Abstract
Aims
The preservation of pancreatic β-cell function is a key point in the treatment of type 2 diabetes mellitus. There is substantial evidence demonstrating that heat-shock protein 90 (Hsp90) is needed for the stabilization and correct folding of client proteins and plays important roles in various biological processes. Here, we revealed the important role of Hsp90 in β-cell function.
Methods
Islets from male Sprague–Dawley rats were isolated to be used for further RT-PCR, Western blot, and insulin secretion test ex vivo in response to different stimuli.
Results
Our results revealed that Hsp90 expression was significantly decreased in isolated rat islets exposed to high glucose, which was involved in glucokinase activation and glucose metabolism, not calcium signaling. Two kinds of Hsp90 inhibitors 17-DMAG and CCT018159 markedly enhanced glucose-stimulated insulin secretion from rat islets, along with increased expressions of genes closely related to β-cell function.
Conclusions
These data indicate that Hsp90 may be involved in high glucose-induced islet function adaptation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pancreatic β-cell dysfunction is the central problem in type 2 diabetes mellitus [1, 2]. The preservation of β-cell function has become a major point of research in this field. Glucose is a major physiological regulator of insulin secretion, and the mechanism currently known about pancreatic β-cell function is mostly from the researches on glucose-stimulated insulin secretion (GSIS) [3]. Acute treatment with high glucose potently increases insulin secretion, and an appropriate extension of time exposed to high glucose also causes enhancement of β-cell secretion capacity [4, 5]. Exploring the signaling pathway of β-cell compensation will provide important insights to better understand the molecular mechanisms underlying β-cell dysfunction and develop novel approaches for prevention and treatment of type 2 diabetes mellitus.
The heat-shock protein Hsp90 forms molecular chaperone complexes with its cofactors and facilitates the maturation of its substrates, termed client proteins. Hsp90 has several isoforms, including cytoplasmic Hsp90α and Hsp90β, as well as the endoplasmic reticulum and mitochondrial isoforms Grp94 and TRAP1 [6]. Initially recognized as a stress-induced protein, Hsp90 has now been identified as an important modulator in oncogenic signaling with the emerging of Hsp90 targets which are critical for cancer cell growth and proliferation [7]. Several Hsp90 inhibitors have entered clinical trials as new anti-cancer drugs, which are expected to be widely used in the future [8]. In addition to its role in tumor regulation, Hsp90 plays an important role in several metabolic pathways [9, 10]. Glucocorticoid receptor (GR), a ligand-activated transcription factor, is a well-known obligate Hsp90 client and mediates the stimulatory effects of glucocorticoid on hepatic gluconeogenesis, lipogenesis, adipogenesis, and lipolysis in adipose tissue. Glucocorticoid treatment inhibits insulin secretion and increases β-cell apoptosis [11]. Hsp90 exerts a critical role in the folding, ligand binding, and nuclear translocation of GR and several other steroid receptors [12]. It has been demonstrated that Hsp90 inhibitors reverse hyperglycemia in the diabetic db/db mice and improve insulin sensitivity in the diet-induced obese mouse model of insulin resistance [13]. However, little is known about the role of Hsp90 in islet function.
Our microarray data showed that high glucose significantly decreased the mRNA expressions of Hsp90aa1 and Hsp90ab1, which coded Hsp90α and Hsp90β, respectively. Given the important role of glucose in the regulation of β-cell function, it is reasonable to assume that Hsp90 proteins may play a role in glucose-induced β-cell function alteration. In this study, we synchronously detected Hsp90aa1 and Hsp90ab1 expressions in rat islets treated with various concentrations of glucose and observed the effect of Hsp90 inhibitors on insulin secretion.
