Abstract
Aims
Stable genetic background makes individuals from the Mediterranean island of Sardinia ideal to define the predictive power of islet-related autoantibodies (IRAs): glutamic acid decarboxylase antibodies (GADA), tyrosine phosphatase-like antibodies (IA-2A), islet cell antibodies (ICA) to identify T1DM progressors. The aims of the present study were: (1) determination of IRAs reference limits in healthy non-diabetic Sardinian schoolchildren (SSc). (2) Predictive power evaluation of IRAs as single or combined determination to identify islet to identify T1DM progressors.
Methods
Between 1986 and 1994, 8448 SSc were tested for IRAs. All were followed up for 10 years. The predictive power of single or combination of IRAs was determined as hazard ratio (HR), sensitivity, specificity, area under the ROC curve, negative and positive predictive value (NPV, PPV).
Results
All 43 progressors to T1DM, but three showed at least one autoantibody positivity. HR for any single-autoantibody positivity was 55.3 times greater when compared to SSc negative for all IRAs. Any single autoantibody performed at least 64.9 % sensitivity with PPV always lower than 16 %. The best performing combination was ICA, plus IA-2A (showing 52.6 % sensitivity, 99.8 % specificity, 0.76 area under the ROC curve, 51.3 % PPV and 99.8 % NPV.
Conclusions
Determination of IRAs reference limits in healthy SSc by standard statistical methods is crucial to establish the power of IRAs as progression markers to T1DM. Our data offer a solid rationale for future testing of ICA and IA-2A as routine laboratory markers to identify individuals at high risk of T1DM in the general population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type 1 diabetes mellitus (T1DM) is a chronic autoimmune disease based on genetic susceptibility, eventually triggered by unknown environmental factors [1].
T1DM incidence worldwide varies from the lowest values reported in China and Venezuela (0.1 per 100,000 inhabitants per year) and South korea (0.7 per 100,000 inhabitants per year) [2, 3], to the highest recorded in Finland (42.9 per 100,000 inhabitants per year) [3, 4]. In Europe, a South–North gradient has been described with lower rates observed in the Mediterranean area (4.6–10.6 per 100,000 inhabitants per year, Northern Greece and Spain, respectively) and high incidence in Scandinavian countries (20.8–42.9 per 100,000 inhabitants in Norway and Finland, respectively. An exception to this trend is the Mediterranean island of Sardinia, where a progressive increase in new cases of T1DM (37 per 100,000 inhabitants per year) has been reported since the 1960’s, reaching values close to those of Finland [3, 5–7].
Genetic analysis has proven that the historical, geographical and cultural isolation of Sardinia from European and African populations has maintained the genome background of Sardinians stable for centuries [8, 9]. The identification of Sardinia as an area with an elevated T1DM incidence renders the island an ideal epidemiological environment to design studies aimed at identifying individuals at risk of developing the disease [10].
Clinical onset of T1DM is preceded by a preclinical phase in which the presence of several circulating islet cells has been described. These include the heterogeneous islet cell cytoplasm autoantibodies (ICA) [11] and directed to target islet antigens such as insulin [12], glutamic acid decarboxylase (GADA) [13] and the protein tyrosine phosphatase-like IA-2 (IA-2A) [14, 15]. Preclinical detection of ICA has been proven to predict T1DM in individuals at risk, such as those carrying HLA susceptibility haplotypes or first-degree relatives [16]. Even in individuals at risk, the predictive value of diabetes-related antibodies is dependent on titre and on the detection of multiple circulating antibody specificities [17]. However, the occurrence of T1DM is sporadic in about 90 % of cases, and studies on unselected individuals suggest that multiple islet autoantibody determination could have a significant predictive value in the general population [17–19].
Retrospective data on children with family history or HLA risk of developing T1DM who associated dietary supplements of vitamin D or omega-3 fatty acids showed a reduced hazard ratio of developing islet autoimmunity [20]. However, T1DM occurs mostly in the general population and future prevention therapeutic trials need to be carried out on individuals truly at risk identified from the population at large using tests with high predictive values.
