Abstract
Purpose
Current solutions for navigated spine surgery remain hampered by restrictions in surgical workflow as well as a limited versatility and applicability. Against this background, we report the first experience of navigated spinal instrumentation with the mobile AIRO® intraoperative computed tomography (iCT) scanner.
Methods
AIRO® iCT was used for navigated posterior spinal instrumentation of 170 screws in 23 consecutive patients operated on in our Department between the first use of the system in May 2014 and August 2014. The indications for AIRO® were based on the surgical region, anatomical complexity and the need for >3 segment instrumentation. Following navigated screw insertion, screw positions were confirmed intraoperatively by a second iCT scan. CT data on screw placement accuracy were retrospectively reviewed and analyzed by an independent observer.
Results
AIRO®-based spinal navigation was easy to implement and successfully accomplished in all patients, adding around 18–34 min to the net surgery time. A systematic description of the authors’ approach, setup in the OR and workflow integration of the AIRO® is presented. Analysis of screw placement accuracy revealed 9 (5.3 %) screws with minor pedicle breaches (<2 mm). A total of 7 screws (4.1 %) were misplaced >2 mm, resulting in an accuracy rate of 95.9 %.
Conclusions
The AIRO® system is an easy-to-use and versatile iCT for navigated spinal instrumentation and provides high pedicle screw accuracy rates. Although the authors’ experience suggests that the learning curve associated with AIRO®-based spinal navigation is steep, a systematic user-based approach to the technology is required.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Recent studies and meta-analysis have evidenced the superior accuracy of navigated spinal instrumentation compared to non-navigated techniques with further improvement through additional implementation of intraoperative computed tomography (iCT) [1–9]. Despite their hallmark characteristic of permitting immediate assessment of incorrect screw positioning with the chance of direct revision, however, currently available iCT setups remain hampered for one or more reasons, such as their limited versatility due to permanently installed iCT scanners in a single dedicated OR suite, the small iCT gantry size or a limited scan volume [1, 5, 8–13]. Moreover, currently available full-fledged iCT/navigation setups usually require cumbersome surface- or point-matching for referencing, which may obstruct surgical workflow [1, 5, 8–10]. In particular for novice operators, initial experience with spinal image guidance may, therefore, not immediately demonstrate the same high accuracy rates regarding screw positioning widely quoted in the literature.
Recently, a new mobile iCT has become commercially available with focus on combining full-fledged iCT imaging and navigation, while at the same time improving versatility and optimizing surgical workflow. Against this background, the aim of the present study was to report on accuracy and workflow of navigated spinal instrumentation with the mobile AIRO® iCT scanner.
Materials and methods
Patients and surgical indications
From May 2014 to August 2014, iCT data of 23 patients (11 female, 12 male; median age 68, range 31 to 83 years) with surgical indication for navigated posterior instrumentation were acquired. The surgical indications were degenerative disease (10/23; 43 %), tumors (8/23; 35 %) and trauma (5/23; 22 %) in anatomically complex deformities, revision cases, regions of the spine that are typically difficult to visualize with conventional iso-C 3D C-arm navigated fluoroscopy, such as the cranio-cervical or cervico-thoracic junction, or cases that required multilevel (>3 segments) instrumentation. One patient had occipito-cervical instability, 3 patients had C1/C2 instability, 3 patients had cervico-thoracic instability, 6 patients had long-segment (>5 segments) thoraco-lumbar instability and 10 patients had lumbar–sacral instability.
General setup of the AIRO® during navigated spinal instrumentation
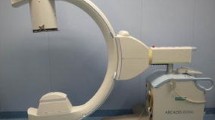
For iCT-based spinal navigation, we used the mobile AIRO® CT scanner (Brainlab AG, Feldkirchen, Germany). This system is designed to function within an existing OR suite and contains a mobile CT gantry (diameter 107 cm; dimensions 30.5 cm × 38 cm), which houses the X-ray tube, 32 slice helical scan detector array, high-voltage generator, air cooling system and battery pack. The AIRO® is controlled by a handheld detachable touchpad, which permits execution of CT scanning. A suspension controlled electrical drive system allows the AIRO® to be maneuvered from OR to OR by a single person (Fig. 1a). Surgery is performed on a mobile, radiolucent, carbon fiber CT examination table (TRUMPF TruSystem 7500, Trumpf Inc., Farmington, Connecticut, USA), which is fixed to the gantry during surgery. The footprint of the entire setup is 1.5 m2. For navigation, the AIRO® is connected to an image-guidance system and infrared tracking camera (BrainLab Curve™, Brainlab AG, Feldkirchen, Germany) for automatic image transfer and image-patient co-registration. A schematic of the general AIRO® setup in our OR suite during surgery and iCT scanning is illustrated in Fig. 1b.
