Abstract
Purpose
Utilizing 2D measurements, previous studies have found that in AIS, increased thoracic Cobb and decreased thoracic kyphosis contribute to pulmonary dysfunction. Recent technology has improved our ability to measure and understand the true 3D deformity in AIS. The purpose of this study was to evaluate which 3D radiographic measures predict pulmonary dysfunction.
Methods
One hundred and sixty-three surgically treated AIS patients with preoperative PFTs (FEV, FVC, TLC) and EOS® imaging were identified at a single center. Each spine was reconstructed in 3D to obtain the true coronal, sagittal, and apical rotational deformities. These were then correlated with the patient’s preoperative PFT measurements. Regression analysis was performed to determine the relative effect of each radiographic measure.
Results
There were 124 thoracic and 39 lumbar major curves. The range of preoperative thoracic and lumbar 3D coronal angle was 11–115° and 11–98°, respectively. The range of preoperative thoracic 3D kyphosis (T5–T12) and thoracic apical vertebral rotation was −56 to 44° and 0–29°, respectively. Increasing thoracic 3D Cobb and thoracic vertebral rotation and decreasing thoracic 3D kyphosis most significantly correlated with decreasing pulmonary function, especially FEV. In patients with the largest degree of thoracic deformity (3D Coronal Cobb > 80°, 3D thoracic lordosis >20°, and absolute apical rotation >25°), the majority of patients had moderate to severe pulmonary impairment (≤65 % predicted). 3D thoracic kyphosis was the most consistent predictor of FEV (r 2 = 0.087), FVC (r 2 = 0.069), and TLC (r 2 = 0.098) impairment.
Conclusions
Larger thoracic coronal, sagittal, and axial deformities increase the risk of pulmonary impairment in patients with AIS. Of these, decreasing 3D thoracic kyphosis is the most consistent predictor. This information can guide surgeons in the decision making process for determining which surgical techniques to utilize and which component of the deformity to focus on.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A significant amount of attention has been directed towards spinal deformity and its effect on pulmonary function. In patients with early onset scoliosis, spinal deformity can profoundly affect pulmonary function, altering lung development and even cause early mortality through respiratory failure [1–3]. This has resulted in recommendations for early interventions to alter the natural history of these patients. In adolescent idiopathic scoliosis (AIS), the focus of most studies has either been directed toward the effect of spinal deformity magnitude or the effect of different surgical approaches on pulmonary function [3–19].
Weinstein et al. demonstrated a direct correlation between thoracic magnitude and pulmonary impairment with significant decrease in respiratory function in curves greater than 100° [20]. Others have supported these findings in their evaluation of patients with scoliosis. In addition to curve magnitude, previous studies have also reported on curve location, the number of vertebrae in the curve, and curve rigidity in relation to pulmonary function [12, 21–23]. Besides coronal plane measurement, thoracic hypokyphosis and chest wall deformity have also been shown to have a negative impact on pulmonary function [24]. Newton et al. reported on the results of pulmonary function testing in 631 patients with AIS [12]. They found that while thoracic magnitude, number of vertebrae in the thoracic curve, thoracic hypokyphosis and coronal imbalance had a statistically significant effect, it represented less than 20 % of the variability on pulmonary function testing.
One of the limitations of many of these studies is the use of two-dimensional radiographs. AIS is known to be a three dimensional spinal deformity [25, 26]. Standard scoliosis films do not allow accurate assessment of the axial plane deformity and frequently underestimate the sagittal plane deformity. The recent development of bi-planar X-ray imaging techniques that allow for 3D reconstruction of the spine have resulted in improved measurement and better understanding of the true coronal, sagittal and apical rotational deformity [27, 28]. The purpose of this study was to evaluate which 3D radiographic measures predict pulmonary dysfunction.
