Abstract
Impaired fasting glucose (IFG) and impaired glucose tolerance (IGT) are two intermediate states in the transition from normal glucose metabolism to type 2 diabetes. Insulin clamp studies have shown that subjects with IGT have increased insulin resistance in skeletal muscle, while subjects with IFG have near normal muscle insulin sensitivity. Because of the central role of altered free fatty acid (FFA) metabolism in the pathogenesis of insulin resistance, we have examined plasma free fatty acid concentration under fasting conditions, and during OGTT in subjects with IGT and IFG. Seventy-one NGT, 70 IGT and 46 IFG subjects were studied. Fasting plasma FFA in IGT subjects was significantly greater than NGT, while subjects with IFG had similar fasting plasma FFA concentration to NGT. However, fasting plasma insulin concentration was significantly increased in IFG subjects compared to NGT while subjects with IGT had near normal fasting plasma insulin levels. The adipocyte insulin resistance index (product of fasting plasma FFA and FPI) was significantly increased in both IFG and IGT subjects compared to NGT. During the OGTT both IFG and IGT subjects suppressed their plasma FFA concentration similarly to NGT subjects, but the post-glucose loads were significantly increased in both IFG and IGT subjects. These data suggest that both subjects with IFG and IGT have increased resistance to the antilipolytic action of insulin. However, under basal conditions, fasting hyperinsulinemia in IFG subjects is sufficient to offset the adipocyte insulin resistance and maintain normal fasting plasma FFA concentration while the lack of increase in FPI in IGT subjects results in an elevated fasting plasma FFA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Insulin resistance is a characteristic feature of type 2 diabetes [1], and much evidence supports an important role for increased plasma free fatty acid (FFA) concentrations in the pathogenesis of the insulin resistance [2]. Diabetic and non-diabetic insulin-resistant subjects have elevated plasma FFA levels compared to insulin-sensitive individuals [3, 4]. In normal healthy subjects, a small physiologic rise in plasma FFA concentration induces muscle insulin resistance and the resultant severity of insulin resistance is similar to that observed in subjects with type 2 diabetes mellitus [5]. Furthermore, reduction of the plasma FFA concentration in type 2 diabetic subjects with acipimox improves the insulin resistance [6]. Impaired fasting glucose (IFG) and impaired glucose tolerance (IGT) represent prediabetic states which are intermediate between normal glucose tolerance and type 2 diabetes [7] and manifest distinct metabolic abnormalities [8]. Recently, we have demonstrated that IGT subjects have severe insulin resistance in skeletal muscle, while individuals with IFG have normal muscle insulin sensitivity [9]. Because elevated plasma FFA levels have been implicated in the pathogenesis of muscle insulin resistance, we hypothesized that differences in the plasma FFA concentration may vary between these two states. Therefore, we have examined plasma FFA levels in IFG and IGT subjects in the fasting state and during an oral glucose tolerance test (OGTT) and compared the values to those in normal glucose tolerant (NGT) subjects.
Methods
All subjects were from the Veterans Administration Genetic Epidemiology Study (VEGAS) who, according to the ADA criteria [10], had isolated impaired fasting glucose (IFG) (FPG = 100–125 mg/dl, and 2 h PG < 140 mg/dl, n = 46), isolated impaired glucose tolerance (IGT) (FPG < 100 mg/dl and 2 h PG = 140–199 mg/dl, n = 70) or normal glucose tolerance NGT (FPG < 100 mg/dl and 2 h PG < 140 mg/dl, n = 71) and had a measurement of plasma free fatty acid concentration (FFA) under basal postabsorptive condition and during an OGTT. All subjects were of Mexican American descent and each subject received an OGTT performed following a 10–12 h overnight fasting. During OGTT, blood samples were collected at −30, −15, 0, 15, 30, 45, 60, 75, 90, 105 and 120 min for the measurement of plasma glucose and insulin concentrations; plasma FFA concentration was measured at −30, −15, 0, 60, 90 and 120 min. All subjects were healthy, and none were taking any medication known to affect glucose tolerance or lipid/FFA metabolism. No subject participated in any strenuous exercise program. Body weight was stable (±1.5 kg) for at least 3 months before study in all subjects. The study protocol was approved by the Institutional Review Board of the University of Texas Health Science Center at San Antonio, and informed written consent was obtained from all subjects before their participation.
