Abstract
Purpose
In this systematic review, we aim to illustrate the current and safe concepts in the assessment, diagnosis and management of herniated lumbar disc (HLD) during pregnancy.
Methods
A systematic review and reporting on the diagnosis, treatment and clinical results of HLD during pregnancy is performed.
Results
The MRI represents the first level and safest diagnostic tool for pregnant women affected by spinal problems allowing for a noninvasive and detailed radiological examination of the spine. The initial management of pregnant women affected by HLD is conservative, and primarily aimed to pain therapy. Whenever radicular pain and progressive neurological deficits unresponsive to medical management occur, surgery should be considered. Few case reports regarding the operative management of HLD in pregnant women have been published up to date. Laminectomy and/or microdiscectomy represent the classical and most commonly used techniques that can be safely performed without affecting pregnancy, delivery, or baby’s health. Endoscopic discectomy may be an alternative. The most adequate timing and surgical position are chosen based on to the fetal gestational age and site of the pathology.
Conclusions
Surgical treatments during pregnancy impose multiple medical and ethical problems. Timely diagnosis by MRI, careful clinical evaluation, and surgical treatment represent safe and effective procedures. Ongoing evolution of surgical, anesthesiological and obstetrical procedures results in favorable outcomes. However, interdisciplinary management and a wide knowledge of pregnancy-related pathologies are crucial for the best outcome for both mother and child.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Spinal diseases are common issues in women of child-bearing age and can be an independent cause of maternal disability during pregnancy. The clinical approach and management of low back pain in pregnant women represent a medical and ethical challenge. Moreover, there is no consensus or guidelines for the treatments of such diseases.
Low back pain is frequently encountered in pregnancy [1]. It affects more than 50% of pregnant women, typically between the 5th and the 7th month of gestation [2]. Due to this high incidence, LBP is most often considered by many physicians as a natural symptom during pregnancy; however, it is actually a serious discomfort that cannot be ignored since 80% of pregnant women find daily activities very challenging and 30% of them require bed rest because of severe pain [3].
Several risk factors have been identified for pregnancy-related LBP including maternal age, parity, physical and psychological work factors, LBP during previous pregnancy or menstruation [2,3,4,5,6,7,8].
Several causative factors for LBP in pregnancy have been suggested. Relaxation of the sacroiliac joint and symphysis pubis plays an important role [9]. High levels of relaxin, secreted by the corpus luteum, might result in widening and sliding mobility of the sacroiliac joints and pubic symphysis with a potential instability leading to pelvic pain [10,11,12]. Relaxin could determine analogous changes in the disks and posterior longitudinal ligament, thus predisposing to disk bulging or herniation [13]. However, the role of relaxin in this contest is still controversial [14, 15].
Mechanical factors also concur to the determination of LBP, including the enlarging gravid uterus which moves the maternal center of gravity anteriorly leading to a compensatory lordosis, and therefore, a straining of the lower back and pelvic girdle [16,17,18].
In approximately 1:10,000 pregnancies a herniated lumbar disc (HLD) can be the cause of intractable pain often associated with sciatic nerve distribution. LaBan et al. diagnosed five symptomatic HLD in a series of 48.760 consecutive deliveries [19]. Symptomatic HLD represents the most common intrinsic spinal pathology during pregnancy and, even if exceedingly rare, it must be promptly explored, since delayed diagnosis and treatment may cause permanent neurologic deficits [19].
Whereas LPB during pregnancy may be treated conservatively, surgery should be taken into consideration whenever radicular pain and progressive neurological deficits do not respond to medical management. When an HLD is identified as the cause of gestational low back pain, additional concerns arise regarding subsequent treatment and management of the delivery. In fact, surgical interventions during pregnancy represent a clinical challenge and require an appropriate treatment strategy. Several case reports on the management of HLD in pregnant patients have been published up to date [20, 21], but there is no consensus yet on the best treatment options and on the timing of surgery or associated obstetrical interventions.
Questions such as “which is the correct clinical and radiological approach?”; “which are the conservative treatment options?”; “what is the most appropriate surgical treatment according to the gestational age?” still remain unanswered. Indeed, the clinical condition of mother and fetus, the stage of pregnancy, as well as the severity of symptomatology make the decision controversial.
