Abstract
Purpose
Postoperative pain following scoliosis correction surgery is severe and usually requires prolonged intravenous opioid therapy. Regional anesthesia options are limited and include intrathecal opioid and epidural analgesia; however, they remain little used because of side effects and inconsistent efficacy. We describe a novel multimodal anesthetic regimen incorporating bilateral bi-level erector spinae plane (ESP) blocks together with a combination of several evidence-based intraoperative opioid-sparing analgesic strategies.
Methods
Two healthy young adult patients with idiopathic scoliosis underwent posterior spinal fusion involving 12 vertebral levels (T2–L1 and T3–L2). Bilateral single-injection ESP blocks were performed at two levels (T4 and T10) prior to incision. Intraoperatively, patients received intravenous dexamethasone and infusions of dexmedetomidine and ketamine for multimodal analgesia. Remifentanil was omitted from the total intravenous anesthetic regimen to avoid opioid-induced hyperalgesia.
Results
Both patients had minimal pain on emergence. They transitioned successfully to oral analgesia on the first postoperative day, with modest opioid requirements, no side effects, and low pain scores throughout their hospital stay.
Conclusion
Bilateral bi-level ESP blocks are a simple method of providing pre-emptive regional analgesia in extensive multi-level spine surgery. Integration of ESP blocks into a multimodal regimen that employs other opioid-sparing strategies may have additive, and potentially synergistic, benefits in improving postoperative analgesia and reducing opioid requirements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Posterior spinal fusion for scoliosis correction is extremely painful and usually requires prolonged and significant opioid use for adequate perioperative analgesia [1, 2]. Regional anesthesia is an important component of multimodal analgesia but the options are limited. Intrathecal or epidural opioid injection and epidural local anesthetic infusion are reported but little used because of logistical complexity, side effects, and inconsistent analgesic efficacy [3,4,5,6]. The erector spinae plane (ESP) block was originally described for thoracoabdominal analgesia by anesthetizing ventral rami of spinal nerves [7, 8]. However, it also effectively blocks the dorsal rami innervating the back (Fig. 1) [9,10,11,12]. In this report, we describe a unique anesthetic regimen for opioid-sparing analgesia after posterior spinal fusion spanning 12 vertebral segments. This incorporates (1) bilateral ESP blocks performed at two levels (T4 and T10) and (2) a combination of intraoperative pharmacologic adjuncts previously shown to improve analgesic outcomes in spine surgery when used in isolation [13,14,15,16]. The benefits of this comprehensive multimodal approach are illustrated in two healthy young patients undergoing idiopathic scoliosis correction. Written informed consent was obtained from both patients for inclusion in this report.
The erector spinae plane block is performed by injection of local anesthetic into the musculofascial plane between the deep aspect of the erector spinae muscle and the transverse processes. This local anesthetic (green oval) spreads to effectively anesthetize the branches of the dorsal rami of the spinal nerves that travel through this plane. Image adapted and used with permission from Maria Fernanda Rojas Gomez
Case description
Perioperative anesthetic regimen
Prior to induction of general anesthesia, bilateral ESP blocks were performed at T4 and T10 vertebral levels by an experienced regional anesthesiologist. These levels were chosen by dividing the extent of the planned incision into two and injecting at the approximate midpoint of each half (Fig. 2). In each ESP block, a 22-gauge needle was inserted in-plane to a linear-array ultrasound transducer placed in a longitudinal orientation over the tip of the transverse processes. The needle was directed in a caudal-to-cranial direction at T4 and a cranial-to-caudal direction at T10. Entry into the musculofascial plane between transverse process and erector spinae muscle was confirmed by hydrolocation with 1–2 mL of 5% dextrose, following which 15–20 mL of 0.25% bupivacaine with epinephrine 5 mcg/mL was injected (Fig. 3). The volume administered was calculated so as not to exceed a total bupivacaine dose of 3 mg/kg.
