Abstract
Purpose
To provide treatment guidelines for patients with long bone metastatic disease based on a systematic review of the literature and to propose an algorithm to guide orthopedic surgeons in decision-making for these patients.
Materials and methods
We performed a computerized literature search in MEDLINE, EMBASE and Scopus for studies on patients with long bone metastases. We used the key words “long bones”, “metastasis” and “treatment” for published studies that evaluated any treatment for long bone metastases. The articles found were then studied to determine the accuracy of surgical treatments for long bone metastases in every anatomic location, regardless of cancer type, stage and grade of the oncologic disease. Guidelines inferred from this literature review were collected, and an algorithm was proposed.
Results
There was no clear evidence to support excision of a long bone metastatic lesion at the same surgical setting with internal fixation or prosthetic reconstruction. However, en bloc resection of an isolated bone metastasis may have a beneficial effect on survival. The life expectancy of the patients should be considered for any surgical treatment. Internal fixation preferably with reconstruction nails is indicated for meta-diaphyseal lesions; their rate of mechanical failure and complications ranges from 2 to 22 %. Prosthetic reconstruction is indicated for extensive lytic lesions or pathologic fractures in a meta-epiphyseal locations; their rate of mechanical failure and complications ranges from 3.7 to 35 %. Most of the internal fixation-related complications occur more than 1 year after treatment, in contrast to prosthetic reconstruction-related complications that may occur earlier.
Conclusions
Intramedullary nail fixation or prosthetic reconstruction should be chosen on the basis of the location of the lesion, the extent of bone destruction and the stability of the construct to outlast the expected life of the patient. Implant-related complication is similar but may occur earlier with prosthetic reconstructions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Currently, the prevalence of metastatic bone disease in developed countries is more than 280,000 new cases per year [1]; this number is expected to rise as patients with cancer live longer [2]. Bone is the third most common site of metastatic disease after the lung and the liver. The most common primary cancers are lung, breast, prostate, thyroid and renal cancer. Postmortem analysis shows that approximately 70 % of all patients with breast and prostate cancer and 35–42 % of patients with lung, thyroid and renal cancer have skeletal metastases [3]. The financial burden of treating patients with metastatic bone disease in the USA per year estimates approximately 12.6 billion dollars, which accounts for 17 % of the total annual cost of cancer treatments [4].
In the past, the occurrence of a pathological fracture through bone sarcomas or skeletal metastases was an absolute contraindication for limb salvage; in this setting, treatment traditionally consisted of amputation proximal to the most superior aspect of the fracture hematoma. Currently, the decision for limb salvage surgery has been reconsidered [1–12]. Although malignant primary bone tumors are usually referred for treatment at tertiary tumors centers, patients with metastatic bone disease may be treated by general orthopedic surgeons at community hospitals [2]. In any case, surgical considerations of patients with bone metastases should include multiple factors such as the location of the tumor within bone and the skeleton, the histology of the primary tumor, the pain, comorbidities and expected life span of the patients. Impending or actual pathological fractures of the long bones in patients with primary and metastatic bone tumors are a difficult and challenging complication that may alter management and prognosis, decrease quality of life and jeopardize survival [1–12]. In general, stabilization of an impending or pathological fracture involves techniques and concepts that differ from those used for patients with non-pathologic fractures [2]. Bone involved by metastatic cancer is weakened and requires stabilization or reconstruction with an implant or prosthetic device that should last the remaining of patient’s life and to stabilize the entire diseased bone [2]. Surgical complications should be minimized, as complications in cancer patients are unwanted. Methyl methacrylate is often used in combination with prostheses or intramedullary devices to supplement the fixation [2]. Radiation therapy and embolization often are used as a local adjuvant after surgical treatment or for palliation [13, 14].