The aim of this narrative review about HLD cases during pregnancy is to describe the current approaches in the assessment, diagnosis and management of this challenging disorder.
Materials and methods
The systematic review was performed following the PRISMA (preferred reporting items for systematic reviews and meta-analyses) statement. The PRISMA search algorithm is shown in Fig. 1.
We performed a search using the keyword ‘herniated lumbar disc’ and ‘low back pain’ in combination with ‘pregnancy’, with no limit regarding the year of publication. The following databases were accessed on 2nd February 2017: PubMed (http://www.ncbi.nlm.nih.gov/sites/entrez/); Ovid (http://www.ovid.com); Cochrane Reviews (http://www.cochrane.org/reviews/), Google Scholar. Only publications in English were considered in the current review. In addition, the search was extended by screening the reference list of all the articles. The investigator (ADM) read the abstract of each publication identified; if an abstract was not available, the article was excluded. We included studies, which reported outcome after conservative and surgical treatments of pregnant women affected by HLD (Table 1).
Studies on other musculoskeletal disorders during pregnancy were excluded. Studies on animals and cadavers, and in vitro studies, technical notes, letters to editors and articles not specifically reporting outcomes were also excluded. To qualify, an article would have to have been published in peer-reviewed journals.
These selection criteria allowed excluding papers based on the text of the abstract. We obtained full-text versions if the abstract did not permit to include or exclude the study. If there was doubt about inclusion of an article, the senior authors made a collegial decision. The publications thus selected were examined by two authors (FR, GV).
We posed the following questions: (1) which is the correct clinical and radiological approach?; (2) which are the conservative treatment options?; (3) what is the most appropriate surgical treatment according to the gestational age?
Results
The literature search and cross-referencing resulted in 125 references, of which 95 were rejected due to off topic abstract of the results (Fig. 1). After reading the remaining full-text articles, another 10 articles were excluded for failing to fulfil the inclusion criteria. The remaining 20 articles were included in the present study. All the characteristics of the studies are shown in Table 1. A total of 35 patients were reported in the included studies. Age varied from 27 years old to 41 years old (average 32.6 years old). Gestational age ranged from 6 weeks to 36 weeks (average 23.8 weeks). 13 patients were affected by L4–L5 HLD and 22 patients by L5–S1 HLD. Out of 35 women, 34 had no sequela and gave birth to healthy babies.
Which is the correct clinical and radiological approach?
Pregnant women affected by HLD usually experience LBP during the early stages of pregnancy that gradually decreases [22, 23]. The typical clinical presentation is unilateral leg pain that, unlike PGP, involves the dermatome of the affected root radiating to the legs or feet. Paresthesias, reflex changes and associated muscle weakness, as well as a positive straight leg-raising test may vary based on the involved root [24].
LBP must be distinguished from pelvic girdle pain (PGP) and combined pain [4, 5]. PGP is a specific type of LBP defined as the pain experienced between the posterior iliac crest and the gluteal fold, mainly close to the sacroiliac joints with or without radiation to the posterior thigh. The pain may also occur in the symphysis pubis with or without anterior thigh pain [5, 6].
Fewer than 2% of HLD during pregnancy causes cauda equina syndrome or severe and/or progressive deficits [25]. Cauda equina syndrome is an urgent condition caused by compression of the lumbosacral nerve roots characterized by radiating pain or numbness at legs, paralysis, sexual dysfunction and bladder or bowel impairment ranging from painless urinary retention to urine and faecal incontinence. Physical examination shows a positive straight leg raising test, reduced rectal sphincter tone, saddle anesthesia, and decreased deep tendon reflexes [26]. The clinical presentation can be acute or more gradual in onset [27]. Tay et al. divided the patients into 3 groups based on the onset of symptoms: acute onset with no previous symptoms, patients with history of back pain and last episode of cauda equina syndrome, and patients with gradually progressive symptoms going from back pain and sciatica to paralysis of the cauda equina [28].
