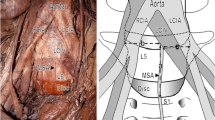
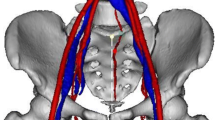
Abstract
Purpose
Pelvic and sacral surgeries are considered technically difficult due to the complex multidimensional anatomy and the presence of significant neurovascular structures. Knowledge of the key neurovascular anatomy is essential for safe and effective execution of partial and complete sacral resections. The goal of this anatomic, cadaveric study is to describe the pertinent neurovascular anatomy during these procedures.
Methods
Three embalmed human cadaveric specimens were used. Sacrectomies and sacroiliac joint resections were simulated and the structures at risk were identified. Both anterior and posterior approaches were evaluated.
Results
During sacroiliac joint resection, L5 nerve roots are at high risk for iatrogenic injury; the vasculatures at greatest risk are the common iliac vessels and internal iliac vessels with L5–S1 and S1–S2 high sacrectomies. Minor bleeding risk is associated with S2–S3 osteotomy because of the potential to damage superior gluteal vessels. S3–S4 osteotomy presents a low risk of bleeding. Adjacent nerve roots proximal to the resection level are at high risk during higher sacrectomies.
Conclusions
Several sacrectomy techniques are available and selection often depends on the specific case and surgeon preference; nevertheless, anatomic knowledge is extremely important. Considering the highly variable anatomic relations of the vascular bundles, a preoperative evaluation with CT or MRI with vascular reconstruction may be helpful to decrease bleeding risk by preemptively binding the internal iliac vessels in cases where higher tumors are present. To decrease the risk of damaging nerve roots, it is recommended to perform the resection as close to the involved foramina as possible.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Primary sacral tumors are relatively rare; chondroma being the most common, followed by giant cell tumor; other malignancies such as osteosarcoma, chondrosarcoma, and Ewing Sarcoma are rare in the sacral area [1]. While medical therapies are demonstrating more interesting results, en bloc resection with wide margins is still the mainstay for treatment [2].
Due to its anatomic characteristics, sacrectomy is considered a technically demanding surgery, especially when the tumor is proximal to S3, where vascular bundles and nerve roots are at high risk during surgery.
Several studies are available in the literature describing the anatomic relations of the vascular bundles within the low-lumbosacral spine; in effect, it is possible to note extremely high variability. The majority analyze the relationship between the L5–S1 disk and major vessels bifurcation, likely because these vessels are at risk during posterior discectomy and anterior lateral interbody fusion (ALIF).
Vaccaro et al. evidences that the aortic bifurcation is at the L4 level in 18 of 30 cases, lower in prone position in 1 of 30 cases, which is not a statistically significant distal migration in supine position; the confluence of the common iliac veins was identified at L4 in 15 of 30 cases, at L4–L5 in 6 of 30 cases and at L5 in 8 of 30 cases; nevertheless, the caudal migration was consistently more important in supine position, when considering 17 of 30 cases at the L5 level [3]. The common iliac vessels are near the midline at the L4–L5 disk level, but more lateral at the L5–S1 disk [3]. No sex-related differences were found [3].
Pirrò et al. [4] published another interesting study on 42 cadaveric dissections, reporting the aortic bifurcation at L4 in 50 % of cases, but at L5 in 39 % of cases; the iliocava junction was at L5 in 64 % of cases, L4 in 12 % and at S1 in 10 %; they evidence that the variability of the iliocava confluence complicates the anterior approach to the lumbosacral spine.
Capellades et al. [5] pointed their attention to the iliocava junction, confirming it is located at the L5–S1 disk level in 18.05 % of patients from a population of 134 who underwent MRI for low back pain.
Fatu et al. [6] studied the relationship of the internal iliac artery; they dissected 100 cases and verified that the origin of the internal iliac artery is at the level of the sacroiliac joint in females and internal to that in males; its length and ending is variable. Several papers are also present describing the anatomy and variations of the distal rami [7–9].
Even considering the variability of the lumbopelvic vascular bundles, knowledge of the key neurovascular anatomy is essential for safe and effective execution of partial and complete sacral resections.
