/jc.2017-02498
Roussel R, Riveline JP, Vicaut E et al (2021) Important drop rate of acute diabetes complications in people with type 1 or type 2 diabetes after initiation of flash glucose monitoring in france: the RELIEF study. Diabetes Care 44(6):1368–76. https://doi.org/10.2337/dc20-1690
Tsur A, Cahn A, Israel M et al (2021) Impact of flash glucose monitoring on glucose control and hospitalization in type 1 diabetes: a nationwide cohort study. Diabetes Metab Res Rev 37(1):e3355. https://doi.org/10.1002/dmrr.3355
van Beers CA, DeVries JH, Kleijer SJ et al (2016) Continuous glucose monitoring for patients with type 1 diabetes and impaired awareness of hypoglycaemia (IN CONTROL): a randomised, open-label, crossover trial. Lancet Diabetes Endocrinol 4(11):893–902. https://doi.org/10.1016/S2213-8587(16)30193-0
Beck RW, Riddlesworth T, Ruedy K, DIAMOND Study Group et al (2017) Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: the diamond randomized clinical trial. JAMA 317(4):371–378. https://doi.org/10.1001/jama.2016.19975
Nathanson D, Svensson A-M, Miftaraj M et al (2021) Effect of flash glucose monitoring in adults with type 1 diabetes: a nationwide, longitudinal observational study of 14,372 flash users compared with 7691 glucose sensor naive controls. Diabetologia 64(7):1595–1603. https://doi.org/10.1007/s00125-021-05437-z
Mustonen J, Rautiainen P, Lamidi ML et al (2022) Marked improvement in A1C levels after initiation of intermittently scanned continuous glucose monitoring is maintained over 4 years in patients with type 1 diabetes. Diabetes Spectr 35(4):469–475. https://doi.org/10.2337/ds21-0087
Kekäläinen P, Tirkkonen H, Laatikainen T (2016) How are metabolic control targets of patients with type 1 diabetes mellitus achieved in daily practice in the area with high diabetes prevalence? Diabetes Res Clin Pract 115:9–16. https://doi.org/10.1016/j.diabres.2016.03.005
Bolinder J, Antuna R, Geelhoed-Duijvestijn P et al (2016) Novel glucose-sensing technology and hypoglycaemia in type 1 diabetes: a multicentre, non-masked, randomised controlled trial. Lancet 388:2254–2263. https://doi.org/10.1016/S0140-6736(16)31535-5
Paris I, Henry C, Pirard F et al (2018) The new FreeStyle libre flash glucose monitoring system improves the glycemic control in a cohort of people with type 1 diabetes followed in real-life conditions over a period of one year. Endocrinol Diabetes Metab 1:e00023. https://doi.org/10.1002/edm2.23
Evans M, Welsh Z, Ells S et al (2020) The impact of flash glucose monitoring on glycaemic control as measured by HbA1c: a meta-analysis of clinical trials and real-world observational studies. Diabetes Ther 11(1):83–95. https://doi.org/10.1007/s13300-019-00720-0. (Epub 2019 Oct 31)
Deshmukh H, Wilmot EG, Gregory R, Barnes D et al (2020) Effect of flash glucose monitoring on glycemic control, hypoglycemia, diabetes-related distress, and resource utilization in the association of british clinical diabetologists (Abcd) nationwide audit. Diabetes Care 43(9):2153–2160. https://doi.org/10.2337/dc20-0738
Charleer S, De Block C, Van Huffel L et al (2020) Quality of life and glucose control after 1 year of nationwide reimbursement of intermittently scanned continuous glucose monitoring in adults living with type 1 diabetes (FUTURE): a prospective observational real-world cohort study. Diabetes Care 43(2):389–397. https://doi.org/10.2337/dc19-1610
Davis TME, Dwyer P, England M et al (2020) Efficacy of intermittently scanned continuous glucose monitoring in the prevention of recurrent severe hypoglycemia. Diabetes Technol Ther 22(5):367–373. https://doi.org/10.1089/dia.2019.0331
Stimson RH, Dover AR, Ritchie SA et al (2020) HbA1c response and hospital admissions following commencement of flash glucose monitoring in adults with type 1 diabetes. BMJ Open Diabetes Res Care 8(1):e001292. https://doi.org/10.1136/bmjdrc-2020-001292
Visser MM, Charleer S, Fieuws S et al (2021) Comparing real-time and intermittently scanned continuous glucose monitoring in adults with type 1 diabetes (ALERTT1): a 6-month, prospective, multicentre, randomised controlled trial. Lancet 397(10291):2275–2283. https://doi.org/10.1016/S0140-6736(21)00789-3. (Epub 2021 Jun 2)