Similar symptoms can occur in pregnancy, mainly during the third trimester. There exists, therefore, a potential for misdiagnosis and consequent delay in treatment. Indeed, pregnancy increases mechanical and positional stress of the spine causing lumbosacral pain and leading more than 50% of expectant women to complain at some stage of their pregnancy [29].
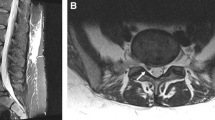
Therefore, when a pregnant woman complains of LBP or radiating pain, an obstetrician should always consider HLD since a delayed diagnosis and treatment may cause permanent neurologic deficits [30]. Most of all, when leg motor weakness progresses with unilateral radiating pain, or cauda equina syndrome is suspected, a neurological physical exam including rectal and sensory examination of the perineum and sacral area is mandatory. In particular, an MRI should be performed as soon as possible whenever the cauda equina syndrome is suspected since it is potentially reversible if managed acutely [31]. Pregnancy at any stage does not represent a contraindication to MRI. Indeed, MRI has no adverse effects on the progression of pregnancy or on the development of the fetus [23] and it is the safest exam to be performed on expectant women with spinal problems [20]. Garmel et al. reported on three pregnant women with HLD. In all cases MRI was a useful diagnostic tool and all failed conservative treatment and required surgery resulting in good clinical outcome both for the newborn and the mother.
MRI represents the first level and safest diagnostic tool for pregnant women affected by spinal problems [20, 22]. It has been shown to be a safe procedure during pregnancy [23], allowing for a noninvasive and detailed radiological examination of the spine without exposing the developing fetus to the hazardous effects of ionizing radiation (Fig. 2). Indeed, Evans et al. [23], have showed no increased rate of complications such as infertility or low-birth-weight infants in MRI technicians compared to control pregnant women. Electromyography (EMG) may represent a useful exam to confirm the presence of a compression on the motor component of a spinal root.
Plain radiography or computed tomography should not be used as first diagnostic tool in pregnant women with spinal problems to minimize radiation exposure to the fetus. Even though most of diagnostic procedures do not involve fetal exposure above 0.05 Gy (threshold level for teratogenic risk), and the risk of congenital anomalies after exposure to ionizing radiation is strictly connected to the gestational age at the time of exposure and radiation dose [24], radiographic examinations should be avoided during the first trimester.
Which are the conservative treatment options?
The initial management of pregnant women affected by HLD is always conservative, and is primarily aimed to pain reduction, either with the use of analgesics, or by reducing pressure on the nerve roots. Indeed, when LBP with or without radiation to the lower limbs is the main symptom in absence of motor weakness or severe cauda equina/spinal cord compromise, the conservative treatment including bed rest, analgesics, muscle relaxants, and physical therapy is suggested [16, 20]. With this approach, most pregnant women can deliver with no further complications.
Decreased physical activity, low-heeled shoes, and bed rest in supine position with raised feet to decrease lumbar lordosis or hip flexion may relieve muscle spasm and pain [32]. Whenever the pain decreases, selected exercises to reinforce the tone of abdominal and back muscles are recommended [33, 34]. A lumbosacral brace can help relieving symptoms.
Particular attention should be given to the pharmacological approach in pregnancy, since it may determine temporary or permanent effects on the fetus. A valid criterion for the selection of pain medications in pregnancy is taking into account the FDA category of the drug. As a general rule, FDA categories A and B drugs should be preferred for administration in pregnant women: for example, antibiotics such as Cephalosporin and Ampicillin, are listed in the FDA category B and have no teratogenic effects [35]. On the contrary, non-steroidal anti-inflammatory drugs (NSAIDs) are under the FDA categories C or D and should be avoided during pregnancy because of their potential for fetal congenital anomalies [36]. Pethidine and acetaminophen can be safely used for pain control during pregnancy. In case of uncontrolled pain, an epidural injection of steroids can be considered in women during the second or third trimester of pregnancy. Nerve or regional blocks may represent valid alternatives to the use of analgesics [37]. Steroids are usually administered to women in pregnancy since these help the development of the lungs of the fetus [38]; however, they should only be administered for short periods of time, because long term administration poses high risk of negative fetal effects including: decreased fetal body weight, breathing movements, fetal heart rate variation and an increased risk of early onset neonatal sepsis [39]. Even though muscle relaxants are commonly administered in LBP patients, most should be avoided in pregnancy. Cyclobenzaprine, under FDA category B, is the only available muscle relaxant for pregnant women [40].
