Abstract
Aims
Food addiction (FA) is conceptualized as a behavioral pattern that is similar in some ways to addictions to alcohol and other substances. This disorder has not been well studied among patients with type 2 diabetes (T2DM). We aimed to analyze if there is any relationship between FA and clinical or psychological variables among patients with T2DM.
Methods
Three hundred patients with T2DM were analyzed cross-sectionally. Participants were evaluated for the presence of FA by completing the Yale Food Addiction Scale 2.0 questionnaire.
Results
29.3% of patients screened positive for FA. Patients with FA had a greater BMI (33.41 ± 7.5 vs. 31.6 ± 5.9 kg/m2; p = 0.04). HbA1c was higher among individuals with FA (7.9 ± 4.4 vs. 7.6 ± 1.4%, p = 0.008). The proportion of subjects with diabetic retinopathy, neuropathy and nephropathy was greater among patients with criteria for FA compared with patients without this condition (25% vs. 13.2%, 29.5% vs. 21.8% and 32% vs. 22.3%; p = 0.03, p = 0.05 and p = 0.05, respectively). The percentage of patients with FA with significant depressive symptoms was also greater (36.4% vs. 18.5%; p = 0.002).
Conclusions
The presence of FA among T2DM patients implied a worse glycaemic control. Microvascular complications and depressive symptoms were higher among these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type 2 diabetes (T2DM) has become a global epidemic, with its associated high morbidity and mortality rates [1]. On the other hand, the prevalence of obesity has increased worldwide during the past few decades, and it is closely related to T2DM [1, 2]. Both, T2DM and obesity, have documented strong associations with psychiatric diseases, such as depression and eating disorders [3,4,5]. The co-occurrence of a psychiatric condition among subjects with diabetes and/ or obesity has been related to higher incidences of unhealthy behaviours (tobacco abuse, lack of physical activity or unhealthy diets), poor metabolic outcomes and higher risk of complications [6,7,8,9].
In the 1950s, Randolph first defined the term food addiction (FA) in the scientific literature, which referred to individuals could not control food consumption rationally and had developed tolerance, withdrawal, cravings, and other behavioural features that were similar to addictions to other substances, such as alcohol or cocaine [10]. The behaviours are generally characterized by the high consumption of highly palatable foods, such as sugary, salty, fatty and processed foods. Since then, interest in the concepts of food addiction is growing progressively, and there is increasing evidence of similarities between substance dependence and excessive food intake [10,11,12,13].
Currently, the most generally accepted tool for assessing FA is the Yale Food Addiction Scale 2.0 (YFAS 2.0). The YFAS 2.0 was introduced as a valid and a reliable self-reported measurement for the assessment of FA symptomatology paralleling the DSM-V Substance Disorder criteria [11,12,13,14,15,16].
Moreover, the prevalence of FA ranges from 5.4 to 10% in general population to 15.2–19.6% in obese treatment-seeking adults, to 42% in bariatric surgery candidates and up to 57% in subjects with obesity and a concomitant binge eating disorder [17,18,19,20,21,22,23]. However, data regarding prevalence rates among subjects with T2DM are scarce and may vary from 8.6 to 70.7% [23,24,25]. Besides, to our knowledge, studies assessing the impact of FA on different outcomes among individuals with T2DM have been focused mainly on psychological parameters, such as anxiety, depressive disorder or quality of life. However, data regarding the effects of FA among subjects with T2DM on metabolic control or diabetic complications are lacking.
The aim of our study was to investigate the frequency and the severity of FA among patients with T2DM. Also, we wanted to analyse whether there were any differences in clinical, biochemical and psychological outcomes among patients with FA and diabetes compared with patients with T2DM without this condition.
Subjects, materials and methods
Participants
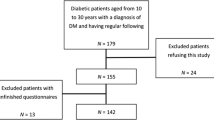
A cross-sectional study was designed where three hundred individuals with a diagnosis of T2DM were recruited randomly, using serial selection from the Endocrine Department of a tertiary centre in Palma de Mallorca (Balearic Islands, Spain).