We evaluated the predictive power of single and combined diabetes related to identify individuals at risk of developing T1DM in the general population. Taking advantage of the high incidence of T1DM in Sardinia and its geographical peculiarities, serum samples were collected from 8448 healthy Sardinian schoolchildren (SSc) at enrolment, and ICA, GADA and IA-2A were successively measured. All SSc enrolled were followed up for a period of up to 10 years.
Methods
Study design
A total of 8448 healthy SSc (3972 girls, 4476 boys, aged 4–17 years) were recruited from primary (4366 SSc) and middle schools (4082 SSc) in 36 randomly selected towns in the four provinces of Sardinia. Specifically, Cagliari (south; eight towns; 2080 schoolchildren), Oristano (mid-west; five towns; 1730 schoolchildren), Nuoro (mid-east; 12 towns; 1661 schoolchildren) and Sassari (north; 11 towns; 2977 schoolchildren) representing 70–89 % of all registered schoolchildren. Twenty-four schoolchildren with T1DM at screening (prevalence 2.8/1000) were excluded from the study. Enrolment was carried out between 1986 and 1994 in the same cohort of SSc analysed in a previous report on the incidence of goitre in Sardinia [21]. First-degree family history (FH) for T1DM (diagnosis of diabetes according to the ADA) and thyroid diseases (TD) were obtained by a questionnaire completed by the parents of 3331 (39.4 %) SSc. In order to ascertain diagnostic criteria for T1DM and TD, the authors (AL, FV, GS) personally validated all questionnaires by telephone calls. T1DM FH data were also obtained from the Sardinian Eurodiab ACE register with a final estimated completeness of ascertainment of 91.3 % calculated by “capture–recapture” method [22]. Overall, 16 SSc had a sibling with T1DM, but only one showed ICA positivity (45 JDFu). All SSc were followed up yearly for at least 10 years by authors’ telephone calls (AL, FV, GS), general practitioners and the Eurodiab ACE register [22].
Sample collection
Blood samples were collected by venipuncture in 10 ml plain Vacutainer tubes (Becton–Dickinson, Vacutainer®, Le Pont-De-Claix, France). The blood was clotted at room temperature, and sera were separated within 2 h after sampling. Serum aliquots of 100 µl were stored at −20 °C until tested.
ICA detection
After heating the serum to 56 °C to denature interfering proteins, ICA were detected by means of undiluted sera using indirect immunofluorescence on 4-µm unfixed cryostat sections of group 0 human pancreas [11]. Quantification in Juvenile Diabetes Foundation units (JDFu) was performed applying serial dilution in parallel with a standard, calibrated against an international reference sample of 80 JDFu according to recommendation of the 2nd international ICA workshop [23]. Local standards of 13, 48 and 80 JDFu were also included in each assay. End-point titre of test samples was converted to JDFu by comparison with a standard curve of log2JDFu versus log2 of end-point titre of standard sera [24]. Variation coefficients between assays for control sera of 13, 48 and 80 JDFu used in 10 consecutive assays were 10, 22 and 4 %, respectively. The threshold of ICA detection was 4 JDFu.
GADA and IA-2A detection
Serum GADA [25] and IA-2A [26] levels were determined by a radio-binding assay and retested if higher than 95th centile. Levels were converted into arbitrary units (AU) by extrapolation from a standard curve. Our laboratory achieved 100 % sensitivity, specificity and validity in radio immunoassays for the 3rd International GAD and IA2 Proficiency Programs in 1997 [27].