Methods
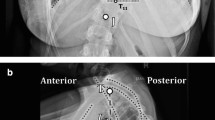
One hundred and sixty-three surgically treated AIS patients with preoperative pulmonary function tests (PFTs) and EOS images (EOS imaging, Paris, France) were identified at a single center. The pre-operative spine images are all standardly obtained at this center and include simultaneous, upright, bi-planar X-ray images (anteroposterior and lateral), which allow for 3D reconstructions of the spine to be performed. Angle of trunk rotation, as measured by Scoliometer, was also collected. 2D coronal Cobb and T5–T12 kyphosis measurements were obtained. Each spine was reconstructed in 3D to obtain the true coronal, sagittal, and apical rotational deformity measurements utilizing sterEOS software. 3D sagittal kyphosis was regionally defined between T5 and T12. To obtain the 3D reconstructions, a trained operator generated a full spine (thoracic and lumbar) reconstruction by morphing a model of each vertebra from T1 to L5 to match that of the patient’s vertebra as observed on both the posterior–anterior and lateral radiographs [27, 29]. This method has been shown to be an accurate and reliable assessment of the scoliotic spine [27, 30–34].
As described by Newton et al. [29]. custom Matlab (Mathworks, Natick, MA) software was used to create a local reference frame for each vertebra and disc, which allows for segmental evaluation of one aspect of the deformity (e.g., kyphosis) by removing the other two dimensions (e.g., coronal and axial rotation). Local segmental kyphosis measurements from T5 to T12 were summed to obtain a 3D measure of kyphosis. In the same manner, local vertebral Cobb measurements were summed between bounding vertebrae to obtain a 3D measure of Cobb. For example, for a curve between T4 and T11, T4 to T11 vertebra measurements and T4–T5 to T10–T11 disc measurements would be summed to get the 3D Cobb. Clockwise axial rotation was defined as positive.
These 3D values of coronal Cobb, kyphosis, and axial rotation were then correlated to the patient’s corresponding preoperative forced expiratory volume (FEV), forced vital capacity (FVC), and total lung capacity (TLC) pulmonary function measurements. Each of the three measures of deformity were also evaluated in an incremental fashion to identify the percentage of patients with moderate–severe pulmonary impairment as defined by the American Thoracic Society [35]. Moderate impairment was defined as having <65 % but >50 % of any predicted PFT value and severe was defined as <50 %. Normal/Mild impairment was defined as >65 % predicted. Multivariate linear regression analysis was performed to determine the relative effect on pulmonary function of each radiographic measure and the thoracic Scoliometer measurement. SPSS v.12 was utilized for all analyses and alpha was set at p < 0.05 to declare significance (SPSS Inc., Chicago, IL).
Results
There were 124 thoracic and 39 lumbar major curves. There were 141 females and 22 males, and the average age of the cohort was 15 ± 2 years. The range of preoperative thoracic and lumbar coronal 3D Cobb angles was 11–115° and 11–98°, respectively. The average curve flexibility on 2D bending radiographs for the entire cohort was 26 ± 21 % (range 0–92 %) for the thoracic curves and 67 ± 23 % (range 12–100 %). The range of preoperative 3D thoracic kyphosis (T5–T12) and thoracic apical vertebral rotation was −56 to 44° and 0–29°, respectively. The range of preoperative thoracic and lumbar 2D Cobb angles was 5–118° and 14–98°, respectively. The range of 2D T5–T12 kyphosis was −11 to 62°.
The correlation between 3D radiographic and pulmonary function values can be found in Table 1. Increasing thoracic 3D Cobb was associated with decreasing FVC, TLC, and especially FEV values (Fig. 1). Increasing axial rotation of the thoracic spine was also associated with decreasing pulmonary function values (Fig. 2). Decreasing 3D thoracic kyphosis was associated with decreasing pulmonary function values, again, most significantly with FEV (Fig. 3). Thoracic rib prominence obtained via Scoliometer also significantly correlated with PFT values, with increasing rib prominence associated with decreasing pulmonary function (Fig. 4). Regression analysis found thoracic kyphosis to be the most consistent predictor of FEV (r 2 = 0.087), FVC (r 2 = 0.069), and TLC (r 2 = 0.098) impairment.