Patients were excluded from the study if they had type 1 diabetes or gestational diabetes, were individuals with a diagnosis of T2DM in the preceding 12 months, were T2DM patients in pregnancy or lactation, had alcohol or substance dependence or were individuals who had an uncontrolled medical condition other than diabetes. Also, all patients included in the study had both a blood test in the prior three months and a funduscopic exam in the last twelve months. Written informed consent was obtained from all patients prior to study participation. The study was approved by the Ethics Committee of the hospital.
Assessment of food addiction symptomatology
The Yale Food Addiction Scale 2.0 (YFAS 2.0) is a revision of the original Yale Food Addiction Scale and measures addictive eating behaviors that reflect the updated DSM-5 criteria for a substance use disorder. In this new version of the YFAS, the threshold for meeting criteria for addictive eating lowered, similar to the threshold for meeting DSM-5 criteria for a substance use disorder, and four additional criteria were added to the YFAS 2.0. The Spanish version of the YFAS 2.0 was used to assess food addiction symptomatology [15]. The validation of the original English version shows an adequate internal reliability and good convergent and incremental validity in predicting binge eating [16, 26]. It consists of a self-report 35-item scale designed to measure FA symptoms over the past 12 months using a Likert scale. Participants answered 35 questions based on a 0–6 scale, indicating frequency of which they experienced symptoms that correlated with food addiction. The frequency of experienced symptoms is stratified in the test according to the following numbers on the Likert scale: 0, less than once a month; (1) once a month; (2) 2–3 times a month; (3) once a week; (4) 2–3 times a week; (5) 4–6 times a week; and (6) every day. The YFAS 2.0 measure focuses on operationalizing addictive eating behaviors, which parallel the symptoms of substance dependence criteria listed in the DSM-V. YFAS 2.0 has two scoring options: one is FA diagnosis (dichotomous food addiction classification), and the other is the food addiction symptom score severity. The 11 food criteria include: (1) substance taken in larger amount and for longer period than intended: (2) persistent desire or repeated unsuccessful attempt to quit: (3) much time/activity to obtain, use and recover; (4) important social, occupational or recreation activities given up or reduced; (5) continued use despite knowledge of adverse consequences; (6) tolerance (marked increased in amount and significant decrease in effect); (7) characteristic withdrawal symptoms: (8) continued use despite social or interpersonal problems; (9) failure to fulfill major role obligations; (10) use in physical hazardous situations; and (11) craving, or a strong desire to urge or use. The FA diagnosis criteria are met when individuals endorse more than two symptoms over the past year, as well as meeting the threshold for at least one of the two symptoms of “clinical significance”, defined as impairment as a result of their behaviors. If individuals meet at least two symptoms but do not meet criteria for clinical significance, they are not diagnosed with FA. Moreover, there is also a specification of the levels of severity: mild (presence of 2–3 symptoms), moderate (presence of 4–5 symptoms) and severe (presence of 6 or more symptoms). YFAS is a reliable and valid scale for both bariatric surgery patients and non-clinical obese samples [12].
Assessment for depressive symptoms
To identify depressive syndrome, all participating patients rated the presence and severity of depressive symptoms by using the Spanish version of the Beck Depression Inventory (BDI), a 21-item questionnaire that assesses mood over the previous month [26]. Total scores range from 0 to 63, with higher scores indicating greater symptoms of depression. The Beck Depression Inventory has been widely used as a screening tool for major depression in the general population. In this setting, a cutoff score equal to or greater than thirteen is indicative of significant depression. However, among people with diabetes, a cutoff score equal to or greater than sixteen for the entire 21-item measure exhibited the best balance between sensitivity and positive predictive value when prediction values were extrapolated to a diabetic population with a depression prevalence rate of 20%. This cutoff would be able to capture more than seventy percent of the subjects diagnosed with major depression yet provide more than seventy percent of certainty that a person screening positive actually has this condition. Thus, a score in BDI ≥ 16 was considered as positive for significant symptoms of depression among subjects with diabetes [26, 27].