a Setup of the AIRO® iCT in the OR. Positioning of the AIRO® is performed by one person with the help of a suspension controlled battery powered electrical drive system. b Schematic of the OR setup for navigated spinal instrumentation with the mobile AIRO® iCT. To permit the full range of surgical access during surgery, (left) the OR table is positioned perpendicular to the iCT gantry. For iCT scanning, the operating table is manually turned 90° clockwise in order to allow positioning within the iCT gantry (right). N nurse instrumentation trays, A anesthesiology unit, Nav navigation system, C navigation camera, 1 surgeon, 2 surgeon, 3 scrub nurse, 4 circulating nurse, 5 anesthesiologist, 6 anesthesiologic nurse, 7 radiology technician
Surgery and preparation for iCT scanning
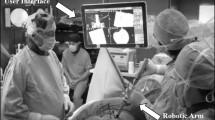
After induction of anesthesia, patients were positioned prone on the iCT examination/OR table without the need for repositioning during iCT scanning. For surgeries from the cervical to mid-thoracic level, the patient was positioned to allow headfirst entry into the iCT gantry with the head fixed in a radiolucent carbon fiber Mayfield clamp (Trumpf X-RAY, Trumpf Inc., Farmington, Connecticut, USA) (Fig. 2a). For surgeries below the mid-thoracic level, the patient was oriented for feet-first entry into the iCT gantry with the head positioned in a foam-cushioned headrest.
a–d Intraoperative setup, e navigation and f CT images acquired with the AIRO® iCT for navigated instrumentation at the cervico-thoracic junction. a View through the CT gantry showing the patients’ position on the carbon OR table. b Intraoperative setup during the surgical approach before iCT scanning. c Attachment of the navigation tracking device to a thoracic spinous process at the lower end of the surgical opening. d The AIRO® iCT in scanning position after 90° rotation of the operating table. e Real-time screenshot of navigated entry-point identification before tapping and drilling of the left Th3 pedicle. f Intraoperative CT screenshot following multilevel screw insertion from C3 to Th4 with focus on the Th3 level for direct intraoperative verification of correct screw placement
Surgical exposure was carried out in routine fashion (Fig. 2b). After completion, the navigation tracking device (Brainlab AG, Feldkirchen, Germany) was clamped 1–2 spinous processes cranial or caudal to the levels of instrumentation (Fig. 2c). Next, the nurses’ instrumentation trays and the anesthesiology unit were moved to the far side of the OR and the patient was positioned within the iCT gantry by manually rotating the OR Table 90° clockwise.
Intraoperative CT scanning
After definition of the scan parameters, the scan area was programmed with the assistance of laser guidance. The navigation camera was adjusted to allow co-registration of the tracking device and the registration fiducials mounted on the AIRO® gantry. Next, the OR personal exited the room and the iCT scan was executed by an in-house radiology technician on call. Through collaboration with the Department of Radiology, this technician is authorized to execute the scan by order of the 24/7 in-house radiologists with technical CT qualification. The time between calling the technician to his/her arrival in the OR (approximately 3–5 min) is bridged by prepping for the iCT procedure, which results in little to no delay to perform the scans. During image acquisition, the iCT gantry moved over the patient, with the position of the catheters and ventilation tube remaining stationary (Fig. 2d). All iCT scanning was performed under apnea ventilation following pre-oxygenation to reduce motion artifacts.