What is the most appropriate surgical treatment according to the gestational age?
There are no general consensus or treatment guidelines for spinal surgery in pregnant women; however, when dealing with a pregnant woman affected by HLD, the surgical indications reflect those for ordinary patients [20]. Most spine surgeries can safely be performed without affecting either pregnancy, delivery, or baby’s health.
In women after 34–36 weeks of gestation, the delivery can be induced or a cesarean section can be performed with same day standard elective surgery for HLD. However, whenever cauda equina syndrome or a progressive motor weakness are present, urgent surgery is mandatory regardless of the stage of pregnancy, sometimes following the cesarean section under the same anesthesia [41]. However, if neurologic symptoms are minimal and not progressing, the surgical treatment can be delayed to after delivery. Surgery can also be performed at any stage of pregnancy when disabling pain is not responsive to conservative treatment [20].
Few case reports regarding the operative management of HLD in pregnant women have been published up to date (Table 1) [1,2,3,4,5,6,7,8,9,10] Laminectomy and/or microdiscectomy represent the classical and most commonly performed surgical techniques [20, 41, 42]. Kim et al. described an endoscopic discectomy as the surgical treatment for HLD in pregnancy. Attention should be posed to limiting or avoiding exposure to radiation which can increase the risk of congenital anomalies [20].
The patient’s position is one of the main issues of lumbar spine surgery during pregnancy, since most surgeons are familiar to the prone position. However, the most adequate surgical position could be modified according to the fetal gestational age and side of the pathology [20]. The prone position can still be used during the first trimester of pregnancy and at the beginning of the second one [43]. However, after the second trimester a left lateral decubitus can be chosen to avoid aortocaval compression by the gravid uterus, which may take place when laying on the right side [21]. Alternatively, surgery for HLD in pregnant women can be performed on the Relton–Hall laminectomy frame, an equivalent four-poster or bow frame [20]. These tools avoid any pressure on the uterus and fetus since the upper posts are applied to the sternum and the lower to the anterior iliac crests (Fig. 3) [42]. The authors demonstrated that pregnancy is not a contraindication to epidural or general anesthesia and surgical intervention [42]. Anesthesiological management aims at maternal safety, optimal analgesia and prevention of hypotension, hypoxia, and preterm labour. Epidural anesthesia has been proven to be the safest procedure at any stage of gestation and it is, indeed, used in most vaginal deliveries and cesarean sections [20, 42]. Intraoperative fetal heart monitoring may be essential after 23 weeks of gestation to look after the fetus and to evaluate the eventual abnormal heart rate patterns. Monitoring between 20th and 23rd week is still controversial, and it is not indicated before the 20th week [44].
Discussion
Most of the available literature about HLD in pregnancy is related to the management of cauda equina syndrome or of severe and/or progressive neurologic deficits due to HLD (Table 1) that represent a surgical emergency in both pregnant and non-pregnant patients. To avoid permanent sequelae, the diagnosis in a pregnant woman should be made as soon as possible after the onset of symptoms. A delay in neural decompression may result in permanent neurologic deficits [19]. Ashkan et al. [29] reported on a 37 year old patient at 20 weeks of gestation that underwent surgery for HLD at L5–S1, who ended up with a loss of sensation in the sacral area still present one year after surgery. Timothy et al. [30] reported persisting urinary and faecal incontinence despite surgery in a case where diagnosis was delayed in a primigravida. Therefore, a rigorous and accurate neurological examination is highly recommended when a cauda equina syndrome is suspected, to promote early treatment, and in fact a delay in the diagnosis led to persistent neurological problems despite surgical treatment.