Statistical analysis
Data were described as mean and standard deviation (SD) or counts and percentages. Reference limits were computed as the 2.5th and the 97.5th percentile of their distribution, overall and separately for gender and age groups (≤10/>10 years), together with their 90 % confidence intervals (CI). Positivity was defined as an antibody level above the upper reference limit [28]. Incidence rates of diabetes, and their 95 % CI, were computed per 100,000 inhabitants per year. As prevalence of T1DM, [29] in Sardinia was assumed that reported by Frongia et al. of 4.59 per 1000 (95 % CI 4.07–5.17 [30]. Cox regression was used to assess the predictive role of positive antibodies or their association. The assumption of proportional hazard was verified by means of a test on Schoenfeld residuals. Finally, the sensitivity, specificity, area under the ROC curve, positive and negative predictive value of each antibody or their combination were computed to measure their ability to predict diabetes at 10 years. Stata 10 (StataCorp, College Station, TX, USA) was used for computation. A two-sided p value <0.05 was considered statistically significant.
Ethical approval
Approval for the study was granted by the Sardinia Research Ethical Committee of the University of Cagliari, Italy. Written consent was obtained from the Italian Ministry of Health, the Italian Ministry of Education, all school boards, local parent committees, school headmasters and parents for the physical examination and blood sampling of the schoolchildren.
Results
Reference limits and positivity of islet cell autoantibodies in healthy Sardinian schoolchildren
ICA, GADA and IA-2A were tested in all 8448 healthy SSc recruited on blood taken at the time of enrolment. Upper and lower references limits (97.5th and 2.5th, respectively) and their 90 % CI for ICA, GADA and IA-2A were calculated overall and according to gender and age groups.
Particularly, upper limit for ICA was 9 JDFu in males over 10 years and in females both under or over 10 years, while it was 11 JDFu in males equal or under 10 years; upper limit for GADA was 6 AU and 7 AU, respectively in females and males aged equal or under 10 years, while it was 8 AU in females and males over 10 years; finally, upper limit for IA-2A was 2 AU in females and males equal or under 10 years, while it was 3 AU in females and males over 10 years.
Positivity for at least one autoantibody was found in 477 SSc (5.6 %). ICA, GADA or IA-2A positivity was detected in 208 (2.46 %), 198 (2.34 %) and 164 (1.94 %) SSc, respectively. Forty-six SSc (0.54 %) showed positivity for two autoantibodies. Forty-three SSc (0.51 %) were positive for ICA and GADA, 39 (0.46 %) for ICA and IA-2A and 46 (0.54 %) showed positivity for GADA and IA-2A. Positivity for all the autoantibodies tested was found in 28 (0.3 %) out of 8448 SSc enrolled on the study.
Diagnostic ability of islet cell autoantibodies to predict the occurrence of T1DM within 10 years of testing
All SSc were followed up for at least 10 years (median 168 months). The yearly rate T1DM incidence per 100,000 healthy Sardinian schoolchildren was 3/100,000 (95 % CI 0.9–8.3) per year without any autoantibody, 140/100,000 (95 % CI 75–260) per year with one autoantibody positivity, 2100/100,000 (95 % CI 1200–4000) per year with two autoantibodies, and 7600/100,000 (95 % CI 4800–12,000) per year with three autoantibodies.
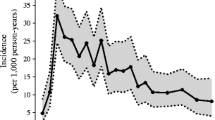
As shown in the life-table (Fig. 1), forty-three SSc developed T1DM [29] after a median follow-up of 48 months (25th–75th percentiles, 24–84), corresponding to a rate of 36 cases per 100,000 inhabitants per year (95 % CI, 26–48), as also reported in the Sardinian diabetes register [6]. Their mean age at the time of recruitment was 15.8 years (SD 5.1; range 7.2–26.7); 26 (60 %) were females and 17 (40 %) males. Three (0.04 %) out of 7897 SSc negative for all tests developed T1DM, as compared to 10 (2.10 %) positive for only one autoantibody, 12 (26.1 %) positive for two autoantibodies and 18 (64.3 %) positive for ICA, GADA and IA-2A. In Table 1, the hazard ratio of developing T1DM after 10 years from autoantibody assessment is shown: the positivity for any of the three significantly increased by 55-fold the risk of developing T1DM within 10 years from testing, when compared to negativity to all. A further significant increase in risk (by 14 and 3 times, respectively) was observed for the presence of 2 versus 1 and 3 versus 2 autoantibodies.