Pathological long bones fractures pose many difficulties to the most experienced surgeons. Their management may alter prognosis, affecting both the quality of life and survival of the patients [12]. Previous studies have reported on the diagnosis, management and survival of cancer patients with metastatic bone disease and pathological fractures [2–38]; their results cannot be decisive regarding the optimal management of the patients and the survival of the reconstructions because of the lack of control groups and short term survival of these patients. Moreover, studies that propose treatment guidelines for cancer patients with bone metastases are limited [12, 39], and most surgeons treat these patients according to standard practice for fractures or their own preference. Therefore, to enhance the literature, this study aims to provide practical guidelines for the treatment of patients with long bone metastases based on a systematic review of the literature and to propose an algorithm to guide the treating orthopedic surgeons in their decision-making for these patients. We believe that the findings of the present article and the proposed treatment algorithm based on the experience and current practice of a tertiary tumor center with a relatively large experience on the treatment of tumor patients would be useful for the treatment approach of patients with long bone metastases.
Materials and methods
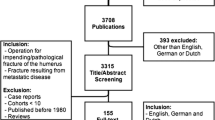
The search strategy included a computerized literature search in MEDLINE, EMBASE and Scopus. A systematic review of clinical studies that evaluated any treatment of long bone metastases was done using as keywords “long bones”, “metastases” and “treatment”. The reference lists of the selected papers were also checked for further relevant publications. Papers in other than English language and publication date before 2000 were excluded. The articles were then censored if they were case reports, review or opinion articles, or technique descriptions. From a total of 398 articles found, 19 articles were related to treatment of a clinical cohort of patients with metastatic disease of the long bones (Table 1) [2, 6–9, 21, 24, 25, 27–37]. There was no disagreement between the authors of the present study regarding the level of evidence of the included 19 articles. These articles were studied to determine the accuracy of surgical treatments for long bone metastases in every anatomic location, regardless of cancer type, stage and grade of the oncologic disease. Last, guidelines for the treatments of long bone metastases were inferred from the review analysis.
Results
The most common primary cancer was breast followed by lung, prostate, renal and thyroid cancer; the femur was the most commonly affected site. Long bone metastatic lesions did not always require surgical intervention; the size and location of the metastatic lesion, the general health status and expected survival of the patients influenced the decision-making regarding the optimal treatment. The indications for surgery of long bone metastases were an impending or an actual pathological fracture and/or severe, constant pain; patients were operated if an expected survival of more than 6 weeks was estimated. Patients with a long bone metastasis to the lower extremities were more commonly operated compared to those with a long bone metastasis to the upper extremities.
There was no clear evidence to support resection of the metastatic lesion at the same surgical setting with internal fixation or prosthetic reconstruction of the metastatic lesion. The overall incidence of local disease progression was 11.5 %, while subsequent development of new lesions in the diseased bone was only 1 %. However, wide resection of a solitary bone metastasis, especially from renal cancer was associated with a more beneficial effect on survival of the patients compared with that of patients treated with less than a wide resection.
Internal fixation preferably with a reconstruction type intramedullary nail was indicated for meta-diaphyseal lesions; their rate of mechanical failure ranged from 2 to 22 %, but perioperative deaths from cardiopulmonary complications ranged from 1 to 10 %. Prosthetic reconstruction was indicated for extensive lytic lesions or pathological fractures in meta-epiphyseal locations; their rate of mechanical failure rate was less than 3.7 %, with an overall complications rate ranging from 6 to 35 %. Most of the internal fixation-related complications occurred more than 1 year after diagnosis and treatment, in contrast to prosthetic reconstruction-related complications that occurred earlier.
Local adjuvants were extensively used as per surgeons’ own preference. Cement augmentation was used for additional mechanical stability of the fixation. Radiation therapy or embolization has been used to slow local disease progression and avoid any subsequent failure of the fixation.
Discussion
Improvements in imaging, surgery and medical oncology treatments have greatly increased the survival of cancer patients [3–5], including those with metastatic disease [6]. However, the occurrence of a skeletal event from cancer significantly influences the quality of life of these patients. In these cases, the indications for surgical treatment vary from pain to impending or actual pathological fracture, and seem to differ between countries [5]; in the USA, up to 71 % of patients with metastatic cancers have been treated due to impending pathological fractures compared to only 18 % in the Nordic countries [6]. Decisions regarding potential surgery for metastatic bone disease require reliable data about the patients’ expected survival and quality of life [15].