Sociodemographic and clinical parameters
Demographic information was gathered during the eligibility period and included age, gender and ethnicity. Other features of diabetes such as diabetes duration, current treatment for T2DM, presence of diabetic complications (coronary heart disease, vasculopathy, cerebrovascular disease, neuropathy, retinopathy, nephropathy and diabetic foot), frequency of hypoglycaemias, admissions due to acute complications related to diabetes or for any other reason during the previous year, and other cardiovascular risk factors (tobacco use, alcohol abuse, hypertension and dyslipidemia) were recorded by clinical interview. Furthermore, the revision of clinical reports was also performed in order to have additional information.
Anthropometric parameters
Height and weight were measured while each participant was wearing indoor clothing without shoes. Body mass index (BMI) was calculated as weight divided by height squared. The World Health Organisation (WHO) and the National Institute for Health and Clinical Excellence (NICE) define the following cut-points for BMI: < 18.5 kg/m2 underweight, 18.5–24.9 kg/m2 healthy weight, 25.0–29.9 kg/m2 overweight, and ≥ 30 kg/m2 obese [28]. Waist circumference was measured at the midpoint between the lowest rib and the iliac crest.
Metabolic profile
Blood samples were drawn following the visit for the determination of these variables: blood count, coagulation, fasting glucose, glycated hemoglobin (HbA1c), total cholesterol, HDL cholesterol, LDL-cholesterol, triglycerides, plasma creatinine, hepatic profile and thyroid profile. All measurements were taken after an overnight fast (at least 8 h fasting). Early morning urine samples were collected to determine random albumin-to-creatinine ratio.
In terms of glycaemic control, patients were categorized based on the latest consensus of the American Diabetes Association and the European Association for the Study of Diabetes [29].
Statistical analyses
Initial analyses were descriptive and included calculation of mean and standard deviation (SD) for continuous variables and as frequencies for categorical variables. The distribution of the sample was analyzed by the Kolmogorov–Smirnov test and normality plots. Comparison between the two groups was analyzed by an unpaired Student’s test for variables with a normal distribution and the Wilcoxon test for the variables without a normal distribution. A p value < 0.05 on the two-tailed test was considered to indicate statistical significance. Data were analyzed using SPSS version 16 statistical software (SPSS Inc., Chicago, IL, USA).
Results
According to the YFAS 2.0, 88 patients with T2DM met criteria for FA, in other words, 29.3% of the sample scored positive for FA. When taking into account the severity of FA among these patients, 43.3% (38/88) had mild FA, 20.5% (18/88) scored for moderate FA, and the remaining 36.5% (32/88) had criteria for severe FA.
Patients with criteria for FA had a higher BMI compared with the group without FA (33.41 ± 7.5 vs. 31.6 ± 5.9 kg/m2; p = 0.04). Moreover, when we divided individuals with FA depending on the severity of symptoms, BMI increased as the degree of FA did (33.2 ± 6.6 kg/m2 for mild food addiction and 39.4 ± 9.1 kg/m2 for severe FA; p = 0.04). However, we could not find any differences between the two groups regarding waist circumference; this anthropometrical parameter was greater among patients with severe food addiction compared with subjects without any abnormal eating pattern (108.4 ± 11.7 vs. 114.1 ± 8.8 cm: p = 0.018). No significant differences were seen in any other of the clinical variables assessed.