Garmel et al. [45] reported on the outcomes of three pregnant women with progressive back pain, paresthesias, and urinary retention due to HLD. All of them did well postoperatively, with improvement of symptoms and delivery at or near term. Brown et al. [42] reported on three pregnant patients with HLD causing cauda equina syndrome and severe neurologic deficits. They were operated on in prone position under epidural anesthesia. Surgery consisted of one-level hemilaminectomy, partial facetectomy, and discectomy.
Hakan et al. [46] described the case of a 34-year-old woman at 25-week gestation with a 2-day history of low back pain, numbness in her buttocks, urinary and fecal incontinence caused by a large extruded disk fragment at L5–S1. She underwent L5–S1 discectomy under general anesthesia on prone position, delivering a healthy infant at term.
Surgery for lumbar disk herniation in pregnancy has also been performed in patients without cauda equina syndrome, with intractable pain and neurological deficits being the main indications for surgery.
Fahy et al. [47] reported on two cases of patients affected by HLD. One patient was a 32 year old woman at 32 weeks of gestation affected by L4–L5 HLD with a footdrop. She underwent an L4–L5 discectomy under general anesthesia in prone position. Treatment was effective and she gave birth to a healthy baby. The other patient was a 31-year-old lady at 33 weeks of gestation affected by an L4–L5 HLD. She underwent a L4–L5 discectomy under general anesthesia in prone position. Vougioukas et al. [21] reported on two cases of pregnant women affected by HLD, who underwent discectomy during pregnancy. They did not recommend prone positioning after the 12th week of gestation and operated on them in left lateral decubitus position to avoid the risk of compression of the inferior vena cava, by employing the intraperioperative cardiotocography. Han et al. [20] reported on the cases of six women who underwent surgery for HLD. For patients in the first trimester, the prone position was used and for patients in the second and third trimester, the lateral decubitus position was preferred. Five patients with HLDs underwent partial hemilaminectomy and discectomy during pregnancy and maintained the pregnancy. Conversely, one patient underwent a posterior lumbar interbody fusion and an abortion was performed 6 days after surgery because the patient had a CT scan at a gestational age of 9 weeks and there was a risk of fetal anomaly.
LaBan et al. [22] described six patients, all of whom without antecedent history LBP, who were managed for HLD. One out of six patients required surgery. After a miscarriage, another patient underwent a discectomy for intractable pain.
Ashkan et al. described two cases with HLD. A 37-year-old woman affected by an L5–Sl HLD who underwent emergency discectomy. And a 29-year-old woman affected by a central L5–S1 HLD with marked thecal displacement that underwent urgent L5–S1 discectomy [29].
Martel et al. reported on a patient operated under general anesthesia in the last trimester of pregnancy, at 27 and 4 weeks’ gestation. Fetal heart tones were monitored preoperatively and postoperatively, but no intraoperative fetal heart rate monitoring was performed [48].
Geftler et al. treated a 33-year-old woman at 36 week gestation affected by an L4–L5 HLD. Immediately following the cesarean section, they performed a partial laminoforaminotomy and discectomy at L4–L5 with completely regression of the symptomatology [49].
Recently, novel treatments have been proposed in this context, like the endoscopic discectomy performed by Kim et al. and Hayakawa et al. that performed a microendoscopic discectomy on patients affected of HLD during the third trimester of pregnancy. This alternative treatment may represent an option for HLD during pregnancy [50, 51].
Symptomatic HLD, even if rare, is an emergent situation and only a small portion of these patients present with cauda equina syndrome. However, the incidence of HLD is increasing, probably not because of pregnancy [52] but as a result of the increased mean age of the pregnant women, and of the increased incidence of obesity [53]. The average age of primiparous women has significantly increased in the United States and Europe in the past two decades. The prevalence of primiparous women older than 30 years of age has increased from 17.7 to 30.2% [53]. In recent reports, almost all patients were older than 30 years of age [20, 42]. In association to the advancing maternal age, physiological and anatomical changes may predispose to lumbar disk herniation [54, 55]. Surgical treatments during pregnancy impose multiple medical and ethical problems. Physicians should be aware of the increasing risk of HLD in pregnant women. Indeed, if such pathology is missed or diagnosed late, the prognosis may be poor or result in permanent neurologic deficits. Timely diagnosis by MRI, careful clinical evaluation, and surgical treatment represents safe and effective procedures.