In Table 2, the diagnostic ability of positive ICA, GADA or IA-2A (>97.5th percentile) to identify occurrence of type 1 diabetes mellitus according to age and gender within 10 years from autoantibody assessment is reported. Prevalence of type 1 diabetes mellitus at 10 years, estimated from the data, was 0.44 % (95 % CI 32.0–61.7).
Among the three islet cell-related autoantibodies tested, the best performing single positivity was IA-2A, with the highest area under the ROC curve of 0.84 and positive predictive value (PPV) of 15.9 % (Table 2). Among the double positivity, ICA plus IA-2A performed better when compared to ICA plus GADA or GADA plus IA-2A, with higher PPV (51.3 %), but comparable areas under the ROC curve. Positivity to all three tested showed the best PPV of 60.7 %. Finally, as shown in Table 2, the sensitivity tended to decrease with the increased number of positive antibodies, while the specificity modestly increased. Correspondingly, the area under the ROC curve decreased from 0.84 to 0.72, although it still reflected a satisfying diagnostic ability of the single or their combination. Life-table analysis of cumulative risk of T1DM within 10 years in 8448 school children in relation to numbers of additional positive is showed in Fig. 1.
Discussion
The stable genetic background of Sardinians created an ideal environment to define reference limits of ICA, GADA and IA-2A in our 8448 healthy schoolchildren. A standard statistical method accepted worldwide in hospital laboratories for routine clinical determinations of islet cell autoantibodies was employed [28]. However, data from high risk children do not allow for our findings to be generalised, which represents a limitation of the present study.
The aim of this work was to establish the power of single and multiple antibodies to predict T1DM progression in the general population. Several long-term reports on non-diabetic schoolchildren tested islet-related autoantibodies report variable results. Three studies used ICA positivity as the main predictive test. Bruining et al. reported on 4806 Dutch schoolchildren aged 5–19 years within 10-year follow-up. Relative risk of developing T1DM for ICA positive individuals was 95 %; however, ICA positivity predicted T1DM in only four out of eight progressors [31].
Schatz et al. [32] reported on 3854 healthy Florida schoolchildren aged 5–18 years showing 100 % sensitivity and 18 % PPV for ICA greater than 10 JDFu. Ten ICA positive children all developed T1DM after 2–8.2 years follow-up. Lévy-Marchal et al. [33] studied 13,380 non-diabetic French schoolchildren aged 6–17 years. Five children developed T1DM after 4 years from enrolment; all had ICA greater than 20 JDFu. One out of three studies [31] reported that ICA positivity identifies less than 50 % progressors to T1DM. An explanation could be that ICA determination is known to be the most operator dependent test among all islet autoantibody assays. Several prediction studies used instead a multiple autoantibody approach (Table 3). Genovese et al. [34] assessed IAA, GADA and 37 k antibodies in 81 ICA positive English schoolchildren and observed that the three progressors to T1DM were positive for 37 k antibody, suggesting these antibodies as a tool predictive for future T1DM in the general population. Bonifacio et al. [35] measured ICA, antibodies to GAD 65, to 37,000/40,000 M(r) islet tryptic fragments, and ICA 69 in 100 newly diagnosed T1DM patients, in 27 pre T1DM, and in 83 control subjects, and concluded that none of the single antibody specificities are as sensitive as ICA, but that a combination of antibodies to GAD65 and to 37,000/40,000 M(r) islet tryptic fragments has the potential to identify more than 90 % of future cases of T1DM. Bingley et al. [36] indirectly estimated the predictive value of multiple autoantibodies, in 