In patients with the largest degree of thoracic deformity (3D Coronal Cobb >80°, 3D thoracic lordosis >20°, and absolute apical rotation >25°), the majority of patients had moderate to severe pulmonary impairment (≤65 % predicted). The number of patients with pulmonary impairment in relation to 3D thoracic Cobb, 3D thoracic kyphosis, and thoracic apical rotation can be found in Figs. 5, 6, 7, respectively.
Discussion
There have been extensive studies evaluating the relationship of spinal deformity and its effect on pulmonary function [1–16, 20–23]. Specifically, there has been concern about the associated decline of respiratory function with increasing severity of curve magnitude. Furthermore, studies have found increasing effect with sagittal plane deformity, especially the hypokyphosis seen with AIS [12]. As treatment methods evolved, studies began focusing on the surgical approach and the negative effect of procedures that violated the thoracic cage [16, 17].
This study supports many of the previously published reports demonstrating a decline in pulmonary function with increasing spinal deformity. In the coronal plane, a larger thoracic Cobb correlated with decreased pulmonary function. The greatest decline in PFT’s was found in thoracic curves greater than 80 degrees. In the sagittal plane, 3D reconstructions from biplanar X-ray imaging allowed more accurate assessment of the true thoracic kyphosis than in previous studies. As expected, increasing loss of 3D thoracic kyphosis resulted in a greater decline in PFTs. Interestingly, thoracic kyphosis was the most consistent radiographic predictor of pulmonary dysfunction with the greatest declines seen in frankly lordotic thoracic spines. These 3D reconstructions also allowed for a more reliable assessment of axial plane deformity on respiratory function. Similar to the coronal plane, greater axial rotation correlated with a decline in PFTs.
This study most closely supports the findings already published by Newton et al. [12]. In their study of 631 AIS patients, the magnitude of the 2D thoracic curve, the number of vertebrae in the thoracic curve, 2D thoracic hypokyphosis, and coronal imbalance significantly correlated with a decline in pulmonary function testing. While supporting the coronal findings, our study provides more accurate assessment of the sagittal deformity and also demonstrates the effect of axial deformity. In their discussion, Newton et al. did suggest that axial rotation may have an effect on PFT through its effect on chest wall shape. However, they questioned all previous studies due to the low reliability of rotation assessment at that time. Similar to that study, our correlations also demonstrate that these radiographic measures are not the only factors involved in the variability of pulmonary function testing.
This study did not evaluate all of the potential factors that may affect pulmonary function as many of them do not relate to the measurement of spinal deformity in 3D. Previous studies have found that in addition to what was analyzed in the current study, pulmonary dysfunction is likely influenced by a greater number of vertebrae in the thoracic curve, a more cephalad curve location, and increased age [12, 22, 36]. Coronal imbalance and curve rigidity have both been found to be associated with decreased pulmonary function; however, the rigidity of the thoracic curve in relation to pulmonary function has had mixed findings [21, 23]. Numerous studies have evaluated various aspects of the impact scoliosis has on the thoracic cage [37] and, in turn, how that affects pulmonary function [38]. These measures were not assessed in the current study as our utilization of 3D reconstructions did not provide a more accurate assessment of these variables than that of the previous studies.
Other studies have attempted to relate pulmonary function in AIS to some 3D measures utilizing CT scans. However, the majority of these studies focused on lung volumes and chest wall dimensions. Chun et al. found that patients with increasing spinal deformity had greater discrepancy between their concave and convex lung volumes suggesting an effect on lung function [39]. Yu et al. subsequently demonstrated that patients with AIS, as compared to normal controls, had lower lung volumes that correlated with lower PFT’s [40]. Interestingly, when lung volumes were assessed by postoperative CT scan, Sarwahi et al. was unable to demonstrate any improvement in lung volumes or concave–convex lung ratios following surgical correction of AIS [41]. They suggested that any postoperative improvement in lung function was related to dynamic rather than static factors.