Navigated screw insertion with automatic patient-image co-registration
After completion of the scan, the acquired image data were automatically transferred to the in-house DICOM system and the intraoperative image-guidance platform (Brainlab Spinal Navigation Software Version 3.0 and Brainlab Curve™, Brainlab AG, Feldkirchen, Germany). The patient was rotated 90° counterclockwise into the surgery position and an image-guided surface probe was used to validate the automatic registration. Next, a navigated drill guide (Brainlab AG, Feldkirchen, Germany) was used to define the image-guided trajectory of pediculation in axial, coronal and sagittal real-time views (Fig. 2e). A power drill (Stryker Cordless Driver, Kalamazoo, Michigan, USA) with a 2.6 mm drill bit (Brainlab AG, Feldkirchen, Germany) was then inserted through the drill guide and advanced to a desired depth by the assisting surgeon. After removal of the drill, a guide wire was inserted and the drill guide was removed from the surgical field. Next, the pedicle was tapped with a cannulated tap and cannulated pedicle screws with a diameter of 3.5–7.5 mm (cervical spine: neon3™, Ulrich Medical, Ulm, Germany; thoraco-lumbar-sacral spine: CD Horizon® Legacy™ Spinal System, Medtronic, Minneapolis, Minnesota, USA) were inserted over the guide wire without additional image guidance. In 3 cases (n = 11 screws), cervical pedicle screws were placed with a non-cannulated system (Aesculap S4® Cervical, Aesculap AG, Tuttlingen, Germany) and the assistance of a navigated tap (Brainlab AG, Feldkirchen, Germany) (Fig. 3). In all cases, screw length and diameter were determined intraoperatively, based on the scale readings superimposed onto the simulated navigation trajectory of the drill guide.
AIRO®-based navigated pedicle screw tapping and insertion at the upper- and mid-cervical level. Axial (left), sagittal (center) and coronal (right) real-time screenshots during the final stages of navigated tapping (upper panels) of the right C2 pedicle with corresponding iCT images to judge the result of screw insertion before closure (lower panels)
Evaluation of workflow and screw position
The times required for the setup and execution of the AIRO® iCT were noted for each procedure.
Following the instrumentation, all screws were evaluated by a second iCT. Final screw positions were retrospectively graded by an independent observer according to the previous classification systems of the cervical [14] and thoraco-lumbar-sacral [5, 8, 15] spine. The main objective was to determine the screw accuracy rate, which we calculated from the percentage of screws (a) placed completely within the pedicle [correct (c)] or (b) showing minor pedicle perforations (m.p.; <2 mm) (Table 1).
Results
Intraoperative applicability
Intraoperative CT-based navigated spinal instrumentation with the AIRO® resulted in no case of wrong level surgery. The large gantry diameter permitted iCT in obese patients and in patients who were positioned in a Mayfield clamp. The high image quality and resolution permitted radiographic visualization of all levels of the spine and its surrounding structures without difficulties. Automatic image-patient co-registration was successful in all cases and provided seamless workflow integration. There was no need for a repeat iCT scan due to faulty co-registration or a wrong-level scan. Up to 11 vertebrae were instrumented after a single registration sequence without a significant increase in screw misplacement >2 mm.
Intraoperative workflow
Compared to non-navigated spinal instrumentation, the additional steps and average time for navigated instrumentation with the AIRO® iCT include:
-
1.
Removal of the mobile column for the regular OR Table (2–3 min).
-
2.
Positioning of the AIRO® (5–7 min).
-
3.
Setup of the image-guidance system (5–6 min).
-
4.
Surgical prep-time before iCT navigation scan (3–7 min).
-
5.
iCT prep-time before iCT navigation scan (3–5 min).
-
6.
iCT scan (median scan time 10:51 s; range 7:45–17:23 s).
-
7.
Prep-time before resuming surgery (2–4 min).
-
8.
Second iCT scan after screw insertion (9–17 min; steps 4–7).
Thus, the total additional time required for setup and navigated instrumentation with the AIRO® was determined between 30 and 50 min with an additional mere surgical time (steps 4–8) between 18 and 34 min. The longer time specification is representative of the first 3 surgeries, whereas the shorter time specification represents the following 20 procedures.
Accuracy of image guidance
Hardware-related artifacts were minor to none and irrelevant for the assessment of screw positions, which is demonstrated in the following exemplary cases:
-
Cervico-thoracic instrumentation (Fig. 2).
-
Cervical pedicle screw placement (Fig. 3).
-
Long-segment thoracic instrumentation (Fig. 4).