Materials and methods
Reagents
Bovine serum albumin (BSA) and collagenase type XI were purchased from Sigma (St Louis, MO, USA). RPMI 1640 medium, fetal bovine serum (FBS), and other culture reagents were obtained from Gibco Life Technologies (Grand Island, NY). Rat insulin RIA kit was obtained from Mercodia (St Charles, MO, USA). Rabbit anti-Hsp90α antibody was purchased from Millipore Technologies (Billerica, MA, USA). Rabbit anti-β-actin antibody was purchased from Cell Signaling Technology (Beverly, MA, USA). CCT018159 and 17-DMAG were obtained from Calbiochem (San Diego, CA).
Islet isolation and treatment
Islet was isolated from male Sprague–Dawley rats by in situ collagenase infusion and separated by density gradient centrifugation as described previously [4]. Freshly isolated rat islets were transferred to 24-well plates (10 islets per well) or 6-well plates (400 islets per well) and cultured in RPMI 1640 containing 100 μg/ml streptomycin sulfate, 100 U/ml penicillin G sodium, and 0.25 % BSA at 37 °C and 5 % CO2.
Insulin secretion
Cultured islets were washed once in Krebs–Ringer bicarbonate (KRB) buffer [128.8 mmol/l NaCl, 4.8 mmol/l KCl, 1.2 mmol/l KH2PO4, 1.2 mmol/l MgSO4, 2.5 mmol/l CaCl2, 5 mmol/l NaHCO3, and 10 mmol/l Hepes, pH 7.4 with 0.25 % BSA] containing 3.3 mmol/l glucose and then were preincubated in 1 ml of the same medium at 37 °C for 30 min. This buffer was then replaced with 1 ml of prewarmed KRB containing other additions as indicated for a further 60 min at 37 °C. An aliquot was then removed for analysis of insulin secretion. Islets were extracted with 0.18 N HCl in 75 % acid–ethanol solution for insulin content and subsequent normalization.
Immunoblotting
Four hundred islets cultured in 6-well plates were washed twice with ice-cold PBS and then placed immediately in lysis buffer containing 25 mmol/l Hepes(pH 7.4), 1 % Nonidet P-40, 100 mmol/l NaCl, 2 % glycerol, 5 mmol/l NaF, 1 mmol/l EDTA, 1 mmol/l Na3VO4, 1 mmol/l NaPPi, 1 mmol/l phenylmethylsulfonyl fluoride (PMSF), 10 μg/ml aprotinin, 5 μg/ml leupeptin, and 5 μg/ml pepstatin. Lysates were gently mixed for 10 min at 4 °C and then centrifuged at 14,000g for 15 min at 4 °C. Measurement of protein concentrations was performed according to the method of Bradford, using BSA as the standard. Proteins were separated by SDS-PAGE on 8 % polyacrylamide gels. After electrophoresis, the proteins were transferred from the gel to PVDF-Plus membranes (Bio-Rad). Blots were blocked with 5 % BSA. Primary antibodies were detected with donkey anti-rabbit at 1:2000 for 1 h at room temperature. Blotted membrane was developed with ECL Advance (Cell Signaling Technology, Boston, MA) and imaged with a LAS-4000 Super CCD Remote Control Science Imaging System (Fuji, JAP).
Quantitative real-time PCR
Total RNA was extracted from isolated islets using TRIzol (Invitrogen) according to the manufacturer’s instructions. Quantitative real-time PCR (qRT-PCR) was performed using a SYBR Green Premix Ex Taq (Takara, Shiga, Japan). Equal amounts of cDNA were analyzed in triplicate for each run. Values were chosen in the linear range of amplification, and the comparative Ct method was used to assess differences in gene expressions between samples. The results of relative expression were normalized to 18-s mRNA levels in each sample. All primers were synthesized by Shanghai Biological Engineering Technology & Services Co., Ltd. The sequences of primers used are shown in Supplemental Table 1.
Immunofluorescence staining
The rat paraffin-embedded pancreatic sections were blocked with PBS contained 5 % BSA for 1 h and then incubated overnight at 4 °C with rabbit anti-Hsp90 (1:100) and guinea pig anti-insulin (1:400) primary antibodies followed by stained with rhodamine-coupled anti-rabbit IgG and FITC-labeled anti-guinea pig IgG secondary antibodies at room temperature for 2 h in a wet chamber protected from light. Images were collected and analyzed using a fluorescence microscope (Olympus BX51; Olympus America, Inc., Melville, NY).