The advantage of this study is the more accurate assessment of the three dimensional deformity seen in AIS in an upright posture. While not able to provide exact 3D spine rendering or 3D rib cage assessments, recent advances in biplanar X-ray technology and computer modeling have allowed scoliosis physicians to more easily see spinal deformity in 3D without the radiation associated with computed tomography. Even before the current use of 3D imaging, studies have shown the underestimation of thoracic hypokyphosis seen with a standard lateral radiograph. A significant portion of our patients were found to be frankly lordotic across the thoracic spine. In the axial plane, prior to 3D reconstructions, surrogate measures were used to categorize vertebral rotation as opposed to providing an absolute measure [42–44].
One of the limitations of this study is the reliability with which the PFTs were obtained. Utilizing normative data based on height is a concern when evaluating patients that have a condition that is known to affect their height. This may have resulted in a slight underestimation of the effect of the scoliosis on pulmonary function. In addition, scoliosis may also affect a patient’s activity level especially in the more severe cases which may be a confounding variable. It is likely that patients who engage in fewer sports and exercise activities will have decreased PFTs compared to their more active counterparts.
Conclusion
Larger thoracic 3D coronal, 3D sagittal, and axial deformities increase the risk of pulmonary impairment in patients with AIS. Of these, decreased 3D thoracic kyphosis is the most consistent predictor. This information can guide surgeons in the decision making process for determining which surgical techniques to utilize and which component of the deformity to focus on. Further analysis is needed to determine the value of improvement in these radiographic measures following surgery on the potential for improved pulmonary function.
References
Gillingham BL, Fan RA, Akbarnia BA (2006) Early onset idiopathic scoliosis. J Am Acad Orthop Surg 14:101–112
Goldberg CJ, Gillic I, Connaughton O, Moore DP, Fogarty EE, Canny GJ, Dowling FE (2003) Respiratory function and cosmesis at maturity in infantile-onset scoliosis. Spine (Phila Pa 1976) 28:2397–2406
Redding G, Song K, Inscore S, Effmann E, Campbell R (2008) Lung function asymmetry in children with congenital and infantile scoliosis. Spine J 8:639–644
Chen SH, Huang TJ, Lee YY, Hsu RW (2002) Pulmonary function after thoracoplasty in adolescent idiopathic scoliosis. Clin Orthop Relat Res 399:152–161
Gagnon S, Jodoin A, Martin R (1989) Pulmonary function test study and after spinal fusion in young idiopathic scoliosis. Spine (Phila Pa 1976) 14:486–490
Graham EJ, Lenke LG, Lowe TG, Betz RR, Bridwell KH, Kong Y, Blanke K (2000) Prospective pulmonary function evaluation following open thoracotomy for anterior spinal fusion in adolescent idiopathic scoliosis. Spine (Phila Pa 1976) 25:2319–2325
Kim YJ, Lenke LG, Bridwell KH, Kim KL, Steger-May K (2005) Pulmonary function in adolescent idiopathic scoliosis relative to the surgical procedure. J Bone Joint Surg Am 87:1534–1541
Kinnear WJ, Kinnear GC, Watson L, Webb JK, Johnston ID (1992) Pulmonary function after spinal surgery for idiopathic scoliosis. Spine (Phila Pa 1976) 17:708–713
Kumano K, Tsuyama N (1982) Pulmonary function before and after surgical correction of scoliosis. J Bone Joint Surg Am 64(2):242–248
Lenke LG, Bridwell KH, Blanke K, Baldus C (1995) Analysis of pulmonary function and chest cage dimension changes after thoracoplasty in idiopathic scoliosis. Spine (Phila Pa 1976) 20:1343–1350