The goal of this anatomic, cadaveric study is to describe the pertinent neurovascular anatomy found during these procedures. Special attention is paid to the Gigli saw technique, used for sacroiliac joint resections and osteotomes for transverse cuts [10, 11]. Though this anatomy is readily available in atlases, this study is unique, describing the pertinent surgical anatomy as it relates specifically to sacral and sacroiliac resection.
Materials and methods
Three embalmed human cadaveric specimens were used. Sacrectomies and sacroiliac joint resections were simulated and the structures at risk were identified. Both anterior and posterior approaches were evaluated. In supine position, a xypho-pubic skin incision was performed. A bilateral subcostal incision, a pubic transverse incision, and a symphysis resection were made to increase exposure; after dislocation and resection of the intestine, the retroperitoneal area was exposed. The anterior vascular bundles were dissected to identify the aorta and vena cava, the iliac vessels, and the internal and external iliac vessels. A bilateral L5–S4 dissection was performed from the foramina to the greater sciatic notch.
The cadavers were then placed in prone position; midline skin incisions extending from L3 to the coccyx were performed; two transverse incisions were proximally and distally placed, increasing exposure. The posterior aspect of L4 and L5 vertebras, sacrum, and coccyges was visualized. The dissection was laterally extended beyond the posterior iliac spines and sacroiliac joints.
The L5 transverse processes were cut and two moldable wires were inserted from the proximal edge of the sacroiliac joints, anteriorly up to the distal edge, and taken back to simulate placement of the Gigli saw through the sacroiliac joints during the total sacrectomy or sacroiliac joint resection.
Successively, the bodies were turned in supine position and the path of the wires described, demonstrating the risk to vascular bundles and nerve roots.
After performing a L5–S4 laminectomy in prone position, the transverse osteotomies were simulated at S3–S4, S2–S3, S1–S2, and L5–S1; the risks to vessels and nerves were described from an anterior point of view.
The present study was conducted respecting the ethical standards in the Helsinki Declaration of 1975, as revised in 2008, as well as the national law.
Results
During sacroiliac resection (Fig. 1a), the Gigli saw contacted L5, S1 and S2 nerve roots, with vessels coming from and to the internal iliac vascular bundles.
a The posterior aspect of the sacrum: the transverse processes’ fractures allow better access to the proximal sacroiliac joints; two wires simulating a Gigli saw were positioned to proceed with resection. b The anterior aspect of the right L5, S1, S2 nerve roots: the gluteal vessels were resected and removed to better visualize the relation of the sacral plexus to the sacral wings; arrows a correctly positioned Gigli saw. c The anterior aspect of the proximal left sacral wing: arrows a Gigli saw entrapping L5 nerve root, which would have been cut during resection
In 4 of 6 cases, the wire was passed without difficulty (Fig. 1b); in one case (Fig. 1c), the L5 left root should have been cut, and in the remaining case, L5 should have been spared. S1 would have been cut without consequence to the patient because it had already been proximally cut to include the S1 vertebral body during resection.
The L5–S1 osteotomy is extremely dangerous for the internal iliac vessels; the right internal iliac artery would have been at risk in all three right side hemi-body incisions. On the left side, the iliac artery would have been at risk in one case, the internal iliac vein in another, and the common iliac vein in yet another case where the cava bifurcation was extremely distal, covering the anterior part of the L5 body (Fig. 2a, b).
a The posterior aspect of the sacrum: the L5 roots were identified and spared; the conus medullaris (CM) was bound between the L5 and S1 foramina; arrows the osteotomes positioned for cutting the lateral aspect of L5–S1. b The correspondent anterior osteotomes are shown from an anterior point of view: the iliac internal artery (IIA) would have been at risk on the right side, and the common iliac vein (CIV) on the left (completely covering the left osteotome); in this case, the cava vein bifurcation was particularly distal. c The posterior aspect of the sacrum: the S1 roots were identified and spared; the CM was bound between S1 and S2; arrows the osteotomes positioned for cutting the lateral aspect of S1–S2. d Anterior view of the sacrum: during this dissection, the main vessels are relatively safe; nevertheless, significant bleeding can derive from damaging small branches of the internal iliac vessels, as shown in the picture (black arrow, laterally to the right osteotome, it is possible to note the superior gluteal artery); on the right side, the white arrow evidences the proximal cut damaging the corresponding S1 root
The S1–S2 osteotomy is dangerous to the gluteal vessels, most notably, when anterior binding of the internal iliac vessels is not performed. In 1 of 6 cases, injury to the superior gluteal artery would have been a risk (Fig. 2c, d). Proximal to the resection level, nerve roots can be damaged if the incision is made too near the proximal foramina (Fig. 2d, left osteotome).