Statistical analysis
All results are expressed as mean ± SEM. Differences were analyzed using ANOVA for multiple groups followed by Dunnett test or the Student’s test for two groups. Statistical significance was set at the level p < 0.05.
Results
High glucose inhibits Hsp90 expression in rat islets
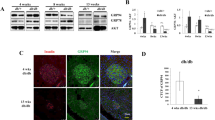
Immunofluorescence data from rat pancreas section showed that Hsp90α was highly expressed in the cytoplasm of pancreatic β-cells (Fig. 1a). Our microarray results revealed that the expressions of two Hsp90 family members Hsp90aa1 and Hsp90ab1 were decreased by 53 and 68 % in rat islets treated with 16.7 mmol/l glucose for 24 h compared to 3.3 mmol/l glucose, respectively. qRT-PCR results validated the reduction of Hsp90aa1 and Hsp90ab1 expressions in the same samples used in the microarray analysis. Compared with 3.3 mmol/l glucose, treatment with 16.7 mmol/l glucose decreased the mRNA expressions of Hsp90aa1 and Hsp90ab1 by 80.21 and 59.25 %, respectively (Fig. 1b, c). Immunoblotting data demonstrated that the protein expression of Hsp90α was also inhibited by 16.7 mmol/l glucose (Fig. 1d).
Hsp90aa1 and Hsp90ab1 expressions in isolated rat islets exposed to high glucose. a Immunofluorescence illustrated that Hsp90α was highly expressed in islet β-cells (scale bar 20 μm). Isolated rat islets were pretreated with 3.3 or 16.7 mM glucose for 24 h, and the mRNA expressions of Hsp90aa1 (b) and Hsp90ab1 (c) were detected by qRT-PCR. The protein level of Hsp90α was detected by Western blot (d). e, f Isolated rat islets were pretreated with 3.3, 5.6, 8.3, 11.1, and 16.7 mM glucose for 24 h, and the mRNA expressions of Hsp90aa1 and Hsp90ab1 were measured by qRT-PCR. Data were given as mean ± SEM for three separate experiments. *p < 0.05; **p < 0.01 versus 3.3 mM glucose
To measure the dose-dependent effect of glucose on Hsp90 gene expression, we treated isolated rat islets with 3.3, 5.6, 8.3, 11.1, and 16.7 mmol/l glucose for 24 h. The results showed that 5.6 mmol/l glucose already decreased Hsp90aa1 and Hsp90ab1 mRNA levels, and more than 8.3 mmol/l glucose did not further decrease the expression of the two genes (Fig. 1e, f).
Activation of glucokinase-suppressed Hsp90 expression
Calcium was shown to have important roles in both glucose-stimulated insulin exocytosis and transcriptional regulation in β-cells [14]. To assess whether calcium was involved in high glucose-regulated Hsp90 gene expression, rat islets were treated with tolbutamide (a KATP channel blocker) and BayK8664 (an L-type calcium channel agonist) at 3.3 mmol/l glucose. Two drugs had no effect on Hsp90aa1 and Hsp90ab1 mRNA expressions at basal glucose concentration (Fig. 2a, b). Diazoxide (a KATP channel opener) and nifedipine (an L-type calcium channel blocker) could not block high glucose-reduced Hsp90aa1 and Hsp90ab1 mRNA expressions in rat islets (Fig. 2a, b).