Calculations
Since plasma insulin concentration is a strong inhibitory stimulus for lipolysis, the basal lipolytic insulin resistance index was calculated as the product of the fasting plasma insulin concentration and the log transformation of the fasting plasma FFA concentration [11]. Experimental validation of this index previously has been published [11]. Over the range of plasma insulin concentrations from 6 ± 1 μU/ml (fasting) to 10 ± 1 μU/ml (4 mU/m2 min euglycemic insulin clamp) to 22 ± 2 μU/ml (10 mU/m2 min euglycemic insulin clamp), the suppression of plasma FFA concentration (log transformed) and the suppression of plasma FFA (14C-palmitate) turnover (log transformed) is linear. Furthermore, over this range of plasma insulin concentrations, the correlation between the plasma FFA concentration and plasma FFA turnover is highly correlated (r = 0.96, p < 0.0001). Therefore, the product of the fasting plasma insulin and fasting plasma FFA (log transformed) concentrations over this range of hyperinsulinemia provides a valid index of adipocyte sensitivity to the antilipolytic effect of insulin.
Analytical techniques
Plasma glucose was measured by the glucose oxidase reaction (Glucose Oxidase Analyzer, Beckman, Fullerton, CA). Plasma insulin concentration was measured by radioimmunoassay. Plasma FFA was measured by a colorimetric assay (Wako Chemicals, Germany).
Statistical analysis
Data are presented as the mean ± standard deviation. To compare the mean of the groups ANOVA was used. When the means were significantly different, post hock testing was made with student t test. To adjust for possible covariates (sex, cholesterol and triglycerides), ANCOVA was used. Statistical significance was considered at p < 0.05. All statistical analysis were performed with JMP Ver 5.1.
Results
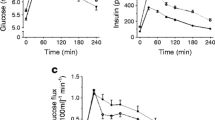
Table 1 presents the clinical and anthropometric characteristics of the three study groups, which were well matched for age, waist circumference and BMI. IFG is more common in males than females [7], not surprisingly, there was male predominance in the IFG group (p = 0.02 with chi square). Fasting plasma insulin concentration was different amongst the three study groups (Fig. 1) (p = 0.02 with ANOVA). Subjects with IFG had a significantly higher (p < 0.01) fasting plasma insulin concentration compared to subjects with IGT or NGT. Fasting plasma FFA concentration also differed amongst the three study groups (Fig. 1) (p = 0.006 with ANOVA). After adjustment for gender, fasting plasma FFA concentration differed amongst the three study groups (p = 0.01 with ANCOVA). Subjects with IFG had similar fasting plasma FFA concentration to subjects with NGT (668 ± 203 vs. 692 ± 201 μM, respectively, p = 0.37). However, IGT subjects had a significantly higher fasting plasma FFA concentration compared to NGT (780 ± 231 μM, p = 0.0008) and IFG (p < 0.001) subjects. The adipocyte insulin resistance index (product of fasting plasma FFA (log transformed) and fasting plasma insulin concentration) was different amongst the three study groups (p = 0.006 with ANOVA) and was significantly increased in both IFG (p = 0.01) and IGT (p < 0.01) subjects compared to NGT subjects (53 ± 48, 39 ± 29, and 29 ± 20, respectively) (Fig. 1), and the difference remained statistically different after adjustment for covariates (p = 0.03 with ANCOVA). Suppression of plasma FFA concentration during the OGTT was similar in all three groups (Fig. 2). However, the mean plasma concentration during the 2-h OGTT was significantly increased in IGT 190 ± 10 and IFG 226 ± 23 μM compared to 168 ± 10 μM in NGT subjects (p < 0.05 by ANOVA).
Discussion
Resistance of the peripheral tissues to the action of insulin plays a pivotal role in the pathogenesis of type 2 diabetes [1]. Much attention has focused on the insulin resistance in skeletal muscle because of its association with the metabolic syndrome and cardiovascular disease [12]. In this study, we demonstrate that subjects with IGT also manifest moderate-severe insulin resistance in adipocytes. In NGT subjects, the dose response curve for the inhibition of lipolysis by insulin is shifted to the left compared to dose response curve for insulin’s stimulatory effect on muscle glucose uptake [11]. Thus, small increases in plasma insulin concentration have a marked effect to inhibit lipolysis and to reduce the plasma FFA concentration. Over the range of plasma insulin concentrations from 4 to 24 μU/ml, we have shown that ability of insulin to inhibit lipolysis and reduce the plasma FFA concentration is linear [13]. Therefore, the product of the fasting plasma insulin and FFA concentrations provides a quantitative measure of the fat cells sensitivity to the antilipolytic effect of insulin. The adipocyte insulin resistance index was significantly increased in subjects with IGT compared to NGT, indicating an enhanced rate of lipolysis despite a normal fasting plasma insulin concentration. The increased fasting plasma FFA concentration in IGT subjects may contribute to the muscle insulin resistance that has been demonstrated in this prediabetic group with the euglycemic insulin clamp [9, 14, 15].
Subjects with IFG also manifested adipocyte insulin resistance as demonstrated by an increase in the adipocyte insulin resistance index (FPI X FFA). However, unlike subjects with IGT, their fasting plasma FFA concentration is maintained within the normal range. This is explained by the significant increase in fasting plasma insulin concentration, which is sufficient to offset the adipocyte insulin resistance and prevent the basal rate of lipolysis from increasing. We postulate that the normal fasting plasma FFA concentration in IFG subjects, in part, contributes to the normal insulin sensitivity that has been reported in this group [9, 14, 15].