The S2–S3 and S3–S4 distal osteotomies were performed for each case, without evident risk to the vascular bundles or roots (Fig. 3).
a The posterior aspect of the sacrum: the S2 roots were identified and spared; the CM was bound between S2 and S3 nerve roots onset; arrows the osteotomes positioned for cutting the lateral aspect of S2–S3. b The anterior view of the sacrum: S2, S3, and S4 roots were more horizontal than proximal, indicating a transverse osteotomy for that level; arrows the osteotomes; slight bleeding was associated with the middle sacral vessels plexus dissection. c The posterior aspect of the sacrum: the S3 roots were identified and spared, the S4 roots and the filum terminalis, bound, and cut; arrows the osteotomes positioned for cutting the lateral aspect of S3–S4. d S3–S4 dissection from an anterior point of view: at that level, the roots are quite horizontal and the vascular plexus is less represented; arrows the distant osteotomes positioned for cutting the lateral parts of the sacrum
Discussion
Pelvic and sacral surgeries are considered technically difficult due to the complex multidimensional anatomy of the area and the presence of neurovascular structures, which can be easily damaged. Several techniques are available and selection often depends on the specific case, and surgeon preference [12–15]. Independent from technique, knowledge of the anatomy helps to decrease intraoperative and postoperative complications, assuring better functional outcomes for the patient.
Sacroiliac joint resection is necessary for both total en bloc sacrectomy and resection of tumors located in the medial iliac bone. Resecting the L5 process is suggested to better visualize the proximal aspect of the sacroiliac joint and eventually to view, and spare the L5 nerve root. During the distal dissection, careful attention must be paid to avoid damaging the superior gluteal artery. The procedure is safer in cases where the internal iliac vessels were already bound during an anterior approach. In the present anatomical study, we simulated osteotomy using the Gigli saw technique; to reduce the risk of root and vessel entrapment, the saw is positioned as close to the bone as possible. Due to its anatomical position, the L5 root is at particular risk; it adheres to the superior and anterior sacral faces where it can easily be cut, especially in the presence of L5 body anterior osteophytes, which decrease its mobility (Fig. 4).
Unfortunately, no data are present in the literature about the effective risk of damaging L5, probably because it is not easily identified during surgery.
In the present study, an S1 root was entrapped in the simulated cut; although, it would not have been a problem, because it was proximally included in the high resection of the conus, previously.
The level of transverse cuts depends on the level of tumor. Wide resection is necessary for every primary sacral tumor. It is suggested to cut inside the first healthy level; obviously, the roots distal to the cut have to be tied, but attention must also be made not to damage the proximal pair, passing through the anterior foramina, which can be at risk when the lateral edges of the sacrum are cut. To reduce the risk of damaging the more proximal roots at the resection level, it is advisable to place osteotomes as close as possible to the distal foramen. At that level, the proximal root is more lateralized. It is always important to consider the oncological margin, maintaining slight osteotome convergence can be helpful to reduce further damage.
The L5–S1 and S1–S2 proximal osteotomies bear significant vascular risk during surgery. The gluteal bundles are fixed to the internal iliac arteries and vein in the lateral part of the cutting field to reduce bleeding risk. An anterior approach to bind the internal vessels is suggested before posterior resection. In every case, especially if the posterior approach is adopted, attention has to be made to complete the ventral part of osteotomies [14, 16].
The L5–S1 osteotomy is extremely dangerous, not for hypothetical damage to L5 roots, which can be avoided with good lateral dissection, but because during the lateral L5–S1 disk or S1 body cut, the internal iliac vessels can be easily damaged. In case of a distal cava bifurcation, even the common iliac veins can be at risk (Fig. 2b); internal iliac vessels are more at risk during S1–S2 resection.