Lenke LG, Newton PO, Marks MC, Blanke KM, Sides B, Kim YJ, Bridwell KH (2004) Prospective pulmonary function comparison of open versus endoscopic anterior fusion combined with posterior fusion in adolescent idiopathic scoliosis. Spine (Phila Pa 1976) 29:2055–2060
Newton PO, Faro FD, Gollogly S, Betz RR, Lenke LG, Lowe TG (2005) Results of preoperative pulmonary function testing of adolescents with idiopathic scoliosis. A study of six hundred and thirty-one patients. J Bone Joint Surg Am 87:1937–1946
Vedantam R, Lenke LG, Bridwell KH, Haas J, Linville DA (2000) A prospective evaluation of pulmonary function in patients with adolescent idiopathic scoliosis relative to the surgical approach used for spinal arthrodesis. Spine (Phila Pa 1976) 25:82–90
Wong CA, Cole AA, Watson L, Webb JK, Johnston ID, Kinnear WJ (1996) Pulmonary function before and after anterior spinal surgery in adult idiopathic scoliosis. Thorax 51:534–536
Wood KB, Schendel MJ, Dekutoski MB, Boachie-Adjei O, Heithoff KH (1996) Thoracic volume changes in scoliosis surgery. Spine (Phila Pa 1976) 21:718–723
Yaszay B, Jazayeri R, Lonner B (2009) The effect of surgical approaches on pulmonary function in adolescent idiopathic scoliosis. J Spinal Disord Tech 22:278–283
Gitelman Y, Lenke LG, Bridwell KH, Auerbach JD, Sides BA (2011) Pulmonary function in adolescent idiopathic scoliosis relative to the surgical procedure: a 10-year follow-up analysis. Spine (Phila Pa 1976) 36:1665–1672
Johnston CE, Richards BS, Sucato DJ, Bridwell KH, Lenke LG, Erickson M (2011) Correlation of preoperative deformity magnitude and pulmonary function tests in adolescent idiopathic scoliosis. Spine (Phila Pa 1976) 36:1096–1102
Verma K, Lonner BS, Kean KE, LE Dean, Valdevit A (2011) Maximal pulmonary recovery after spinal fusion for adolescent idiopathic scoliosis: how do anterior approaches compare? Spine (Phila Pa 1976) 36:1086–1095
Weinstein SL, Zavala DC, Ponseti IV (1981) Idiopathic scoliosis: long-term follow-up and prognosis in untreated patients. J Bone Joint Surg Am 63:702–712
Jackson RP, Simmons EH, Stripinis D (1989) Coronal and sagittal plane spinal deformities correlating with back pain and pulmonary function in adult idiopathic scoliosis. Spine (Phila Pa 1976) 14:1391–1397
Kearon C, Viviani GR, Kirkley A, Killian KJ (1993) Factors determining pulmonary function in adolescent idiopathic thoracic scoliosis. Am Rev Respir Dis 148:288–294
Upadhyay SS, Mullaji AB, Luk KD, Leong JC (1995) Relation of spinal and thoracic cage deformities and their flexibilities with altered pulmonary functions in adolescent idiopathic scoliosis. Spine (Phila Pa 1976) 20:2415–2420
Aaro S, Ohlund C (1984) Scoliosis and pulmonary function. Spine (Phila Pa 1976) 9:220–222
Labelle H, Aubin CE, Jackson R, Lenke L, Newton P, Parent S (2011) Seeing the spine in 3D: how will it change what we do? J Pediatr Orthop 31:S37–S45
Stokes IA (1994) Three-dimensional terminology of spinal deformity. A report presented to the Scoliosis Research Society by the Scoliosis Research Society Working Group on 3-D terminology of spinal deformity. Spine (Phila Pa 1976) 19:236–248
Glaser DA, Doan J, Newton PO (2012) Comparison of 3-dimensional spinal reconstruction accuracy: biplanar radiographs with EOS versus computed tomography. Spine (Phila Pa 1976) 37:1391–1397
Somoskeoy S, Tunyogi-Csapo M, Bogyo C, Illes T (2012) Accuracy and reliability of coronal and sagittal spinal curvature data based on patient-specific three-dimensional models created by the EOS 2D/3D imaging system. Spine J 12:1052–1059
Newton PO, Fujimori T, Doan J, Reighard FG, Bastrom TP, Misaghi A (2015) Defining the “three-dimensional sagittal plane” in thoracic adolescent idiopathic scoliosis. J Bone Joint Surg Am 97:1694–1701