Fig. 4 AIRO®-based navigation for long-segment thoracic instrumentation. Real-time screenshots following iCT scanning before (left) and after (right) navigated screw insertion from Th3/4/5 to Th8/9/10 demonstrate the large AIRO® field of view and the possibility of accurate multilevel screw insertion after only a single registration scan
Independent review of post-instrumentation iCT scans of 170 pedicle screws revealed 9 (5.3 %) minor perforations below 2 mm and 7 (4.1 %) misplaced screws >2–4 mm. No screws were misplaced >4 mm (Table 1). Of the 7 misplaced screws, 3 (Th9, L5, L5) were displaced laterally, 3 (Th3, L4, L5) were displaced medially and 1 screw (C1) was displaced cranially. The median distance of misplaced screws to the navigation tracking device was 2 (range 1–8) segments. Diameter and height of the 6 perforated pedicles were greater than the diameter of the implanted screws, even in the perforated Th3 pedicle with the smallest diameter (Th3: width 5.6 mm/height 11.1 mm; screw diameter: 4.5 mm; displacement: 2.2 mm medial). In all cases, notion was made to leave the screws in place, because less than half of the screw diameter had penetrated the pedicle wall and there was no impingement of adjacent neurovascular structures or concern regarding biomechanical stability.
Discussion
Accuracy and workflow
As with any new technology, iCT-based spinal navigation underlies a learning curve that may force novice surgeons to abandon this technique early. In our experience, however, navigated spinal instrumentation with the AIRO® iCT was efficient, easy to implement and characterized by a steep learning curve, which is supported by the much shorter time that we required after the first 3 procedures had been performed. It should be noted, however, that our previous experience with iso-C 3D C-arm navigation may have facilitated certain steps of the procedure dealing with the spinal navigation, such as the setup of the image-guidance system and the tracking camera, draping, clamping of the tracking device and handling of the navigated instruments. Unfortunately, reports investigating the learning curve associated with iCT image guidance are sparse but it has been hypothesized that merely 5 cases may be necessary for surgeons with image-guidance skills to obtain sufficient experience with this technology [16]. Recent statistical evidence of the learning curve for 3D scan acquisition, pedicle screw placement and accuracy underlines that routine use of spinal navigation is critical to overcome the learning curve, establish normal workflow and improve screw accuracy rates [17]. Despite initial concerns that routine use of image guidance could negatively affect the training of young spinal surgeons, so far navigated instrumentation has had a beneficial training effect in our department, which is also in line with the general experience in cranial navigation. During surgery, for example, residents first identify the screw entry points based on anatomical landmarks before using image guidance for confirmation, which provides the benefit of immediate feedback of anatomical knowledge and facilitates screw insertions by only fluoroscopic guidance in other cases where navigation and/or iCT are unavailable due to full-capacity use elsewhere or in late-night emergency procedures.
The screw misplacement rate of approximately 4 % that we noted in our 23 consecutive patients appears in line with the numbers quoted in previous reports [3, 5, 7, 13]. Although this may partly be due to our previous experience with navigated spinal instrumentation, we believe that the good accuracy rates were largely due to the following factors: First, the mobility and easy handling of the AIRO® contributed to a non-disrupted surgical workflow. Second, the AIRO® reliably delivered CT-quality images, even in obese patients or in regions of the spine, which are difficult for radiographic visualization, such as the cranio-cervical and cervico-thoracic junction. Moreover, the high scan resolution and detail quality resulted in only negligible hardware artifacts, which is an important safety feature since it allows precise intraoperative screw assessment with the possibility of direct navigated revision when needed. Importantly, AIRO®-based spinal instrumentation also increases the safety and comfort of the OR personal, due to a 1:10 radiation exposure reduction to the surgeon [18] and the obsoleteness to wear lead-shielded vests compared to non-navigated fluoroscopic screw insertion.
AIRO® iCT for navigated spinal instrumentation
Although improper pedicle screw placement rarely results in permanent neurological injury, misplaced screws often result in persistent pain and surgical re-intervention, which can be avoided by the use of image-guided spinal navigation [2–4]. Against this background, navigated spinal instrumentation based on iCT scanning has gained increasing attention over the past years [1, 5, 8–10]. However, the advantages of contemporary iCT solutions, such as the higher image quality, unlimited field of view and immediate intraoperative verification of accurate screw placement still remain hampered by the limited versatility of a permanently installed iCT in a dedicated OR suite next to its small gantry size and cumbersome handling.
In our initial experience of 23 patients, AIRO®-based spinal navigation offered a versatile alternative to a conventional iCT, with the hallmark characteristics of a large gantry size, high mobility and easy handling, which allowed utilization in multiple OR suites for increased flexibility and efficiency. In our department, for example, 2 out of 6 OR suites have been converted to support the use of the AIRO® iCT by removal of the fixed columns for the previous OR tables. Next to a more flexible planning of OR schedule, this should also increase (cost-) efficiency in the OR [12], which must be kept in mind due to the high cost of the AIRO® technology compared to an iso-C 3D C-arm, for example. Currently, one of the main problems is that these higher costs—not only for AIRO® but also for other imaging/navigation solutions such as the O-Arm® or even iso-C 3D C-arm navigation—are not reimbursed compared to non-navigated (fluoroscopic) instrumentation. Therefore, future studies must prove which technology has the best cost/benefit ratio.