Pedersen-Bjergaard U, Færch L, Allingbjerg ML et al (2015) The influence of new European Union driver’s license legislation on reporting of severe hypoglycemia by patients with type 1 diabetes. Diabetes Care 38(1):29–33. https://doi.org/10.2337/dc14-1417. (Epub 2014 Oct 6 PMID: 25288675)
Wang H, Donnan PT, Leese CJ, Duncan E et al (2017) Temporal changes in frequency of severe hypoglycemia treated by emergency medical services in types 1 and 2 diabetes: a population-based data-linkage cohort study. Clin Diabetes Endocrinol 15(3):7. https://doi.org/10.1186/s40842-017-0045-0
Leese GP, Wang J, Broomhall J, Kelly P et al (2003) Frequency of severe hypoglycemia requiring emergency treatment in type 1 and type 2 diabetes: a population-based study of health service resource use. Diabetes Care 26(4):1176–1180. https://doi.org/10.2337/diacare.26.4.1176
Shivers JP, Mackowiak L, Anhalt H et al (2013) Turn it off: diabetes device alarm fatigue considerations for the present and the future. J Diabetes Sci Technol 7(3):789–94. https://doi.org/10.1177/193229681300700324
Hayek Al, Al Dawish MA (2021) frequency of diabetic ketoacidosis in patients with type 1 diabetes using FreeStyle libre: a retrospective chart review. Adv Ther 38(6):3314–3324. https://doi.org/10.1007/s12325-021-01765-z
Riveline JP, Roussel R, Vicaut E (2022) Reduced rate of acute diabetes events with flash glucose monitoring is sustained for 2 years after initiation: extended outcomes from the relief study. Diabetes Technol Ther. https://doi.org/10.1089/dia.2022.0085. (Epub ahead of print)
Weinstock RS, Xing D, Maahs DM, Michels A, for the T1D Exchange Clinic Network et al (2013) Severe Hypoglycemia and diabetic ketoacidosis in adults with type 1 diabetes: results from the T1D exchange clinic registry. J Clin Endocrinol Metabol 98(8):3411–3419. https://doi.org/10.1210/jc.2013-1589
Gibb FW, Teoh WL, Graham J et al (2016) Risk of death following admission to a UK hospital with diabetic ketoacidosis. Diabetologia 59(10):2082–7. https://doi.org/10.1007/s00125-016-4034-0. (Epub 2016 Jul 11)
Acknowledgements
None.
Funding
Open access funding provided by University of Eastern Finland (UEF) including Kuopio University Hospital. The funding for this study came from the Siun sote fund and was partly funded by the Research Committee of the Kuopio University Hospital Catchment Area for the State Research Funding (project QCARE, Joensuu, Finland), the Strategic Research Council at the Academy of Finland (project IMPRO, 312703 and 336325), and the Finnish Diabetes Association. No funding from Abbott Diabetes was included.
Author information
Authors and Affiliations
Contributions
JMu, PR, JMa, and TL planned the study design, JMu edited the data from the electronic database, and ML researched data and contributed to the statistical methods and results. PL verified the analytical methods. JMu wrote and edited various versions of the manuscript. All authors participated in the interpretation of the data, contributed to the discussion, and reviewed and commented the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Janne Martikainen is a founding partner of ESiOR. This company was not involved in carrying out this research. The other authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies using human or animal subjects performed by any of the authors.
Informed consent statement
According to the Finnish legislation, register authorities give permissions to use register data including sensitive individual information (e.g., health data) to study specified research questions to named individuals who have signed a pledge of secrecy. The joint municipal authority for North Karelia Social and Health services (Siun sote) has given a permission for the use of the data. The Finnish legislation do not require ethics approcla nor informed consent for register-based research when study is solely based on registers (involving no contact with the study subjects) and the study is considered to be of public health importance.
Additional information
Managed by Massimo Federici.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mustonen, J., Rautiainen, P., Lamidi, ML. et al. The use of isCGM leads to marked reduction in severe hypoglycemia requiring emergency medical service or hospital admission and diabetic ketoacidosis in adult type 1 diabetes patients. Acta Diabetol 60, 891–898 (2023). https://doi.org/10.1007/s00592-023-02079-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-023-02079-y