High glucose-suppressed Hsp90 expression is related to glucose metabolism. a, b Rat islets were incubated with 4 μM BayK8664 and 100 μM tolbutamide (Tol) in the presence of 3.3 mM glucose (Glu) or 250 μM diazoxide (Dia) and 10 μM nifedipine (Nif) in the presence of 16.7 mM glucose. Hsp90aa1 and Hsp90ab1 mRNA expressions were detected by qRT-PCR. c, d Hsp90aa1 and Hsp90ab1 mRNA expressions in isolated rat islets incubated with glucose (Glu), 2-deoxyglucose (2-DG), and 3-O-methylglucose (3-OMG) for 24 h. e, f Hsp90aa1 and Hsp90ab1 mRNA expressions are decreased by 3 μM GKA50 at 3.3 mM glucose in rat islets. Results were expressed as mean ± SEM for three separate experiments. **P < 0.01 versus 3.3 mM glucose
We further observed whether glucose metabolism is required to induce the downregulation of Hsp90 genes. Two non-metabolizable glucose analogues 3-O-methylglucose (3-OMG) and 2-deoxyglucose (2-DG) were unable to decrease Hsp90aa1 and Hsp90ab1 expressions at the concentration of 16.7 mmol/l (Fig. 2c, d). Several glucokinase activators, including GKA 50, are developed for the treatment of type 2 diabetes [15]. At the concentration of 3.3 mmol/l glucose, Hsp90aa1 and Hsp90ab1 mRNA expressions were significantly suppressed by 3 μmol/l GKA 50 (Fig. 2e, f). These results suggest that glucose metabolism is necessary for glucose-inhibited expression of Hsp90aa1 and Hsp90ab1.
Augmentation of insulin secretion by Hsp90 inhibitors
Two different kinds of Hsp90 inhibitors were used to observe the effect of Hsp90 inhibition on insulin secretion from rat islets. 17-DMAG is a potent anti-tumor analogue of 17-AAG that binds to the ATPase site of human Hsp90α with high affinity, excellent bioavailability, and aqueous solubility. CCT018159, a pyrazole-resorcinol derivative, was identified through high-throughput drug screening using an ATPase assay, which can deplete Hsp90 client proteins [8, 16]. We incubated rat islets with 1 μmol/l 17-DMAG or 20 μmol/l CCT018159 at 5.6 mmol/l glucose for 24 h. The insulin secretion was increased by 2.22-fold in 17-DMAG treatment group and 1.84-fold in CCT018159 treatment group (Fig. 3a). When rat islets were challenged with 16.7 mmol/l glucose for 1 h in the presence or absence of the two inhibitors, GSIS was enhanced 2.02-fold by 17-DMAG and 92 % by CCT018159 (Fig. 3b).
Inhibition of Hsp90 potentiates glucose-stimulated insulin secretion. a Isolated rat islets were incubated with 1 μM 17-DMAG and 20 μM CCT018159 in the presence of 5.6 mM glucose for 24 h. The culture medium was taken for insulin assay. b Isolated rat islets were preincubated with the indicated Hsp90 inhibitors for 24 h and then stimulated with 3.3 or 16.7 mM glucose for 1 h. The culture medium was taken for insulin assay. Data were given as mean ± SEM for three separate experiments. *p < 0.05; **p < 0.01 versus control (CON)
Effect of Hsp90 inhibitors on the expression of genes involved in islet function
To further explore the mechanism underlying Hsp90 inhibitors-enhanced insulin secretion, we treated rat islets with 17-DMAG and CCT018159 for 24 h at the concentration of 5.6 mmol/l glucose and analyzed the expressions of genes important for β-cell function. Treatment with 17-DMAG or CCT018159 resulted in significant increases in insulin receptor substrate 2 (IRS2), proprotein convertases 1(PCSK1), and PCSK2 mRNA levels, with increased expressions of transcription factor MafA. The transcript levels of glucose sensor glucose transporter 2 (GLUT2) and insulin exocytosis-related gene SNAP25 were also elevated in islets treated with CCT018159. However, no changes were observed in insulin 1(INS1), INS2, and pancreatic and duodenal homeobox 1 (PDX1) expressions (Fig. 4). These results suggest that Hsp90 inhibitors enhance β-cell secreting function via increasing expressions of multiple genes involved in glucose sensing, proinsulin processing, and exocytosis.