Routine postoperative CT imaging after spinal instrumentation is not mandatory and remains a matter of debate. However, our results suggest that AIRO® iCT represents a valid and reliable method for assessment of screw positioning—at the surgeons discretion—with the chance for immediate intraoperative revision. Further, post-instrumentation iCT might also be helpful to determine the degree of decompression or debulking in complex trauma or tumor cases, in addition to benefiting a teaching environment. Also, AIRO®-based spinal navigation should not be associated with additional radiation exposure compared to our current protocol for navigated instrumentation with iso-C 3D C-arm fluoroscopy (Fig. 5). In Departments without routine postoperative CT imaging, the iCT scan after screw insertion could of course be omitted. In any case, we encourage a close collaboration with the Radiologists/Neuroradiologists to ensure radiological diagnostics of iCT findings and for assistance in radiation protection.
Flow chart illustrating the typical radiation exposure for navigated spinal instrumentation. The current standard of navigated spinal instrumentation (iso-C 3D C-arm fluoroscopy with automatic patient-image co-registration; left) in our department is compared to our current AIRO® iCT protocol (right), which compensates for additional intraoperative fluoroscopy as well as for pre- and postoperative CT scanning
Limitations and pitfalls
It is well known that the risk of inaccuracy increases with decreasing dimension of the pedicle isthmus [19]. However, in each of the 6 perforated pedicles in our series, the dimension of the pedicle isthmus was greater than the screw diameter. This suggests that our inaccuracies are rather caused by misleading navigation than by too small anatomical structures. Although the number of pedicle breaches was too low for statistical analysis, possible explanations could be biomechanical instability in trauma or tumor cases, soft tissue tension with relative movement of the spine during positioning of the drill guide, loosening or insufficient fixation of the navigation tracking device as well as instrumentation at a large distance from the tracking device with a only a single iCT registration scan. Consequently, the virtual reality must be compared to the surgical site at any time in addition to real-time fluoroscopic plausibility checks when in doubt, particularly in critical regions (i.e., cervical spine and regions with small pedicle isthmus dimensions). In the AIRO® setup that we described, for example, fluoroscopy can be easily integrated even at a later time point during surgery. Therefore, surgical landmarks and fluoroscopy should always remain an invaluable gold standard for pedicle screw insertion and the surgeon should use routine cases to familiarize himself with spinal navigation to translate safe applicability to complex settings.
In conclusion, our findings suggest that AIRO® is efficient, safe and simple to use. The frequent exposure of the entire OR team to the technology should facilitate the development of a systematic and professional approach with the goal to optimize workflow and clinical results for the benefit of our patients. Importantly, spinal navigation must always be regarded as an adjunct to thorough knowledge of spinal anatomy and not as a substitute for it. Therefore, residents and spinal surgeons must also be proficient in using fluoroscopic/free-hand screw insertion techniques to recognize pitfalls and limitations of navigated instrumentation [20] and to deal with the situation if for any reason navigation should not be available.