Distal to S2–S3, osteotomies have minor vascular risk; gluteal vessels are relatively fair, so resection is advisable only by posterior approach without binding the internal iliac vessels (Fig. 3). Also, the risk to proximal nerve roots is lower due to the more horizontal pathway (Fig. 3b, d). Nevertheless, distal cutting is always advisable at this level if it does not compromise the wide margin.
The S2–S3 and S3–S4 distal osteotomies are relatively safe for the main vascular bundles, which are more proximal, and for S3, and S4 nerve roots. At those levels, the sacroiliac joints are proximal, and the lateral sacral edges are very convergent to the coccyges. The nerve roots exit horizontally to reach the sacral notch laterally, merging into the sciatic nerve. To the best of our knowledge, this is the first cadaveric study showing the anatomic bases of possible surgical complications related to sacral and sacroiliac resection. Further anatomic studies with a high number of dissections could be helpful to better define the anatomy and normal variability between individuals.
References
Unni KK, Inwards CY (2010) Dahlin’s Bone Tumors. Lippincott Williams & Wilkins, Philadelphia
Bergh P, Gunterberg B, Meis-Kindblom JM et al (2001) Prognostic factors and outcome of pelvic, sacral, and spinal chondrosarcomas: a center-based study of 69 cases. Cancer 91(7):1201–1212
Vaccaro AR, Kepler CK, Rihn JA et al (2012) Anatomical relationships of the anterior blood vessels to the lower lumbar intervertebral discs: analysis based on magnetic resonance imaging of patients in the prone position. J Bone Joint Surg Am 94(12):1088–1094
Pirró N, Ciampi D, Champsaur P et al (2005) The anatomical relationship of the iliocava junction to the lumbosacral spine and the aortic bifurcation. Surg Radiol Anat 27(2):137–141
Capellades J, Pellisé F, Rovira A et al (2000) Magnetic resonance anatomic study of iliocava junction and left iliac vein positions related to L5–S1 disc. Spine 25(13):1695–1700
Fătu C, Puişoru M, Fătu IC (2006) Morphometry of the internal iliac artery in different ethnic groups. Ann Anat 188(6):541–546
Sañudo JR, Mirapeix R, Rodriguez-Niedenführ M et al (2011) Obturator artery revisited. Int Urogynecol J 22(10):1313–1318
Pathi SD, Castellanos ME, Corton MM (2009) Variability of the retropubic space anatomy in female cadavers. Am J Obstet Gynecol 201(5):524.e1–524.e5
Holub Z, Lukac J, Kliment L et al (2005) Variability of the origin of the uterine artery: laparoscopic surgical observation. J Obstet Gynaecol Res 31(2):158–163
Zoccali C, Zoccali G, Bakaloudis G et al (2013) A new technique to perform pelvic osteotomy using Gigli saw. J Surg Oncol 108(2):136
Brunori A, Bruni P, Greco R et al (1995) Celebrating the centennial (1894–1994): Leonardo Gigli and his wire saw. J Neurosurg 82(6):1086–1090
Clarke MJ, Dasenbrock H, Bydon A et al (2012) Posterior-only approach for en bloc sacrectomy: clinical outcomes in 36 consecutive patients. Neurosurgery 71(2):357–364
Zhang HY, Thongtrangan I, Balabhadra RS et al (2003) Surgical techniques for total sacrectomy and spinopelvic reconstruction. Neurosurg Focus 15(2):E5
Gokaslan ZL, Romsdahl MM, Kroll SS et al (1997) Total sacrectomy and Galveston L-rod reconstruction for malignant neoplasms. Technical note. J Neurosurg 87(5):781–787
Fourney DR, Rhines LD, Hentschel SJ et al (2005) En bloc resection of primary sacral tumors: classification of surgical approaches and outcome. J Neurosurg Spine 3(2):111–122
Clarke MJ, Dasenbrock H, Bydon A et al (2012) Posterior-only approach for en bloc sacrectomy: clinical outcomes in 36 consecutive patients. Neurosurgery 71(2):357–364
Conflict of interest
The authors declare the absence of any conflict of interest or financial relationships pertaining to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zoccali, C., Skoch, J., Patel, A. et al. The surgical neurovascular anatomy relating to partial and complete sacral and sacroiliac resections: a cadaveric, anatomic study. Eur Spine J 24, 1109–1113 (2015). https://doi.org/10.1007/s00586-015-3815-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-3815-3