Effect of Hsp90 inhibitors on the mRNA expressions of islet function-related genes. Rat islets were isolated and cultured with 1 μM 17-DMAG and 20 μM CCT018159 at 5.6 mM glucose for 24 h. mRNA expressions of islet function-related genes were detected by qRT-PCR. Data were given as mean ± SEM for three separate experiments. *p < 0.05 versus control (CON)
Discussion
Glucose is the most essential factor in regulating β-cell function. The effect of glucose on β-cell function depends on its concentration and exposure time. High glucose was validated to promote insulin secretion through extremely complex metabolic pathways [3]. However, continuous overstimulation of the β-cell by high glucose could lead to depletion of insulin stores, apoptosis, and finally deterioration of β-cell function [17]. As a molecule chaperone, Hsp90 is involved in the regulation of a variety of metabolic pathways [9, 10]. In this study, high glucose significantly decreased Hsp90aa1 and Hsp90ab1 expressions in rat islets. Two Hsp90 inhibitors potentiated GSIS from rat islets, showing an effect similar to the prolonged treatment with high glucose. This suggests that Hsp90 may be involved in high glucose-induced islet functional compensation.
Glucose metabolism is essential for the stimulation of insulin secretion and regulation of gene transcription in β-cells [18, 19]. Our data demonstrated that high glucose downregulated Hsp90aa1 and Hsp90ab1 expressions. Two non-metabolizable glucose analogues 3-OMG and 2-DG had no impact on the expression of Hsp90, while glucokinase agonist GKA50 attenuated Hsp90aa1 and Hsp90ab1 mRNA levels. These data suggest that glucose metabolism is involved in the downregulation of Hsp90 genes mediated by high glucose. No obvious changes in Hsp90 expression were observed in islets treated with BayK8664, nifedipine, diazoxide, or tolbutamide, which clamped cytosolic calcium at high or low level via manipulating the open or close of L-type calcium and KATP channels [20, 21]. This indicates that calcium signaling has little effect on high glucose-reduced Hsp90aa1 and Hsp90ab1 expressions.
Given the broad spectrum of client proteins, it is not surprising that Hsp90 is essential for various cellular processes [22, 23]. Acetylation of Hsp90 promotes the dissociation of p23 from the GR·Hsp90 heterocomplex, and the receptor becomes unable to bind steroid or translocate into the nucleus [24]. Histone deacetylase 6 (HDAC6)-mediated Hsp90 deacetylation allows the chaperone complex to interact with nascent GR, which is required for the proper folding and maturation of GR. Upon hormone binding, the GR dissociates from the chaperone complex and translocates into the nucleus, where it activates the transcription of genes involved in hepatic gluconeogenesis [25]. This suggests that Hsp90 plays an important role in the regulation of metabolic pathways in hepatocytes. In the present study, we provided evidence that high glucose decreased Hsp90 gene expression in rat islets, indicating that Hsp90 may be involved in glucose-potentiated islet function. In support of this assumption, two Hsp90 inhibitors amplified GSIS from rat islets. It is possible that high expression of Hsp90 at lower blood glucose level inhibits insulin secretion to protect against hypoglycemia.
The whole event of GSIS in β-cell is related to a complicated molecular network, including the regulation of insulin synthesis and exocytosis. Many effectors have been found to be involved in GSIS, such as IRS2, MafA, PCSK1, and PCSK2 [26–28]. In this current study, we found that Hsp90 inhibitors exerted a stimulatory effect on the expressions of these genes. CCT018159 treatment resulted in significant increases in IRS2, PCSK1, PCSK2, MafA, GLUT2, and SNAP25 mRNA levels, which may be related to the potentiated insulin secretion. A large number of the Hsp90-modulated client proteins belong to the protein kinase family, representing one of the largest groups of known Hsp90 partners. Accordingly, the inhibition of the Hsp90 is pursued as a novel therapeutic strategy for treating several types of cancer. Hsp90 inhibitors are currently being evaluated in clinical trials. Before they are administered to perform anti-tumor on diabetic patients, any possible effects on islet function should be identified.