Patients with metastatic bone disease are often treated at community hospitals [2]. To our opinion and the related literature, these patients should be better referred for treatment at tertiary tumor centers [1–6, 12]. In cases that a musculoskeletal tumor trained orthopedic surgeon will not be the treating physician, general orthopedic surgeons should be familiar with the management of these patients. More importantly, the treating orthopedic surgeon should know that a metastatic bone lesion may not reliably heal [2]. As the treatment of metastatic bone disease is multidisciplinary, it is imperative that orthopedic surgeons are involved at an early stage and not only follow the patients with pathological fractures, as early treatment, management of pain, and improvement in the functional status is particularly important for cancer patients, especially those with a short life expectancy [6].
Prognostic life expectancy of patients with skeletal metastasis is important for the choice of the appropriate treatment. Previous studies by orthopedic surgeons [40–42] and radiation oncologists [43–45] reported on the prognostic factors for patients with skeletal metastasis. Tomita et al. [40] and Bauer et al. [42] involved analysis of patients who had undergone surgery. Tokuhashi et al. [41] studied both surgically and conservatively treated patients. In a recent prospective study, the number of surgeries performed was approximately 7 % of that of radiotherapies [46]. Therefore, previously published studied in orthopedic oncology literature might not be representative of patients with bone metastasis [47]. Katagiri et al. [47] identified six significant prognostic factors for survival: the primary lesion, visceral or cerebral metastases, abnormal laboratory data, poor performance status, previous chemotherapy and multiple skeletal metastases. The prognostic score was calculated by adding together all the scores for individual factors. With a prognostic score of ≥7, the survival rate was 27 % at 6 months and only 6 % at 1 year; in contrast, patients with a prognostic score of ≤ 3 had a survival rate of 91 % at 1 year and 78 % at 2 years. This scoring system was able to predict the survival rates of patients with skeletal metastases more accurately than previous scoring systems and may be useful for selecting an optimal treatment [47].
Mirels’ scoring system [16] seems to be the most detailed used classification system for the evaluation and decision-making for treatment of a long bone metastatic lesion (Fig. 1) [2]. This system is based on the size, site and imaging of the lesion, and the presence of pain [16]; a score higher than 8 points is associated with an impending fracture and indicates the need for prophylactic surgery [16]. However, this method does not take into account the life expectancy of the patient and which type of prophylactic surgery is better to use [38]. Conventional implants may not achieve rigid fixation in patients with long bone metastasis. Acrylic bone cement augmentation and/or adjuvant treatments, such as embolization or radiation therapy, are often mandatory to obtain local disease control and to avoid mechanical failure of the implant [2].
There is controversy regarding the optimal surgical treatment for patients with a solitary bone metastasis, especially for patients with renal cancer [6, 17–20]. Some authors reported that en bloc resection of a solitary metastasis was a positive prognostic factor for an improved survival [6, 17]; however, other authors reported that en bloc resection did not affect the survival of the patients [18–20]. Additionally, there is no agreement that en bloc resection of a metastasis in patients with multiple metastases affects the survival of the patients when compared with intralesional surgery [6]. There is also controversy regarding the complications and reoperation rates after surgery for bone metastases. Some authors reported that the complications rate seems to be lower after en bloc resection compared to marginal or intralesional surgery [6, 7, 21, 22]. We concur that en bloc resection of a metastatic lesion may prevent local progression of the disease and therefore mechanical complications and reoperations [6, 8, 23–25]. However, most complications after intramedullary nailing for long bone metastases occur late, more than 1 year after diagnosis and treatment, which should be considered in treatment decision-making [7]. In contrast, prosthetic reconstruction usually allows for immediate postoperative stability and weight bearing, and has been associated with lower mechanical failure rates compared to intramedullary nailing [8, 23–25]. Therefore, prosthetic reconstruction seems to be the more durable surgical option for patients with a longer expectancy survival, especially if e bloc resection of the metastatic lesion can be obtained [24, 26].