Carreau JH, Bastrom T, Petcharaporn M, Schulte C, Marks M, Illés T, Somoskeöy S, Newton PO (2014) Computer-generated, three-dimensional spine model from biplanar radiographs: a validity study in idiopathic scoliosis curves greater than 50°. Spine Deform 2:81–88
Newton PO, Khandwala Y, Bartley CE, Reighard FG, Bastrom TP, Yaszay B (2016) New EOS imaging protocol allows a substantial reduction in radiation exposure for scoliosis patients. Spine Deform 4:138–144
Ilharreborde B, Steffen JS, Nectoux E, Vital JM, Mazda K, Skalli W, Obeid I (2011) Angle measurement reproducibility using EOS three-dimensional reconstructions in adolescent idiopathic scoliosis treated by posterior instrumentation. Spine 36:E1306–E1313
Somoskeöy S, Tunyogi-Csapó M, Bogyó C, Illés T (2012) Accuracy and reliability of coronal and sagittal spinal curvature data based on patient-specific three-dimensional models created by the EOS 2D/3D imaging system. Spine J 12:1052–1059
Al-Aubaidi Z, Lebel D, Oudjhane K, Zeller R (2013) Three-dimensional imaging of the spine using the EOS system: is it reliable? A comparative study using computed tomography imaging. J Pediatr Orthop B 22:409–412
Murray J, Nadel J, Boushey H, Mason R (eds) (2000) Textbook of Respiratory Medicine. Saunders, Philadelphia
Johari J, Sharifudin MA, Ab Rahman A, Omar AS, Abdullah AT, Nor S, Lam WC, Yusof MI (2016) Relationship between pulmonary function and degree of spinal deformity, location of apical vertebrae and age among adolescent idiopathic scoliosis patients. Singapore Med J 57:33–38
Harris JA, Mayer OH, Shah SA, Campbell RM Jr, Balasubramanian S (2014) A comprehensive review of thoracic deformity parameters in scoliosis. Eur Spine J 23:2594–2602
Tsiligiannis T, Grivas T (2012) Pulmonary function in children with idiopathic scoliosis. Scoliosis 7:7
Chun EM, Suh SW, Modi HN, Kang EY, Hong SJ, Song HR (2008) The change in ratio of convex and concave lung volume in adolescent idiopathic scoliosis: a 3D CT scan based cross sectional study of effect of severity of curve on convex and concave lung volumes in 99 cases. Eur Spine J 17:224–229
Yu W, Song K, Zhang Y, Zheng GQ, Dong T (2014) Relationship between lung volume and pulmonary function in patients with adolescent idiopathic scoliosis: computed tomographic-based three-dimensional volumetric reconstruction of lung parenchyma. J Spinal Disord Tech (Epub ahead of print)
Sarwahi V, Sugarman EP, Wollowick AL, Amaral TD, Harmon ED, Thornhill B (2014) Scoliosis surgery in patients with adolescent idiopathic scoliosis does not alter lung volume: a 3-dimensional computed tomography-based study. Spine (Phila Pa 1976) 39:E399–E405
Kuklo TR, Potter BK, Polly DW Jr, O’Brien MF, Schroeder TM, Lenke LG (2005) Reliability analysis for manual adolescent idiopathic scoliosis measurements. Spine (Phila Pa 1976) 30:444–454
Lam GC, Hill DL, Le LH, Raso JV, Lou EH (2008) Vertebral rotation measurement: a summary and comparison of common radiographic and CT methods. Scoliosis 3:16
Stokes IA, Bigalow LC, Moreland (1986) Measurement of axial rotation of vertebrae in scoliosis. Spine (Phila Pa 1976) 11:213–218
Acknowledgments
Research support is gratefully acknowledged from the Rady Children’s Foundation Assaraf Family Research Fund and the Setting Scoliosis Straight Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Study conducted Rady Children’s Hospital, San Diego.
Rights and permissions
About this article
Cite this article
Yaszay, B., Bastrom, T.P., Bartley, C.E. et al. The effects of the three-dimensional deformity of adolescent idiopathic scoliosis on pulmonary function. Eur Spine J 26, 1658–1664 (2017). https://doi.org/10.1007/s00586-016-4694-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4694-y