2855 schoolchildren and in 256 new diagnosed T1DM children living in the Oxford region (UK), concluding that although this estimate model would reach a satisfactory potential means of identification, it should be confirmed by actual prospective studies before the application in field strategies of prediction. The Karlsburg study [19] evaluated the predictive value of ICA, GADA, IA-2A and IAA in 9419 non-diabetic German schoolchildren. Six individuals developed T1DM after 19 ± 10 months (mean ± SD). All progressors had ICA greater than 40 JDFu and two or more autoantibody positivity higher than 99.8th centile. A Sweden study [37] reported on 1031 healthy schoolchildren screened for ICA, GADA and IA-2A with 10-year follow-up. Six children developed T1DM and all but 1 had at least 1 autoantibody positivity. ICA and GADA positivity resulted as the best performing combination with 40 % PPV. Kulmala et al. [38] published a study on 3652 non-diabetic Finnish schoolchildren screened for ICA, GADA, IA-2A and IAA. Four individuals developed T1DM after a median follow-up of 5.3 years. All progressors were positive for two or more autoantibodies. Sensitivity for any autoantibody tested was 100 %. PPV of ICA greater than 20 JDFu was 29 %. IA-2A and GADA positivity showed 19 and 11 % PPV, respectively. Combination of two or more autoantibody positivity performed 19 % PPV. The Washington State Diabetes Prediction Study [18] screened 3000 healthy schoolchildren for ICA, GADA, IA-2A and IAA. Six individuals progressed to T1DM within 10 years from enrolment and all of them were positive for two or more islet cell autoantibodies. PPV for multiple autoantibody testing was 50 %. From these studies, we learn that single-autoantibody sensitivity of either ICA or radio binding ranges from 50 to 100 %.
A further Finnish study recently published by Knip et al. [39] evaluated the T1DM predictive power of GADA, and subsequently, IA-2A in the cohort of 3475 non-diabetic previously enrolled who were followed for a further 27 years: GADA and/or IA-2A showed a 61 % sensitivity, 99 % specificity, 22 % PPV, and GADA and IA-2A reported 39 % sensitivity, 100 % specificity and an impressive 100 % PPV. These data from the general population at the highest risk of developing T1DM, might give strength to our results, obtained in Sardinia, the second region in the world at highest risk of developing T1DM.
Our data are consistent with the majority of these works showing sensitivity for any single autoantibody tested always greater than 64.9 % and PPV lower than 16 %. The risk of developing T1DM in our non-diabetic 8448 SSc within 10 years from enrolment was 55.3 times higher in positives to any single autoantibody tested and increased by 14.5 times for any double autoantibody positivity. Only 3 out of 43 (7 %) SSc that developed T1DM in 10 years were negative to all. Specificity to develop T1DM in SSc positive to any single autoantibody was always greater than 97.8 %. PPV greater than 50 % is a recognised successful achievement. Our data show that the combination of ICA and IA-2A is an effective approach when large populations need to be screened to identify T1DM progressors. Single ICA or IA-2A positivity gave 70 % sensitivity and 98 % specificity. A combination of ICA and IA-2A positivity reached a PPV of 51.3 %.
Conclusion
The combination of ICA and IA-2A is an effective approach when large populations need to be screened to identify T1DM progressors. Their triple (ICA, GADA, IA-2A) combination reached a PPV of 60.7 % and a NPV of 99.7. These data give a solid rationale to continue multiple autoantibody screening among young Sardinians. It could identify individuals potentially eligible for enrolment in clinical trials aimed at reducing the risk of progression from islet autoimmunity towards clinical T1DM [38–41].