References
Gutke A, Ostgaard HC, Oberg B (2008) Predicting persistent pregnancy-related low back pain. Spine (Phila Pa 1976) 33:E386–E393
Ansari NN, Hasson S, Naghdi S, Keyhani S, Jalaie S (2010) Low back pain during pregnancy in Iranian women: prevalence and risk factors. Physiother Theory Pract 26:40–48
Mens JM, Vleeming A, Stoeckart R, Stam HJ, Snijders CJ (1996) Understanding peripartum pelvic pain. Implications of a patient survey. Spine (Phila Pa 1976) 21:1363–1369
Ostgaard HC, Roos-Hansson E, Zetherstrom G (1996) Regression of back and posterior pelvic pain after pregnancy. Spine (Phila Pa 1976) 21:2777–2780
Vleeming A, Albert HB, Ostgaard HC, Sturesson B, Stuge B (2008) European guidelines for the diagnosis and treatment of pelvic girdle pain. Eur Spine J 17:794–819
Wu WH, Meijer OG, Uegaki K, Mens JM, van Dieën JH, Wuisman PI, Ostgaard HC (2004) Pregnancy-related pelvic girdle pain (PPP), I: terminology, clinical presentation, and prevalence. Eur Spine J 13:575–589
Mohseni-Bandpei MA, Fakhri M, Ahmad-Shirvani M, Bagheri-Nessami M, Khalilian AR, Shayesteh-Azar M, Mohseni-Bandpei H (2009) Low back pain in 1,100 Iranian pregnant women: prevalence and risk factors. Spine J 9:795–801
Ostgaard HC, Andersson GB, Wennergren M (1991) The impact of low back and pelvic pain in pregnancy on the pregnancy outcome. Acta Obstet Gynecol Scand 70:21–24
Wright JL (1952) Relaxation of the pelvic joints in pregnancy; a report of three cases. N Z Med J 51:377–380
Unemori EN, Amento EP (1990) Relaxin modulates synthesis and secretion of procollagenase and collagen by human dermal fibroblasts. J Biol Chem 265:10681–10685
Kristiansson P, Svardsudd K, von Schoultz B (1996) Serum relaxin, symphyseal pain, and back pain during pregnancy. Am J Obstet Gynecol 175:1342–1347
MacLennan AH, Nicolson R, Green RC, Bath M (1986) Serum relaxin and pelvic pain of pregnancy. Lancet 2:243–245
O’Connell JE (1960) Lumbar disc protrusions in pregnancy. J Neurol Neurosurg Psychiatry 23:138–141
Marnach ML, Ramin KD, Ramsey PS, Song SW, Stensland JJ, An KN (2003) Characterization of the relationship between joint laxity and maternal hormones in pregnancy. Obstet Gynecol 101:331–335
Schauberger CW, Rooney BL, Goldsmith L, Shenton D, Silva PD, Schaper A (1996) Peripheral joint laxity increases in pregnancy but does not correlate with serum relaxin levels. Am J Obstet Gynecol 174:667–671
Ritchie JR (2003) Orthopedic considerations during pregnancy. Clin Obstet Gynecol 46:456–466
Mantle MJ, Greenwood RM, Currey HL (1977) Backache in pregnancy. Rheumatol Rehabil 16:95–101
Betsch M, Wehrle R, Dor L, Rapp W, Jungbluth P, Hakimi M, Wild M (2015) Spinal posture and pelvic position during pregnancy: a prospective rasterstereographic pilot study. Eur Spine J 24(6):1282–1288
LaBan MM, Perrin JC, Latimer FR (1983) Pregnancy and the herniated lumbar disc. Arch Phys Med Rehabil 64:319–321
Han IH, Kuh SU, Kim JH, Chin DK, Kim KS, Yoon YS, Jin BH, Cho YE (2008) Clinical approach and surgical strategy for spinal diseases in pregnant women: a report of ten cases. Spine (Phila Pa 1976) 33:E614–E619
Vougioukas VI, Kyroussis G, Gläsker S, Tatagiba M, Scheufler KM (2004) Neurosurgical interventions during pregnancy and the puerperium: clinical considerations and management. Acta Neurochir (Wien) 146:1287–1291