When assessing glycaemic parameters, patients with FA had greater fasting plasma glucose levels compared with patients without FA (164.6 ± 72.8 vs. 140.5 ± 38.2 mg/dL; p = 0.034). Also, HbA1c was higher among individuals with FA compared with T2DM patients without FA (7.9 ± 4.4 vs. 7.6 ± 1.4%; p = 0.008). Furthermore, the worse the FA severity, the higher the HbA1c was (7.5 ± 1.3% for mild FA vs. 7.9 ± 1.3% for severe FA; p = 0.01). Also, albumin-to-creatinine ratio was higher among people with any degree of FA compared with patients with diabetes and without FA (217.5 ± 158.6 vs. 138.1 ± 189.8 mg/dL; p = 0.01). However, lipid profile, renal and hepatic functions were comparable between the two groups. These data are summarized in Table 1.
When we assessed for acute complications related to T2DM, the proportion of patients who had one or more clinically significant hypoglycaemias during the last year was lower among individuals with FA than people without FA (8% vs. 19%; p = 0.022). However, no differences were seen between the two groups either with admissions related to diabetes or admissions for any other disease during the last year.
When taking into account microvascular complications related to T2DM, the proportion of patients with diabetic retinopathy, neuropathy and nephropathy was greater among patients with criteria for FA compared with patients without this condition (25% vs. 13.2%, 29.5% vs. 21.8% and 32% vs. 22.3%; p = 0.03, p = 0.05 and p = 0.05, respectively). Furthermore, the frequency of patients with any degree of diabetic retinopathy was greater among individuals with moderate to severe FA compared with patients without FA (33.3% and 34.4% vs. 13.2%; p = 0.03). Besides, the proportion of patients with diabetic neuropathy was also greater among patients with moderate and severe FA compared with individuals without FA (44.4% and 40.6% vs. 21.8%; p = 0.05). Also, the percentage of people with T2DM with nephropathy was higher among patients with moderate and severe FA in comparison with people without this condition (25% and 38.9% vs. 22.3%; p = 0.05). No differences were seen when comparing individuals without FA and people with mild FA. However, we could not find any differences between subjects with criteria for FA and patients with diabetes and without FA regarding other cardiovascular risk factors or macrovascular complications. These data are summarized in Table 2.
On the other hand, the proportion of people with T2DM with lower adherence to healthy dietary habits was higher among those who had FA compared with individuals without food addiction (21.6% vs. 56.4%; p < 0.0001). However, no differences regarding the adherence to a completion of more than 150 min of exercise per week or the proportion of subjects on an insulin regimen were seen between the two groups.
When taking into account psychological variables, the frequency of patients with a depressive disorder was significantly greater among subjects with FA compared with individuals without FA (36.4% vs. 18.5%; p = 0.002). Conversely, the proportion of patients taking antidepressants was greater among those with FA compared with individuals without FA (29.5% vs. 17.5%; p = 0.03). These data are summarized in Table 3.
Figure 1 is a representation of all significant differences between subjects with and without FA.
Discussion
The frequency of FA among our patients was within the range of prevalence previously reported among subjects with T2DM, which may vary from 8.6 to 70.2% [23, 24, 30]. These prevalence rates compare to 5–11.4% reported for general population and 19.9% weighted mean prevalence [25]. These prevalence rates of FA among subjects with T2DM are more comparable to those reported for patients with obesity than other subgroups [20, 21]. However, worldwide estimates of FA prevalence among subjects with T2DM appear to vary among ethnic subgroups, region and participants [23, 24].
Subjects with T2DM and FA had a greater BMI and waist circumference compared with subjects without FA. This fact could reflect the difficulty of these patients to adhere to healthy dietary habits as is reflected in our study. Patients with diabetes need to restraint foods high in sugar in order to achieve a proper glycaemic control, but, actually, this type of foods has some biochemical properties with the potential to cause this addictive process [9, 10, 12, 31]. Therefore, these addiction-related problematic behaviours are, in some cases, sufficient to cause clinically significant consequences, such as an increase in weight [29]. Actually, FA is one risk factor for obesity and it might affect outcomes of weight loss programs [32]. On the other hand, and as suggested in previous studies, BMI may be a consequence of disinhibited eating behaviour rather than a factor related to FA [15, 33]. Moreover, FA has previously been associated with increased BMI in people with T2DM [34,35,36].