References
Tormenti MJ, Kostov DB, Gardner PA et al (2010) Intraoperative computed tomography image-guided navigation for posterior thoracolumbar spinal instrumentation in spinal deformity surgery. Neurosurg Focus 28:E11. doi:10.3171/2010.1.FOCUS09275
Shin BJ, James AR, Njoku IU, Hartl R (2012) Pedicle screw navigation: a systematic review and meta-analysis of perforation risk for computer-navigated versus freehand insertion. J Neurosurg Spine 17:113–122. doi:10.3171/2012.5.SPINE11399
Gelalis ID, Paschos NK, Pakos EE et al (2012) Accuracy of pedicle screw placement: a systematic review of prospective in vivo studies comparing free hand, fluoroscopy guidance and navigation techniques. Eur Spine J 21:247–255. doi:10.1007/s00586-011-2011-3
Kosmopoulos V, Schizas C (2007) Pedicle screw placement accuracy: a meta-analysis. Spine (Phila Pa 1976) 32:E111–E120
Waschke A, Walter J, Duenisch P et al (2013) CT-navigation versus fluoroscopy-guided placement of pedicle screws at the thoracolumbar spine: single center experience of 4,500 screws. Eur Spine J 22:654–660. doi:10.1007/s00586-012-2509-3
Ishikawa Y, Kanemura T, Yoshida G et al (2010) Clinical accuracy of three-dimensional fluoroscopy-based computer-assisted cervical pedicle screw placement: a retrospective comparative study of conventional versus computer-assisted cervical pedicle screw placement. J Neurosurg Spine 13:606–611. doi:10.3171/2010.5.SPINE09993
Tian W, Liu Y, Zheng S, Lv Y (2013) Accuracy of lower cervical pedicle screw placement with assistance of distinct navigation systems: a human cadaveric study. Eur Spine J 22:148–155. doi:10.1007/s00586-012-2494-6
Zausinger S, Scheder B, Uhl E et al (2009) Intraoperative computed tomography with integrated navigation system in spinal stabilizations. Spine (Phila Pa 1976) 34:2919–2926
Scheufler K-M, Franke J, Eckardt A, Dohmen H (2011) Accuracy of image-guided pedicle screw placement using intraoperative computed tomography-based navigation with automated referencing, part I: cervicothoracic spine. Neurosurgery 69:782–795. doi:10.1227/NEU.0b013e318222ae16 (discussion 795)
Barsa P, Frohlich R, Benes V, Suchomel P (2014) Intraoperative portable CT-scanner based spinal navigation—a feasibility and safety study. Acta Neurochir (Wien). doi:10.1007/s00701-014-2184-8
Van de Kelft E, Costa F, Van der Planken D, Schils F (2012) A prospective multicenter registry on the accuracy of pedicle screw placement in the thoracic, lumbar, and sacral levels with the use of the O-arm imaging system and Stealth Station Navigation. Spine (Phila Pa 1976) 37:E1580–E1587
Costa F, Cardia A, Ortolina A et al (2011) Spinal navigation: standard preoperative versus intraoperative computed tomography data set acquisition for computer-guidance system: radiological and clinical study in 100 consecutive patients. Spine (Phila Pa 1976) 36:2094–2098. doi:10.1097/BRS.0b013e318201129d
Rivkin MA, Yocom SS (2014) Thoracolumbar instrumentation with CT-guided navigation (O-arm) in 270 consecutive patients: accuracy rates and lessons learned. Neurosurg Focus 36:E7. doi:10.3171/2014.1.FOCUS13499
Neo M, Sakamoto T, Fujibayashi S, Nakamura T (2005) The clinical risk of vertebral artery injury from cervical pedicle screws inserted in degenerative vertebrae. Spine (Phila Pa 1976) 30:2800–2805
Learch TJ, Massie JB, Pathria MN et al (2004) Assessment of pedicle screw placement utilizing conventional radiography and computed tomography: a proposed systematic approach to improve accuracy of interpretation. Spine (Phila Pa 1976) 29:767–773
Nottmeier EW, Crosby TL (2007) Timing of paired points and surface matching registration in three-dimensional (3D) image-guided spinal surgery. J Spinal Disord Tech 20:268–270
Ryang Y-M, Villard J, Obermuller T et al (2014) Learning curve of 3D-fluoroscopy image-guided pedicle screw placement in the thoracolumbar spine. Spine J. doi:10.1016/j.spinee.2014.10.003
Villard J, Ryang Y-M, Demetriades AK et al (2014) Radiation exposure to the surgeon and the patient during posterior lumbar spinal instrumentation: a prospective randomized comparison of navigated versus non-navigated freehand techniques. Spine 39:1004–1009. doi:10.1097/BRS.0000000000000351
Rampersaud YR, Simon DA, Foley KT (2001) Accuracy requirements for image-guided spinal pedicle screw placement. Spine (Phila Pa 1976) 26:352–359
Rahmathulla G, Nottmeier EW, Pirris SM et al (2014) Intraoperative image-guided spinal navigation: technical pitfalls and their avoidance. Neurosurg Focus 36:E3. doi:10.3171/2014.1.FOCUS13516
Conflict of interest
Peter Vajkoczy is a consultant for Aesculap and Ulrich Medical.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hecht, N., Kamphuis, M., Czabanka, M. et al. Accuracy and workflow of navigated spinal instrumentation with the mobile AIRO® CT scanner. Eur Spine J 25, 716–723 (2016). https://doi.org/10.1007/s00586-015-3814-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-3814-4