LaBan MM, Rapp NS, von Oeyen P, Meerschaert JR (1995) The lumbar herniated disk of pregnancy: a report of six cases identified by magnetic resonance imaging. Arch Phys Med Rehabil 76:476–479
Evans JA, Savitz DA, Kanal E, Gillen J (1993) Infertility and pregnancy outcome among magnetic resonance imaging workers. J Occup Med 35:1191–1195
De Santis M, Di Gianantonio E, Straface G, Cavaliere AF, Caruso A, Schiavon F, Berletti R, Clementi M (2005) Ionizing radiations in pregnancy and teratogenesis: a review of literature. Reprod Toxicol 20:323–329
Mousavi SJ, Parnianpour M, Vleeming A (2007) Pregnancy related pelvic girdle pain and low back pain in an Iranian population. Spine (Phila Pa 1976) 32:E100–E104
Dinning TA, Schaeffer HR (1993) Discogenic compression of the cauda equina: a surgical emergency. Aust N Z J Surg 63:927–934
Kostuik JP, Harrington I, Alexander D, Rand W, Evans D (1986) Cauda equina syndrome and lumbar disc herniation. J Bone Jt Surg Am 68:386–391
Tay EC, Chacha PB (1979) Midline prolapse of a lumbar intervertebral disc with compression of the cauda equina. J Bone Jt Surg Br 61:43–46
Ashkan K, Casey AT, Powell M, Crockard HA (1998) Back pain during pregnancy and after childbirth: an unusual cause not to miss. J R Soc Med 91:88–90
Timothy J, Anthony R, Tyagi A, Porter D, van Hille PT (1999) A case of delayed diagnosis of the cauda equina syndrome in pregnancy. Aust N Z J Obstet Gynaecol 39:260–261
O’Laoire SA, Crockard HA, Thomas DG (1981) Prognosis for sphincter recovery after operation for cauda equina compression owing to lumbar disc prolapse. Br Med J (Clin Res Ed) 282:1852–1854
Jacobson H (1991) Protecting the back during pregnancy. AAOHN J 39:286–291
Spankus JD (1965) The cause and treatment of low back pain during pregnancy. Wis Med J 64:303–304
McIntyre IN, Broadhurst NA (1996) Effective treatment of low back pain in pregnancy. Aust Fam Physician 25(9 suppl 2):S65–S67
Dashe JS, Gilstrap LC 3rd (1997) Antibiotic use in pregnancy. Obstet Gynecol Clin North Am 24:617–629
Ofori B, Oraichi D, Blais L, Rey E, Bérard A (2006) Risk of congenital anomalies in pregnant users of non-steroidal anti-inflammatory drugs: a nested case-control study. Birth Defects Res B Dev Reprod Toxicol 77:268–279
Torstensson T, Lindgren A, Kristiansson P (2009) Corticosteroid injection treatment to the ischiadic spine reduced pain in women with long-lasting sacral low back pain with onset during pregnancy: a randomized, double blind, controlled trial. Spine (Phila Pa 1976) 34:2254–2258
Crowther CA, McKinlay CJ, Middleton P, Harding JE (2015) Repeat doses of prenatal corticosteroids for women at risk of preterm birth for improving neonatal health outcomes. Cochrane Database Syst Rev. doi:10.1002/14651858.CD003935.pub4
Mariotti V, Marconi AM, Pardi G (2004) Undesired effects of steroids during pregnancy. J Matern Fetal Neonatal Med 16(Suppl 2):5–7
Childers MK, Borenstein D, Brown RL, Gershon S, Hale ME, Petri M, Wan GJ, Laudadio C, Harrison DD (2005) Low-dose cyclobenzaprine versus combination therapy with ibuprofen for acute neck or back pain with muscle spasm: a randomized trial. Curr Med Res Opin 21:1485–1493
Brown MD, Brookfield KF (2004) Lumbar disc excision and cesarean delivery during the same anesthesia. A case report. J Bone Jt Surg Am 86(9):2030–2032
Brown MD, Levi AD (2001) Surgery for lumbar disc herniation during pregnancy. Spine (Phila Pa 1976) 26:440–443