Ultrasound images of injection at the transverse processes (TP) of T5 and T10 vertebrae in the second patient. Note that the erector spinae muscle (ESM) is significantly thicker and the TP deeper at the T10 level. The needle has to be advanced at a steeper trajectory and is less visible as a result (indicated by dotted line). Nevertheless, local anesthetic (LA) spread is clearly visible as a hypoechoic layer that lifts the ESM off the TP
Following block completion, general anesthesia was induced and maintained using intravenous (IV) infusions of propofol 80–130 mcg/kg/min, dexmedetomidine 0.2–0.4 mcg/kg/min, and ketamine 0.5 mg/kg/h (Table 1), titrated to achieve hemodynamic stability and adequate anesthetic depth (Entropy™ (GE Healthcare, Helsinki, Finland) values between 45 and 65). Dexmedetomidine and ketamine infusions were stopped at the commencement of wound closure. Local anesthetic infiltration of the wound was not performed. Somatosensory and motor evoked potential monitoring was routine in all cases.
Postoperative analgesia consisted of oral acetaminophen 1 g 6-hourly, and IV patient-controlled analgesia (PCA) with hydromorphone (bolus 0.2–0.4 mg, lockout interval 5 min, no background infusion). Patients were assessed daily by the acute pain service team and IV-PCA was converted to oral opioid therapy when deemed appropriate.
Case 1
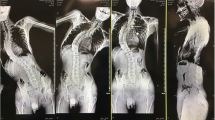
A 22-year-old man presented for T2–L1 scoliosis correction and instrumented fusion (Fig. 4a, b). IV midazolam 2 mg was administered for sedation and the ESP blocks performed in the sitting position. General anesthesia was induced with IV fentanyl 150mcg, lidocaine 100 mg, propofol 150 mg, and rocuronium 50 mg. IV dexamethasone 8 mg and hydromorphone 1 mg were administered prior to incision (Table 1). Surgery lasted 5 h and was uneventful with estimated blood loss (EBL) of 1195 mL. Additional intraoperative analgesics included IV ketorolac 30 mg and fentanyl 100mcg administered at completion of wound closure. The patient was extubated awake 105 min after cessation of dexmedetomidine and ketamine and reported no pain in his back. He required no analgesics in the PACU and was able to lift both legs and roll over on his side for wound inspection without discomfort. The first PCA demand occurred 1 h after extubation, and the IV-PCA was discontinued on the morning of postoperative day (POD) 1, with the patient having used IV hydromorphone 4.4 mg over the preceding 15 h. He did not experience any opioid-related side effects. Analgesia was provided by oral acetaminophen 1 g 6-hourly and hydromorphone 2–4 mg 2-hourly as needed until hospital discharge on POD5. Opioid consumption and pain scores are summarized in Table 1.
Case 2
A 21-year-old woman presented for T3–L2 scoliosis correction and instrumented fusion (Fig. 4c, d). Oral acetaminophen 1 g and celebrex 200 mg was administered upon hospital admission. IV midazolam 2 mg was administered for sedation and the ESP blocks performed in the prone position. General anesthesia was induced with IV midazolam 2 mg, propofol 300 mg, lidocaine 100 mg, and rocuronium 40 mg. IV dexamethasone 10 mg was administered prior to incision. Surgery lasted four hours and was uneventful with EBL 1900 mL. No additional intraoperative analgesics were administered. The patient was extubated awake 50 min after cessation of dexmedetomidine and ketamine and reported mild pain, for which she received IV hydromorphone 1 mg in PACU. Shortly thereafter, she reported no pain in the back and was able to perform straight-leg raises without discomfort. No further analgesics were administered in PACU and her first PCA demand was initiated 2 h after extubation. The IV-PCA was discontinued on the morning of POD1, with the patient having used IV hydromorphone 4.8 mg over the preceding 18 h. She did not experience any opioid-related side effects. Analgesia was provided by oral acetaminophen 1 g 6-hourly and hydromorphone 2–4 mg 2-hourly as needed until hospital discharge on POD4. Opioid consumption and pain scores are summarized in Table 1.
Discussion
Most patients undergoing posterior spinal fusion for scoliosis correction require IV-PCA opioid for 36 h or more and report moderate-to-severe pain over the first several postoperative days [1, 2]. This brief report suggests that the application of ESP blocks, combined with intraoperative use of multiple non-opioid analgesic modalities, can significantly improve the pain trajectory and reduce opioid consumption.