In conclusion, high glucose can decrease Hsp90aa1 and Hsp90ab1 expressions in rat islets, which is associated with glucose metabolism. Moreover, the inhibition of Hsp90 markedly enhances GSIS, along with increased expressions of several genes related to glucose sensing, insulin processing, and exocytosis. The results add support to the potentially protective role of Hsp90 inhibitors on islet function and may open a new perspective in application of Hsp90 inhibitors into the clinical therapy for type 2 diabetes mellitus.
References
Ashcroft FM, Rorsman P (2012) Diabetes mellitus and the beta cell: the last ten years. Cell 148(6):1160–1171. doi:10.1016/j.cell.2012.02.010
Fu Z, Gilbert ER, Liu D (2013) Regulation of insulin synthesis and secretion and pancreatic Beta-cell dysfunction in diabetes. Curr Diabetes Rev 9(1):25–53
Henquin JC (2009) Regulation of insulin secretion: a matter of phase control and amplitude modulation. Diabetologia 52(5):739–751. doi:10.1007/s00125-009-1314-y
Deng R, Nie A, Jian F et al (2014) Acute exposure of beta-cells to troglitazone decreases insulin hypersecretion via activating AMPK. Biochim Biophys Acta 1840(1):577–585. doi:10.1016/j.bbagen.2013.10.021
Corkey BE (2012) Banting lecture 2011: hyperinsulinemia: cause or consequence? Diabetes 61(1):4–13. doi:10.2337/db11-1483
Zuehlke AD, Beebe K, Neckers L, Prince T (2015) Regulation and function of the human HSP90AA1 gene. Gene 570(1):8–16. doi:10.1016/j.gene.2015.06.018
Khurana N, Bhattacharyya S (2015) Hsp90, the concertmaster: tuning transcription. Front Oncol 5:100. doi:10.3389/fonc.2015.00100
Sidera K, Patsavoudi E (2014) HSP90 inhibitors: current development and potential in cancer therapy. Recent Pat Anti-Cancer Drug Discov 9(1):1–20
Murphy PJ, Morishima Y, Kovacs JJ, Yao TP, Pratt WB (2005) Regulation of the dynamics of Hsp90 action on the glucocorticoid receptor by acetylation/deacetylation of the chaperone. J Biol Chem 280(40):33792–33799. doi:10.1074/jbc.M506997200
Yan FF, Pratt EB, Chen PC et al (2010) Role of Hsp90 in biogenesis of the beta-cell ATP-sensitive potassium channel complex. Mol Biol Cell 21(12):1945–1954. doi:10.1091/mbc.E10-02-0116
Patel R, Williams-Dautovich J, Cummins CL (2014) Minireview: new molecular mediators of glucocorticoid receptor activity in metabolic tissues. Mol Endocrinol 28(7):999–1011. doi:10.1210/me.2014-1062
Kirschke E, Goswami D, Southworth D, Griffin PR, Agard DA (2014) Glucocorticoid receptor function regulated by coordinated action of the Hsp90 and Hsp70 chaperone cycles. Cell 157(7):1685–1697. doi:10.1016/j.cell.2014.04.038
Lee JH, Gao J, Kosinski PA et al (2013) Heat shock protein 90 (HSP90) inhibitors activate the heat shock factor 1 (HSF1) stress response pathway and improve glucose regulation in diabetic mice. Biochem Biophys Res Commun 430(3):1109–1113. doi:10.1016/j.bbrc.2012.12.029
Heit JJ, Apelqvist AA, Gu X et al (2006) Calcineurin/NFAT signalling regulates pancreatic beta-cell growth and function. Nature 443(7109):345–349. doi:10.1038/nature05097
Johnson D, Shepherd RM, Gill D, Gorman T, Smith DM, Dunne MJ (2007) Glucose-dependent modulation of insulin secretion and intracellular calcium ions by GKA50, a glucokinase activator. Diabetes 56(6):1694–1702. doi:10.2337/db07-0026
Khandelwal A, Crowley VM, Blagg BS (2015) Natural product inspired N-terminal Hsp90 inhibitors: from bench to bedside? Med Res Rev. doi:10.1002/med.21351