Despite the adipocyte insulin resistance observed in the fasting state, the suppression of plasma FFA concentration during the OGTT in both IFG and IGT was similar to that in NGT. The normal suppression of plasma FFA concentration during the OGTT can be explained by the high plasma insulin concentration achieved during the OGTT (>90 μU/ml) (Table 1), which is more than sufficient to overcome the adipocyte insulin resistance and suppress lipolysis. This is consistent with previous studies which have demonstrated that a plasma insulin concentration of ∼50 μU/ml causes a near complete suppression of plasma FFA levels in type 2 diabetic subjects [13, 16, 17].
References
DeFronzo RA (2004) Pathogenesis of type 2 diabetes mellitus. Med Clin North Am 8:787–835
Bays H, Mandarino L, DeFronzo RA (2004) Role of the adipocyte, free fatty acids, and ectopic fat in pathogenesis of type 2 diabetes mellitus: peroxisomal proliferator-activated receptor agonists provide a rational therapeutic approach. J Clin Endocrinol Metab 89:463–478
Schalch DS, Kipnis DM (1965) Abnormalities in carbohydrate tolerance associated with elevated plasma nonesterified fatty acids. J Clin Invest 44:2010–2020
Reaven GM (1995) The fourth musketeer—from Alexandre Dumas to Claude Bernard. Diabetologia 38:3–13
Belfort R, Mandarino L, Kashyap S, Wirfel K, Pratipanawatr T, Berria R, Defronzo RA, Cusi K (2005) Dose-response effect of elevated plasma free fatty acid on insulin signaling. Diabetes 54:1640–1648
Bajaj M, Suraamornkul S, Romanelli A, Cline GW, Mandarino LJ, Shulman GI, DeFronzo RA (2005) Effect of a sustained reduction in plasma free fatty acid concentration on intramuscular long-chain fatty acyl-CoAs and insulin action in type 2 diabetic patients. Diabetes 54:3148–3153
Unwin N, Shaw J, Zimmet P, Alberti KGMM (2002) Impaired glucose tolerance and impaired fasting glycemia: the current status on definition and intervention. Diabet Med 19:708–723
Abdul-Ghani MA, Tripathy D, DeFronzo RA (2006) Contributions of beta cell dysfunction and insulin resistance to the pathogenesis of impaired glucose tolerance and impaired fasting glucose. Diabetes Care 29:1130–1139
Abdul-Ghani MA, Tripathy D, Jenckinson C, Ritchardson D, DeFronzo RA (2006) Insulin secretion and insulin action in subjects with impaired fasting glucose and impaired glucose tolerance: results from the Veterans Administration Genetic Epidemiology Study (VEGAS). Diabetes 55:1430–1435
The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (1997) Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 20:1183–1197
Groop LC, Bonadonna RC, Del Prato S, Ratheiser K, Zyck K, Ferrannini E, DeFronzo RA (1989) Glucose and free fatty acid metabolism in non-insulin-dependent diabetes mellitus. Evidence for multiple sites of insulin resistance. J Clin Invest 84:205–213
DeFronzo RA (1997) Insulin resistance: a multifaceted syndrome responsible for NIDDM, obesity, hypertension, dyslipidaemia and atherosclerosis. Neth J Med 50:191–197
Bonadonna RC, Groop LC, Zych K, Shank M, DeFronzo RA (1990) Dose-dependent effect of insulin on plasma free fatty acid turnover and oxidation in humans. Am J Physiol 259:E736–E750
Weyer C, Bogardus C, Pratley RE (1999) Metabolic characteristics of individuals with impaired fasting glucose and/or impaired glucose tolerance. Diabetes 48:2197–2203
Wasada T, Kuroki H, Katsumori K, Arii H, Sato A, Aoki K, Jimba S, Hanai G (2004) Who are more insulin resistant, people with IFG or people with IGT? Diabetologia 47:758–759
Robinson C, Tamborlane WV, Maggs DG, Enoksson S, Sherwin RS, Silver D, Shulman GI, Caprio S (1998) Effect of insulin on glycerol production in obese adolescents. Am J Physiol 274:E737–E743
Campbell PJ, Carlson MG, Nurjhan N (1994) Fat metabolism in human obesity. Am J Physiol 266:E600–E605
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdul-Ghani, M.A., Molina-Carrion, M., Jani, R. et al. Adipocytes in subjects with impaired fasting glucose and impaired glucose tolerance are resistant to the anti-lipolytic effect of insulin. Acta Diabetol 45, 147–150 (2008). https://doi.org/10.1007/s00592-008-0033-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-008-0033-z