Abbreviations
- SSc:
-
Sardinian schoolchildren
- FH:
-
Family history
- TD:
-
Thyroid disease
- T1DM:
-
Type 1 diabetes mellitus
- IRAs:
-
Islet-related autoantibodies
- ICA:
-
Islet cell antibodies
- GADA:
-
Glutamic acid decarboxylase antibodies
- IA-2A:
-
Protein tyrosinephosphatase-like IA-2
- IAA:
-
Insulin autoantibodies
References
Hattersley AT (1997) Genes versus environment in insulin-dependent diabetes: the phoney war. Lancet 349:147–148
Ko KW, Yang SW, Cho NH (1994) The incidence of IDDM in Seoul from 1985 to 1988. Diabetes Care 17:1473–1475
Soltesz G, Patterson CC, Dahlquist G (2007) Worldwide childhood type 1 diabetes incidence–what can we learn from epidemiology? Pediatr Diabetes 8(Suppl 6):6–14
Green A, Gale EA, Patterson CC (1992) Incidence of childhood-onset insulin-dependent diabetes mellitus: the EURODIAB ACE study. Lancet 339:905–909
Songini M, Loche M, Muntoni S et al (1993) Increasing prevalence of juvenile onset type 1 (insulin-dependent) diabetes mellitus in Sardinia: the military service approach. Diabetologia 36:547–552
Casu A, Pascutto C, Bernardinelli L, Songini M (2004) Type 1 diabetes among sardinian children is increasing: the Sardinian diabetes register for children aged 0–14 years (1989–1999). Diabetes Care 27:1623–1629
Casu A, Trucco M, Pietropaolo M (2005) A look to the future: prediction, prevention, and cure including islet transplantation and stem cell therapy. Pediatr Clin North Am 52:1779–1804
Cavalli-Sforza LL, Piazza P (1993) Human genomic diversity in Europe: a summary of recent research and prospects for the future. Eur J Hum Genet 1:3–18
Cappello N, Rendine S, Griffo R et al (1996) Genetic analysis of Sardinia: I. data on 12 polymorphisms in 21 linguistic domains. Ann Hum Genet 60:125–141
Bottazzo GF, Loviselli A, Velluzzi F et al (1997) The “Sardinia-IDDM study”: an attempt to unravel the cause of insulin-dependent diabetes mellitus in one of the countries with the highest incidence of the disease in the world. Ann Ist Super Sanita 33:417–424
Bottazzo GF, Florin-Christensen A, Doniach D (1974) Islet-cell antibodies in diabetes mellitus with autoimmune polyendocrine deficiencies. Lancet 2:1279–1283
Palmer JP, Asplin CM, Clemons P et al (1983) Insulin antibodies in insulin-dependent diabetics before insulin treatment. Science 222:1337–1339
Baekkeskov S, Aanstoot HJ, Christgau S et al (1990) Identification of the 64K autoantigen in insulin-dependent diabetes as the GABA-synthesizing enzyme glutamic acid decarboxylase. Nature 347:151–156
Payton MA, Hawkes CJ, Christie MR (1995) Relationship of the 37,000- and 40,000-M(r) tryptic fragments of islet antigens in insulin-dependent diabetes to the protein tyrosine phosphatase-like molecule IA-2 (ICA512). J Clin Invest 96:1506–1511
Bonifacio E, Lampasona V, Genovese S, Ferrari M, Bosi E (1995) Identification of protein tyrosine phosphatase-like IA2 (islet cell antigen 512) as the insulin-dependent diabetes-related 37/40K autoantigen and a target of islet-cell antibodies. J Immunol 155:5419–5426
Seissler J, Hatziagelaki E, Scherbaum WA (2001) Modern concepts for the prediction of type 1 diabetes. Exp Clin Endocrinol Diabetes 109(Suppl 2):S304–S316
Taplin CE, Barker JM (2008) Autoantibodies in type 1 diabetes. Autoimmunity 41:11–18
LaGasse JM, Brantley MS, Leech NJ et al (2002) Successful prospective prediction of type 1 diabetes in schoolchildren through multiple defined autoantibodies: an 8-year follow-up of the Washington State Diabetes Prediction Study. Diabetes Care 25:505–511
Strebelow M, Schlosser M, Ziegler B, Rjasanowski I, Ziegler M (1999) Karlsburg type I diabetes risk study of a general population: frequencies and interactions of the four major type I diabetes-associated autoantibodies studied in 9419 schoolchildren. Diabetologia 42:661–670