Intrathecal or epidural opioid injection and surgically-inserted epidural catheters are alternative regional anesthesia strategies; however, they have several significant limitations. The analgesic duration of intrathecal opioid is dose-dependent, limited to 12–24 h, and must be weighed against side effects of pruritus, nausea and vomiting, sedation, and respiratory depression [3, 4]. Epidural opioid is associated with similar adverse effects and may be less effective [4]. Epidural analgesia with local anesthetic infusion is resource-intensive, and concerns include adverse effects of epidural opioids, hypotension, and leg weakness. Analgesia is often incomplete and significant benefits are only seen if two catheters are placed [5, 6]; this is likely due to the extent of surgery and surgical disruption of the epidural space. Local anesthetic wound infiltration at closure is a simple and commonly used option; however, a recent meta-analysis found that the analgesic benefit was modest and not evident beyond the first few postoperative hours [17].
The ESP block, on the other hand, has few adverse effects and provides effective analgesia by blocking the dorsal rami of spinal nerves as they course through the musculofascial plane where local anesthetic is deposited [9,10,11,12]. Physical spread over at least 4–6 vertebral levels in the erector spinae plane from a single injection of 20 mL [7, 8, 10, 11, 18] makes coverage of the entire surgical incision in scoliosis surgery feasible with two injections per side. The risk of local anesthetic systemic toxicity is minimized by the addition of epinephrine and by observing recommended maximum bupivacaine dose limits. Unlike surgical wound infiltration, ESP blocks can also be performed prior to incision, which minimizes intraoperative opioid requirements, pain “windup” and central sensitization [19], and contributes to preventive analgesia [20]. Although physical evidence of epidural and paravertebral spread of local anesthetic has been described with the ESP block [18, 21], there was no interference with evoked potential monitoring which is consistent with other reports [9]. Similarly, hypotension related to local anesthetic-induced sympathectomy has never been reported [22]. The most likely explanation is that the actual mass of local anesthetic reaching the epidural space is insufficient to produce a clinically-detectable effect; nevertheless, this should be a consideration in cases at high risk of intraoperative neurological compromise. We performed the ESP blocks in a dedicated block room primarily to maximize operating room efficiency; however they may also be performed after anesthetic induction and prone positioning. This adds little risk to the procedure, and may be more acceptable in young or highly anxious patients. One caveat to this approach is that there may be insufficient time for complete block onset with the long-acting local anesthetics (bupivacaine or ropivacaine) before surgical incision. In this case, the addition of lidocaine to the local anesthetic mixture may help to speed onset [23].
Preoperative performance mandates the use of single-injection blocks instead of a continuous technique. We elected not to surgically place catheters at wound closure due to uncertainty regarding the adequacy of cranial–caudal spread in the now-disrupted tissue plane, and to minimize the complexity of the postoperative analgesic regimen. Studies in other patient populations indicate that a single-injection ESP block provides effective analgesia for at least 8–12 h [22, 24]. We addressed this limitation of fixed analgesic duration by concomitant use of an intraoperative multimodal regimen incorporating agents that have been individually shown to reduce postoperative pain scores and opioid requirements for up to 48 h. Employing pre-emptive multimodal analgesia has been shown to significantly contribute to improved analgesia in spine surgery [25, 26]; however, the combination of dexamethasone, dexmedetomidine, and ketamine has not previously been reported.
Intravenous dexmedetomidine [27] and dexamethasone [28] both prolong the duration of local anesthetic effect, and they may even be synergistic in this regard [29]. In addition, each drug has a systemic analgesic effect, reducing postoperative pain scores and opioid consumption for 24–48 h [30, 31]. In the case of dexmedetomidine, this is attributed to central effects at alpha-2 receptors in the dorsal horn, as well as anti-inflammatory effects which reduce the stress response to surgery and peripheral sensitization [19, 32]. Further benefit may be gained by substituting it for remifentanil, which has been implicated in exacerbating postoperative pain via opioid-induced hyperalgesia [33]. A reduction in pain scores and opioid requirements for up to 48 h following major spine surgery was observed when dexmedetomidine was used instead of remifentanil [15, 16]. Finally, subanesthetic doses of intraoperative ketamine also reduce postoperative opioid consumption without significant side effects [16, 34], with an analgesic benefit lasting up to 48 h [14]. The higher postoperative opioid consumption and pain scores in our patients in the 48–72-h period compared to the 24–48-h period may reflect offset of these analgesic benefits but also increased physical activity.