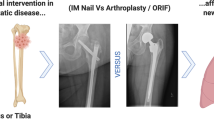
Based on the analysis of the literature [2, 6–9, 21, 24, 25, 27–37, 39], the questions raised by this manuscript are still challenging. After decades of treating tumor patients with different methods and approaches, we obtained great experience for the management of these patients, and we propose treatment guidelines for patients with long bone pathological fractures from metastatic bone tumors (Fig. 2). Surgeons should follow oncological principles in treating tumor patients. Referral to a specialized orthopedic oncology center is generally recommended. Treatment decision requires complete staging and oncological principles. Intramedullary fixation or prosthetic reconstruction should be chosen on the basis of the location of the lesion, the extent of bone destruction and the ability of the construct to last the life expectancy of the patients. If surgical treatment is necessary, the life expectancy of the patients should be considered. We consider expected survival of less than 6 weeks as a relative contraindication for surgical management of any impending or actual pathological fractures, especially if in the upper extremity. For patients with a life expectancy of 3–12 months, we opt for less invasive surgical reconstruction procedures that do not need prolonged rehabilitation [5]. For patients with longer life expectancy (>12 months), we aim for en bloc resection of a metastasis and opt for durable reconstruction procedures such as prosthetic reconstructions, even if associated with longer rehabilitation times [5]. The histology, response to chemotherapy and radiation therapy of the primary cancer, the presence of visceral metastases, the number of skeletal metastases and the overall health status of the patients seem to be the most important predictors of survival [5, 6]. We believe that this algorithm based on our experience and analysis of the literature, would be helpful for decision-making and treatment approach of patients with long bone pathological fractures from metastatic bone tumors.
Mirels’ classification for impending pathological fractures [16]
References
Cheung FH (2014) The practicing orthopedic surgeon’s guide to managing long bone metastases. Orthop Clin North Am 45:109–119
Weber KL, Randall RL, Grossman S, Parvizi J (2006) Management of lower-extremity bone metastasis. J Bone Jt Surg Am 88(Suppl 4):11–19
Coleman RE (2006) Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res 12(20 Pt 2):6243s–6249s
Schulman KL, Kohles J (2007) Economic burden of metastatic bone disease in the US. Cancer 109:2334–2342
Forsberg JA, Eberhardt J, Boland PJ, Wedin R, Healey JH (2011) Estimating survival in patients with operable skeletal metastases: an application of a bayesian belief network. PLoS One 6:e19956
Ratasvuori M, Wedin R, Hansen BH, Keller J, Trovik C, Zaikova O et al (2014) Prognostic role of en-bloc resection and late onset of bone metastasis in patients with bone-seeking carcinomas of the kidney, breast, lung, and prostate: SSG study on 672 operated skeletal metastases. J Surg Oncol 110:360–365
Harvey N, Ahlmann ER, Allison DC, Wang L, Menendez LR (2012) Endoprostheses last longer than intramedullary devices in proximal femur metastases. Clin Orthop Relat Res 470:684–691
Wedin R, Bauer HCF (2005) Surgical treatment of skeletal metastatic lesions of the proximal femur: endoprosthesis or reconstruction nail? J Bone Jt Surg Br 87:1653–1657
Ward WG, Holsenbeck S, Dorey FJ, Spang J, Howe D (2003) Metastatic disease of the femur: surgical treatment. Clin Orthop Relat Res 415(Suppl):S230–S244
Hunt KJ, Gollogly S, Randall RL (2006) Surgical fixation of pathologic fractures: an evaluation of evolving treatment methods. Bull Hosp Jt Dis 63(3–4):77–82