Norris JM, Yin X, Lamb MM et al (2007) Omega-3 polyunsaturated fatty acid intake and islet autoimmunity in children at increased risk for type 1 diabetes. JAMA 298:1420–1428
Loviselli A, Velluzzi F, Mossa P et al (2001) The Sardinian Autoimmunity Study: 3. Studies on circulating antithyroid antibodies in Sardinian schoolchildren: relationship to goiter prevalence and thyroid function. Thyroid 11:849–857
Songini M, Bernardinelli L, Clayton D et al (1998) The Sardinian IDDM study: 1. Epidemiology and geographical distribution of IDDM in Sardinia during 1989 to 1994. Diabetologia 41:221–227
Palmer JP, Wilkin TJ, Kurtz AB, Bonifacio E (1990) The third international workshop on the standardisation of insulin autoantibody measurement. Diabetologia 33:60–61
Bonifacio E, Lernmark A, Dawkins RL (1988) Serum exchange and use of dilutions have improved precision of measurement of islet cell antibodies. J Immunol Methods 106:83–88
Petersen JS, Hejnaes KR, Moody A et al (1994) Detection of GAD65 antibodies in diabetes and other autoimmune diseases using a simple radioligand assay. Diabetes 43:459–467
Seissler J, Morgenthaler NG, Achenbach P et al (1996) Combined screening for to IA-2 and antibodies to glutamic acid decarboxylase in first degree relatives of patients with IDDM. The DENIS Study Group. Deutsche Nikotinamid Interventions-Studie. Diabetologia 39:1351–1356
Verge CF, Stenger D, Bonifacio E et al (1998) Combined use of (IA-2 autoantibody, GAD autoantibody, insulin autoantibody, cytoplasmic islet cell antibodies) in type 1 diabetes: combinatorial islet autoantibody workshop. Diabetes 47:1857–1866
Reed AH, Henry RJ, Mason WB (1971) Influence of statistical method used on the resulting estimate of normal range. Clin Chem 17:275–284
American Diabetes Association (2010) Diagnosis and classification of diabetes mellitus. Diabetes Care 33(Suppl 1): S62–S69
Frongia O, Mastinu F, Sechi GM (1997) Prevalence and 4-year incidence of insulin-dependent diabetes mellitus in the province of Oristano (Sardinia, Italy). Acta Diabetol 34:199–205
Bruining GJ, Molenaar JL, Grobbee DE et al (1989) Ten-year follow-up study of islet-cell antibodies and childhood diabetes mellitus. Lancet 1:1100–1103
Schatz D, Krischer J, Horne G et al (1994) Islet cell antibodies predict insulin-dependent diabetes in United States school age children as powerfully as in unaffected relatives. J Clin Invest 93:2403–2407
Levy-Marchal C, Dubois F, Noel M, Tichet J, Czernichow P (1995) Immunogenetic determinants and prediction of IDDM in French schoolchildren. Diabetes 44:1029–1032
Genovese S, Bingley PJ, Bonifacio E et al (1994) Combined analysis of IDDM-related autoantibodies in healthy schoolchildren. Lancet 10. 344(8924):756
Bonifacio E, Genovese S, Braghi S et al (1995) Islet autoantibody markers in IDDM: risk assessment strategies yielding high sensitivity. Diabetologia 38(7):816–822
Bingley PJ, Bonifacio E, Williams AJ et al (1997) Prediction of IDDM in the general population: strategies based on combinations of autoantibody markers. Diabetes 46(11):1701–1710
Samuelsson U, Sundkvist G, Borg H, Fernlund P, Ludvigsson J (2001) Islet autoantibodies in the prediction of diabetes in school children. Diabetes Res Clin Pract 51:51–57
Kulmala P, Rahko J, Savola K et al (2001) Beta-cell autoimmunity, genetic susceptibility, and progression to type 1 diabetes in unaffected schoolchildren. Diabetes Care 24:171–173
Knip M, Korhonen S, Kulmala P et al (2010) Prediction of type 1 diabetes in the general population. Diabetes Care 33(6):1206–1212
Lebastchi J, Deng S, Lebastchi AH et al (2013) Immune therapy and beta-cell death in type 1 diabetes. Diabetes 62:1676–1680
Jimenez-Gonzalez M, Jaques F, Rodriguez S et al (2013) Cardiotrophin 1 protects beta cells from apoptosis and prevents streptozotocin-induced diabetes in a mouse model. Diabetologia 56:838–846
Acknowledgment
S.M. was supported by Ministero della Salute (Rome, Italy, Grant No. ICS 120.2/RF99.17).