In summary, this case report serves as proof of concept for a novel multimodal opioid-sparing analgesic strategy for scoliosis correction surgery. ESP blocks are relatively simple and safe compared to other regional anesthetic techniques in spine surgery, and the multimodal combination of intraoperative pharmacological adjuncts offers additive, and potentially synergistic, benefits. Further research, including randomized controlled trials, is warranted to confirm these preliminary observations, and to investigate whether the strategy will provide similar opioid-sparing analgesia in other types of spine surgery.
References
Kwan MK, Chiu CK, Chan TS et al (2017) Trajectory of postoperative wound pain within the first 2 weeks following posterior spinal fusion surgery in adolescent idiopathic scoliosis patients. Spine 42:838–843
Hiller A, Helenius I, Nurmi E et al (2012) Acetaminophen improves analgesia but does not reduce opioid requirement after major spine surgery in children and adolescents. Spine 37:E1225–E1231
Tripi PA, Poe-Kochert C, Potzman J, Son-Hing JP, Thompson GH (2008) Intrathecal morphine for postoperative analgesia in patients with idiopathic scoliosis undergoing posterior spinal fusion. Spine 33:2248–2251
Hong RA, Gibbons KM, Li GY, Holman A, Voepel-Lewis T (2017) A retrospective comparison of intrathecal morphine and epidural hydromorphone for analgesia following posterior spinal fusion in adolescents with idiopathic scoliosis. Paediatr Anaesth 27:91–97
Taenzer AH, Clark C (2010) Efficacy of postoperative epidural analgesia in adolescent scoliosis surgery: a meta-analysis. Paediatr Anaesth 20:135–143
Klatt JWB, Mickelson J, Hung M, Durcan S, Miller C, Smith JT (2013) A randomized prospective evaluation of 3 techniques of postoperative pain management after posterior spinal instrumentation and fusion. Spine 38:1626–1631
Forero M, Adhikary SD, Lopez H, Tsui C, Chin KJ (2016) The erector spinae plane block: a novel analgesic technique in thoracic neuropathic pain. Reg Anesth Pain Med 41:621–627
Chin KJ, Adhikary S, Sarwani N, Forero M (2017) The analgesic efficacy of pre-operative bilateral erector spinae plane (ESP) blocks in patients having ventral hernia repair. Anaesthesia 72:452–460
Melvin JP, Schrot RJ, Chu GM, Chin KJ (2018) Low thoracic erector spinae plane block for perioperative analgesia in lumbosacral spine surgery: a case series. Can J Anaesth 65:1057–1065
Ivanusic J, Konishi Y, Barrington MJ (2018) A cadaveric study investigating the mechanism of action of erector spinae blockade. Reg Anesth Pain Med 43:567–571
Yang H-M, Choi YJ, Kwon H-J, O J, Cho TH, Kim SH (2018) Comparison of injectate spread and nerve involvement between retrolaminar and erector spinae plane blocks in the thoracic region: a cadaveric study. Anaesthesia 73:1244–1250
Chin KJ, Lewis S (2019) Opioid-free analgesia for posterior spinal fusion surgery using erector spinae plane (ESP) blocks in a multimodal anesthetic regimen. Spine 44:E379–E383
Sharma S, Balireddy RK, Vorenkamp KE, Durieux ME (2012) Beyond opioid patient-controlled analgesia: a systematic review of analgesia after major spine surgery. Reg Anesth Pain Med 37:79–98
Loftus RW, Yeager MP, Clark JA et al (2010) Intraoperative ketamine reduces perioperative opiate consumption in opiate-dependent patients with chronic back pain undergoing back surgery. Anesthesiology 113:639–646
Hwang W, Lee J, Park J, Joo J (2015) Dexmedetomidine versus remifentanil in postoperative pain control after spinal surgery: a randomized controlled study. BMC Anesthesiol 15:21
Garg N, Panda NB, Gandhi KA et al (2016) Comparison of small dose ketamine and dexmedetomidine infusion for postoperative analgesia in spine surgery: a prospective randomized double-blind placebo controlled study. J Neurosurg Anesthesiol 28:27–31
Perera AP, Chari A, Kostusiak M, Khan AA, Luoma AM, Casey ATH (2017) Intramuscular local anesthetic infiltration at closure for postoperative analgesia in lumbar spine surgery: a systematic review and meta-analysis. Spine 42:1088–1095