Glycaemic control was worse among subjects with FA, possibly explained in part by a lack of adherence to healthy dietary habits. We also found that microvascular complications were more frequent among patients with FA, with no increase in the frequency of macrovascular disease. It is well established that a poor glycaemic control exerts a greater negative effect on microvascular disease compared with macrovascular complications, where other cardiovascular risk factors, such as dyslipidaemia and hypertension, are more relevant [37]. Furthermore, the frequency of hypoglycaemias was lower, probably influenced by the amount of carbohydrates consumed per day. To date, no studies assessing these variables have been conducted.
When taking into account psychological variables, both depressive disorder and the use of antidepressant therapy were more prevalent among subjects with FA. The bidirectional relationship is well established between T2DM and depressive disorder. In fact, individuals with T2DM are twice as likely to suffer from depression compared with people without this condition [7, 34, 38–40]. Moreover, FA is more frequent among subjects with some psychologic disorder, such as depressive disorder, anxiety, attention deficit and hyperactivity disorder or posttraumatic stress. It has been hypothesized that the uncontrolled intake of sugar could lead to either neurobiological alterations in the reward syndrome, motivation or impulse control, the same way as drug abuse does [12, 24, 41]. The presence of FA among subjects with T2DM could worsen depressive symptomatology due to the frustration and sense of guilty secondary to the difficulty to achieve a proper glycaemic control due to a lack of adherence to a healthy diet. Besides, not only a poor glycaemic control but the presence of depressive symptoms among these patients could lead to an increase in the frequency of complications related to diabetes. Also, these patients with food addiction had a higher BMI, and it is known that obesity is one of the strongest predictors of depression in patients with T2DM, with risk of depression being 20–50% higher among subjects with obesity than normal weight individuals [23, 42].
The present study evidenced new and interesting findings, but some limitations should be taken into account. As a cross-sectional study, we cannot make any definitive conclusions regarding a causal relationship between either metabolic control, complications related to diabetes or depression. We used self-report assessments that may have questionable reliability and validity, and be limited in determining a clinical diagnosis. However, despite acknowledging these limitations, previous research has produced valid results using a similar methodology. Also, we did not assess the presence or absence of factors indicating eating disorders or binge eating disorder. It has been reported that higher FA scores are associated with bingeing eating-disorders subtypes and with increased severity of other abnormal eating patterns and psychopathology [15]. Also, we could not observe a gradual increase in many of the parameters included in the study because the number of subjects with criteria for moderate food addiction was small and, therefore, could interfere with the results. Finally, we could not generalize our results to other populations, because all our patients were Caucasian.
We consider these findings stimulate and justify the routine screening of FA among patients with T2DM, implementing standardized self-report tools, such as YFAS 2.0. Furthermore, it could be more productive to focus treatments on FA, rather than attempting to treat the potential consequences of FA among subjects with T2DM [11, 13, 34].
Our results emphasize for the first time that food addiction is very prevalent among patients with T2DM, and it may exert negative effects on metabolic control and clinical outcomes. Therefore, it is extremely important to identify this psychological condition in order to treat it promptly. Future research should also continue to explore both the relationship between all these variables and FA and potential targets for treatment.