van Doorn R, Stapert JW (2000) Treatment of impending and actual pathological femoral fractures with the long Gamma nail in The Netherlands. Eur J Surg Acta Chir 166:247–254
Ruggieri P, Mavrogenis AF, Casadei R, Errani C, Angelini A, Calabrò T, Pala E, Mercuri M (2010) Protocol of surgical treatment of long bone pathological fractures. Injury 41(11):1161–1167
Facchini G, Di Tullio P, Battaglia M, Bartalena T, Tetta C, Errani C, Mavrogenis AF, Rossi G (2016) Palliative embolization for metastases of the spine. Eur J Orthop Surg Traumatol 26(3):247–252
Rossi G, Mavrogenis AF, Casadei R, Bianchi G, Romagnoli C, Rimondi E, Ruggieri P (2013) Embolisation of bone metastases from renal cancer. Radiol Med 118(2):291–302
Nathan SS, Healey JH, Mellano D, Hoang B, Lewis I, Morris CD, Athanasian EA, Boland PJ (2005) Survival in patients operated on for pathologic fracture: implications for end-of-life orthopedic care. J Clin Oncol 23(25):6072–6082
Mirels H (1989) Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin Orthop Relat Res 249:256–264
Fottner A, Szalantzy M, Wirthmann L, Stähler M, Baur-Melnyk A, Jansson V et al (2010) Bone metastases from renal cell carcinoma: patient survival after surgical treatment. BMC Musculoskelet Disord 11:145
Lin PP, Mirza AN, Lewis VO, Cannon CP, Tu S-M, Tannir NM et al (2007) Patient survival after surgery for osseous metastases from renal cell carcinoma. J Bone Jt Surg Am 89:1794–1801
Evenski A, Ramasunder S, Fox W, Mounasamy V, Temple HT (2012) Treatment and survival of osseous renal cell carcinoma metastases. J Surg Oncol 106:850–855
Ruggieri P, Mavrogenis AF, Angelini A, Mercuri M (2011) Metastases of the pelvis: does resection improve survival? Orthopedics 34(7):e236–e244
Steensma M, Boland PJ, Morris CD, Athanasian E, Healey JH (2012) Endoprosthetic treatment is more durable for pathologic proximal femur fractures. Clin Orthop Relat Res 470:920–926
Van der Linden YM, Dijkstra PDS, Kroon HM, Lok JJ, Noordijk EM, Leer JWH et al (2004) Comparative analysis of risk factors for pathological fracture with femoral metastases. J Bone Jt Surg Br 86:566–573
Cannon CP, Mirza AN, Lin PP, Lewis VO, Yasko AW (2008) Proximal femoral endoprosthesis for the treatment of metastatic. Orthopedics 31:361
Chandrasekar CR, Grimer RJ, Carter SR, Tillman RM, Abudu A, Buckley L (2009) Modular endoprosthetic replacement for tumours of the proximal femur. J Bone Jt Surg Br 91:108–112
Selek H, Başarir K, Yildiz Y, Sağlik Y (2008) Cemented endoprosthetic replacement for metastatic bone disease in the proximal femur. J Arthroplasty 23:112–117
Park DH, Jaiswal PK, Al-Hakim W, Aston WJS, Pollock RC, Skinner JA et al (2007) The use of massive endoprostheses for the treatment of bone metastases. Sarcoma 2007:62151
Nilsson J, Gustafson P (2008) Surgery for metastatic lesions of the femur: good outcome after 245 operations in 216 patients. Injury 39(4):404–410
Wedin R, Hansen BH, Laitinen M, Trovik C, Zaikova O, Bergh P, Kalén A, Schwarz-Lausten G, Vult von Steyern F, Walloe A, Keller J, Weiss RJ (2012) Complications and survival after surgical treatment of 214 metastatic lesions of the humerus. J Shoulder Elbow Surg 21(8):1049–1055
Scotti C, Camnasio F, Peretti GM, Fontana F, Fraschini G (2008) Modular prostheses in the treatment of proximal humerus metastases: review of 40 cases. J Orthop Traumatol 9(1):5–10
Miller BJ, Soni EE, Gibbs CP, Scarborough MT (2011) Intramedullary nails for long bone metastases: why do they fail? Orthopedics 34(4):274
Potter BK, Chow VE, Adams SC, Letson GD, Temple HT (2009) Endoprosthetic proximal femur replacement: metastatic versus primary tumors. Surg Oncol 18(4):343–349