Adhikary SD, Bernard S, Lopez H, Chin KJ (2018) Erector spinae plane block versus retrolaminar block: a magnetic resonance imaging and anatomical study. Reg Anesth Pain Med 43:756–762
Kelly DJ, Ahmad M, Brull SJ (2001) Preemptive analgesia I: physiological pathways and pharmacological modalities. Can J Anaesth 48:1000–1010
Katz J, Clarke H, Seltzer Z (2011) Review article: preventive analgesia: quo vadimus? Anesth Analg 113:1242–1253
Vidal E, Giménez H, Forero M, Fajardo M (2018) Erector spinae plane block: a cadaver study to determine its mechanism of action. Rev Esp Anestesiol Reanim 65:514–519
Tulgar S, Selvi O, Senturk O, Serifsoy TE, Thomas DT (2019) Ultrasound-guided erector spinae plane block: indications, complications, and effects on acute and chronic pain based on a single-center experience. Cureus 11:e3815
Cuvillon P, Nouvellon E, Ripart J et al (2009) A comparison of the pharmacodynamics and pharmacokinetics of bupivacaine, ropivacaine (with epinephrine) and their equal volume mixtures with lidocaine used for femoral and sciatic nerve blocks: a double-blind randomized study. Anesth Analg 108:641–649
Krishna SN, Chauhan S, Bhoi D et al (2019) Bilateral erector spinae plane block for acute post-surgical pain in adult cardiac surgical patients: a randomized controlled trial. J Cardiothorac Vasc Anesth 33:368–375
Mathiesen O, Dahl B, Thomsen BA et al (2013) A comprehensive multimodal pain treatment reduces opioid consumption after multilevel spine surgery. Eur Spine J 22:2089–2096
Lee BH, Park J-O, Suk K-S et al (2013) Pre-emptive and multi-modal perioperative pain management may improve quality of life in patients undergoing spinal surgery. Pain Physician 16:E217–226
Abdallah FW, Dwyer T, Chan VWS et al (2016) IV and perineural dexmedetomidine similarly prolong the duration of analgesia after interscalene brachial plexus block: a randomized, three-arm, triple-masked placebo-controlled. Trial Anesthesiol 124:683–695
Pehora C, Pearson AM, Kaushal A, Crawford MW, Johnston B (2017) Dexamethasone as an adjuvant to peripheral nerve block. Cochrane Database Syst Rev 11:CD011770
Kang R, Jeong JS, Yoo JC et al (2019) Improvement in postoperative pain control by combined use of intravenous dexamethasone with intravenous dexmedetomidine after interscalene brachial plexus block for arthroscopic shoulder surgery: a randomised controlled trial. Eur J Anaesthesiol 36(5):360–368
Waldron NH, Jones CA, Gan TJ, Allen TK, Habib AS (2013) Impact of perioperative dexamethasone on postoperative analgesia and side-effects: systematic review and meta-analysis. Br J Anaesth 110:191–200
Schnabel A, Reichl SU, Poepping DM, Kranke P, Pogatzki-Zahn EM, Zahn PK (2013) Efficacy and safety of intraoperative dexmedetomidine for acute postoperative pain in children: a meta-analysis of randomized controlled trials. Paediatr Anaesth 23:170–179
Tonner PH (2017) Additives used to reduce perioperative opioid consumption 1: alpha2-agonists. Best Pract Res Clin Anaesthesiol 31:505–512
Fletcher D, Martinez V (2014) Opioid-induced hyperalgesia in patients after surgery: a systematic review and a meta-analysis. Br J Anaesth 112:991–1004
Schwenk ES, Viscusi ER, Buvanendran A et al (2018) Consensus guidelines on the use of intravenous ketamine infusions for acute pain management from the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists. Reg Anesth Pain Med 43:456–466
Funding
Dr Ki Jinn Chin is supported by a Merit Award from the Department of Anesthesia, University of Toronto.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chin, K.J., Dinsmore, M.J., Lewis, S. et al. Opioid-sparing multimodal analgesia with bilateral bi-level erector spinae plane blocks in scoliosis surgery: a case report of two patients. Eur Spine J 29 (Suppl 2), 138–144 (2020). https://doi.org/10.1007/s00586-019-06133-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-019-06133-8