References
Cho NH, Shaw JE, Karuranga S et al (2018) IDF diabetes atlas: global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract 138:271–281
World Health Organization (2005) Revised global burden of disease (GBD) 2002 estimates. https://www.who.int/healthinfo/bodgbd2002revised/en/index.html
Roy T, Lloyd CE (2012) Epidemiology of depression and diabetes: a systematic review. J Affect Disord 142(Suppl 1):S8–S21
Meneghini LF, Spadola J, Florez H (2006) Prevalence and associations of binge eating disorder in a multiethnic population with type 2 diabetes. Diabetes Care 29:2760
Allison KC, Wadden TA, Sarwer DB (2006) Night eating syndrome and binge eating disorder among persons seeking bariatric surgery: prevalence and related features. Obesity 14:77S–82S
Mezuk B, Eaton WW, Albrecht S, Golden SH (2013) Depression and type 2 diabetes over the lifespan. Diabetes Care 31:2383–2390
Whitworth SR, Bruce DG, Starkstein SE, Davis WA, Davis TME, Bucks RS (2016) Lifetime depression and anxiety increase prevalent psychological symptoms and worsen glycaemic control in type 2 diabetes: the Fremantle Diabetes Study Phase II. Diabetes Res Clin Pract 122:190–197
Nicolau J, Simó R, Sanchís P et al (2015) Eating disorders are frequent among type 2 diabetic patients and are associated with worse metabolic and psychological outcomes: results from a cross-sectional study in primary and secondary care settings. Acta Diabetol 52:1037–1044
Chien I-C, Lin C-H (2014) Increased risk of diabetes in patients with anxiety disorders: a population-based study. J Psychosom Res 86:47–52
Shriner R, Gold M (2014) Food addiction: an evolving nonlinear science. Nutrients 6:5370–5391
Gearhardt AN, Corbin WR, Brownell KD (2009) Food addiction: an examination of the diagnostic criteria for dependence. J Addict Med 3:1–7
Blumenthal DM, Gold MS (2010) Neurobiology of food addiction. Curr Opin Clin Nutr Metab Care 13:359–365
Gearhardt AN, Davis C, Kuschner R, Brownell KD (2011) The addiction potential of hyperpalatable foods. Curr Drug Abuse Rev 4:140–145
Meule A, Gearhardt AN (2014) Five years of the Yale Food Addiction Scale: taking stock and moving forward. Curr Addict Rep 2014(1):193–205
Granero R, Hilker I, Agüera Z et al (2014) Food addiction in a Spanish sample of eating disorders: DSM-5 diagnostic subtype differentiation and validation data. Eur Eat Disord Rev 22:389–396
Davis R, Olmsted MP, Rockert W (1990) Brief group psychoeductation for bulimia nervosa: assessing the clinical significance of change. J Consult Clin Psych 58:882–885
Meule A, Heckel D, Kübler A (2012) Factor structure and item analysis of the Yale Food Addiction Scale in obese candidates for bariatric surgery. Eur Eat Disord Rev 20:419–422
Meule A, Hermann T, Kübler A (2015) Food addiction in overweight and obese adolescents seeking weight-loss treatment. Eur Eat Disord Rev 23:193–198
Clark SM, Saules KK (2013) Validation of the Food Addiction Scale among weight-loss surgery population. Eat Behav 14:216–219
Imperatori C, Innamorati M, Contardi A et al (2014) The association among food addiction, binge eating severity and psychopathology in obese and overweight patient attending low-energy-diet therapy. Compr Psychiatry 55:1358–1362
Eichen DM, Lent MR, Goldbacher E, Foster GD (2013) Exploration of food addiction in overweight and obese treatment-seeking adults. Appetite 67:22–24
Pursey KM, Stanwell P, Gearhardt AN, Collins CE, Burrows TL (2014) The prevalence of food addiction as assessed by the Yale Food Addiction Scale: a systematic review. Nutrients 21:4552–4590
Lee-Raymond K, Lovell GP (2016) Food addiction associations with psychological distress among people with type 2 diabetes. J Diabetes Complicat 30:651–656
Yang F, Liu A, Li Y et al (2017) Food addiction in patients with newly diagnosed type 2 diabetes in northeast China. Front Endocrinol (Lausanne) 8:218
Brunault P, Ballon N, Gaillard P, Réveillère C, Courtois T (2014) Validation of the French version of the Yale Food Addiction Scale: an examination of its factor structure, reliability, and construct validity in a nonclinical sample. Can J Psychiatry 59:276–284