Conflict of interest
The authors declare that there is no conflict of interest associated with this manuscript.
Ethical standard
The study protocol conformed to the principles of the Declaration of Helsinki and was approved by Department of Medical Sciences “Mario Aresu” University of Cagliari, Ethics Committee.
Human and Animal Rights
This article does not contain any studies with human or animal subjects performed by any of the authors.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Managed by Massimo Porta.
The Sardinian Autoimmunity Study Group is given in the “Appendix”.
Appendix
Appendix
The author group is part of the Sardinia Autoimmunity Study Groups’, which include:
General Management
Overall Scientific and Administrative Responsibility: M. Songini (Cagliari), GF. Bottazzo (Rome)
General Co - ordinator: M. Locatelli (Rome);
The Immnunological and Genetic Study Group: Responsible: G.F. Bottazzo (Cagliari-Rome), E. Bonifacio (Milano).
Local Overall Responsibility: M. Songini (Cagliari)
The Epidemiology Study Group, The Statistical Group: Responsible: M. Songini (Cagliari);
The School Children Study Group: Responsibles: A. Loviselli, F. Velluzzi (Cagliari);
Co - ordinator: S. Mariotti (Cagliari);
Physicians and Laboratory Personnel: A. Pilo, G. Pinna, G. Secci, A. Taberlet.
Field workers: P. Mele, M.A. Calia, R. Mastinu, A. Pilleri, S. Serra, M. Diliberto, E. Rizzolo, N. Costantini.
Family GPs in the Provinces: L. Curreli, Serramanna (Ca), A. Trincas, Cabras (Or), M. Lara, S. Melis, L. Melis, G. Uggias, M. Incollu, Baunei (Og), S. Serra, Urzulei (Og), A. Seoni, S. Monni, Villagrande (Og), M. Muntoni, Ulassai (Og), M. Moi, Perdasdefogu (Og), F. Calzone, I. Ena, A. Manca, Bitti (Og), A. Lai, E. Floris, Tertenia (Og), M. Orrù, G. Melis, G. Mou, A. Deplano, Jerzu (Og).
CAD Sardinia:CAD Cagliari, CAD Sassari, CAD Oristano, CAD Nuoro, CAD Lanusei-Tortolì, CAD Olbia, CAD Tempio, CAD Carbonia, CAD Iglesias, CAD Quartu S.E., CAD Senorbì, CAD San Gavino M., CAD Bosa, CAD Isili, CAD Alghero, CAD Tempio Pausania, CAD Ozieri, CAD Santa Teresa di Gallura, CAD LA Maddalena, CAD Arzachena, CAD Bono, CAD Muravera, CAD Siniscola-Orosei, CAD Oristano.
Rights and permissions
About this article
Cite this article
Velluzzi, F., Secci, G., Sepe, V. et al. Prediction of type 1 diabetes in Sardinian schoolchildren using islet cell autoantibodies: 10-year follow-up of the Sardinian schoolchildren type 1 diabetes prediction study. Acta Diabetol 53, 73–79 (2016). https://doi.org/10.1007/s00592-015-0751-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-015-0751-y