Kathirgamanathan A, Jardine AD, Levy DM, Grevitt MP (2006) Lumbar disc surgery in the third trimester–with the fetus in utero. Int J Obstet Anesth 15:181–182
Katz JD, Hook R, Barash PG (1976) Fetal heart rate monitoring in pregnant patients undergoing surgery. Am J Obstet Gynecol 125:267–269
Garmel SH, Guzelian GA, D’Alton JG, D’Alton ME (1997) Lumbar disk disease in pregnancy. Obstet Gynecol 89:821–822
Hakan T (2012) Lumbar disk herniation presented with cauda equina syndrome in a pregnant woman. J Neurosci Rural Pract 3:197–199
Fahy UM, Oni M, Findlay D, Sell P (1998) Surgical management of herniated lumbar disc in pregnancy. J Obstet Gynaecol 18:544–545
Martel CG, Volpi-Abadie J, Ural K (2015) Anesthetic management of the parturient for lumbar disc surgery in the prone position. Ochsner J 15:259–261
Geftler A, Sasson A, Shelef I, Perry ZH, Atar D (2015) Cauda equina syndrome in a 36 week gravida patient. Isr Med Assoc J 17:522–523
Kim HS, Kim SW, Lee SM, Shin H (2007) Endoscopic discectomy for the cauda equina syndrome during third trimester of pregnancy. J Korean Neurosurg Soc 42:419–420
Hayakawa K, Mizutani J, Suzuki N, Haas C, Kondo A, Otsuka S, Fukuoka M, Otsuka T (2017) Surgical management of the pregnant patient with lumbar disc herniation in the latter stage of the second trimester. Spine (Phila Pa 1976) 42:E186–E189
Weinreb JC, Wolbarsht LB, Cohen JM, Brown CE, Maravilla KR (1989) Prevalence of lumbosacral intervertebral disk abnormalities on MR images in pregnant and asymptomatic nonpregnant women. Radiology 170:125–128
Parrish KM, Holt VL, Easterling TR, Connell FA, LoGerfo JP (1994) Effect of changes in maternal age, parity, and birth weight distribution on primary cesarean delivery rates. JAMA 271:443–447
Splendiani A, D’Orazio F, Patriarca L, Arrigoni F, Caranci F, Fonio P, Brunese L, Barile A, Di Cesare E, Masciocchi C (2017) Imaging of post-operative spine in intervertebral disc pathology. Musculoskelet Surg. 101(Suppl 1):75–84
Caranci F, Leone G, Ugga L, Cesarano E, Capasso R, Schipani S, Bianco A, Fonio P, Briganti F, Brunese L (2017) Imaging of post-surgical treatment and of related complications in spinal trauma. Musculoskelet Surg 101(Suppl 1):63–73
Iyilikçi L, Erbayraktar S, Tural AN, Celik M, Sannav S (2004) Anesthetic management of lumbar discectomy in a pregnant patient. J Anesth 18(1):45–47
Abou-Shameh MA, Dosani D, Gopal S, McLaren AG (2006) Lumbar discectomy in pregnancy. Int J Gynaecol Obstet 92(2):167–169
Al-areibi A, Coveney L, Singh S, Katsiris S (2007) Case report: anesthetic management for sequential Cesarean delivery and laminectomy. Can J Anaesth 54(6):471–474
Gupta P, Gurumurthy M, Gangineni K, Anarabasu A, Keay SD (2008) Acute presentation of cauda equina syndrome in the third trimester of pregnancy. Eur J Obstet Gynecol Reprod Biol 140(2):279–281
Acknowledgements
The authors gratefully acknowledge Prof. Roberta Aronica for English revision of the current manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Funding
None.
Rights and permissions
About this article
Cite this article
Di Martino, A., Russo, F., Denaro, L. et al. How to treat lumbar disc herniation in pregnancy? A systematic review on current standards. Eur Spine J 26 (Suppl 4), 496–504 (2017). https://doi.org/10.1007/s00586-017-5040-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-017-5040-8