Beck AT, Steer RA, Brown GA (1996) Beck depression inventory-II (BDI-II). Pearson, Oxford
Lustman PJ, Clouse RE, Griffith LS et al (1997) Screening for depression in diabetes using the beck depression inventory. Psychosom Med 59:24–31
World Health Organization (2014) Obesity and overweight. Fact sheet no 311. Update August 2014
Davies MJ, D'Alessio DA, Fradkin J et al (2018) Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 61:2461–2498
Lee-Raymond K, Lovell GP (2015) Food addiction symptomatology, impulsivity, mood and body mass index in people with type 2 diabetes. Appetite 95:383–389
Cathelain S, Brunault P, Ballon N, Réveillère C, Courtois R (2016) L’addiction à l’alimentation: définition, mesure et limites du concept. Facteurs associés et implications cliniques et thérapeutiques. Presse Med 45:1154–1163
Pedram P, Wadden D, Amini P, Gulliver W, Randell E, Cahill F (2013) Food addiction: its prevalence and significant association with obesity in the general population. PLoS ONE 8:1–6
Agüera Z, Wolz I, Sánchez IM et al (2016) Adicción a la comida: un constructo controvertido. C Med Psicosom 117:17–30
Raymond KL, Kannis-Dymand L, Lovell GP (2016) A graduated food addiction classification approach significantly differentiates obesity among people with type 2 diabetes. J Health Psychol 23:1–9
Burmeister JM, Hinman N, Koball A, Hoffman DA, Carels RA (2013) Food addiction in adults seeking weight loss treatment. Implications for psychosocial health and weight loss. Appetite 60:103–110
Lerma-Cabrera JM, Carvajal F, Lopez-Legarrea P (2016) Food addiction as a new piece of the obesity framework. Nutr J 15:124–126
Zheng Y, Ley SH, Hu FB (2018) Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol 14:88–98
Collins MM, Corcorant P, Perry IJ (2008) Anxiety and depression symptoms in patients with diabetes. Diabet Med 26:153–161
Tovilla-Zárate C, Juárez- Rojo I, Jiménez YP, Jiménez MA, Vázquez S, Bermúdez-Ocaña D (2011) Prevalence of anxiety and depression among outpatients with type 2 diabetes in the Mexican population. PLoS ONE 7:5
Nicolau J, Simó R, Sanchís P et al (2016) Prevalence and clinical correlators of undiagnosed significant depressive symptoms among individuals with type 2 diabetes in a Mediterranean population. Exp Clin Endocrinol Diabetes 124:1–7
Dagher A (2009) The neurobiology of appetite: hunger as addiction. Int J Obes 33(Suppl 2):S30–S33
Svenningsson I, Björkelund C, Marklund B, Gedda B (2012) Anxiety and depression in obese and normal-weight individuals with diabetes type 2: a gender perspective. Scand J Caring Sci 26:349–354
Acknowledgments
None.
Author information
Authors and Affiliations
Contributions
JN wrote the manuscript, researched data and gave final approval of the version. JMR, IR and RF researched data and helped writing the article. PS made the statistics and reviewed the manuscript. AB contributed to the discussion. LM contributed to the discussion, reviewed the manuscript and gave final approval of the version. All authors have approved the final article.
Corresponding author
Ethics declarations
Conflict of interest
All authors have no relevant conflicts of interest to disclose.
Ethical Standard Statement
The study was approved by the Ethical Committee of the hospital.
Informed consent
Written informed consent was obtained from all patients prior to study participation. The study was approved by the Ethics Committee of the hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the topical collection Health Education and Psycho-Social Aspects, managed by Massimo Porta and Marina Trento.
Rights and permissions
About this article
Cite this article
Nicolau, J., Romerosa, J.M., Rodríguez, I. et al. Associations of food addiction with metabolic control, medical complications and depression among patients with type 2 diabetes. Acta Diabetol 57, 1093–1100 (2020). https://doi.org/10.1007/s00592-020-01519-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-020-01519-3