Weiss RJ, Ekström W, Hansen BH, Keller J, Laitinen M, Trovik C, Zaikova O, Wedin R (2013) Pathological subtrochanteric fractures in 194 patients: a comparison of outcome after surgical treatment of pathological and non-pathological fractures. J Surg Oncol 107(5):498–504
Finstein JL, King JJ, Fox EJ, Ogilvie CM, Lackman RD (2007) Bipolar proximal femoral replacement prostheses for musculoskeletal neoplasms. Clin Orthop Relat Res 459:66–75
Sarahrudi K, Greitbauer M, Platzer P, Hausmann JT, Heinz T, Vécsei V (2009) Surgical treatment of metastatic fractures of the femur: a retrospective analysis of 142 patients. J Trauma 66(4):1158–1163
Zacherl M, Gruber G, Glehr M, Ofner-Kopeinig P, Radl R, Greitbauer M, Vecsei V, Windhager R (2011) Surgery for pathological proximal femoral fractures, excluding femoral head and neck fractures: resection vs. stabilisation. Int Orthop 35(10):1537–1543
Manoso MW, Frassica DA, Lietman ES, Frassica FJ (2007) Proximal femoral replacement for metastatic bone disease. Orthopedics 30(5):384–388
Alvi HM, Damron TA (2013) Prophylactic stabilization for bone metastases, myeloma, or lymphoma: do we need to protect the entire bone? Clin Orthop Relat Res 471(3):706–714
Mavrogenis AF, Pala E, Romagnoli C, Romantini M, Calabro T, Ruggieri P (2012) Survival analysis of patients with femoral metastases. J Surg Oncol 105(2):135–141
Willeumier JJ, van der Linden YM, van de Sande MAJ, Dijkstra PDS (2016) Treatment of pathological fractures of the long bones. EFORT Open Rev 1:136–145
Tomita K, Kawahara N, Kobayashi T, Yoshida A, Murakami H, Akamaru T (2001) Surgical strategy for spinal metastases. Spine (Phila Pa 1976) 26(3):298–306
Tokuhashi Y, Matsuzaki H, Oda H, Oshima M, Ryu J (2005) A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine (Phila Pa 1976) 30(19):2186–2191
Bauer HC, Wedin R (1995) Survival after surgery for spinal and extremity metastases. Prognostication in 241 patients. Acta Orthop Scand 66(2):143–146
Rades D, Fehlauer F, Schulte R, Veninga T, Stalpers LJ, Basic H, Bajrovic A, Hoskin PJ, Tribius S, Wildfang I, Rudat V, Engenhart-Cabilic R, Karstens JH, Alberti W, Dunst J, Schild SE (2006) Prognostic factors for local control and survival after radiotherapy of metastatic spinal cord compression. J Clin Oncol 24(21):3388–3393
van der Linden YM, Dijkstra SP, Vonk EJ, Marijnen CA, Leer JW, Dutch Bone Metastasis Study Group (2005) Prediction of survival in patients with metastases in the spinal column: results based on a randomized trial of radiotherapy. Cancer 103(2):320–328
Mizumoto M, Harada H, Asakura H, Hashimoto T, Furutani K, Hashii H, Takagi T, Katagiri H, Takahashi M, Nishimura T (2008) Prognostic factors and a scoring system for survival after radiotherapy for metastases to the spinal column: a review of 544 patients at Shizuoka Cancer Center Hospital. Cancer 113(10):2816–2822
Lipton A, Fizazi K, Stopeck AT, Henry DH, Brown JE, Yardley DA, Richardson GE, Siena S, Maroto P, Clemens M, Bilynskyy B, Charu V, Beuzeboc P, Rader M, Viniegra M, Saad F, Ke C, Braun A, Jun S (2012) Superiority of denosumab to zoledronic acid for prevention of skeletal-related events: a combined analysis of 3 pivotal, randomised, phase 3 trials. Eur J Cancer 48(16):3082–3092
Katagiri H, Okada R, Takagi T, Takahashi M, Murata H, Harada H, Nishimura T, Asakura H, Ogawa H (2014) New prognostic factors and scoring system for patients with skeletal metastasis. Cancer Med 3(5):1359–1367
Acknowledgments
The authors thank Cristina Ghinelli, medical illustrator, for the design of the figures for this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Errani, C., Mavrogenis, A.F., Cevolani, L. et al. Treatment for long bone metastases based on a systematic literature review. Eur J Orthop Surg Traumatol 27, 205–211 (2017). https://doi.org/10.1007/s00590-016-1857-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-016-1857-9