Poitout V, Robertson RP (2008) Glucolipotoxicity: fuel excess and beta-cell dysfunction. Endocr Rev 29(3):351–366. doi:10.1210/er.2007-0023
Prentki M, Matschinsky FM, Madiraju SR (2013) Metabolic signaling in fuel-induced insulin secretion. Cell Metab 18(2):162–185. doi:10.1016/j.cmet.2013.05.018
Porat S, Weinberg-Corem N, Tornovsky-Babaey S et al (2011) Control of pancreatic beta cell regeneration by glucose metabolism. Cell Metab 13(4):440–449. doi:10.1016/j.cmet.2011.02.012
Ravier MA, Cheng-Xue R, Palmer AE, Henquin JC, Gilon P (2010) Subplasmalemmal Ca(2+) measurements in mouse pancreatic beta cells support the existence of an amplifying effect of glucose on insulin secretion. Diabetologia 53(9):1947–1957. doi:10.1007/s00125-010-1775-z
Ashcroft SJ, Ashcroft FM (1992) The sulfonylurea receptor. Biochim Biophys Acta 1175(1):45–59
Erlejman AG, Lagadari M, Toneatto J, Piwien-Pilipuk G, Galigniana MD (2014) Regulatory role of the 90-kDa-heat-shock protein (Hsp90) and associated factors on gene expression. Biochim Biophys Acta 1839(2):71–87. doi:10.1016/j.bbagrm.2013.12.006
Taipale M, Jarosz DF, Lindquist S (2010) HSP90 at the hub of protein homeostasis: emerging mechanistic insights. Nat Rev Mol Cell Biol 11(7):515–528. doi:10.1038/nrm2918
Kadiyala V, Smith CL (2014) Minireview: the versatile roles of lysine deacetylases in steroid receptor signaling. Mol Endocrinol 28(5):607–621. doi:10.1210/me.2014-1002
Kadiyala V, Patrick NM, Mathieu W et al (2013) Class I lysine deacetylases facilitate glucocorticoid-induced transcription. J Biol Chem 288(40):28900–28912. doi:10.1074/jbc.M113.505115
Shao S, Fang Z, Yu X, Zhang M (2009) Transcription factors involved in glucose-stimulated insulin secretion of pancreatic beta cells. Biochem Biophys Res Commun 384(4):401–404. doi:10.1016/j.bbrc.2009.04.135
Andrali SS, Sampley ML, Vanderford NL, Ozcan S (2008) Glucose regulation of insulin gene expression in pancreatic beta-cells. Biochem J 415(1):1–10. doi:10.1042/BJ20081029
Flanagan SE, Clauin S, Bellanne-Chantelot C et al (2009) Update of mutations in the genes encoding the pancreatic beta-cell K(ATP) channel subunits Kir6.2 (KCNJ11) and sulfonylurea receptor 1 (ABCC8) in diabetes mellitus and hyperinsulinism. Hum Mutat 30(2):170–180. doi:10.1002/humu.20838
Acknowledgments
This work was funded by grants from the National Natural Science Foundation of China (81170720, 81270910, 81370876, 81471030, and 81570693).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All experimental procedures related to animal handling in the present study were adhered to the approval obtained from the Laboratory Animal Ethics Committee of Ruijin Hospital.
Human and animal rights
This article does not contain any studies with human subjects performed by any of the authors. All institutional and national guidelines for the care and use of laboratory animals were followed.
Informed consent
No informed consent.
Additional information
Managed by Antonio Secchi.
Xue Yang and Yuqing Zhang have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, X., Zhang, Y., Xu, W. et al. Potential role of Hsp90 in rat islet function under the condition of high glucose. Acta Diabetol 53, 621–628 (2016). https://doi.org/10.1007/s00592-016-0852-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-016-0852-2